Thyroid Autoimmune Disease: Demonstration of Thyroid Antigen-Specific B Cells and Recombination-Activating Gene Expression in Chemokine-Containing Active Intrathyroidal Germinal Centers (original) (raw)
Abstract
Autoimmune thyroid disease—Hashimoto thyroiditis and Graves’ disease—patients produce high levels of thyroid autoantibodies and contain lymphoid tissue that resembles secondary lymphoid follicles (LFs). We compared the specificity, structure, and function of tonsil and lymph node LFs with those of the intrathyroidal LFs to assess the latter’s capability to contribute to autoimmune response. Thyroglobulin and thyroperoxidase binding to LFs indicated that most intrathyroidal LFs were committed to response to thyroid self-antigens and were associated to higher levels of antibodies to thyroglobulin, thyroperoxidase, and thyroid-stimulating hormone receptor. Intrathyroidal LFs were microanatomically very similar to canonical LFs, ie, they had well-developed germinal centers with mantle, light, and dark zones and each of these zones contained B and T lymphocytes, follicular dendritic and interdigitating dendritic cells with typical phenotypes. Careful assessment of proliferation (Ki67) and apoptosis (terminal dUTP nick-end labeling) indicators and of the occurrence of secondary immunoglobulin gene rearrangements (RAG1 and RAG2) confirmed the parallelism. Unexpected high levels of RAG expression suggested that receptor revision occurs in intrathyroidal LFs and may contribute to generate high-affinity thyroid autoantibodies. Well-formed high endothelial venules and a congruent pattern of adhesion molecules and chemokine expression in intrathyroidal LFs were also detected. These data suggest that ectopic intrathyroidal LFs contain all of the elements needed to drive the autoimmune response and also that their microenvironment may favor the expansion and perpetuation of autoimmune response.
Autoimmune thyroid disease (AITD) is a term that includes the various clinical forms of autoimmune thyroiditis, such as classical Hashimoto’s thyroiditis (HT), Graves’ disease (GD), and primary myxedema. An almost invariable feature of AITD is the production of antibodies to at least one of the main thyroid-specific autoantigens, ie, thyroglobulin (Tg), the main protein of the colloid; thyroperoxidase (TPO), the enzyme that catalyzes iodine organification, and the receptor for thyrotropin (TSH-R). 1 Other thyroid-specific autoantigens, such as the newly described sodium iodine symporter 2 and minor colloid antigens, are still being characterized but they do not seem to be the dominant targets of humoral autoimmune response. Thyroid autoantibodies are good clinical markers of disease, and TSH-R antibodies, also known as thyroid-stimulating immunoglobulins, are the direct cause of hyperthyroidism in GD patients and one of the best examples of pathogenic autoantibodies. 3 Taking advantage of their high titer, the availability of purified antigens and of surgically removed tissue, thyroid autoantibodies were subjected to such an exhaustive scrutiny that they arguably became the best-characterized human autoantibodies. A wide variety of techniques have been used, including the generation of human monoclonal antibodies both by hybridoma 4,5 and combinatorial techniques using as source B cells from autoimmune thyroids and the corresponding regional lymph nodes (LNs). 6-8 Epitope restriction typical of human autoantibodies, first suggested for Tg antibodies, 9 has been repeatedly confirmed using human and mouse monoclonal antibodies and recombinant mutated forms of thyroid antigens. 10-13 Genetic analysis provided evidence of restricted Ig V gene family usage and extensive somatic hypermutation, as corresponds to high-affinity antibodies produced in the course of antigen-driven responses. 12-14 The analysis of the T cell response using a variety of approaches also suggests that AITD is caused by antigen-driven responses. 15
Histologically, HT is characterized by lymphocytic infiltration that may progressively replace thyroid follicles. Lymphoid infiltrates are also present in GD glands, but in this condition most of the thyroid remains intact except for the signs of hyperfunction. Cases that share features of HT and GD occur are occasionally observed. 16 Lymphoid infiltrates often organize themselves as follicle-like structures containing germinal centers (GCs) similar to those in secondary lymphoid follicles (LFs) of LNs. Intrathyroidal B lymphocytes can synthesize Tg, TPO, and TSH-R antibodies in vitro, and this suggested that they are an important source of thyroid autoantibodies. 17-21
Ectopic or extranodal secondary LFs are found in other autoimmune lesions, such as rheumatoid arthritis synovium, 22-24 myasthenia gravis thymus, 25 and Sjögren’s disease salivary glands, 26 but also in chronically infected tissues such as hepatitis C liver 27 and Helicobacter pylori gastritis mucosa. 28 The formation of extranodal LFs might be a normal development in the course of a maintained immune response but it is not known if it contributes to response effectiveness. In autoimmune diseases it has been interpreted as a sign of the intensity of the response but not as a significant step in their pathogenesis. The real frequency of lymphoid follicle formation in autoimmune tissue has been difficult to estimate, because these structures are irregularly distributed, scattered all over the affected tissue, and can be easily missed during routine histopathological examination.
Lymphoid follicles with GCs are crucial sites in the development of the anamnestic immune responses because they are the sites where cells undergo somatic hypermutation and affinity maturation. Newly formed GCs are oligoclonal B cell populations derived from one to three B cell clones. 29,30 Somatic hypermutation can generate autoreactive B cells and requires the existence of tolerance mechanisms to keep them under control. 31,32 The discovery of RAG and Tdt expression in GCs has suggested that secondary VDJ rearrangement may be another process that contributes to GC function. 33-35
The presence of well-organized B cell structures in AITD glands may be relevant to pathogenesis, not only for the production of autoantibodies but also for the development and maintenance of autoimmune response. B cells in intrathyroidal LFs are in a privileged location to capture large amounts of self-antigens and to present them to T lymphocytes. It has been suggested that, as they are outside the limits of lymphoid organs, they may bypass normal peripheral tolerance mechanisms more easily. 32 The importance of extranodal LF formation for the development of autoimmune disease was established in recent experiments by Ludewig and colleagues 36 using the RIP-GP mouse model; these authors reported a positive correlation between neogenesis of lymphoid tissue and development of autoimmune diabetes. The prevention of diabetes in NOD mice incorporating a Igμ null mutation 37 constitutes compelling evidence for the role of B cell in endocrine autoimmune disease.
Previous studies by ourselves, 38 and by other authors have described the surprisingly frequent occurrence and complex organization of intrathyroidal LFs. This prompted the present study, whose results indicate that intrathyroidal LFs in AITD patients are indeed functional. Our data include first evidence that chemokines capable of organizing and self-perpetuating LFs are generated in AITD glands and that secondary rearrangement of immunoglobulin genes may take place in these structures. In addition, we have found that intrathyroidal secondary LFs are more prevalent than previously estimated, correlate with autoantibody titer, and seem to result from the expansion of a few seeding B cells that are specific for thyroid autoantigens.
Materials and Methods
Patients
Thyroid tissue was obtained from 67 patients, 35 with GD, 8 with HT, 22 with multinodular goiter (MNG), and 2 previously healthy multiorgan cadaveric donors. MNG glands were considered nonautoimmune thyroid tissue. Clinical diagnosis was based on usual thyroid function tests, including free thyroxine, tri-iodothyronine, and TSH plasma levels. TPO and Tg antibodies were measured by enzyme-linked immunosorbent assay (Immunowell, San Diego, CA), normal ranges are 42 to 100 and 67 to 115 IU/ml, respectively. Antibodies to TSH-R were measured by radioimmunoassay (Brahms Diagnostica, Berlin, Germany) and all values >1.5 IU/L were considered positive. In the statistical analysis, negative samples were assigned the value to the lower limit of detection of the corresponding assay (ie, 24.0, 36.0, and 1.0 IU, respectively). Diagnoses were confirmed by histopathological examination of the glands. Tissue samples from one thymus, six palatine tonsils (PTs), and five LNs were used as reference lymphoid tissue.
Several blocks from most specimens were treated separately; some were formalin-fixed for standard histopathology tests and other were snap-frozen in isopentane and stored at −70°C. Series of sequential cryostat sections (4 μm) obtained from frozen blocks of thyroid, tonsil, thymus, and LN were used for immunofluorescence staining, terminal dUTP nick-end labeling techniques, and also for RNA extraction under RNase-free conditions.
Dispersed thyroid cells and intrathyroidal lymphocytes from 42 (26 GD, 3 HT, 13 MNG) glands were prepared by enzymatic digestion as described elsewhere. 39 The cells were filtered through a 500-μm mesh and cultured in RPMI 1640, 10% fetal calf serum, and antibiotics. Adherent cells, which included thyroid follicular cells, were separated from infiltrating thyroid lymphocytes by adherence to plastic after 18 to 24 hours of culture and separately cryopreserved in fetal calf serum containing 10% dimethyl sulfoxide. The lymphocyte-to-thyroid follicular cell ratio was assessed by fluorescence-activated cell sorting as described elsewhere. 39
Identification of Germinal Centers, Immunofluorescence Staining, and Image Analysis
Thyroid blocks, with an average weight of 5 g, were systematically screened for LFs by examining 1 out of 10 sequential cryostat sections stained by hematoxylin and eosin (H&E) and confirmed by either immunofluorescence staining with peanut agglutinin and anti-IgD (cryostat sections) 40 or with anti-CD20 (formalin-fixed sections). Ten to 80 consecutive 4-μm sections were stained for phenotypic markers, adhesion molecules, and related functional markers by simple or double-indirect immunofluorescence or by immunohistochemistry technique following published protocols 41 and using the antibodies listed in Table 1 ▶ . Epithelial cells were identified with high titer (1:105) anti-TPO patient’s serum. Affinity-purified fluorochrome-labeled conjugated antisera (specific for some IgG subclasses) were used as secondary antibodies (all from Southern Biotechnology, Birmingham, AL). In all cases, the controls included both using nonimmune sera or unrelated mAb as primary antibodies and testing the effect of omitting each of the layers. Biotin-labeled TPO and Tg (a kind gift from Pharmacia-Upjohn, Freiburg, Germany) and fluorescein isothiocyanate-streptavidin were used to detect B and plasma cells bearing Ig that were specific for these antigens. The controls included blocking staining with unlabeled antigens.
Table 1.
List of Lectins and Antibodies Used in the Characterization of Thyroidal GCs
| Molecule | Antibody | Source | Host | Isotype |
|---|---|---|---|---|
| Bcl-2 | Bcl-2 | Zymed | Mouse | IgG1 |
| CD19 | Leu-12 | B-D | Mouse | IgG1 |
| CD19 | A3B1 | HCPB | Mouse | IgG2a |
| CD154 | CD154 | De-Cheng Shen | Mouse | IgM |
| CD20 | 93-1B3 | HCPB | Mouse | IgG1 |
| CD20 | LE | Dako | Mouse | IgG |
| CD21L | 7D6 | P. Garrone | Mouse | IgG1 |
| CD23 | Leu-10 | B-D | Mouse | IgG1 |
| CD3 | UCHT1 | ATCC | Mouse | IgG1 |
| CD3 | Cris-7 | HCPB | Mouse | IgG |
| CD38 | RM3A5 | HCPB | Mouse | IgM |
| CD4 | RFT4 | RFHSM | Mouse | IgG1 |
| CD40 | HB14 | T.F. Tedder | Mouse | IgG1 |
| CD5 | 33-1C6 | HCPB | Mouse | IgG2a |
| CD5 | CD5 | Caltag | Mouse | IgG1 |
| CD50 | 101-1D2 | HCPB | Mouse | IgG1 |
| CD50 | 152-2D11 | HCPB | Mouse | IgG1 |
| CD54 | RM4A3 | HCPB | Mouse | IgG |
| CD71 | 120-2A3 | HCPB | Mouse | IgG1 |
| CD77 | 38.13 | IGR | Rat | IgM |
| CD79 | HM47 | HCPB | Mouse | IgG |
| CD8 | RFT8 | RFHSM | Mouse | IgG1 |
| CD83 | HB15 | T.F. Tedder | Mouse | IgG2 |
| CD95/Fas | DX2 | Pharmingen | Mouse | IgG1 |
| CD95L/FasL | NOK-1 | Pharmingen | Mouse | |
| CLA | HECA-452 | Pharmingen | Rabbit | IgG |
| Ki-67 | Ki-67 | Roche | Mouse | IgG1 |
| FVIII | FVIII | ATCC | Rabbit | |
| HLA Class I | W6/32 | Dakopatts | Mouse | IgG2a |
| HLA Class II | Edu-1 | HCPB | Mouse | IgG2b |
| hlgD | hlgD | Caltag | Goat | |
| hlgG | hlgG | Southern-Biotech | Mouse | |
| hlgM | A1B1 | HCPB | Mouse | IgG |
| hlgs | hlgs | Southern | Goat | |
| PNA | NO | Sigma | Peanut | NO |
The slides were examined blindly by two independent observers (MPA and RPB or MJO), using either UV or transmitted light microscopy (Axioplan II, Zeiss, Wetzlar, Germany). To better compare the distribution of different markers in some double-immunofluorescence experiments, photomicrographs of the red and green fluorescence images were digitized and then superimposed using commercial software (Photoshop; Adobe, San Jose, CA). Images from some preparations were acquired and deconvolved using a digital confocal microscopy system (Openlab; Improvision, Coventry, UK) to improve resolution but natural colors were maintained. The figure legend indicates whether the images have been processed. To compare better the LFs from thyroid autoimmune glands, LNs and tonsils, we measured the GC and mantle zone (MZ) areas, the perimeter, and the maximal and minimal diameters using the Openlab morphometry module.
TdT-Mediated dUTP Nick-End Labeling Assay
Apoptosis was detected by using a commercial kit (In situ cell death detection; Boehringer Mannheim, Mannheim, Germany) following the manufacturer’s instructions. Two substrates were used and the protocol was adjusted accordingly; 1) sections from formalin-fixed paraffin-embedded blocks (4 μm) were dewaxed, rehydrated, and digested with proteinase K (20 μg/ml), and 2) cryostat sections (4 μm) were fixed in 4% paraformaldehyde and permeabilized with 0.1% Triton X-100/0.1% sodium citrate solution. dNTP-fluorescein isothiocyanate was detected with an anti-fluorescein alkaline phosphatase-conjugated antiserum and Fast red. The sections were counterstained with Mayer’s hematoxylin for 30 seconds. Apoptotic nuclei were counted using the Openlab software and expressed as number of positive nuclei per mm2.
Microdissection, RNA Extraction, and Reverse Transcriptase-Polymerase Chain Reaction (RT-PCR) Amplification
To study the RNA from the LFs, these structures were identified by H&E staining and microdissected from 4 to 10 consecutive thyroidal sections under a stereomicroscope (SV8, Zeiss). RNA was extracted from sections devoid of LFs and from control normal lymphoid organs. To study chemokine expression, RNA was extracted from frozen tissue blocks. Genomic DNA was removed by digestion with DNase-I and the RNA was reprecipitated with 0.4 μmol/L of nuclease-free glycogen (Boehringer-Mannheim) as a carrier; then, it was quantified in a spectrophotometer at OD260 nm. cDNA was prepared in a final volume of 10 μl by mixing 200 ng to 1 μg of total RNA, 20 pmol Oligo-(dT15) (Pharmacia-Biotech), 40 U/μl RNase inhibitor (Clontech), 10 mmol/L dNTP mix (Pharmacia-Biotech), 0.1 mol/L dithiothrcitol (DTT), 1× SuperScript buffer and 200 U/μl of SuperScript-II enzyme. For RAG2, the antisense primer used for the retrotranscription and for PCR amplification was the same. The reaction was performed at 42°C for 45 minutes and the product was diluted 5 to 10 times in TE (10mM TRIS/1mM EDTA, (TE) pH 8) for subsequent experiments. For PCR reactions 1 to 3 μl of cDNA was added to the mixture containing 0.2 mmol/L dNTP, 0.3 μmol/L each primer, 1× PCR buffer and 300 mU of DynaZyme II (Finnzymes, Oy, Finland) to a final volume of 20 μl in hot start. The following programs and primers were used: 1) GAPDH, 30 cycles at 94°C for 30 seconds; denaturation, 65°C for 45 seconds; annealing/elongation (sense primer 5′-TCTTCTTTTGC GTCGCCAG-3′, antisense primer 5′-AGCCCCAGCCTTCTCCA-3′), amplicon 371 bp. 2) RAG1 35 cycles at 94°C for 30 seconds; denaturation, 56°C for 30 seconds; annealing at 72°C for 40 seconds; elongation (sense primer 5′-ACTTTCCCTTCATCCTGCTTA-3′, antisense primer 5′-TTTTTCTCCTCCTCTTGCTTC-3′), amplicon 653 bp. 3) RAG2, 30 cycles of denaturation at 94°C for 30 seconds; annealing at 63°C for 30 seconds; extension at 72°C for 40 seconds (sense primer 5′-GCCACAGTCATAGTGGGCAGTCA G-3′, antisense primer 5′-CAAAGGGAGTGGAATCCCCTGG-3′), amplimer 534 bp. 4) SDF-1, 30 cycles of denaturation at 94°C for 30 seconds; annealing at 60°C for 20 seconds; and extension at 72°C for 30 seconds (sense primer 5′-GTCGTGGTCGTGCTGGTC-3′, antisense primer 5′-CGGGCTACAATCTGCAGG-3′), amplicon 155 bp. 5) SLC, 35 cycles at 94°C for 30 seconds; denaturation, 65°C for 45 seconds; annealing/elongation (sense primer 5′-AAGGCAGTGATGGAGGGG-3′, antisense primer 5′-CTGGGCTGGT TTCTGTGG-3′), amplicon 238 bp. 6) BLC, 35 cycles at 94°C for 30 seconds; denaturation, 51°C for 20 seconds; annealing at 72°C for 30 seconds; elongation (sense primer 5′-CGACATCTCTGCTTCTC-3′, antisense primer 5′-ACTTCCATCATTCTTTG-3′), amplicon 255 bp. All amplifications were followed by a final extension at 72°C for 7 minutes. The PCR products were analyzed by 1 to 3.5% agarose gel electrophoresis and transferred to nylon membranes (Hybond+, Amersham). The membranes were hybridized with specific labeled probes [γ-32P]ATP 10μCi/μl) at high stringency conditions (RAG1 5′-CCCTTACTGTTGAGACTGC-3′; RAG2 5′-GGACAAAA AGGCTGGCCCAA-3′; SDF-1 5′-TGCCTCAGCGACGGG-3; SLC 5′-CTTGGTT CCTGCTTCCG-3′; BLC 5′-ACAACCATTCCCACGG-3′). In the case of RAG1, both primers annealed to the same exon (exon 2) so a sample of unretrotranscribed RNA was introduced as an additional control. To estimate the amount of chemokine message in tissue samples, hybridized membranes were exposed and the autoradiographies were either counted on a Phosphorimager (BioRad, Richmond, CA) using Quantity One software or assessed by densitometry (TDI Systems). The results were normalized according to the amount of GAPDH message as estimated by densitometry of gels stained with ethidium bromide. Preliminary experiments were performed to establish the conditions under which the reactions were within the exponential phase of amplification. To better compare the levels of the different chemokines and refer them to a physiological substrate, the values obtained from thyroid samples were divided by the average value from PT for each chemokine. Therefore, the results are given as percentages of reference PT values.
Statistical Analysis
Data distribution was first analyzed using the Sigma Stat software (Microsoft Corp., Seattle, WA). Parametric (Student’s _t_-test) and nonparametric (Mann-Whitney) tests were applied to normal and nonnormally distributed data, as indicated.
Results
Most AITD Glands Contain Typical Secondary Lymphoid Follicles that Are Large and Tonsil-Like in HT and Smaller and Lymph Node-Like in GD
Typical secondary LFs, similar to those found in lymphoid organs were easily detected by screening H&E sections. Eight of 8 (100%) glands from HT, 14 of 26 (53.8%) from GD, and 1 of 22 (4.5%) from MNG patients contained LFs (Table 2) ▶ .
Table 2.
Classification of Thyroid Glands According the AITD, Autoantibody Titers, Percent of Leukocyte Infiltration and Presence of Germinal Centers
| Sample | AITD | αTq Ul/ml | αTPO Ul/ml | αTSHR Ul/l | % LI | S (N of GC) |
|---|---|---|---|---|---|---|
| 228 | GD | -ve | -ve | 80.0 | ND | 1 (11)/2 (1) |
| 255 | GD | -ve | 25 | 20 | ND | 1 (1) |
| 257 | GD | 598.0 | 1155.0 | 67.0 | 70.2 | 1 (3) |
| 258 | GD | -ve | -ve | 118.0 | ND | 1 (1) |
| 278 | GD | -ve | -ve | 16.0 | ND | 1 (1) |
| 373 | GD | 172.1 | 39.2 | 178.0 | 27.7 | 1 (1) |
| 378 | GD | 177.3 | 152.3 | 1.0 | 30.0 | 1 (1) |
| 381 | GD | 162.8 | 48.1 | ND | 19.5 | NO |
| 389 | GD | 66.8 | 143.6 | 37.5 | 9.6 | NO |
| 390 | GD | 28.3 | -ve | 8.4 | 7.03 | NO |
| 391 | GD | ND | 25 | ND | 4.9 | NO |
| 393 | GD | 35 | 42.0 | 62.0 | 43.4 | 1 (1) |
| 394 | GD | -ve | 43.5 | 38.0 | 4.8 | NO |
| 403 | GD | 36 | 550.0 | -ve | 38.8 | 2 (3) |
| 412 | GD | 239 | 124.3 | 8.8 | 20.0 | 1 (4) |
| 413 | GD | 37.2 | 39.4 | 20.6 | 5.2 | NO |
| 416 | GD | 56.4 | 40.5 | -ve | 9.2 | NO |
| 417 | GD | -ve | 25 | 75.0 | 9.5 | NO |
| 423 | GD | 70 | 53 | 15.5 | 10.2 | NO |
| 424 | GD | -ve | -ve | 4.1 | 54.5 | NO |
| 425 | GD | -ve | 28 | 4.0 | 11.4 | NO |
| 426 | GD | 1265 | 1001 | 85.9 | 45.4 | 1 (2)? |
| 427 | GD | 190 | 1001 | 14.9 | ND | 1 (1) |
| 429 | GD | -ve | 7000 | -ve | 50 | 3 (2) |
| 430 | GD | -ve | 46 | 10.1 | 10.6 | 1 (1) |
| 290 | HT | 584.0 | 766.0 | -ve | ND | 2 (1) |
| 384 | HT | ND | ND | ND | 66.6 | 1 (5) |
| 385 | HT | 774.0 | 105000 | -ve | 76.6 | 1 (1) |
| BL | HT | ND | ND | ND | ND | 1 (10) |
| JC | HT | ND | ND | ND | ND | 1 (5) |
| RP | HT | ND | ND | ND | ND | 1 (10) |
| AP1 | HT | ND | ND | ND | ND | 1 (4) |
| AP2 | HT | ND | ND | ND | ND | 1 (11) |
| 359 | MNG | -ve | -ve | -ve | 5.2 | NO |
| 360 | MNG | -ve | -ve | 2.5 | 1.3 | NO |
| 361 | MNG | -ve | -ve | 7 | 0.3 | NO |
| 362 | MNG | -ve | -ve | 1.6 | 6.1 | NO |
| 364* | MNG | -ve | -ve | 1.2 | 12.0 | 1 (1) |
| 376 | MNG | -ve | 37.9 | 1.4 | 3.5 | NO |
| 379 | MNG | -ve | -ve | 1.2 | 6.8 | NO |
| 395 | MNG | -ve | -ve | -ve | 7.2 | NO |
| 396 | MNG | -ve | 36 | -ve | 3.9 | NO |
| 399 | MNG | -ve | 45.9 | 1.2 | 21.7 | NO |
| 409 | MNG | 27.5 | 41 | -ve | 20.6 | NO |
| 419 | MNG | 36 | 23 | -ve | 11.7 | NO |
| 420 | MNG | -ve | -ve | -ve | 7.5 | NO |
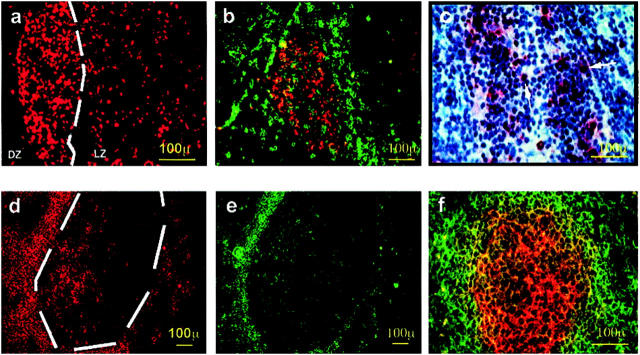
Intrathyroidal secondary LFs possessed a MZ and well-formed GCs with obvious signs of activity (eg, lymphoblasts in mitosis). The GCs appeared in areas of heavy infiltration, but relatively isolated GCs were also found, and often they were so close to the thyroid follicles that the mantle lymphocytes were adjacent to the thyroid epithelium. Nests of epithelial cells often remained in the middle of dense infiltrates, apparently unaffected by the surrounding lymphocytes. The cells forming the intrathyroidal GCs were polarized in a dark zone containing lymphoblasts with big nuclei and two or three nucleoli, and a light zone surrounded by small lymphocytes with the features of centrocytes. Large dendritic cells with elongated nuclei were present in the GC and the MZ, whereas plasma cells were scattered all over the infiltrate (Figure 1a) ▶ . This cellular distribution was confirmed by staining for a series of phenotypic markers. Among them, anti-CD20 (Figure 1b) ▶ and peanut agglutinin-fluorescein isothiocyanate (Figure 1c) ▶ specifically labeled the GCs. Peanut agglutinin stained both the GCs and the vascular endothelium, a feature that was useful to trace a given GC in consecutive sections. The MZ consisted of typical CD19low IgD+ IgM+ CD38− CD23−/low+ follicular B lymphocytes (data not shown), but some areas were rich in CD3+ cells, predominantly of the CD5+ CD3+ CD4+ phenotype but also containing CD3+ CD8+ cells; these areas corresponded to the T-cell rich MZ areas observed in lymphoid node LFs (Figure 1 ▶ ; d, e, and f). Staining for CD83 (Figure 1g) ▶ revealed a rich network of DCs that included extensive areas of the MZ and of the surrounding infiltrate, but normally did not reach the inner GC area. Immunofluorescence staining confirmed the presence within the GC of the two characteristic B cell populations: IgM−/low IgD− CD23− CD77+ CD38+ centroblasts and CD77+ CD38+ CD23+ centrocytes. A framework of follicular dendritic cells, polarized toward the light zone, was identified by mAb 7D6 against the long form of CD21(CD21L) (Figure 1h) ▶ .
Figure 1.
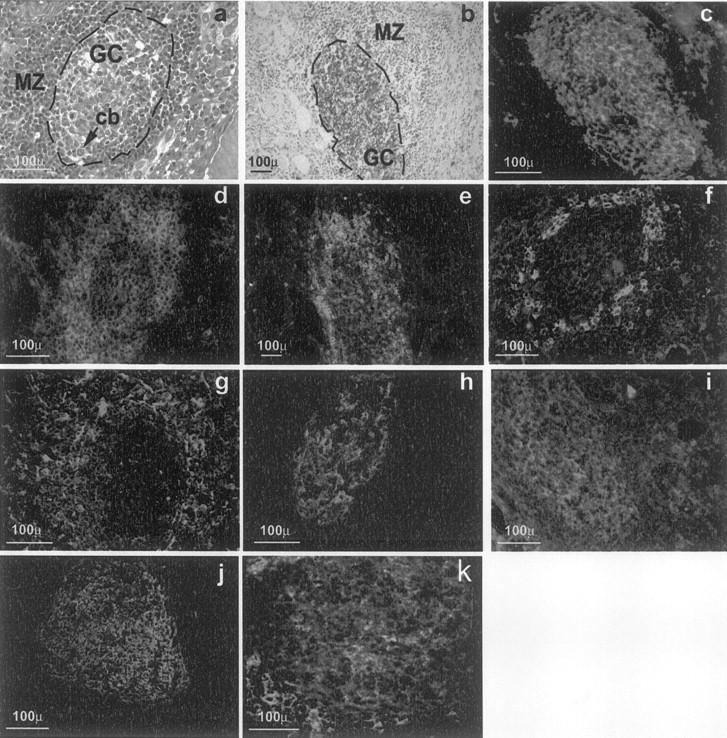
Morphology of intrathyroidal secondary LFs in AITD. a: A section from a formalin-fixed paraffin-embedded block from a HT gland. b to h: Sections from frozen blocks from GD glands. g was deconvolved to improve image definition (see text for details), the other images are standard micrographs. a: H&E staining showing a typical secondary follicle with a GC and a well-formed MZ (cb, centroblasts undergoing mitosis). b: CD20 on B cells of the GC (brown) using the immunoperoxidase technique and counterstained with hematoxylin (GD, case TB228). c: Direct immunofluorescence with peanut agglutinin-fluorescein isothiocyanate, showing positive staining of centroblasts and follicular dendritic cells (GD, case TB378). d: Staining for CD3+ reveals abundant T cells in the MZ with scattered cells inside the GC (GD, case TB228). e: Demonstration of abundant CD4+ T among the T cells in the MZ (GD, case TB228). f: Staining for CD8 shows moderately abundant CD8+ T lymphocytes in the MZ (GD, case TB378). g: Staining for CD 83 highlights the network of mature dendritic cells in the MZ (GD, case TB278). h: Staining for the long form of CD21 reveals the network of follicular dendritic cells in the CG and their polarization toward the light zone (GD, case TB373). i: CD38, as marker of centrocytes. j: CD23 staining revealing the area occupied by the GC. k: CD77 staining as an additional GC marker .
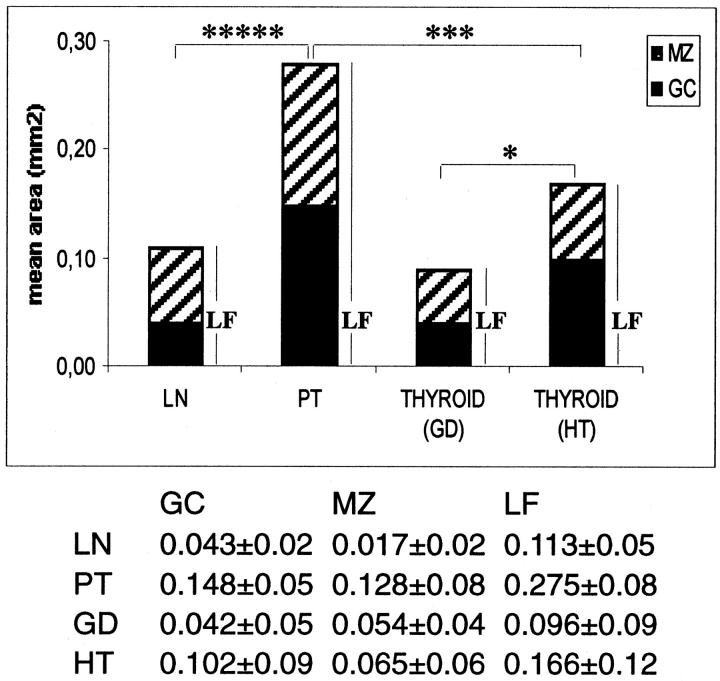
LFs from different glands were found to differ in size and complexity, and a correlation with diagnosis was quickly discovered. LFs in HT glands were large and similar to LFs in tonsils, whereas LFs in GD glands were smaller and similar to LN LFs. The measurement of 122 LFs from a random selection of samples (26 in tonsil, 39 in LN, 20 in GD, 37 in HT samples) confirmed this fact (Figure 2) ▶ . GC and LF areas in GD and HT glands were significantly different (P = 0.0020 and P = 0.0174, respectively, _t_-test). As expected, the presence of LFs was associated by the presence of extensive lymphoid infiltration (P < 0.000005, Mann-Whitney). LFs were detected in heavily infiltrated glands (defined as containing >25% of lymphocytes over total amount of dispersed cells, as assessed by flow cytometry of 24-hour thyroid primary cultures (data not shown), but this relationship was not symmetrical; some glands with extensive infiltration lacked LFs.
Figure 2.
Morphometric analysis of LFs from HT and GD compared with LFs from LNs and PTs. Solid bar, area occupied by the GC; hatched bar, area occupied by the MZ. Number of LFs studied: LNs, 39; PTs, 26; GDs, 20; and HTs, 37. The asterisks indicate significant differences between total LF areas: ****, P < 0.001; ***, P < 0.00.5; *, P < 0.05. The table below gives numerical values ± SD.
AITD Intrathyroidal Lymphoid Follicles Are Committed to TPO and Tg Antibody Production
Both in the whole study group and in the AITD group, the presence of intrathyroidal LFs was associated to significantly higher levels of TPO (P = 0.00860, Mann-Whitney test) but not of Tg autoantibodies, although a tendency toward association was observed (P = 0.053). In the GD group, intrathyroidal LFs were associated to higher levels of TSH-R antibodies (P = 0.0146, Mann-Whitney test). The specificity of the B cells in intrathyroidal LFs was demonstrated by the binding of Tg and TPO to LFs in a HT gland (16 of 28 LFs) and in four GD glands (17 of 27; Figure 3b ▶ ). Two types of cells bound thyroid antigens: numerous small cells with a distribution similar to that of GC B cells and strongly positive cells with a strong cytoplasmic staining scattered all over the LFs and the diffuse lymphoid infiltrates. Double immunofluorescence revealed that most cells that bound to TPO or Tg were IgG+. The distribution of Tg+ or TPO+ cells was markedly uneven among the LFs, which tended to either be totally negative or contain numerous positive cells. This suggested that, as in canonical LFs, intrathyroidal CGs consist of an oligoclonal B cell population. Blocking experiments with unlabeled TPO and Tg gave negative results, thus confirming the specificity of the reaction.
Figure 3.
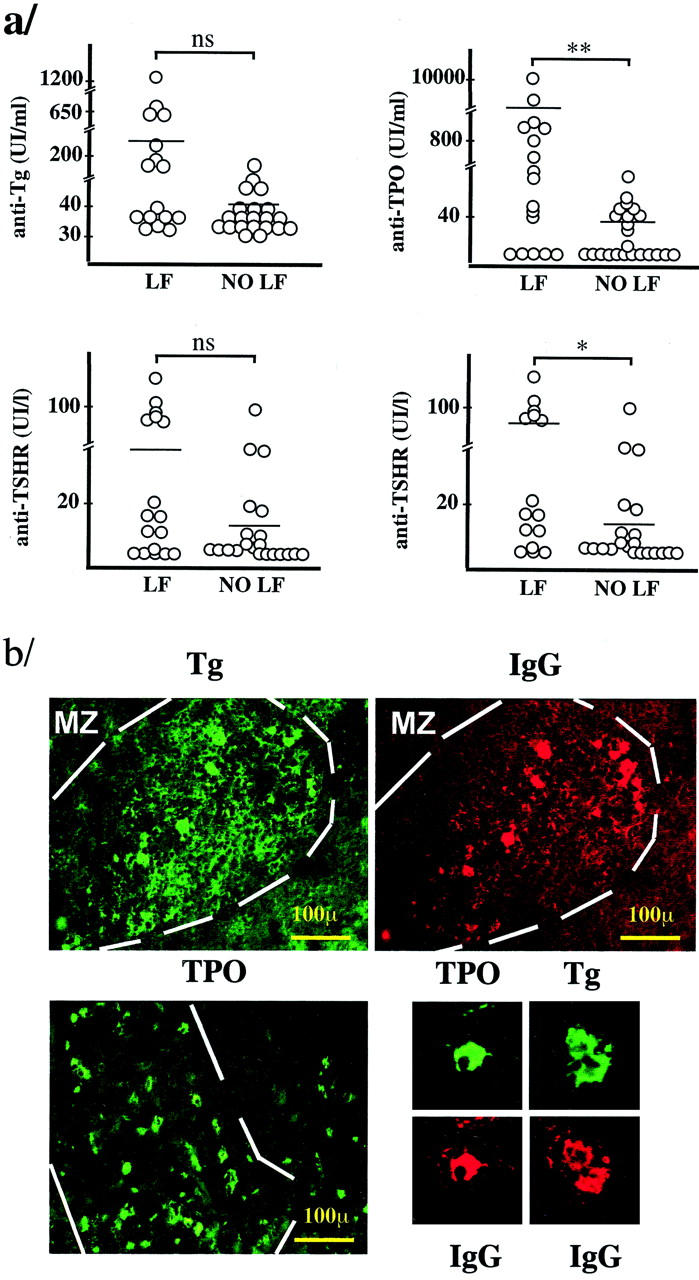
a: Relationship between thyroid antibody titer and the presence of intrathyroidal LFs. TSHR, thyrotropin receptor; LF, group of thyroid glands containing LFs; NO LF, group of thyroid glands without LFs. The plotted data, except for those at bottom right, correspond to all of the patients in Table 2 ▶ . At the bottom right, only the data from GD patients were plotted (*, P < 0.05; **, P < 0.01; ns, not significant P value, Mann-Whitney test). b: Demonstration of the specificity of B and plasma cells in the intrathyroidal LFs. Double immunofluorescence using biotinylated Tg (green, top left) and anti-IgG (red, top right), demonstrating binding of Tg to the same cells that are stained for IgG. Positive lymphocytes only appear in the LFs, whereas plasma cells are present both in the LFs and in the diffuse infiltrate. Bottom left: Binding of biotinylated TPO to abundant cells in the LFs and also to some cells in the diffuse infiltrate. Note the presence of membrane and cytoplasmic staining that correspond to lymphocytes and plasma cells, respectively. Bottom right: Examples of plasma cells stained for TPO and Tg in double immunofluorescence with IgG (GD, case 378).
AITD Lymphoid Follicles Express the Molecules Required for Secondary Lymphoid Follicle-Specific Processes, Including Recombinase-Associated Gene Products RAG1 and RAG2
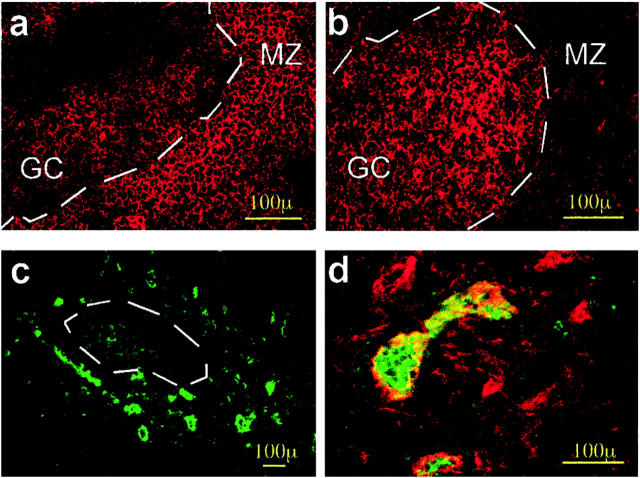
Proliferation in the LFs is linked to somatic hypermutation and Ig repertoire diversification. AITD GCs contained many cells positive for the proliferation marker Ki67, which were oriented toward the dark zone; this was also the case in control tonsil and LN tissue samples (Figure 4a) ▶ . Two-color immunofluorescence with anti-IgM antibodies confirmed that most Ki67-positive cells were lymphoblasts with slight or no IgM staining. Cell proliferation is high in AITD GCs, especially in HT glands; the level of proliferation is similar to that detected in tonsil GCs and well above that of LNs (Figure 4b) ▶ . Collaboration between B and T lymphocytes through CD40-CD40L interaction is central to GC functions. 42 As in lymphoid LFs, 43 intrathyroidal LFs contained in their GCs a small polarized population of CD40+ B cells and some scattered B lymphocytes bearing the IgD+CD38−CD40L+ phenotype in the T-cell-rich area. CD40 was expressed by a population of large-sized dendritic cells located outside the LFs, in the areas of diffuse lymphocytic infiltration where T lymphocytes predominate. These cells probably correspond to the dendritic cells stained by CD83 in other sections and are equivalent to the dendritic interdigitating cells described in the paracortical area of LNs (data not shown).
Figure 4.
Proliferation and apoptosis in thyroid LFs. a: Immunofluorescence staining for Ki67 in an intrathyroidal LF, showing abundant proliferating cells polarized toward the dark zone (GD, case TB228). b: Double-immunofluorescence staining for IgD (green) and Ki67 (red), demonstrating that most proliferating cells are in the light zone of the GC and are either negative or slightly positive for IgD, as in the case of centroblasts. c: Demonstration of the existence of apoptotic cells by the terminal dUTP nick-end labeling technique and hematoxylin counterstain. Most positive cells were found in the GC area; no positive cells were seen in the epithelium of the thyroid follicles (data not shown). d and e: Double immunofluorescence for bcl-2 and IgD. The level of expression of apoptosis inhibitor bcl-2 is high in the MZ but is also expressed in the GC cells. The staining for IgD shows the distribution of mature B cells in the MZ. f: Double exposure of double-immunofluorescence staining for IgD (green) and CD95/Fas (red). Notice that Fas is mainly expressed in the CG, whereas IgD is expressed in the MZ, as is the case in normal LNs.
Apoptosis, which is an intrinsic feature of LN GCs, was detected by the terminal dUTP nick-end labeling technique in intrathyroidal LFs, especially in those from HT glands (Figure 4c) ▶ . Positive nuclei were located mainly in the light zone, but some labeled nuclei were detected in the rest of the GC areas as well as outside them, but not in the thyroid follicular epithelium, as reported. 44 The number of apoptotic cells per square mm in LFs from HT was as high as in the tonsils (370.2 ± 186 versus 325 ± 99.4; P = not significant) and significantly higher than in GD glands (126.4 ± 57.6) (HT versus GD; P < 10−4, _t_-test). B-lymphocyte apoptosis in lymphoid organ LFs is triggered by CD95-CD95L (Fas/FasL) interaction and is modulated by the expression of Bcl-2 and other anti-apoptotic factors. As expected, centroblasts were Fas+ Bcl2− (Figure 4, d and f) ▶ whereas most centrocytes were Fas− Bcl-2+. The IgD+ naïve B cells in the MZ were also bcl-2-positive. In general, the distribution of these two molecules was similar to that observed in tonsils and LNs, but in intrathyroidal LFs the level of Bcl-2 seemed to be higher than in lymphoid organ LFs.
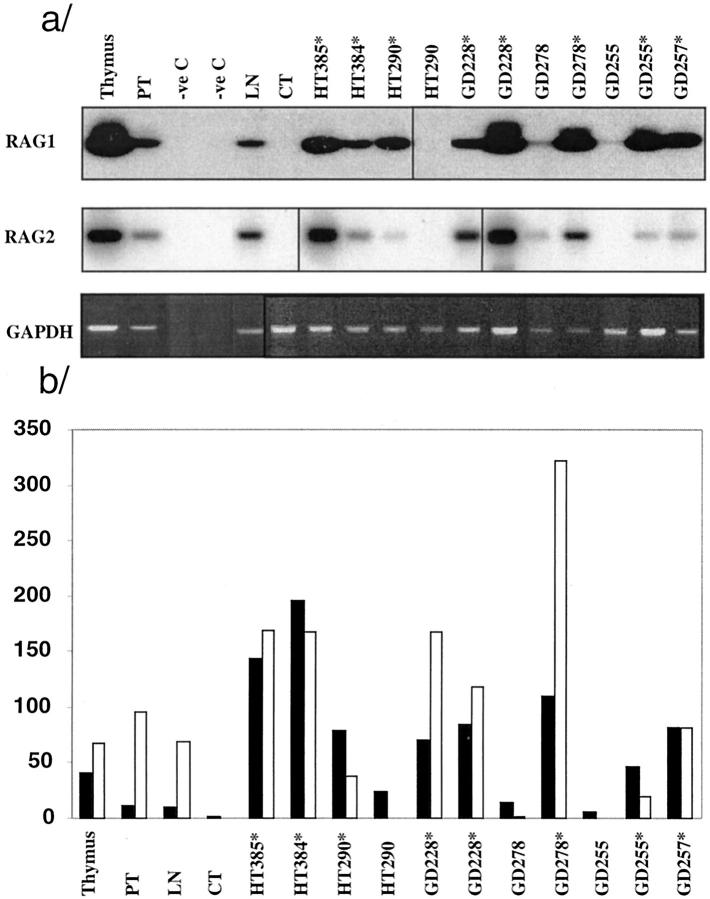
It has been reported that the RAG1 and RAG2 recombinase genes are expressed in secondary lymphoid organs during active immune response (see Discussion). We assessed the expression of RAG1 and RAG2 in AITD glands by RT-PCR and Southern blotting using thymus, tonsil, and LN as positive controls. RNA was extracted from 4 to 10 consecutive cryostat sections and, when feasible, intrathyroidal GCs were microdissected. All sections containing GCs were positive for RAG1 and RAG2 (HT, n = 3; GD, n = 4) irrespective of diagnosis, whereas sections that did not contain GCs were negative (HT, n = 1) (Figure 5) ▶ . Based on partially normalized densitometric data, the levels of RAG1 and RAG2 mRNA levels in most glands containing LFs were several times higher than in tonsil and LN samples, and in some cases higher than in samples from the thymus. Tissue samples obtained from the same glands but without LFs showed lower RAG1 and RAG2 levels. There were no clear differences between HT and GD glands.
Figure 5.
RAG1 and RAG2 mRNA expression measured by RT-PCR Southern blot in HT and GD glands. a: Southern blot of RT-PCR products amplified with RAG1 and RAG2 primers and hybridized with the corresponding oligoprobes; bottom, RT-PCR for control GAPDH; only partial normalization was achieved because of the small amount of available sample. Asterisks indicate samples that contained visible GCs. TMB, thymus; CT, esophagus. b: Graph representing the ratio of densitometry values: RAG1, GAPDH (solid bars); RAG2, GAPHDH (open bars), using an inverted image of the GAPDH gel image.
AITD Lymphoid Follicles Express the Adhesion Molecule and Chemokines Required for Self-Perpetuation as Homing Area
These two categories of molecules determine the development, organization, and self-perpetuation of LFs in the lymphoid organs. The distribution of ICAM-3 (CD50) and ICAM-1 (CD54) was similar to that observed in LN LFs, 45 ie, ICAM-3 was expressed mainly in the MZ and the surrounding area and in a few cells of the GC (both centroblasts and centrocytes), whereas ICAM1 was expressed by GC cells and especially in the follicular dendritic cell-rich area (Figure 6) ▶ . The distribution of CLA (cutaneous lymphocyte-associated antigen), a carbohydrate domain that is the ligand for l-selectin (CD62L) and a marker for high endothelial venules (HEVs), 46 was studied in parallel with that of factor VIII, an endothelial marker. 38 In the rich capillary network that surrounds the intrathyroidal LFs revealed by FVIII staining, ∼20% of the endothelial cells (Figure 6, c and d) ▶ were positive for CLA (Figure 6e) ▶ . Besides, occasional CD3+ CLA+ were detected in the areas of lymphoid infiltration. The endothelium of MNG and GD thyroids lacking LFs was either completely negative or only showed a very low expression of CLA.
Figure 6.
Distribution of adhesion molecules in the thyroidal LFs. a: ICAM-1 membrane staining is mainly present in the MZ cells. b: ICAM3 expression is higher in cells inside the GC and has a reticular pattern similar to that observed for the follicular dendritic cells. c: CD62L-positive HEVs are distributed around the LF. d: Picture resulting from superimposing digitized images for CLA (green) and Factor VIII (red) staining in the same section. Note that the large vessels are positive for both markers, thus indicating the location of the HEV. Cryostat sections from GD, case TB278.
The expression of chemokines known to be important for the formation and maintenance of GCs [ie, stromal cell-derived factor 1 (SDF1 or CXCL12), secondary lymphoid tissue chemokine (SLC or CCL21), and B lymphocyte chemoattractant/B-cell-attracting chemokine 1 (BLC/BCA-1 or CXCL13)], was assessed by semiquantitative RT-PCR. A small selection of glands containing LFs was selected, RNA samples were prepared from total tissue and retrotranscribed as described above. Figure 7a ▶ shows the results from one representative amplification experiment. mRNA from the three chemokines was detected in the positive control and their level of expression was clearly high in the two HT samples. Because the cDNA samples had been normalized by GADPH expression, the observed fourfold increase of SLC and SDF1 levels over PT levels indicates a remarkably active synthesis of chemokine message in the HT glands. BLC message was also higher, but only when compared to normal or MNG thyroid tissue. In the GD gland studied, chemokine expression was not different from that in normal or MNG tissue (Figure 7b) ▶ .
Figure 7.
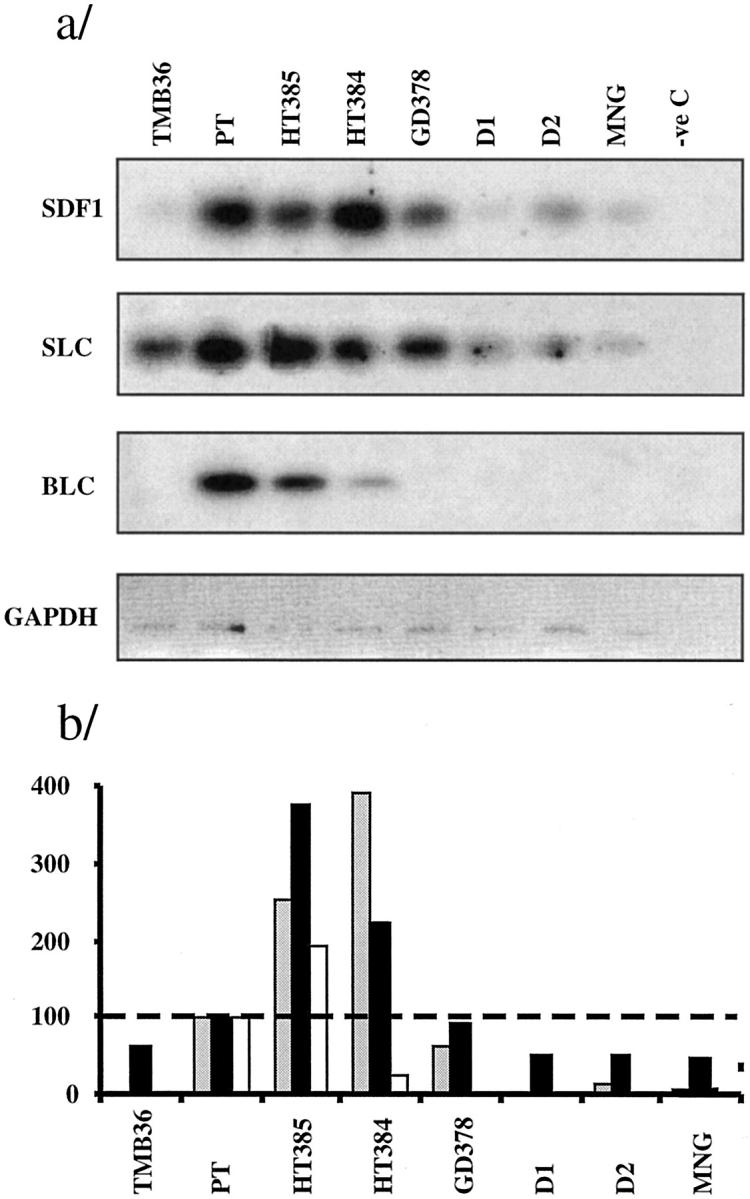
Chemokine expression as assessed by RT-PCR-Southern blot and phosphorimager counting. a: RT-PCR-Southern blots. b: Ratio for normalized chemokine value in each gland: normalized tonsil value, ×100; the broken line at 100% represents the tonsil reference value. Gray bars, SDF1 (CXCL12); solid bars, SLC (CCL21); open bars, BLC (CXCL13). TMB, thymus; D1 and D2, normal donors.
Discussion
Secondary LFs have been studied so intensively in the last decade that it is not feasible to reproduce here even a small fraction of these studies on intrathyroidal LFs. However, by using a series of phenotypic markers we have obtained a fairly complete picture of their architecture, which we have shown to be similar in every respect to that of classical secondary LFs. The analysis of the main processes that take place in GCs through the careful assessment of the expression of molecules that act as indicators of proliferation, apoptosis, and secondary Ig gene rearrangement confirmed the similarity between typical LFs and intrathyroidal LFs. More importantly, Tg and TPO binding to LFs indicates that most intrathyroidal LFs are committed to the response to thyroid self-antigens. Finally, the finding of well-formed HEVs and a coherent pattern of adhesion molecules and chemokine expression support the notion that intrathyroidal LFs have the capability to organize and perpetuate themselves.
Histopathological and immunohistochemistry methods have been applied and, when appropriate, observations were confirmed by confocal digital microscopy (deconvolution-based) and computerized morphometry. The antigen specificity of B and plasma cells was studied using a classical approach: binding of labeled antigen and visualization by immunofluorescence. 47 For some molecules, such as chemokines and RAG1 and RAG2 gene products, we used RT-PCR-Southern blot, but in these cases the substrates were normalized and the LFs were selected by microdissection. Despite many technical difficulties and limited sample availability, human material was used as substrate to ensure the relevance of the findings to human disease. In thyroid patients, late surgery and prolonged treatment with thionamides that are immunomodulators, 48 may be a limitation. However, our results cannot be attributed to this treatment, because intrathyroidal LFs were described well before thionamides were available and HT patients had not been treated with thionamides.
The presence of intrathyroidal LFs in AITD was considered to be a very characteristic feature of HT but not of GD. By their nature, LFs are scattered over the thyroid gland and can be easily missed, especially in GD, where infiltration is much less intense. By using a screening protocol that takes into account the average size of LFs and checking 1 in 10 sections, the possibility of missing LFs was reduced to the minimum. However, because we did not screen >5 g of each gland, our data could still be an underestimate. It is plausible that, if the entire glands had been screened, the frequency of LFs in GD would have been higher, thus implying that LFs are a regular feature. The association between high titer of antibodies against TPO and TSH-R and the presence of intrathyroidal LFs in AITD glands suggests that these structures play a role in antibody production. One possibility is that the formation of intrathyroidal LFs determines transition from self-limited responses to thyroid antigens to AITD, as suggested by the experimental model of Ludewig and colleagues. 36 Our results showed binding of Tg and TPO to B and plasma cells, and their distribution among LFs suggests that most intrathyroidal LFs are actually committed to thyroid antigen immune response. The elucidation of the relative contribution of intrathyroidal LFs to the circulating levels of thyroid autoantibodies requires experiments that are difficult to justify in human patients, but the clinical observation of a quick reduction in autoantibody levels after thyroidectomy in GD patients (A Belfiore, personal communication), 49 suggests that this contribution is important. If the autoimmune response was driven from the regional LNs, the persistence of antigen trapped in their follicular dendritic cell 50 would maintain the response for a much longer period of time.
The similarity of HT and GD LFs to PTs and LN LFs, respectively, similarities that also apply to the levels of proliferation and apoptosis, may indicate a difference in the pathogenesis of each of these two forms of AITD. PTs and intrathyroidal HT LFs show a high degree of activation. It has been reported that PT LFs are activated by lipopolysaccharide and other bacterial products present in the pharynx. 51 Current evidence and our data suggest that intrathyroidal LFs are stimulated by high concentrations of thyroid proteins, thus explaining their state of activation. It is relevant that B lymphomas that arise from HT glands have features similar to those of the MALT lymphomas, 52 which are caused by monoclonal expansion of B cells chronically stimulated by antigens of the gastrointestinal tract. Similarly, B lymphomas in HT patients, which are histopathologically similar to the MALT lymphomas, could result from chronic stimulation of GC lymphocytes by thyroid autoantigens. 53 The intrathyroidal LFs from GD patients are less active, and this may be because of a lower concentration of TSH-R, the most important self-antigen in this entity. Besides, as TSH-R is also expressed outside the thyroid, 3 the response to it may be less dependent on intrathyroidal stimulation. In fact, it has been proposed that GD is a systemic disease. 54
The complete parallelism between intrathyroidal LFs and secondary LFs in peripheral lymphoid organs suggests similar functional capabilities, probably including those not specifically studied in this work, such as Ig class switch. RAG1 and RAG2 expression, the visualization of HEV and the detection of the main chemokines that determine the organization of LFs have not been reported before and need to be discussed.
The RAG1 and RAG2 recombination activation genes play a central role in the rearrangement of V(D)J gene segments during lymphocyte development in the primary lymphoid organs. 55 The finding of RAG1 and RAG2 expression in splenic B cells stimulated in vitro with LPS+IL-4 and in GC B cells of immunized mice suggested that at least some peripheral B cells can be induced to re-express or up-regulate these genes. 33,34,56-58 This RAG re-expression was initially interpreted as evidence for the occurrence of peripheral receptor editing (receptor revision) aimed at rescuing autoreactive B cells generated by somatic hypermutation. The demonstration that Ig cross-linking inhibited RAG expression in human tonsil B cells led to the suggestion that receptor revision was rather aimed at rescuing cells whose receptor lost affinity during somatic hypermutation. 59 Contrary to the former data, more recent experiments using GFP as a reporter of RAG2 induction have suggested that RAG expression in GCs may be because of the presence of a small percentage of immature RAG-expressing B cells in the spleen and LNs. 60 In any case, these immature B cells probably contribute to the immune response and RAG expression should now be considered a feature of active GCs. 61 RAG expression in intrathyroidal GCs was remarkably high, in some cases well above the levels detected in thymus, and probably indicates the high activity of these GCs. Our present data do not allow us to establish which of the following two not mutually exclusive alternatives explains the high level of expression of RAG: 1) a very active receptor revision and 2) the presence of a high proportion of immature B cells or, in general, of recent bone marrow emigrants. We believe that the phenotype of B cells in intrathyroidal LFs and the cloning of B cells/plasma cells bearing high-affinity antibodies to Tg and TPO from these cells favor the first alternative. Plausibly, B lymphocytes recruited from blood into intrathyroidal LFs have low-affinity receptors 62 and may require extensive receptor revision to generate high-affinity B cells. Interestingly, evidence for over-active receptor revision has been found in human systemic lupus erythematosus (SLE) 63 and, more recently, in ectopic LFs from rheumatoid arthritis synovium. 64
Selective lymphocyte migration into peripheral lymphoid organs is regulated by adhesion molecules and chemokines. HEVs are specialized structures that allow rapid and selective lymphocyte trafficking from the blood into the secondary lymphoid tissues. Cutaneous lymphocyte-associated antigen (CLA), a HEV addressin, was detected in intrathyroidal small vessels that are morphologically similar to LN HEV. Secondary lymphoid tissue chemokine (SLC), a HEV-expressed chemokine that activates lymphocyte integrins, was also detected in intrathyroidal LFs. The distribution of HEVs around the intrathyroidal GCs indicates that this process takes place in the thyroid and in the LN. In addition to cell recruitment, the formation of LFs requires cytokines and chemokines that provide the signals for tissue organization. 65 Among them, SDF-1 66 and BLC act synergistically: the former directs the migration of naive B cells to the GC and the latter induces the subsequent exit of activated B cells. 67 Responsiveness to SDF-1 seems to be regulated during B lymphocyte activation and correlates with the location of B cells within the secondary lymphoid organ. The relatively high level of transcription of these chemokines found by us in intrathyroidal LFs underlines the parallelism between canonical and intrathyroidal LFs, and the high level of activation of the latter. We are currently working to determine the cell source of each of these chemokines, and whether the location of their receptors is in accordance with the suggestion that intrathyroidal GCs can organize and perpetuate themselves (MP Armengol, manuscript in preparation).
The major question that arises from this work is whether intrathyroidal LFs containing GCs are involved in pathogenesis. It is difficult to answer this question, because the role of the physiological ectopic LFs that appear during the response to infection has not yet been elucidated. It is not clear what advantage is obtained by the immune system with the placement of these advanced posts in the target tissue. The LFs in areas very rich in antigen and outside the lymphoid tissue might allow a fast expansion of the response, which thus would reach a high level of intensity. The recruitment of weakly autoreactive B cells in the thyroid and the extensive revision of the receptors in these cells (which thus would acquire high-affinity surface Igs), may be important for the production of high-affinity autoantibodies. In a normal LF, autoreactive B cells would be eliminated by apoptosis as they would encounter soluble antigen 31 but this does not happen to the B cells in intrathyroidal GCs despite the availability of very high levels of antigen, thus suggesting that the microenvironment in ectopic LFs is permissive for autoimmune responses. Later, self-antigen presentation by autoreactive B cells and perhaps also by both classical and rogue antigen presenting cells (APCs), such as thyrocytes expressing high levels of histocompatibility leucocyte antigen (HLA) and adhesion molecules 68-70 may be crucial for the maintenance and expansion of the autoimmune response. In this scenario it is easy to predict that autoimmune responses would be more difficult to control once LFs with GCs develop in the target tissue.
The event that initiates AITD still has to be identified, but sudden changes in the level of iodine intake are probably important. 70 It is not difficult to conceive that an initial nonspecific inflammatory response induced by local iodine toxicity could lead to the secretion of inflammatory cytokines and chemokines by macrophages, dendritic cells, and the thyroid epithelial follicular cells themselves. In certain circumstances, endothelial cells may acquire the features of HEVs and recruit B and T lymphocytes whereas some stromal cells evolve into follicular dendritic cells. In this case, the simultaneous onset of the immune response in the LNs is not so crucial, as the lymphocytes in the thyroid glands could promote the immune response.
The possible involvement of intrathyroidal lymphoid tissue in AITD suggests that the local administration of immunosuppressive therapy could control the disease. This is not so important in the case of AITD, because the currently available treatment is overall satisfactory; however, it may be important for other tissue-specific autoimmune diseases because the administration of a local treatment would result in side effects that are less severe than systemic immunosuppressive treatment. Overall, our results support the relevance of the events in the target organ in the pathogenesis of organ-specific autoimmunity.
Acknowledgments
We thank the colleagues cited in the text for their generous donations of antibodies and other reagents (J. F. Tedder, R. Vilella, P. Garrone); C. López for measuring thyroid autoantibodies; M. A. Fernández for his support in microscope image acquisition and processing; Dr. A Alastrué, Dr. Escalante, and Dr. G. Obiols for help in collecting thyroid sample materials; Dr. L. Alcalde, Dr. M. Sospedra, and Dr. F. Vargas for help in tissue processing; Pharmacia-Upjohn for preparing and supplying us with biotinylated Tg and TPO antigens; Dr. J. Verdaguer and M. Cullell for critical revision of the manuscript; and to our colleagues in the Endocrine Division and to Prof. M. Foz for their support and encouragement.
Footnotes
Address reprint requests to Prof. R. Pujol-Borrell, Immunology Unit, Hospital Universitari Germans Trias i Pujol, Ctra. Canyet s/n, P.O. Box 72, 08916 Badalona, (Barcelona), Spain. E-mail: rpujolb@servet.uab.es.
Supported by the “Fondo de Investigaciones Sanitarias” (project 98/1123, to M. P. A.; and grants 98/1123 and 99/1063).
Presented in part at the second International Autoimmunity Congress, Tel Aviv, March, 8 to 11, 1999.
References
- 1.Weetman AP, McGregor AM: Autoimmune thyroid disease: further developments in our understanding. Endocr Rev 1994, 15:788-830 [DOI] [PubMed] [Google Scholar]
- 2.Ajjan RA, Kemp EH, Waterman EA, Watson PF, Endo T, Onaya T, Weetman AP: Detection of binding and blocking autoantibodies to the human sodium-iodide symporter in patients with autoimmune thyroid disease. J Clin Endocrinol Metab 2000, 85:2020-2027 [DOI] [PubMed] [Google Scholar]
- 3.Rapoport B, Chazenbalk GD, Jaume JC, McLachlan SM: The thyrotropin (TSH) receptor: interaction with TSH and autoantibodies Endocr Rev 1998, 19:673-716 [DOI] [PubMed] [Google Scholar]
- 4.Fukuma N, Petersen VB, McLachlan SM, Pegg CA, Rees Smith B: Human monoclonal thyroglobulin autoantibodies of high affinity. I. Production, characterisation and interaction with murine monoclonal thyroglobulin antibodies. Autoimmunity 1991, 10:291-295 [DOI] [PubMed] [Google Scholar]
- 5.Prentice L, Kiso Y, Fukuma N, Horimoto M, Petersen V, Grennan F, Pegg C, Furmaniak J, Rees Smith B: Monoclonal thyroglobulin autoantibodies: variable region analysis and epitope recognition. J Clin Endocrinol Metab 1995, 80:977-986 [DOI] [PubMed] [Google Scholar]
- 6.Portolano S, Seto P, Chazenbalk GD, Nagayama Y, McLachlan SM, Rapoport B: A human Fab fragment specific for thyroid peroxidase generated by cloning thyroid lymphocyte-derived immunoglobulin genes in a bacteriophage lambda library. Biochem Biophys Res Commun 1991, 179:372-377 [DOI] [PubMed] [Google Scholar]
- 7.Hexham JM, Furmaniak J, Pegg C, Burton DR, Smith BR: Cloning of a human autoimmune response: preparation and sequencing of a human anti-thyroglobulin autoantibody using a combinatorial approach. Autoimmunity 1992, 12:135-141 [DOI] [PubMed] [Google Scholar]
- 8.McIntosh RS, Asghar MS, Watson PF, Kemp EH, Weetman AP: Cloning and analysis of IgG kappa and IgG lambda anti-thyroglobulin autoantibodies from a patient with Hashimoto’s thyroiditis: evidence for in vivo antigen-driven repertoire selection. J Immunol 1996, 157:927-935 [PubMed] [Google Scholar]
- 9.Nye L, De Carvalho LP, Roitt IM: An investigation of the clonality of human autoimmune thyroglobulin antibodies and their light chains. Clin Exp Immunol 1981, 46:161-170 [PMC free article] [PubMed] [Google Scholar]
- 10.Finke R, Seto P, Ruf J, Carayon P, Rapoport B: Determination at the molecular level of a B-cell epitope on thyroid peroxidase likely to be associated with autoimmune thyroid disease. J Clin Endocrinol Metab 1991, 73:919-921 [DOI] [PubMed] [Google Scholar]
- 11.Estienne V, McIntosh RS, Ruf J, Asghar MS, Watson PF, Carayon P, Weetman AP: Comparative mapping of cloned human and murine antithyroglobulin antibodies: recognition by human antibodies of an immunodominant region. Thyroid 1998, 8:643-646 [DOI] [PubMed] [Google Scholar]
- 12.Guo J, McIntosh RS, Czarnocka B, Weetman AP, Rapoport B, McLachlan SM: Relationship between autoantibody epitopic recognition and immunoglobulin gene usage. Clin Exp Immunol 1998, 111:408-414 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.McIntosh R, Watson P, Weetman A: Somatic hypermutation in autoimmune thyroid disease. Immunol Rev 1998, 162:219-231 [DOI] [PubMed] [Google Scholar]
- 14.McLachlan SM, Rapoport B: Autoimmune response to the thyroid in humans: thyroid peroxidase—the common autoantigenic denominator. Intern Rev Immunol 2000, 19:587-618 [DOI] [PubMed] [Google Scholar]
- 15.Martin A, Barbesino G, Davies TF: T cell receptors and autoimmune thyroid disease—signposts for T-cell-antigen driven diseases. Int Rev Immunol 1999, 18:111-118 [DOI] [PubMed] [Google Scholar]
- 16.Bottazzo GF, Doniach D: Autoimmune thyroid disease. Annu Rev Med 1986, 37:353-359 [DOI] [PubMed] [Google Scholar]
- 17.McLachlan SM, McGregor A, Smith BR, Hall R: Thyroid-autoantibody synthesis by Hashimoto thyroid lymphocytes. Lancet 1979, 1:162-163 [DOI] [PubMed] [Google Scholar]
- 18.McLachlan SM, Fawcett J, Atherton MC, Thompson P, Baylis P, Smith BR: Thyroid autoantibody synthesis by cultures of thyroid and peripheral blood lymphocytes. II. Effect of thyroglobulin on thyroglobulin antibody synthesis. Clin Exp Immunol 1983, 52:620-628 [PMC free article] [PubMed] [Google Scholar]
- 19.McLachlan SM, Dickinson A, Baylis P, Proctor S, Rees Smith B: Enrichment and depletion of thyroglobulin autoantibody synthesizing lymphocytes. Clin Exp Immunol 1983, 53:397-405 [PMC free article] [PubMed] [Google Scholar]
- 20.Atherton MC, McLachlan SM, Pegg CA, Dickinson A, Baylis P, Young ET, Proctor SJ, Rees Smith B: Thyroid autoantibody synthesis by lymphocytes from different lymphoid organs: fractionation of B cells on density gradients. Immunology 1985, 55:271-279 [PMC free article] [PubMed] [Google Scholar]
- 21.McLachlan SM, Pegg CA, Atherton MC, Middleton SL, Clark F, Rees Smith B: TSH receptor antibody synthesis by thyroid lymphocytes. Clin Endocrinol 1986, 24:223-230 [DOI] [PubMed] [Google Scholar]
- 22.Randen I, Mellbye OJ, Forre O, Natvig JB: The identification of germinal centres and follicular dendritic cell networks in rheumatoid synovial tissue. Scand J Immunol 1995, 41:481-486 [DOI] [PubMed] [Google Scholar]
- 23.Schröder AE, Greiner A, Seyfert C, Berek C: Differentiation of B cells in the nonlymphoid tissue of the synovial membrane of patients with rheumatoid arthritis. Proc Natl Acad Sci USA 1996, 93:221-225 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Wagner UG, Kurtin PJ, Wahner A, Brackertz M, Berry DJ, Goronzy JJ, Weyand CM: The role of CD8+CD40L+ T cells in the formation of germinal centres in rheumatoid synovitis. J Immunol 1998, 161:6390-6397 [PubMed] [Google Scholar]
- 25.Shione H, Fujii Y, Okumura M, Yakeuchi Y, Inoue AM, Matsuda H: Failure to down-regulate Bcl-2 protein in thymic germinal center B cells in myasthenia gravis. Eur J Immunol 1997, 27:805-809 [DOI] [PubMed] [Google Scholar]
- 26.Stott D, Hiepe F, Hummel M, Steinhauser GCB: Antigen-driven clonal proliferation of B cell within the target tissue of an autoimmune disease. J Clin Invest 1998, 102:938-943 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Murakami J, Shimizu Y, Kashii Y, Kato T, Minemura M, Okada K, Nambu S, Takahara T, Higuchi K, Maeda Y, Kumada T, Watanabe A: Functional B-cell response in intrahepatic lymphoid follicles in chronic hepatitis C. Hepatology 1999, 30:143-150 [DOI] [PubMed] [Google Scholar]
- 28.Zaitoun AM: The prevalence of lymphoid follicles in Helicobacter pylori associated gastritis in patients with ulcers and non-ulcer dyspepsia. J Clin Pathol 1995, 48:325-329 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.MacLennan IC: Germinal centers. Annu Rev Immunol 1994, 12:117-139 [DOI] [PubMed] [Google Scholar]
- 30.Liu YJ, Arpin C: Germinal center development. Immunol Rev 1997, 156:111-126 [DOI] [PubMed] [Google Scholar]
- 31.Shokat KM, Goodnow CC: Antigen-induced B-cell death and elimination during germinal-centre immune responses. Nature 1995, 375:334-338 [DOI] [PubMed] [Google Scholar]
- 32.Pulendran B, van Driel R, Nossal GJ: Immunological tolerance in germinal centres. Immunol Today 1997, 18:27-32 [DOI] [PubMed] [Google Scholar]
- 33.Han S, Dillon SR, Zheng B, Shimoda M, Schlissel MS, Kelsoe G: V(D)J recombinase activity in a subset of germinal center B lymphocytes. Science 1997, 278:301-305 [DOI] [PubMed] [Google Scholar]
- 34.Hikida M, Mori M, Takai T, Tomochika K, Hamatani K, Ohmori H: Reexpression of Rag-1 and Rag-2 genes in activated mature mouse B cells. Science 1996, 274:2092-2094 [DOI] [PubMed] [Google Scholar]
- 35.Giachino C, Padovan E, Lanzavecchia A: Re-expression of RAG-1 and RAG-2 genes and evidence for secondary rearrangements in human germinal center B lymphocytes. Eur J Immunol 1998, 28:3506-3513 [DOI] [PubMed] [Google Scholar]
- 36.Ludewig B, Odermatt B, Landmann S, Hengartner H, Zinkernagel RM: Dendritic cells induce autoimmune diabetes and maintain disease via de novo formation of local lymphoid tissue. J Exp Med 1998, 188:1493-1501 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Falcone M, Lee J, Patstone G, Yeung B, Sarvetnick N: B lymphocytes are crucial antigen-presenting cells in the pathogenic autoimmune response to GAD65 antigen in nonobese diabetic mice. J Immunol 1998, 161:1163-1168 [PubMed] [Google Scholar]
- 38.Lucas-Martin A, Foz-Sala M, Todd I, Bottazzo GF, Pujol-Borrell R: Occurrence of thyrocyte HLA class II expression in a wide variety of thyroid diseases: relationship with lymphocytic infiltration and thyroid autoantibodies. J Clin Endocrinol Metab 1988, 66:367-375 [DOI] [PubMed] [Google Scholar]
- 39.Sospedra M, Obiols G, Babi LF, Tolosa EF, Vargas F, Roura-Mir C, Lucas-Martin A, Ercilla G, Pujol-Borrell R: Hyperinducibility of HLA class II expression of thyroid follicular cells from Graves’ disease. A primary defect? J Immunol 1995, 154:4213-4222 [PubMed] [Google Scholar]
- 40.Rose ML, Birbeck MSC, Wallis VJ, Forrester GA, Davies AJS: Peanut lectin binding properties of germinal centres of mouse lymphoid tissue. Nature 1980, :364-366 [DOI] [PubMed] [Google Scholar]
- 41.Sospedra M, Tolosa E, Armengol P, Ashhab Y, Urlinger S, Lucas-Martin A, Foz-Sala M, Jaraquemada D, Pujol-Borrell R: Hyperexpression of transporter in antigen processing-1 (TAP-1) in thyroid glands affected by autoimmunity: a contributing factor to the breach of tolerance to thyroid antigens? Clin Exp Immunol 1997, 109:98-106 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Foy TM, Laman JD, Ledbetter JA, Aruffo A, Claassen E, Noelle RJ: gp39-CD40 interactions are essential for germinal center formation and the development of B cell memory. J Exp Med 1994, 180:157-163 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Grammer AC, McFarland RD, Heaney J, Darnell BF, Lipsky PE: Expression, regulation, and function of B cell-expressed CD154 in germinal centers. J Immunol 1999, 163:4150-4159 [PubMed] [Google Scholar]
- 44.Giordano C, Stassi G, De Maria R, Todaro M, Richiusa P, Papoff G, Ruberti G, Bagnasco M, Testi R, Galluzzo A: Potential involvement of Fas and its ligand in the pathogenesis of Hashimoto’s thyroiditis. Science 1997, 275:960-963 [DOI] [PubMed] [Google Scholar]
- 45.Terol MJ, Cid MC, Lopez-Guillermo A, Juan M, Yague J, Miralles A, Vilella R, Vives J, Cardesa A, Montserrat E, Campo E: Expression of intercellular adhesion molecule-3 (ICAM-3/CD50) in malignant lymphoproliferative disorders and solid tumors. Tissue Antigens 1996, 48:271-277 [DOI] [PubMed] [Google Scholar]
- 46.Tu L, Delahunty MD, Ding H, Luscinskas FW, Tedder TF: The cutaneous lymphocyte antigen is an essential component of the L-selectin ligand induced on human vascular endothelial cells. J Exp Med 1999, 189:241-252 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Kofler R, Wick G: Immunofluorescence localization of thyroglobulin-autoantibody producing cells in various organs of obese strain (OS) chickens. Z Immunol Forsch 1978, 154:88-93 [PubMed] [Google Scholar]
- 48.Ratanachaiyavong SM, McGregor AM: Immunosuppressive effects of antithyroid drugs. Clin Endocrinol Metab 1985, 14:449-466 [DOI] [PubMed] [Google Scholar]
- 49.Pellegriti G, Belfiore A, Giuffrida D, Lupo L, Vigneri R: Outcome of differentiated throid cancer in Grave’s patients. J Clin Endocrinol Metab 1998, 83:2805-2809 [DOI] [PubMed] [Google Scholar]
- 50.Tew JG, Phipps RP, Mandel TE: The maintenance and regulation of the humoral immune response: persisting antigen and the role of follicular antigen-binding dendritic cells as accessory cells. Immunol Rev 1980, 53:175-201 [DOI] [PubMed] [Google Scholar]
- 51.Perry M, Whyte A: Immunology of the tonsils. Immunol Today 1998, 19:414-421 [DOI] [PubMed] [Google Scholar]
- 52.Hykek E, Isaacson PG: Primary B cell lymphoma of the thyroid and relationship to Hashimoto thyroiditis. Hum Pathol 1988, 9:1315-1326 [DOI] [PubMed] [Google Scholar]
- 53.Williams ED: Malignant lymphoma of the thyroid. Clin Endocrinol Metab 1991, 10:379-389 [DOI] [PubMed] [Google Scholar]
- 54.Kiljanski J, Nebes V, Stachura I, Kennerdell JS, Wall JR: Should Graves’ disease be considered a collagen disorder of the thyroid, skeletal muscle and connective tissue? Horm Metab Res 1995, 27:528-532 [DOI] [PubMed] [Google Scholar]
- 55.Nemazee D: Receptor selection in B and T lymphocytes. Annu Rev Immunol 2000, 18:19-51 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Han S, Zheng B, Schatz DG, Spanopoulou E, Kelsoe G: Neoteny in lymphocytes: Rag-1 and Rag-2 expression in germinal center B cells. Science 1996, 374:2094-2097 [DOI] [PubMed] [Google Scholar]
- 57.Papavasiliou F, Casellas R, Suh H, Qin XF, Besmer E, Pelanda R, Nemazee D, Rajewsky K, Nussenzweig MC: V(D)J recombination in mature B cells: a mechanism for altering antibody responses. Science 1997, 278:298-301 [DOI] [PubMed] [Google Scholar]
- 58.Hikida M, Ohmori H: Rearrangement of lambda light chain genes in mature B cells in vitro and in vivo. Function of reexpressed recombination-activating gene (RAG) products. J Exp Med 1998, 187:795-799 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Meffre E, Papavasiliou F, Cohen P, de Bouteiller O, Bell D, Karasuyama H, Schiff C, Banchereau J, Liu YJ, Nussenzweig MC: Antigen receptor engagement turns off the V(D)J recombination machinery in human tonsil B cells. J Exp Med 1998, 188:765-772 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Yu W, Nagaoka H, Jankovic M, Misulovin Z, Suh H, Rolink A, Melchers F, Meffre E, Nussenzweig MC: Continued RAG expression in late stages of B cell development and no apparent re-induction after immunization. Nature 1999, 400:682-687 [DOI] [PubMed] [Google Scholar]
- 61.Monroe RJ, Seidl KJ, Gaertner F, Han S, Chen F, Sekiguchi J, Wang J, Ferrini R, Davidson L, Kelsoe G, Alt FW: RAG2: GFP knockin mice reveal novel aspects of RAG2 expression in primary and peripheral lymphoid tissues. Immunity 1999, 11:201-212 [DOI] [PubMed] [Google Scholar]
- 62.Sospedra M, Ferrer-Francesch X, Dominguez O, Juan M, Foz-Sala M, Pujol-Borrell R: Transcription of a broad range of self-antigens in human thymus suggests a role for central mechanisms in tolerance toward peripheral antigens. J Immunol 1998, 161:5918-5929 [PubMed] [Google Scholar]
- 63.Dorner T, Foster SJ, Farner NL, Lipsky PE: Immunoglobulin kappa chain receptor editing in systemic lupus erythematosus. J Clin Invest 1998, 102:688-694 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Itoh K, Meffre E, Albesiano E, Farber A, Dines D, Stein P, Asnis SE, Furie RA, Jain RI, Chiorazzi N: Immunoglobulin heavy chain variable region gene replacement as a mechanism for receptor revision in rheumatoid arthritis synovial tissue B lymphocytes. J Exp Med 2000, 192:1151-1164 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Ansel KM, Ngo VN, Hyman PL, Luther SA, Forster R, Sedgwick JD, Browning JL, Lipp M, Cyster JG: A chemokine-driven positive feedback loop organizes lymphoid follicles. Nature 2000, 406:309-314 [DOI] [PubMed] [Google Scholar]
- 66.Bleul CC, Schultze JL, Springer TA: B lymphocyte chemotaxis regulated in association with microanatomic localisation, differentiation state, and B cell receptor engagement. J Exp Med 1998, 187:753-762 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Gunn M, N′go VN, Ansel KM, Ekland EH, Jason GC, Williams LT: A B cell homing chemokine made in lymphoid follicles activates Burkitt’s lymphoma receptor-1. Nature 1998, 391:799-803 [DOI] [PubMed] [Google Scholar]
- 68.Pujol-Borrell R, Hanafusa T, Chiovato L, Bottazzo GF: Lectin-induced expression of DR antigen on human cultured follicular thyroid cells. Nature 1983, 303:71-73 [DOI] [PubMed] [Google Scholar]
- 69.Bottazzo GF, Pujol-Borrell R, Hanafusa TMF: Role of aberrant HLA-DR expression and antigen presentation in induction of endocrine autoimmunity. Lancet 1983, 2:1115-1118 [DOI] [PubMed] [Google Scholar]
- 70.Verma S, Hutchings P, Guo J, McLachlan S, Rapoport B, Cooke A: Role of MHC class I expression and CD8(+) T cells in the evolution of iodine-induced thyroiditis in NOD-H2(h4) and NOD mice. Eur J Immunol 2000, 30:1191-1202 [DOI] [PubMed] [Google Scholar]