Vixotrigine (original) (raw)
From Wikipedia, the free encyclopedia
Analgesic drug under development
Pharmaceutical compound
Vixotrigine
 |
|
|---|---|
| Clinical data | |
| Other names | Raxatrigine; CNV1014802; GSK-1014802; BIIB 074 |
| Routes of administration | By mouth |
| ATC code | None |
| Identifiers | |
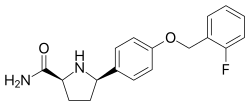
| IUPAC name (2_S_,5_R_)-5-[4-[(2-fluorophenyl)methoxy]phenyl]pyrrolidine-2-carboxamide | |
| CAS Number | 934240-30-9 934240-35-4 (mesylate) |
| PubChem CID | 16046068 |
| ChemSpider | 13174481 |
| UNII | QQS4J85K6Y |
| KEGG | D11179 |
| CompTox Dashboard (EPA) | DTXSID901031867 |
| Chemical and physical data | |
| Formula | C18H19FN2O2 |
| Molar mass | 314.360 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES C1CC(NC1C2=CC=C(C=C2)OCC3=CC=CC=C3F)C(=O)N | |
| InChI InChI=1S/C18H19FN2O2/c19-15-4-2-1-3-13(15)11-23-14-7-5-12(6-8-14)16-9-10-17(21-16)18(20)22/h1-8,16-17,21H,9-11H2,(H2,20,22)/t16-,17+/m1/s1Key:JESCETIFNOFKEU-SJORKVTESA-N |
Vixotrigine (INNTooltip International Nonproprietary Name, USANTooltip United States Adopted Name), formerly known as raxatrigine (INNTooltip International Nonproprietary Name, USANTooltip United States Adopted Name), is an analgesic which is under development by Convergence Pharmaceuticals for the treatment of lumbosacral radiculopathy (sciatica) and trigeminal neuralgia (TGN).[1][2][3] Vixotrigine was originally claimed to be a selective central Nav1.3 blocker, but was subsequently redefined as a selective peripheral Nav1.7 blocker.[_citation needed_] Following this, vixotrigine was redefined once again, as a non-selective voltage-gated sodium channel blocker.[_citation needed_] As of January 2018, it is in phase III clinical trials for trigeminal neuralgia and is in phase II clinical studies for erythromelalgia and neuropathic pain.[4] It was previously under investigation for the treatment of bipolar disorder, but development for this indication was discontinued.[4]
- ^ "CNV1014802". Convergence Pharmaceuticals.
- ^ McMahon S, Koltzenburg M, Tracey I, Turk DC (1 March 2013). Wall & Melzack's Textbook of Pain: Expert Consult - Online. Elsevier Health Sciences. p. 508. ISBN 978-0-7020-5374-0.
- ^ Bagal SK, Chapman ML, Marron BE, Prime R, Storer RI, Swain NA (August 2014). "Recent progress in sodium channel modulators for pain". Bioorganic & Medicinal Chemistry Letters. 24 (16): 3690–3699. doi:10.1016/j.bmcl.2014.06.038. PMID 25060923.
- ^ a b "Vixotrigine - Biogen". Adis Insight. Springer Nature Switzerland AG.