Loss of ileal IgA+ plasma cells and of CD4+ lymphocytes in ileal Peyer's patches of vitamin A deficient rats (original) (raw)
Abstract
Child mortality in diarrhoeal disease is increased significantly by vitamin A deficiency in poor countries. The pathological mechanisms are not known in detail. However, in this paper we report that vitamin A-deficient Wistar rats had much reduced IgA+ plasma cells in the ileal lamina propria (eightfold reduction from 470 cells/mm2, P = 0·009), as well as a prominent reduction of CD4+ cells in the parafollicular regions of ileal Peyer's patches (reduction from 7200 to 105 cells/mm2, P = 0·009). IL-2Ralpha-chain (CD25) positive lymphocytes in the ileal Peyer's patches were also reduced significantly in vitamin A deficiency (from 1400 to 300 cells/mm2, P = 0·009). The density of CD8 cells tended to be increased relative to the control animals (from 5100 to 6000 cells/mm2, not statistically significant). In conclusion, the marked decrease of lamina propria IgA+ plasma cells may be one cause of the high diarrhoeal mortality in vitamin A deficiency. This, in turn, appears to be related to reduced numbers of activated or regulatory CD4+ T cells in Peyer's patches.
Keywords: immunohistochemistry, mucosal immunology, Peyer's patches, small intestine, vitamin A deficiency
Introduction
Vitamin A deficiency (VAD) is one of the main nutritional deficiencies in low-income countries and is associated with increased incidence of infectious diseases [1]. A major cause of increased child death due to VAD is increased mortality in diarrhoeal disease, as shown in several community studies [2–5]. Thus, supplementation with vitamin A has been shown to decrease child mortality rates strongly, with the most clear-cut effects on mortality in diarrhoea.
The degree and extent of bacterial colonization of the small intestine in rats has been shown to be increased by VAD. Normal animals were colonized only in the ileum, while VAD rats were colonized in all parts of the small intestine. Penetration of gut bacteria to lymph nodes and blood as well as mortality in septicaemia was increased by VAD [6]. Intestinal IgA can neutralize bacteria, toxins and viruses in the gut lumen and may also act intracellularly [7]. VAD reduced the levels of serum and biliary IgA [8].
Mucosal IgA producing B cells responding to T cell-dependent antigens, such as bacterial exotoxins, are activated by T helper cells in the Peyer's patches, go through an IgA switch, differentiation and clonal expansion. Class switching and maturation into IgA producing plasma cells has been suggested recently to occur locally in the gut [9]. However, the B cells usually will migrate out from the Peyer's patches via lymphatic vessels to the mesenteric lymph nodes, mature to plasma cells and finally home back to the intestinal lamina propria via the blood [10]. However, it is not clear by what mechanism VAD can affect intestinal IgA levels and contribute to an increased susceptibility to intestinal infections. Therefore, we wanted to investigate the effects of VAD on IgA+ plasma cells in the ileum of rats. Furthermore, we asked if changes of IgA+ cells and biliary IgA by VAD was caused by alterations in the T cell population of ileal Peyer's patches. We have thus compared the ileal immunohistochemistry of vitamin A replete rats with rats on a VAD diet.
Materials and methods
Animals
Weanling male Wistar Furth rats (B&K Universal, Stockholm, Sweden) were fed a vitamin A-deficient or vitamin A-replete diet (Analycen, Lidköping, Sweden). The vitamin A-deficient pellets consisted of 24% casein de-vitaminized by heating at 105°C for 5 days, 61% corn starch, 4% cellulose flour, a 1% mixture of all necessary vitamins except vitamin A, a 4% salt mixture and 6% peanut oil. The vitamin A-replete diet was identical except for the presence of 17 IU/g of vitamin A. All animals appeared to be healthy before sacrifice.
After sacrifice, 5 mm-long sections of ileum containing Peyer's patches were embedded in OCT-medium (Sakura Finetechnical Co., Tokyo, Japan) and frozen in isopentane which had been precooled in liquid nitrogen. The tissue blocks were stored at − 70°C prior to cutting into six µm-thick sections.
The experimental work was approved by the local ethics committee for animal research.
Immunohistochemistry
Sections were cut and then fixed in cold 50% acetone for 30 s in cold 100% acetone for 5 min and air-dried for at least 10 min. Depletion of endogenous peroxidase activity was performed with a PBS solution containing glucose, glucose oxidase and sodium azide. The sections were incubated overnight at 4°C in a humid chamber, with mouse monoclonal antibodies directed against rat CD4 (W3/25) diluted 1/50, rat IL-2R-alfa chain – also known as CD25 (OX39 1/20 and Serotec MCA494G antibodies diluted 1/100 were used together) − or rat CD8 (Ox8, diluted 1/50), rat IgA (biotinylated, MCA191B, diluted 1/5000); and as negative con-trol, a mouse anti-TNP antibody (03001D 1/50; Pharmingen, Stockholm, Sweden). All primary antibodies were diluted in PBS-Tween + 2% BSA. After washing the sections 3 × 5 min in PBS, they were incubated with biotinylated horse antimouse IgG (BA-2001; Vector Laboratories, Burlingame, CA, USA), for 1 h at room temperature.
After three washings the sections were incubated for 30 min with streptavidin linked to horseradish peroxidase (streptABComplex/HRP, Dako, Glostrup, Denmark). For the colour reaction, they were washed again and incubated for 6–7 min in a filtered solution containing 0·18 mg/ml amino ethyl carbazole + 10% DMSO + 0·02 m sodium acetate at pH 5·5. All sections were counterstained with Mayer's haematoxylin and mounted in Gurr Aquamount (BDH). After blinding of samples, the cells were counted using a computer supported image analysis system, Leica Q500MC (Leica, Cambridge, UK) All results were confirmed by a separate observer. Cells stained for IgA, CD4, CD25 or CD8 were recorded as cells/mm2 tissue. The epithelial areas of lamina propria were excluded when counting immune cells. Areas marked for counting were about 0·02 mm2 each and the distribution of cells in the lamina propria and between T cell areas was fairly homogeneous throughout each section.
Statistics
Statistical significance was determined using a two-tailed Mann–Whitney _U_-test.
Results
In vitamin A-deficient rats, the reductions of IgA+ plasma cells in the ileal lamina propria were dramatic and statistically significant (P = 0·009; Figs 1 and 2). The median number of IgA+ plasma cells were reduced almost eightfold, from ca. 470 to 60 IgA+ cells/mm2. Neither vitamin A-deficient or vitamin A-replete animals showed any signs of infiltrating inflammatory cells into the gut mucosa (data not shown).
Fig. 1.

Number of IgA+ plasma cells per mm2 in ileal lamina propria. Each open ring represents one vitamin A-deficient rat (VAD); well-nourished control rats (NORM) are represented by filled squares. Statistical significance was determined by the Mann–Whitney _U_-test, _P_-values are indicated within the figure. Horizontal lines represent the median of each group.
Fig. 2.

IgA+ plasma cells in ileal sections from a vitamin A-deficient rat (top) and a vitamin A-replete rat (bottom).
To determine if the effects of vitamin A deficiency also acted on the regulation of intestinal plasma cells, T helper cells in the ileal Peyer's patches were also studied. Indeed, the median number of CD4+ cells in the T cell areas (parafollicular regions) were reduced seventy-fold, from ca. 7200 to 105 cells per mm2 (Figs 3a,4). The CD4+ cells that were present in VAD rat ileum usually stained more weakly than in replete rats. As shown in Fig. 3b, IL-2 receptor alpha chain (CD25) positive lymphocytes in the T cell areas of ileal Peyer's patches were also significantly reduced by VAD (from 1400 to 300 cells per mm2, P = 0·009). Relative to all parafollicular cells (mainly T cells), CD25 cells decreased from 9·3% to 1·6%, P = 0·009 (Fig. 3c). The median size of the parafollicular regions tended to be reduced in VAD rats (not shown).
Fig. 3.

(a) Number of CD4+ cells per mm2 in the T cell parafollicular areas of Peyer's patches. (b) Number of CD25+ cells per mm2 in T cell areas of Peyer's patches. (c) Frequency of CD25+ cells out of total cells in T cell areas of Peyer's patches. (d) Number of CD8+ cells per mm2 in the T cell areas of Peyer's patches. Statistical significance was determined by the Mann–Whitney _U_-test, _P_-values are indicated within the figure. Horizontal lines represent the median of each group.
Fig. 4.
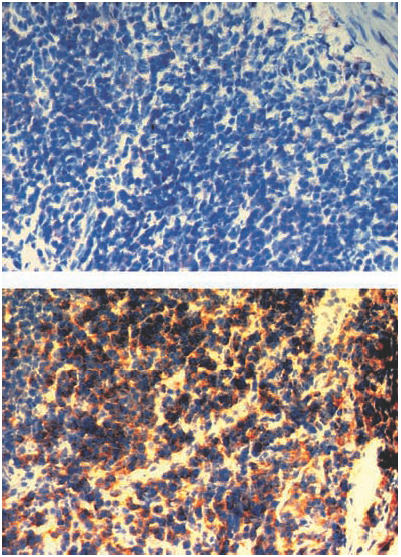
CD4+ cells in sections of ileal Peyer's patches from a vitamin A-deficient rat (top) and a vitamin A-replete rat (bottom).
In contrast, CD8+ in the Peyer's patches tended to increase (from 5100 to 6000 per mm2, P = 0·12) in vitamin A deficiency (Fig. 3d). However, the size of parafollicular areas per section tended to decrease (41% reduction, P = 0·14). Therefore, total numbers of CD8 cells per section tended to be lower in VAD, while the CD8 : CD4 ratio and the density of CD8 cells tended to increase in VAD rats.
In the ileal lamina propria as in the Peyer's patches, CD4+ cells were significantly reduced in VAD (from a median of 1390 to 355 cells per mm2, P < 0·01). However, with regard to CD25+ cells (the median was reduced from 740 to 415 cells per mm2, P = 0·12) or CD8 (from 900 cells to 960 per mm2, not significant), there were no clear effects of VAD in the ileal lamina propria.
Discussion
In the present study, we found reduced numbers of IgA+ plasma cells in the lamina propria in VAD. Furthermore, in the Peyer's patches the CD4+ cells were reduced in number, the numbers of CD8+ cells tended to be slightly increased and the number of CD25+ cells in the T cell areas was reduced. This indicates that the reductions of biliary IgA and of the number of intestinal IgA+ plasma cells in VAD may be caused by reduced activation of T cells in the Peyer's patches and by decreased numbers of Peyer's patch CD4+ T cells.
The literature describing the effect of VAD on the mucosal immune system is limited. However, it has been shown previously that antigen-specific and total IgA of serum and bile was much reduced in vitamin A-deprived rats [8]. According to another report, VAD led instead to increased biliary IgA as well as increased number of CD8+ cells while the number of IgA+ plasma cells in the jejunal lamina propria was reduced. The increase in biliary IgA in that study may have been caused by a higher total bacterial load during VAD, as the rats were colonized with ampicillin-resistant Escherichia coli and the natural gut flora was suppressed with antibiotics [6]. In IgA-deficient humans immune exclusion in the gut is clearly suboptimal, with increased influx of antigens. This has been found despite abundant intestinal IgM production, providing compensatory secretory IgM [11]. While the total number of bacteria in the small intestine as a whole was higher in VAD rats than in normal rats, it was not changed in the ileum [6]. Events in the ileum may therefore reflect more of the direct effects of VAD on the mucosal immune system and be less affected than in the upper small intestine by changes in bacterial load. In the present study, as in previous studies, VAD rats still appeared healthy because they were kept in a pathogen-free environment. However, when VAD rats are exposed to experimental infection they clearly have increased susceptibility to disease [6].
T cell activation is followed by early cell surface expression of the IL-2 receptor (CD25) and its interaction with IL-2 is a critical event in proliferation, differentiation and function of T cells [12]. In primates, 15% of lamina propria lymphocytes are CD25+ compared to <3% in mesenteric lymph nodes [12]. This is due supposedly to the massive antigenic exposure of lymphocytes in the gut and the need for tight regulation of the intestinal immune system. It may be argued that the CD25+ cells could be B cells rather than T cells. However, this is not likely as the CD25+ cells were only present in parafollicular T cell regions, but were absent in the B cell-rich follicular areas. In the present study 9·3% of the cells in the Peyer's patch T cell areas carried CD25. With the VAD diet, activation fell to 1·6%. To our knowledge, CD25 expression has not been analysed previously in Peyer's patches. Moreover, cells expressing the T cell activation marker OX40 have been found in the Peyer's patches of normal mice [13]. Interestingly, in protein-deprived mice, T cell expression of CD28 and CTLA-4 was down-regulated in Peyer's patches, indicating a reduced activation or dysregulation of the T cells [14].
Accordingly, CD25 is involved not only in early T cell activation but is also constitutively present on regulatory CD25+ CD4+ T cells. These cells can, in fact, exert immunosuppressive effects through production of TGF-beta [15]. TGF-beta is also acting as an important switch factor for IgA production in the gut mucosa [16]. Thus, the reduction of intestinal CD25+ cells in VAD may cause both immunological dysregulation in the gut and decreased intestinal IgA due to impaired IgA class switching. In vitamin A-deficient rats CD8+ cells tended to increase somewhat rela-tive to CD4+ cells, due possibly to a disturbance in regulatory CD25+ CD4+ T cells. Notably, the down-regulation of CTLA-4+ T cells seen in protein-deprived mice [14] could also lead to immunological dysregulation as CTLA-4 is expressed on TH3 cells as well as other CD4+ regulatory T cells [17,18].
While studies on the role of VAD for the impairment of gut immunity are scarce, there are some studies of intestinal immunity during protein deprivation and malnutrition. Thus, protein deprivation in mice was shown to cause significant and rapid reductions of B cells, CD4+ cells and CD8+ cells in Peyer's patches, as well as of intestinal IgA [14]. Malnutrition in suckling rat pups also led to reduced numbers of T cells [19,20].
Our data indicate that Peyer's patch T cells may play an important role in the reduction of the numbers of IgA cells and biliary IgA levels during VAD. Reduced numbers of activated or regulatory Peyer's patch T cells, as well as a marked decrease of CD4+ T cells may hamper the activation of B cells in the Peyer's patches and their maturation into IgA-producing plasma cells in the mucosa. This could, in itself, explain the reduced density of IgA cells in ileal lamina propria. Additionally, reduction of CD25+ CD4+ cells could lead to immunological dysregulation in the gut, such that the frequency of pathogen-specific cells decrease relative to immune cells with other specificities. For a definitive answer on the role of CD4+ and CD25+ T cells and plasma cells, reconstitution experiments should be performed by transfer of these cell populations from naive and immunized vitamin A-replete donors. The reduction of CD4+ cells was stronger than the reduction of IgA cells, which may indicate that mucosal IgA responses to T cell-independent antigens could be less sensitive to or partially compensate for the influence of VAD. In mice, T cell-independent IgA forms part of the mucosal response against commensal intestinal bacteria [21].
The effect of VAD on the mucosal immune system is similar to those found during protein malnutrition. In undernourished children, these conditions usually go together [22]. Therefore, the similarities strengthen the likelihood that the effects on local T helper cells and intestinal IgA production are of importance for the host defence in the gut of individuals suffering from malnutrition.
Acknowledgments
We thank Helena Kahu for her skilful technical assistance. The study was supported by grants from the Bergvall foundation, Stockholm, and the Royal Society for Arts and Sciences in Göteborg, Sweden.
REFERENCES
- 1.Wiedermann U, Hanson L, Dahlgren UI. Vitamin A – and the immune system. Immunologist. 1996;4:70–5. [Google Scholar]
- 2.Rahmathullah L, Underwood BA, Thulasiraj RD, et al. Reduced mortality among children in southern India receiving a small weekly dose of vitamin A. N Engl J Med. 1990;323:929–35. doi: 10.1056/NEJM199010043231401. [DOI] [PubMed] [Google Scholar]
- 3.Daulaire NM, Starbuck ES, Houston RM, Church MS, Stukel TA, Pandey MR. Childhood mortality after a high dose of vitamin A in a high risk population. Br Med J. 1992;304:207–10. doi: 10.1136/bmj.304.6821.207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Fawzi WW, Herrera MG, Willett WC, et al. Dietary vitamin A intake and the risk of mortality among children. Am J Clin Nutr. 1994;59:401–8. doi: 10.1093/ajcn/59.2.401. [DOI] [PubMed] [Google Scholar]
- 5.Ross DA, Kirkwood BR, Binka FN, et al. Child morbidity and mortality following vitamin A supplementation in Ghana: time since dosing, number of doses, and time of year. Am J Public Health. 1995;85:1246–51. doi: 10.2105/ajph.85.9.1246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Wiedermann U, Hanson LÅ, Bremell T, Kahu H, Dahlgren UI. Increased translocation of Escherichia coli and development of arthritis in vitamin A deficient rats. Infect Immun. 1995;63:3062–8. doi: 10.1128/iai.63.8.3062-3068.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hayday A, Viney JL. The ins and outs of body surface immunology. Science. 2000;290:97–100. doi: 10.1126/science.290.5489.97. [DOI] [PubMed] [Google Scholar]
- 8.Wiedermann U, Hanson L, Holmgren J, Kahu H, Dahlgren UI. Impaired mucosal antibody response to cholera toxin in vitamin A deficient rats immunized with oral cholera vaccine. Infect Immun. 1993;61:3952–7. doi: 10.1128/iai.61.9.3952-3957.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Fagarasan S, Kinoshita K, Muramatsu M, Ikuta K, Honjo T. In situ class switching and differentiation to IgA-producing cells in the gut lamina propria. Nature. 2001;413:639–43. doi: 10.1038/35098100. [DOI] [PubMed] [Google Scholar]
- 10.Brandtzaeg P, Farstad IN, Johansen F-E, Morton HC, Norderhaug IN, Yamanaka T. The B-cell system of human mucosae and exocrine glands. Immunol Rev. 1999;171:45–87. doi: 10.1111/j.1600-065X.1999.tb01342.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Brandtzaeg P, Nilssen DE. Mucosal aspects of primary B cell deficiency and gastrointestinal infections. Curr Opin Gastroenterol. 1995;11:532–40. [Google Scholar]
- 12.Zeitz M, Greene WC, Peffer NJ, James SP. Lymphocytes isolated from the intestinal lamina propria of normal nonhuman primates have increased expression of genes associated with T cell activation. Gastroenterology. 1988;94:647–55. doi: 10.1016/0016-5085(88)90235-1. [DOI] [PubMed] [Google Scholar]
- 13.Higgins LM, McDonald SA, Whittle N, Crockett N, Shields JG, MacDonald TT. Regulation of T cell activation in vitro and in vivo by targeting the OX40–OX40 ligand interaction: amelioration of ongoing inflammatory bowel disease with an OX40-IgG fusion protein, but not with an OX40 ligand-IgG fusion protein. J Immunol. 1999;162:486–93. [PubMed] [Google Scholar]
- 14.Manhart N, Vierlinger K, Bergmeister H, Boltz-Nitolescu G, Spittler A, Roth E. Influence of short-term malnutrition of mice on the phenotype and costimulatory signals of lymphocytes from spleen and Peyer's patches. Nutrition. 2000;16:197–201. doi: 10.1016/s0899-9007(99)00279-8. [DOI] [PubMed] [Google Scholar]
- 15.Nakamura K, Kitani A, Strober W. Cell contact-dependent immunosuppression by CD4+CD25+ regulatory T cells is mediated by cell surface-bound transforming growth factor beta. J Exp Med. 2001;194:629–44. doi: 10.1084/jem.194.5.629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Weiner HL. Oral tolerance: immune mechanisms and treatment of autoimmune diseases. Immunol Today. 1997;18:335–43. doi: 10.1016/s0167-5699(97)01053-0. [DOI] [PubMed] [Google Scholar]
- 17.Weiner HL. Induction and mechanism of action of transforming growth factor-beta-secreting Th3 regulatory cells. Immunol Rev. 2001;182:207–14. doi: 10.1034/j.1600-065x.2001.1820117.x. [DOI] [PubMed] [Google Scholar]
- 18.Sakaguchi S, Sakaguchi N, Shimizu J, et al. Immunologic tolerance maintained by CD25+ CD4+ regulatory T cells: their common role in controlling autoimmunity, tumor immunity, and transplantation tolerance. Immunol Rev. 2001;182:18–32. doi: 10.1034/j.1600-065x.2001.1820102.x. [DOI] [PubMed] [Google Scholar]
- 19.Fló J, Elías F, Benedetti R, Massouh E. Reversible effects on B and T cells of the gut-associated lymphoid tissues in rats malnourished during suckling: impaired induction of the immune response to intra-Peyer patches immunization with cholera toxin. Clin Immunol Immunopathol. 1996;80:147–54. doi: 10.1006/clin.1996.0108. [DOI] [PubMed] [Google Scholar]
- 20.Fló J, Elías F, Massouh E, Roux ME. Impairment of B and T cell maturation in gut associated lymphoid tissues due to malnutrition during lactation. Dev Comp Immunol. 1994;18:543–55. doi: 10.1016/s0145-305x(06)80008-x. [DOI] [PubMed] [Google Scholar]
- 21.MacPherson AJ, Gatto D, Sainsbury E, Harriman GR, Hengartner H, Zinkernagel RM. A primitive T cell-independent mechanism of intestinal mucosal IgA responses to commensal bacteria. Science. 2000;288:2222–6. doi: 10.1126/science.288.5474.2222. [DOI] [PubMed] [Google Scholar]
- 22.Underwood BA. The role of vitamin A in child growth, development and survival. Adv Exp Med Biol. 1994;352:201–8. doi: 10.1007/978-1-4899-2575-6_16. [DOI] [PubMed] [Google Scholar]