The Complement Inhibitor FUT-175 Suppresses T Cell Autoreactivity in Experimental Autoimmune Encephalomyelitis (original) (raw)
Abstract
Several recent studies have shown that interacting antigen presenting cells and/or T cells produced complement activation products C5a and C3a, are integrally involved in T-cell activation, and promote the generation of myelin oligodendrocyte glycoprotein (MOG35–55)-specific interferon-γ and interleukin-17-producing T cells in experimental autoimmune encephalomyelitis, a rodent model of multiple sclerosis. In this study, we tested whether FUT-175, a clinical pharmaceutical that has been shown to inhibit the formation of C3/C5 convertases, can attenuate myelin-specific T-cell responses, as well as disease severity in experimental autoimmune encephalomyelitis. In vitro, FUT-175 inhibited local C5a/C3a production by antigen presenting cell–T-cell complexes and attenuated MOG35–55-specific Th1 and Th17 responses with little nonspecific cytotoxicity. In vivo administration of FUT-175 delayed experimental autoimmune encephalomyelitis disease onset, lowered clinical scores, decreased central nervous system inflammation, and reduced demyelination. The FUT-175-treated mice exhibited decreased numbers of MOG35–55-specific interferon-γ- and interleukin-17-producing T cells. In addition, results from the FUT-175 treatment of naive recipients of adoptively transferred splenocytes from MOG35–55-immunized mice suggested that the effect of FUT-175 was on MOG-specific cellular responses and not on anti-MOG antibodies. These results argue that complement regulators, which inhibit C5a/C3a production, may have therapeutic efficacy in multiple sclerosis and in other clinical conditions in which T cells drive disease pathogenesis.
In multiple sclerosis (MS), autoreactive T cells attack the myelin sheaths of central nervous system (CNS) nerves. The resulting impairment in neural transmission eventuates in diverse neurological deficits.1 At present, the most widely used therapeutic agents consist of interferon (IFN)-β,2 glatiramer acetate,3 mitoxantrone,4 and natalizumab,5 all of which target T cell activity. Although these agents can slow disease progression, they only have partial effects.
Much of our knowledge about the immunopathogenesis of MS has come from studies of the experimental autoimmune encephalomyelitis (EAE)6 model in rodents. In the most commonly used protocol, C57BL/6 mice are immunized with the encephalitogenic 35–55 peptide of myelin oligodendrocytes glycoprotein (MOG35–55) in complete Freund’s adjuvant together with pertussis toxin. While CNS injury in EAE was thought to be due to myelin specific Th1 cells, which are the major source of IFNγ, IFNγ knockout mice are susceptible to EAE induction7 and systemic antibody depletion of IFNγ exaggerates disease severity.8 Recent studies have shown that both IFNγ-producing Th1 and interleukin (IL)-17 producing Th17 cells contribute to pathology in EAE9 and that Th17 cells may play a greater role than Th1 cells.10 This and other EAE models have not only shown that myelin injury in MS is due to T cell autoimmunity that spread across myelin components, but also have been valuable for testing therapeutic interventions.
Recent work by different groups11,12,13,14,15,16 have documented that complement locally produced by interacting antigen presenting cells (APCs) and/or T cells is integrally involved in T cell responses. These studies showed that early during APC-T cell interactions, both partners locally synthesize the alternative pathway components C3, factor B, and factor D together with C5, factors essential for C5a/C3a generation.13,16 Concurrent with this, both partners down-regulate expression levels of decay accelerating factor (or CD55), a cell surface C3/C5 convertase inhibitor that prevents C3 and C5 activation.17 The lifted restraint on junctional complement activation conferred by the decay-accelerating factor down-regulation allows the local generation of C5a and C3a by both partners. Interaction of these anaphylatoxins with up-regulated C3aR and C5aR on both partners confers G protein-coupled receptor signals that are necessary for both IL-12 and IL-23 production by APCs.16 The increased levels of locally produced C5a/C3a also enhance effector T cell expansion by limiting antigen-induced apoptosis.15 Studies in EAE16 have shown that this C5a/C3a-C5aR/C3aR G protein coupled receptor transduction plays a requisite role in the generation of MOG35–55 specific IFNγ and IL-17 producing T cells.
Nafamostat mesylate (FUT-175) is a small chemical developed in Japan in the early 1980s.18 FUT-175 is a potent serine protease inhibitor18 that has been shown to abolish the enzymatic activities of C1r, C1s, factor D, as well as the Bb component of the alternative pathway C3/C5 convertases (C3bBb/C3bBbC3b), and thereby prevents C3/C5 activation that gives rise to C5a/C3a. Because of its serine protease inhibitory activity and anti-coagulation activity, FUT-175 has been and continues to be used clinically both for treating acute pancreatitis19,20 and for preventing thrombosis in disseminated intravascular coagulation21 and during extracorporeal circulation with safe record.22,23
In this report, we examined whether, and if so how, administration of FUT-175 would impact T cell mediated autoimmune responses in EAE. We found that FUT-175 inhibits antigen specific Th1/Th17 responses while decreases levels of C5a/C3a locally produced by interacting APCs and T cells. Its i.p. administration reduces myelin-specific T cell responses and protects animals from CNS inflammation and demyelination.
Materials and Methods
Mice and Reagents
Six- to eight-week-old female C57BL/B6 mice and OT-II mice were purchased from the Jackson Laboratory (Bar Harbor, ME). MOG35–55 peptide was purchased from Anaspec, Inc (San Jose, CA). All antibodies used were purchased from BD Biosciences (San Jose, CA).
Measurements of Local C5a/C3a Production during APC-T Cell Interactions
The effect of FUT-175 on local C5a and C3a productions by APC-T cell partners during antigen-specific T cell activation were measured by semiquantitative Western blots using rat anti-mouse C5a and rat anti-mouse C3a mAbs, respectively. In brief, 3 × 105 WT C57BL/6 mouse bone marrow-derived dendritic cells were mixed with 5 × 105 purified OT-II mouse CD4+ T cells together with 3 μg/ml OVA323–339 peptide in each well of a 48-well plate. Different concentrations of FUT-175 were added into each well and the plate was incubated at 37°C, 5% CO2; 36 hours later, the supernatants were concentrated using micro ultracentrifuge devices (MW cut off: 3000 Da, Millipore) and separated by a 12.5% SDS-polyacrylamide gel electrophoresis. The locally generated C5a and C3a were detected using 5 μg/ml rat anti-mouse C5a mAb or rat anti-mouse C3a mAb. The amounts of C5a/C3a in the supernatants were quantified using software package MetaMorph 7.5 (Molecular Devices, CA).
FUT-175 in Vitro T Cell Response Inhibition and Cytotoxicity Assays
A total of 6 × 105 spleen cells from MOG35–55 immunized mice were incubated with increasing concentrations of FUT-175 at 37°C, 5% CO2 for 24 hours together with 5 μg/ml MOG35–55 peptide. The minimal concentrations of FUT-175 for inhibiting >50% antigen specific T cell responses were determined by IFNγ/IL-17 ELISPOT assays and cytotoxic effects of FUT-175 at different concentrations were measured by the CellTiter-Blue Cell Viability Assay kit (Promega, Madison, WI) following manufacturer’s protocols.
Induction and Evaluation of EAE
EAE was induced in female C57BL/6 mice (6 to 8 weeks old) by subcutaneous immunization with 200 μg of MOG35–55 peptide emulsified in complete Freund’s adjuvant containing 4 mg/ml heat-killed Mycobacterium tuberculosis H37RA (Sigma-Aldrich, St. Louis, MO). Four hundred nanograms pertussis toxin (List Biolabs, Campbell, CA) was administrated intraperitoneally in 0.1 ml of PBS on days 0 and 2. Clinical severity was assessed daily with a 0 to 5 scoring system (0, no signs; 1, flaccid tail; 2, impaired righting reflex and/or gait; 3, partial hind limb paralysis; 4, total hind limb paralysis; 5, moribund or dead). All mice were housed in a specific pathogen-free barrier facility and screened regularly for pathogens. All studies and procedures were approved by the Institutional Animal Care and Use Committee of Case Western Reserve University.
IFNγ and IL-17 ELISPOT Assays
Mouse spleens were harvested and after red blood cell lysis, single cell suspensions were prepared, and 6 × 105 splenocytes were aliquoted into each well of an ELISPOT plate coated with IFNγ or IL-17 capturing mAb and incubated with different concentrations of MOG35–55 peptide for 24 hours. After developing, IFNγ or IL-17 producing T cells were quantified by ELISPOT assay using the protocol suggested by the manufacturer (Cellular technology Ltd, Cleveland, OH).
In Vivo Treatment with FUT-175
One day after MOG35–55 immunization, mice were randomly divided into two groups. The experimental group was injected i.p. with 10 mg/kg FUT-175 every day, and the control group injected with equal volume of PBS. The treatments continued until the day that mice were sacrificed.
Activated Partial Thromboplastin Time Assay
After systemic treatment by i.p. injection of FUT-175 at 10 mg/kg, mouse blood was withdrawn by cardiac puncture using syringes rinsed with and immediately mixed with 5% sodium citrate so that the final concentration of sodium citrate was 0.4%. The citrate-anticoagulated mouse plasma was further diluted with saline (1:1) and activated partial thromboplastin (aPPT) was measured on an automatic coagulation analyzer by the Clinical Hematology Laboratory of the University Hospital Case Medical Center (Cleveland, OH).
C3b Uptake Assay
Blood samples were acquired from tail veins of PBS and FUT-175 treated mice, then sera were collected after clotting and centrifugation. Conventional C3b uptake assays were performed using antibody sensitized sheep erythrocytes (EshA) to assess the classical pathway and Zymosan (Sigma, MO) to assess the alternative pathway complement activation as described.24
Adoptive Transfer of MOG Reactive T cells and FUT-175 treatment
Naïve C57BL/6 mice were immunized with MOG35–55 peptide in complete Freund’s adjuvant in conjunction of pertussis toxin as described before. Ten days later, spleen and lymph nodes cells were harvested and cultured in RPMI 1640 containing 10% fetal bovine serum together with 25 μg/ml MOG35–55, 10 ng/ml of IL-12 and 10 ng/ml IL-23 for 3 days. Following this, 2 × 107 cells were injected i.v. via tail vein into each naïve C57BL/6 recipients. The recipient mice then were randomly divided into two groups. One group was injected with 10 mg/kg FUT-175 and the other with PBS. Clinical scores and IFNγ/IL-17 producing T cells were measured using the methods described above.
Histology and Histochemical Staining of Spinal Cord
At time of sacrifice, spinal cords were removed and fixed in 4% paraformaldehyde for 24 hours, then embedded in paraffin. Sections were cut at 10 μm on a microtome and stained by H&E to reveal CNS inflammatory infiltrates and Luxol Fast Blue to reveal demyelinated areas. The infiltrated cell numbers and Luxol Fast Blue stained myelin in representative areas were quantified using software package MetaMorph 7.5 (Molecular Devices, Downington, PA).
Statistical Analysis
All experiments were repeated at least twice. To determine whether groups were statistically different, results were compared using the Student’s _t_-test. A P value <0.05 was considered significant.
Results
FUT-175 Suppresses Local C5a/C3a Generation during Antigen-Specific APC-T Cell Interactions
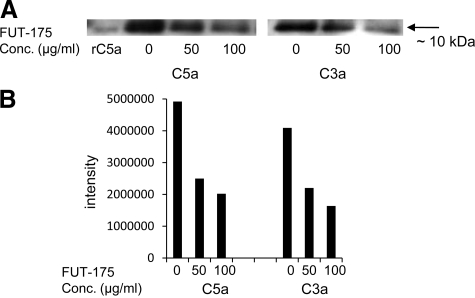
Because of its known complement inhibitory activity, we hypothesized that FUT-175 can inhibit local C5a/C3a generation during antigen specific APC-T cell interactions. To test this, we mixed wild-type bone marrow-derived dendritic cells and OT-II CD4+ T cells together with OVA323–339 peptide. After 36 hours of incubation, we assayed locally generated C5a/C3a in the concentrated culture supernatants by semiquantitative Western blot. As shown in Figure 1, A–B, FUT-175 decreased both local C5a and C3a levels in a dose-dependant manner. At a concentration of 50 μg/ml, it decreases levels of local generated C5a and C3a by >60%.
Figure 1.
FUT-175 inhibits local C5a and C3a productions by APC-T cell partners during antigen specific T cell activation. A: C5a and C3a detections in concentrated culture supernatants from co-cultured bone marrow-derived dendritic cells, OT-II CD4+ T cells, 3 μg/ml OVA323–339 peptide and different concentrations of FUT-175. Recombinant mouse C5a was used as positive control (rC5a). B: C5a and C3a detected in A were semiquantified using the software package MetaMorph 7.5 (Molecular Devices, Downington, PA).
FUT-175 Inhibits MOG35–55-Specific Th1 and Th17 Responses in Vitro
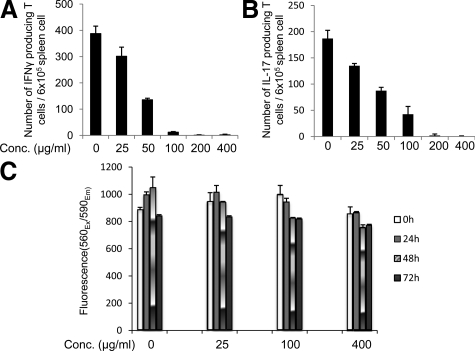
We next tested whether FUT-175 can inhibit the generation of MOG35–55-specific IFNγ and IL-17 producing T cells in vitro by using splenocytes from MOG35–55-immunized mice. To do this, we added increasing concentrations of FUT-175 into microtiter wells containing 6 × 105 spleen cells and 5 μg/ml MOG35–55 peptide and quantitated IFNγ- or IL-17-producing cells by ELISPOT assays. These studies showed that the added FUT-175 reduced the numbers of IFNγ and IL-17 producing T cells in a dose dependent manner (Figure 2, A–B). FUT-175 inhibited the MOG35–55-specific Th1 responses by >70% at a concentration of 50 μg/ml and by >95% at a concentration of 100 μg/ml (Figure 2A) and it inhibited the MOG35–55- specific Th17 response by >50% at a concentration of 50 μg/ml and by >75% at a concentration of 100 μg/ml (Figure 2B). Both MOG35–55-specific Th1 and Th17 responses were inhibited by >99% at a concentration of 200 μg/ml. A cell cytotoxicity assay using the CellTiter-Blue kit (Promega, MI) verified that FUT-175 has little nonspecific cytotoxicity at a concentration as high as 400 μg/ml even after 72 hours of incubation (Figure 2C).
Figure 2.
FUT-175 inhibits MOG35–55-specific T cell responses in vitro: 6 × 105 spleen cells from MOG35–55 immunized mice were aliquoted into each well of a 96 well ELISPOT plate coated with IFNγ- or IL-17-capturing mAb. Then 5 μg/ml MOG35–55 peptide, together with increasing concentrations of FUT-175, was added into the plate and the mixtures were incubated for 24 hours at 37°C, 5% CO2. After developing, numbers of IFNγ (A) and IL-17 (B) producing cells were counted using an ImmunoSPOT Imaging system (Cleveland, OH). C : FUT-175 has little nonspecific cytotoxicity after 72 hours incubation at a concentration as high as 400 μg/ml: 2 × 105 splenocytes from mice with EAE were incubated with 5 μg/ml MOG35–55 peptide and different concentrations of FUT-175 (0, 25,100, 400 μg/ml) at 37°C, 5% CO2. At different time points (0, 24, 48, 72 hours), cell viabilities were assessed by adding 20 μl of indicator dye resazurin from the CellTiter-Blue Cell Viability Assay kit (Promega, MI) and incubating for another 1 hour. After this, fluorescence 560Ex/590Em were measured using a fluorescence plate reader (Molecular Devices, Downington, PA).
FUT-175 Treatment Protects Mice from EAE
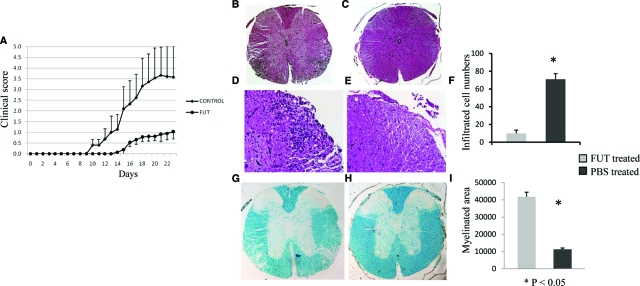
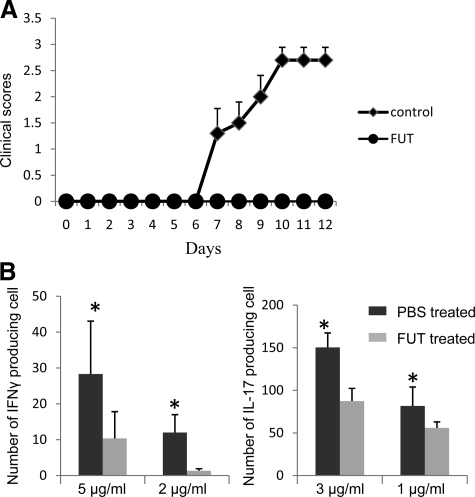
Having established that FUT-175 decreases local C5a/C3a generation during cognate APC-T cell interactions and inhibits MOG35–55 specific Th1/Th17 responses in vitro, we next tested the effects of FUT-175 administration in vivo in EAE. Based on the in vitro data, we estimated that a dosage of 10 mg/kg (Dr. Jeffery Blumer, personal communication) would provide an in vivo drug concentration of ∼100 to 200 μg/ml. We treated wild-type mice (n = 18) with this dosage of FUT-175 starting 1 day after MOG35–55 immunization. As a control, we treated a second set of mice (n = 20) with PBS. The results showed that compared with the PBS-treated control group, the mouse group treated with FUT-175 exhibited delayed disease onset (day15 vs.10) and exhibited much lower clinical scores (1.0 vs. 3.5 at day 24) (Figure 3A). Consistent with this, histopathological examinations showed that FUT-175 suppressed inflammatory cell infiltration in spinal cords (Figure 3, B–E) and reduced demyelination (Figure 3, G and H). Digital imaging analyses documented that while the average number of infiltrated cells in a 10,000 μM2 dorsal area were 71 ± 6 in PBS-treated mice only 10 ± 4 were present in FUT-175 treated mice (P < 0.05) (Figure 3F). Within a 50,000 μM2 area in the white matter, the average Luxol Fast Blue-stained myelin area was 11,246 ± 2689 μM2 in PBS-treated mice compared with 41,783 ± 790 μM2 in FUT-175 treated mice (Figure 3I).
Figure 3.
FUT-175 treatment protects mice from EAE. A : Clinical scores of mice receiving FUT-175 or PBS treatments one day after immunization. Mice were treated with 10 mg/kg FUT-175 or equal volume of PBS i.p. every day 1 day after MOG35–55 immunization. Clinical scores were monitored daily and average scores are shown. (n = 18, P < 0.05) For histological analysis, cervical portion of spinal cords were collected from PBS treated (B,×40 and D, ×200) and FUT-175 treated mice (C, ×40 and E, ×200) 25 days after immunization and processed for H&E staining or Luxol Fast Blue staining (G, H). The average infiltrated cell numbers in a 10,000 μM2 dorsal area were analyzed by the software package MetaMorph 7.5 (Molecular Devices, Downington, PA) and compared (F). The average myelin areas in a 50,000 μmol/L2 white matter area were quantified using the same software package and compared between the PBS- and FUT-175-treated mice (I). *P < 0.05.
FUT-175 Treated Mice Exhibit Dampened Th1/Th17 Responses Against MOG35–55
To determine whether the decreased disease severity in FUT-175 treated mice reflect reduced numbers of MOG35–55 specific IFNγ- and IL-17-producing T cells, we quantitated IFNγ and IL-17 producing T cells in spleens from FUT-175 and PBS treated mice by ELISPOT assays. These assays (Figure 4) showed significantly less IFNγ (96 ± 68 vs. 203 ± 90 spots/105 spleen cells at 5 μg/ml MOG35–55) and IL-17 (89 ± 18 vs. 150 ± 20 spots/105 spleen cells at 3 μg/ml MOG35–55)-producing cells in FUT-175-treated mice than PBS-treated controls (P < 0.05).
Figure 4.
Mice treated with FUT-175 exhibit suppressed MOG35–55-specific T cell responses in vivo. FUT-175-treated mice (n = 18) and control mice (n = 20) were sacrificed 25 days after immunization and T cell responses against MOG35–55 in each group were assayed by IFNγ and IL-17 ELISPOT assays using 6 × 105 spleen cells incubated with different concentrations of MOG35–55 at 37°C and 5% CO2 for 24 hours (*P < 0.05).
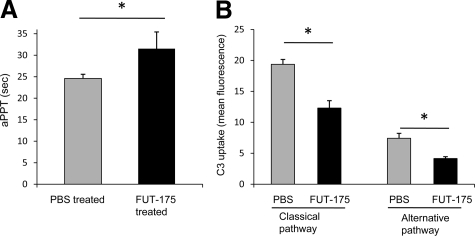
FUT-175-Treated Mice Exhibit Slowed Coagulation and Reduced Systemic Complement Activation
To assess the effect of systemic administration of FUT-175 on coagulation, we measured aPPT of sodium citrate-treated plasma samples from mice received 10 mg/kg FUT-175 or the same volume of saline. These assays (Figure 5A) showed that aPPT was prolonged from 25 ± 1 second in placebo treated mice to 32 ± 4 seconds in systemic FUT-175 treated mice (n = 10 in each group). To assess its effect on systemic complement activation, because sodium citrate interferes with complement activation, we repeated the above described in vivo treatment experiments and acquired blood from mouse tail veins to collect sera for C3b uptake assay. These assays (Figure 5B) showed that as expected, FUT-175 administration inhibited both the classical and the alternative pathways systemic complement activation.
Figure 5.
Effects of FUT-175 treatments on coagulation and systemic complement activation. A : FUT-175 treatments prolonged coagulation time. aPPT were analyzed on an automatic coagulation analyzer using sodium citrate treated plasma samples collected from PBS and 10 mg/kg FUT-175-treated mice (n = 10). B : FUT-175 treatments inhibited systemic complement activation. Deposited C3b levels on EshA (classical pathway) or Zymosan (alternative pathway) were quantitated by flow cytometry using serum samples collected from PBS- and 10 mg/kg FUT-175-treated mice *P < 0.05.
FUT-175 Ameliorates EAE by Inhibiting Pathogenic Cellular Responses
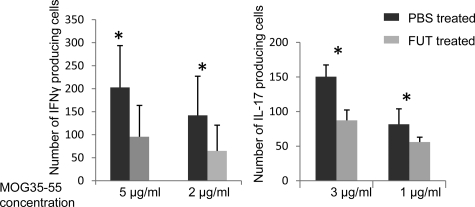
Previous studies25,26 found that C5b-9 membrane attack complexes derived from anti-MOG-antibody activated systemic complement activation can contribute to EAE pathogenesis. In view of these results, we repeated the in vivo treatment studies with FUT-175 or PBS using naïve C57BL/6 recipients of adoptively transferred _in vitro_-amplified spleen cells from donor mice immunized with MOG35–55. As shown in Figure 6, A and B, as compared with the PBS treatment, FUT-175 treatment attenuated EAE severity and suppressed the generation of MOG35–55-specific IFNγ- and IL-17-producing T cells, localizing the FUT-175 effect to MOG35–55-specific cellular responses rather than anti-MOG antibody-initiated systemic complement activation.
Figure 6.
FUT-175 treatments suppress EAE induced by adoptive transferred MOG35–55-reactive T cells in naïve recipient mice. A : After i.v. injection of the _in vitro_-amplified MOG35–55 reactive T cells, naïve recipient mice were randomly divided into two groups (n = 5 in each group). One group received FUT-175 treatments every day (10 mg/kg) while the other group received same volume of PBS. Disease severities were scored daily and average scores are presented. B : Twelve days after adoptive transfer, mice were sacrificed and T cell responses against MOG35–55 were measured and compared between the groups by IFNγ and IL-17 ELISPOT assays. The experiments were repeated with similar results (not shown). *P < 0.05.
Discussion
In this study, we have found that FUT-175, a pharmaceutical that previously was shown to inhibit C1r, C1s, factor D, and the enzymatic Bb component of factor B, suppresses both MOG35–55-specific Th1 and Th17 autoimmunity that leads to EAE. In vitro, FUT-175 treatment inhibited local C5a/C3a production during antigen specific APC-T cell interactions and suppressed MOG35–55-specific Th1/Th17 cell responses in a dose-dependant manner. Cell cytotoxicity assays verified that the T cell inhibitory effect of FUT-175 was not due to nonspecific cytotoxicity. In vivo, FUT-175 treatment delayed EAE disease onset and lowered clinical scores. Histological analysis documented markedly decreased inflammatory cell infiltration and demyelination in spinal cords. ELISPOT assays showed lower numbers of MOG35–55-specific IFNγ- and IL-17-producing T cells in FUT-175 treated mice. Adoptive transfer experiments suggested that FUT-175’s ameliorating effect was on cellular responses and not autoantibody-initiated systemic complement activation.
Although as early as in the 1970s, studies27 showed that depleting complement in guinea pigs by cobra venom factor delayed EAE disease onset and decreased clinical scores compared with the controls, the mechanism was not clarified. Similarly, more recent studies showed that EAE severity in rats was reduced by administration of soluble complement C3 receptor 1, a human C3/C5 convertase inhibitor28 and in mice expressing a transgene encoding soluble complement receptor related protein Y,29 a murine C3/C5 convertase inhibitor. The attenuating effects on EAE severity in both of these studies were attributed to inhibition of membrane attack complex formation. We16 and others13 reported that mice deficient in Daf1, the mouse homologue of human decay accelerating factor, a cell surface complement inhibitor, exhibited heightened MOG35–55-specific Th1/Th17 responses and had markedly more severe EAE. Further studies14,16 indicated that the more severe CNS damage in _Daf1_−/− mice was due to increased generation of C5a/C3a during the induction of MOG35–55 specific T cell responses. These increased C5a/C3a, on interacting with their respective receptors C5aR/C3aR, stimulated more IL-12 and IL-23 production, which in turn promote the pathological Th1/Th17 responses.16 Moreover, the increased C5a/C3a, through up-regulating Bcl-2 expression and inhibiting Fas expression, enhanced differentiated effector T cell expansion by limiting antigen induced apoptosis.15 The new insight revealed by different groups that local complement production by interacting APCs and T cells plays an integral role in effector T cell stimulation and survival11,12,14,16 accounts for the above and other previous findings linking complement to T cell activation,30,31,32 suggesting that complement inhibitors that limit local C5a/C3a generations could be used to inhibit pathological T cell responses in T cell-mediated diseases such as MS.
FUT-175 has been used clinically in Japan and other Asian countries without major side effects. For treating acute pancreatitis, FUT-175 is generally given to patients at doses of 150 to 200 mg either once or twice a day.19,20 For preventing coagulation during extracorporeal circulation, it is given at 20 to 50 mg/hr. Our in vitro studies showed that 200 μg/ml of FUT-175 inhibits the MOG35–55-specific Th1 and Th17 responses by >95% with little nonspecific cytotoxicity (Figure 1) and our in vivo studies showed that FUT-175 protected mice from CNS injury in EAE through suppressing MOG35–55-specific autoreactivity. Although we cannot exclude that other mechanism(s) may also be involved in FUT-175’s effect in ameliorating EAE, because of its wide serine protease inhibitory activity, our above data together with previously published results13,15,16 argue that FUT-175 inhibits MOG-specific Th1/Th17 responses in EAE at least partially by inhibiting local C5a/C3a generation. While the use of FUT-175 itself might not be appropriate for long-term treatment of patients with MS because of its anti-coagulation activity as showed in our aPPT assays, our findings argue that agents that inhibit C5a/C3a generation can effectively attenuate T cell-mediated diseases such as MS. While inhibitors that block the complement cascade at different steps have been explored previously to treat diseases mediated by systemic complement33 and one of them, SOLIRIS (eculizumab), an anti-C5 mAb has been approved for treatment of paroxysmal nocturnal hemoglobinuria,34 the data in this study provide further insight into how to most effectively apply these complement inhibitors for treating T cell-mediated diseases.
Acknowledgments
We thank Dr. Edward Medof for discussion. We are grateful for Dr. Howard Meyerson, Director of Hematopathology, University Hospital Case Medical Center for help on the aPPT assays. We also thank Dr. Scott Howell at the Vision Science Research Center (VSRC) imaging core for digital imaging analysis, and Catherine Doller at the histology core for excellent histology services.
Footnotes
Address reprint requests to Feng Lin, Ph.D., Assistant Professor, Institute of Pathology, Case Western Reserve University School of Medicine, 2085 Adelbert Road, Room 306, Cleveland, OH 44106. E-mail: feng.lin@case.edu.
Supported by National Institute of Health grant NS052471 (F.L.) and National Multiple Sclerosis Society grant RG3664 (F.L.). Q.L. and H.B. were supported in part by Natural Science Foundation of China grant 30671988. The VSRC is supported by NIH grant EY11373.
References
- Sospedra M, Martin R. Immunology of multiple sclerosis. Annu Rev Immunol. 2005;23:683–747. doi: 10.1146/annurev.immunol.23.021704.115707. [DOI] [PubMed] [Google Scholar]
- Guo B, Chang EY, Cheng G. The type I IFN induction pathway constrains Th17-mediated autoimmune inflammation in mice. J Clin Invest. 2008;118:1680–1690. doi: 10.1172/JCI33342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wolinsky JS. The use of glatiramer acetate in the treatment of multiple sclerosis. Adv Neurol. 2006;98:273–292. [PubMed] [Google Scholar]
- Morrissey SP, Le Page E, Edan G. Mitoxantrone in the treatment of multiple sclerosis. Int MSJ. 2005;12:74–87. [PubMed] [Google Scholar]
- Chaudhuri A. Lessons for clinical trials from natalizumab in multiple sclerosis. BMJ. 2006;332:416–419. doi: 10.1136/bmj.332.7538.416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Owens T. Animal models for multiple sclerosis. Adv Neurol. 2006;98:77–89. [PubMed] [Google Scholar]
- Ferber IA, Brocke S, Taylor-Edwards C, Ridgway W, Dinisco C, Steinman L, Dalton D, Fathman CG. Mice with a disrupted IFN-gamma gene are susceptible to the induction of experimental autoimmune encephalomyelitis (EAE). J Immunol. 1996;156:5–7. [PubMed] [Google Scholar]
- Billiau A, Heremans H, Vandekerckhove F, Dijkmans R, Sobis H, Meulepas E, Carton H. Enhancement of experimental allergic encephalomyelitis in mice by antibodies against IFN-gamma. J Immunol. 1988;140:1506–1510. [PubMed] [Google Scholar]
- Kroenke MA, Carlson TJ, Andjelkovic AV, Segal BM. IL-12- and IL-23-modulated T cells induce distinct types of EAE based on histology, CNS chemokine profile, and response to cytokine inhibition. J Exp Med. 2008;205:1535–1541. doi: 10.1084/jem.20080159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Komiyama Y, Nakae S, Matsuki T, Nambu A, Ishigame H, Kakuta S, Sudo K, Iwakura Y. IL-17 plays an important role in the development of experimental autoimmune encephalomyelitis. J Immunol. 2006;177:566–573. doi: 10.4049/jimmunol.177.1.566. [DOI] [PubMed] [Google Scholar]
- Zhou W, Patel H, Li K, Peng Q, Villiers MB, Sacks SH. Macrophages from C3-deficient mice have impaired potency to stimulate alloreactive T cells. Blood. 2006;107:2461–2469. doi: 10.1182/blood-2005-08-3144. [DOI] [PubMed] [Google Scholar]
- Peng Q, Li K, Anderson K, Farrar CA, Lu B, Smith RA, Sacks SH, Zhou W. Local production and activation of complement up-regulates the allostimulatory function of dendritic cells through C3a-C3aR interaction. Blood. 2008;111:2452–2461. doi: 10.1182/blood-2007-06-095018. [DOI] [PubMed] [Google Scholar]
- Heeger PS, Lalli PN, Lin F, Valujskikh A, Liu J, Muqim N, Xu Y, Medof ME. Decay-accelerating factor modulates induction of T cell immunity. J Exp Med. 2005;201:1523–1530. doi: 10.1084/jem.20041967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lalli PN, Strainic MG, Lin F, Medof ME, Heeger PS. Decay accelerating factor can control T cell differentiation into IFN-gamma-producing effector cells via regulating local C5a-induced IL-12 production. J Immunol. 2007;179:5793–5802. doi: 10.4049/jimmunol.179.9.5793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lalli PN, Strainic MG, Yang M, Lin F, Medof ME, Heeger PS. Locally produced C5a binds to T cell-expressed C5aR to enhance effector T-cell expansion by limiting antigen-induced apoptosis. Blood. 2008;112:1759–1766. doi: 10.1182/blood-2008-04-151068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strainic MG, Liu J, Huang D, An F, Lalli PN, Muqim N, Shapiro VS, Dubyak GR, Heeger PS, Medof ME. Locally produced complement fragments C5a and C3a provide both costimulatory and survival signals to naive CD4+ T cells. Immunity. 2008;28:425–435. doi: 10.1016/j.immuni.2008.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Medof ME, Kinoshita T, Nussenzweig V. Inhibition of complement activation on the surface of cells after incorporation of decay-accelerating factor (DAF) into their membranes. J Exp Med. 1984;160:1558–1578. doi: 10.1084/jem.160.5.1558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fujii S, Hitomi Y. New synthetic inhibitors of C1r. C1 esterase, thrombin, plasmin, kallikrein, and trypsin. Biochim Biophys Acta. 1981;661:342–345. doi: 10.1016/0005-2744(81)90023-1. [DOI] [PubMed] [Google Scholar]
- Nakase H, Itani T, Mimura J, Kawasaki T, Komori H, Okazaki K, Chiba T. Successful treatment of severe acute pancreatitis by the combination therapy of continuous arterial infusion of a protease inhibitor and continuous hemofiltration. J Gastroenterol Hepatol. 2001;16:944–945. [PubMed] [Google Scholar]
- Takeda K, Matsuno S, Sunamura M, Kakugawa Y. Continuous regional arterial infusion of protease inhibitor and antibiotics in acute necrotizing pancreatitis. Am J Surg. 1996;171:394–398. doi: 10.1016/S0002-9610(97)89617-1. [DOI] [PubMed] [Google Scholar]
- Kobayashi T, Terao T, Maki M, Ikenoue T. Diagnosis and management of acute obstetrical DIC. Semin Thromb Hemost. 2001;27:161–167. doi: 10.1055/s-2001-14076. [DOI] [PubMed] [Google Scholar]
- Takahashi H, Muto S, Nakazawa E, Yanagiba S, Masunaga Y, Miyata Y, Tamba K, Kusano E, Matsuo M, Matsuo T, Asano Y. Combined treatment with nafamostat mesilate and aspirin prevents heparin-induced thrombocytopenia in a hemodialysis patient. Clin Nephrol. 2003;59:458–462. doi: 10.5414/cnp59458. [DOI] [PubMed] [Google Scholar]
- Nagaya M, Futamura M, Kato J, Niimi N, Fukuta S. Application of a new anticoagulant (Nafamostat Mesilate) to control hemorrhagic complications during extracorporeal membrane oxygenation–a preliminary report. J Pediatr Surg. 1997;32:531–535. doi: 10.1016/s0022-3468(97)90701-6. [DOI] [PubMed] [Google Scholar]
- Lin F, Immormino RM, Shoham M, Medof ME. Bulk production and functional analyses of mouse CD55’s native and deglycosylated active domains. Arch Biochem Biophys. 2001;393:67–72. doi: 10.1006/abbi.2001.2488. [DOI] [PubMed] [Google Scholar]
- Linington C, Lassmann H, Morgan BP, Compston DA. Immunohistochemical localisation of terminal complement component C9 in experimental allergic encephalomyelitis. Acta Neuropathol. 1989;79:78–85. doi: 10.1007/BF00308961. [DOI] [PubMed] [Google Scholar]
- Tran GT, Hodgkinson SJ, Carter N, Killingsworth M, Spicer ST, Hall BM. Attenuation of experimental allergic encephalomyelitis in complement component 6-deficient rats is associated with reduced complement C9 deposition. P-selectin expression, and cellular infiltrate in spinal cords. J Immunol. 2002;168:4293–4300. doi: 10.4049/jimmunol.168.9.4293. [DOI] [PubMed] [Google Scholar]
- Morariu MA, Dalmasso AP. Experimental allergic encephalomyelitis in cobra venom factor-treated and C4-deficient guinea pigs. Ann Neurol. 1978;4:427–430. doi: 10.1002/ana.410040507. [DOI] [PubMed] [Google Scholar]
- Piddlesden SJ, Storch MK, Hibbs M, Freeman AM, Lassmann H, Morgan BP. Soluble recombinant complement receptor 1 inhibits inflammation and demyelination in antibody-mediated demyelinating experimental allergic encephalomyelitis. J Immunol. 1994;152:5477–5484. [PubMed] [Google Scholar]
- Davoust N, Nataf S, Reiman R, Holers MV, Campbell IL, Barnum SR. Central nervous system-targeted expression of the complement inhibitor sCrry prevents experimental allergic encephalomyelitis. J Immunol. 1999;163:6551–6556. [PubMed] [Google Scholar]
- Hopken UE, Lu B, Gerard NP, Gerard C. The C5a chemoattractant receptor mediates mucosal defense to infection. Nature. 1996;383:86–89. doi: 10.1038/383086a0. [DOI] [PubMed] [Google Scholar]
- Szalai AJ, Hu X, Adams JE, Barnum SR. Complement in experimental autoimmune encephalomyelitis revisited: C3 is required for development of maximal disease. Mol Immunol. 2007;44:3132–3136. doi: 10.1016/j.molimm.2007.02.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marsh JE, Farmer CK, Jurcevic S, Wang Y, Carroll MC, Sacks SH. The allogeneic T and B cell response is strongly dependent on complement components C3 and C4. Transplantation. 2001;72:1310–1318. doi: 10.1097/00007890-200110150-00022. [DOI] [PubMed] [Google Scholar]
- Kirschfink M. Targeting complement in therapy. Immunol Rev. 2001;180:177–189. doi: 10.1034/j.1600-065x.2001.1800116.x. [DOI] [PubMed] [Google Scholar]
- Hillmen P, Young NS, Schubert J, Brodsky RA, Socie G, Muus P, Roth A, Szer J, Elebute MO, Nakamura R, Browne P, Risitano AM, Hill A, Schrezenmeier H, Fu CL, Maciejewski J, Rollins SA, Mojcik CF, Rother RP, Luzzatto L. The complement inhibitor eculizumab in paroxysmal nocturnal hemoglobinuria. N Engl J Med. 2006;355:1233–1243. doi: 10.1056/NEJMoa061648. [DOI] [PubMed] [Google Scholar]