High Throughput Screening of Small Molecule Libraries for Modifiers of Radiation Responses (original) (raw)
. Author manuscript; available in PMC: 2012 Aug 1.
Published in final edited form as: Int J Radiat Biol. 2011 Mar 14;87(8):839–845. doi: 10.3109/09553002.2011.560994
Abstract
Purpose
An unbiased approach of drug discovery through high-throughput screening (HTS) of libraries of chemically defined and bioactive small molecule compounds was used to identify modulators of radiation injury with an emphasis on radioprotectors and mitigators rather than radiosensitizers. Assay system endpoints included radiation-induced genotoxicity and DNA damage in yeast and apoptosis in murine lymphocytes. Large-scale data mining of chemically diverse libraries identified agents that were effective with all endpoints. HTS of bioactive compound libraries against murine lymphocytes profiled tetracycline and fluoroquinolone antibiotics and cyclopiazonic acid as having activity, and structure-activity analysis showed a common pharmacophore. Purine nucleosides, the interferon inducer tilorone, and linoleic acid were also identified as potential mitigators of radiation damage that often were also radioprotective. Many of these compounds enhance DNA repair, have anti-inflammatory activity, and stimulate hematopoiesis. Selected compounds within these initial verified hits from both types of libraries identified potent mitigators of lethal whole body irradiation (WBI) in mice.
Conclusion
In spite of the fact that in vitro HTS has limitations and is unable to fully recapitulate all aspects of the complex in vivo acute radiation response, it identified several classes of molecules that had activity as radioprotectors and radiomitigators of the hematopoietic system in vivo. In the future, addition of 3 dimensional (3D) or stem cell cultures or pathway analysis, may improve the power of HTS, but our findings indicate that common, evolutionary conserved, canonical pathways can be identified that could be exploited to mitigate radiation-induced defects.
Keywords: High throughput screening, radioprotector, mitigator, antibiotics
Introduction
High-throughput screening (HTS) platforms can be used in an unbiased way to screen large libraries of small molecule compounds for agents that target specific molecular interactions or pathways. The resulting data can be mined to discover therapeutically active compounds and to generate hypotheses as to active moieties and mechanisms of action. Most often HTS screens use chemical or genetic (small interfering RNA) probes of cell processes in vitro with a spectrophotometric readout, but recent improvements in data acquisition times are allowing at least moderate throughput screening using automated imaging microscopy of multidimensional biological samples, including 3 dimensional (3D) cultures, stem cell spheroids, and zebrafish embryos that expands their potential and relevance to the in vivo situation (Pardo-Martin et al. 2010).
HTS may be used in radiation research to screen for radiosensitizers (Lally et al. 2007), protectors or mitigators. Readouts may be phosphorylation of histone protein H2AX (γ-H2AX) as an index of DNA repair, radiation-induced redistribution of proteins using fluorescent antibodies, radiation-induced signal transduction using reporter gene-luciferase assays, or radiation-induced cell death. The keys to success for HTS approaches are high quality primary data, potent algorithms for data analyses, and industrial strength resources for data storage and mining with chemo- and bio-informatic capabilities.
For small molecule chemical screening, basically two types of libraries are generally employed. One has known biological targets, such as experimental or Food and Drug Administration (FDA) approved drugs, lipids, ion channel blockers, and natural products [e.g. Prestwick (Washington DC, USA), Biomol (Enzo Life Sciences International, Inc., Plymouth Meeting, PA, USA), and Spectrum (Microsource, Gaylordsville, CT, USA) libraries]. These have the advantage that they inform on classes of bioactive agents with common mechanisms and that a considerable amount of information on them is already available. If the agent does not have FDA approval, a known or suspected mechanism is useful for successful development, as is pharmacokinetic, pharmacodynamic and toxicity information.
The other broad group contains synthesized subsets of chemicals distilled from very large libraries of compounds by 3D pharmacophore diversity analysis and selected for breadth of coverage or targeted scaffolds. Some of these have a broad range of diverse pharmacophores, which was defined by Paul Ehrlich in 1909 as “a molecular framework that carries the essential features responsible for a drug’s biological activity” (Ehrlich 1909). Others are enriched in targeted scaffolds directed to generic structures like kinases, proteases, G-protein coupled receptors, or ion channels. Such chemically derived compounds might be expected to uncover core structures that might be pharmaceutically refined into novel lead compounds with further improved efficacy. Although the strategies differ, the ultimate aim of both is to inform on relationships between chemical structures and biological pathways and to identify agents that might be functionally or clinically useful, for example to modify radiation-induced damage.
Choice of Assay Systems
We have used as targets for HTS primarily yeast deletion (DEL) assays for radiation-induced DNA deletion and intrachromosomal recombination in Saccharomyces cerevisiae and murine T lymphocyte apoptosis with viability being measured at 24 hrs by luminescence-based assay of adenosine triphosphate (ATP) concentration (ATPLite, Perkin-Elmer, Waltham, MA, USA) (Kim et al. 2009) to screen for radiation protectors and mitigators. The discriminatory power of the assays was tested in pilot experiments using positive and negative controls to establish the Z’ values (a measure of statistical effect size) as being >0.5 (Zhang et al. 1999).
The HTS employing murine T lymphocytes used radiation-induced apoptosis as an endpoint. There are limits on the time-course over which HTS experiments such as these can be performed and in particular the interval between drug and radiation exposure, which in our case was either 3 hrs before or 1 hr after 2 Gy irradiation. In addition, for practical reasons only one concentration of drug, 10 µM, was used. “Hits” were verified by measuring EC50 (50% effective concentration) values and to determine the size of the “therapeutic window”. In spite of these limitations, HTS data were useful predictors of wider efficacy.
“Hits” were generally identified in a primary screen as > 3 coefficients of variation from the mean. Inevitably, such arbitrary “cherry-picking” of “hits” carries a risk of “throwing the baby out with the bathwater.” Data mining was therefore used to compare hits structurally and functionally with non-hits and to rescue some of the “false negatives.” This was aided by use of the industrial strength database for analysis and mining and a full suite of chemical and biological informatic tools provided by Collaborative Drug Discovery (CDD, San Francisco, CA, USA). These platforms provide not only large-scale chemical and biological data storage but help identify lead candidates by structure activity relationships (SAR) analysis and by providing Lipinski coefficients for their drug like character (molecular weight ≤ ~500 g/mol, log P < 5, no more than 5 hydrogen bond donors, and no more than 10 hydrogen bond acceptors). Also, being able to compare efficacy for a drug across various experimental systems using such databases enables validation of results and exposes possible toxicities.
For example, in our studies, 110 of 8,498 molecules tested in a chemically diverse library were positive as mitigators (>130% viability) of radiation-induced lymphocyte cytotoxicity and 3 of these were strongly positive in the yeast DEL assay for both survival and preventing genomic instability, with the lymphocyte data informing on possible toxicities. Two of these 3, named YEL001 and YEL002 were selected, and YEL002, mitigated against lethal acute hematopoietic radiation syndrome in C3H mice. A novel analog of YEL001 (CJ010) was synthesized using the CDD SAR data mining tools to identify probable functional moieties that also had efficacy in vitro and in vivo (to be published). Furthermore, these compounds decreased rates of radiation-induced leukemia development in mice (to be published) and their anti-carcinogenic qualities were also suggested by a 50–60% reduction of DNA double-strand breaks following exposure to iodine-131 (to be published). The mechanism of action of these lead compounds has yet to be fully elucidated but the fact that they work across species suggests that the pathways they target are highly conserved during evolution.
HTS Profiles of Modulators of Radiation-Induced Lymphocyte Apoptosis
Using bioactive libraries, Kim et al. (2009) were able to detect known compounds that inhibited in vitro radiation-induced lymphocyte apoptosis and that were also effective in vivo. The profiles that were generated informed on canonical circuits of redox changes, DNA damage, and cell death that are activated following irradiation and that are reiterated through inflammatory processes in vivo during wound healing (McBride et al. 2004, Schaue and McBride 2010). The following classes of agents in our opinion fit this broad profile by potentially linking DNA repair, anti-oxidant and anti-inflammatory action, and hematopoiesis.
Purine Nucleosides
The nucleoside adenosine, its derivative deoxyadenosine, and its analog vidarabine, which show 97% structural similarity, were the only nucleoside mitigators of radiation-induced lymphocyte apoptosis out of hundreds of less related analogues. Adenosine is one of nature’s major tissue protectants. It is generally considered to be anti-inflammatory and immunosuppressive, although these characteristics depend on receptor expression and usage. Nucleotides such as adenosine triphosphate, diphosphate, and monophosphate are released from cells following irradiation, as well as in sites of hypoxia, inflammation, and cell death. ATP release into extracellular spaces is probably the primordial “danger” signal emitted by cells and can be affected by alterations in multiple types of membrane channels, including connexin and pannexin hemichannels, maxi-anion channels, volume-regulated anion channels and P2X7 purine receptors (P2X7R), reviewed in (Corriden and Insel 2010). Irradiation causes release of ATP through P2X7R in B16 melanoma cells (Ohshima et al. 2010).
Membrane ecto-nucleotidases convert extracellular nucleotides into adenosine that feeds back information to the cell through P1 (also known as A1, A2, A3) purinergic receptors. Although adenosinergic pathways may be activated in many cell types, in T regulatory (Treg) lymphocytes they are thought to be in part responsible for inhibiting autoimmune reactions and maintaining peripheral immune tolerance, (Antonioli et al. 2008). The enhanced ecto-nucleotidase expression by radiation resistant Treg after irradiation of mice (Schaue, in press) and humans (Mandapathil et al. 2009) may therefore represent a homeostatic response to radiation-induced tissue damage.
The literature clearly shows that purine nucleosides can act in vivo as mitigators of radiation damage, supporting the contention that in vitro HTS can identify classes of agents with similar in vivo activity. Purine nucleosides have previously been shown to diminish the quantity of DNA damage and enhance repair in irradiated leucocytes and mice, and to mitigate death after lethal WBI (Hennig et al. 1996, Hofer et al. 2002). Importantly, mice treated with adenosine and its analog inosine following 12 Gy WBI showed increased survival (Hou et al. 2007), and treatment starting 3 days post-WBI with drugs that elevate extracellular adenosine have been shown to accelerate myelorecovery of mice (Hofer et al. 2002). Interestingly, a fungal Chinese herbal medicinal extract from Cordyceps sinesis that protects mice from bone marrow and intestinal injuries after lethal WBI and enhances the colony-forming ability of both GM-CFU (granulocyte macrophage colony forming units) and osteogenic cells from bone marrow preparations (Liu et al. 2008, Liu et al. 2006) was recently reported to be 3'-adenosine (Zhou et al. 2008). In spite of these reports, nucleotides are of greater significance because of mechanism rather than practical usage as mitigators, which are probably limited by their short half-life.
Antibiotics
The initial most dramatic finding from our HTS runs using murine lymphocytes with drugs given either before or after 2 Gy irradiation was that 8 tetracycline or fluoroquinolone antibiotics were in the top 10 “hits” (Kim et al. 2009); activity obviously independent of their antimicrobial action. These were the only classes of antibiotics that were active in the screen, although many others were represented. This was of obvious interest because these hugely effective broad-spectrum antibiotics are already in the National Stockpile. Using CDD to identify all tetracyclines and fluoroquinolones in the screens and testing over a wide dose range showed that all were in fact radioprotective; some being missed because of high EC50 values. Further data mining showed a strong correlation between their radioprotective and mitigating activities (Figure 1), which was only marginally the case for the libraries as a whole (Kim et al. 2009). Most of these compounds could enhance DNA repair in irradiated human lymphoblastoid cells, as detected by γ-H2AX staining and Comet assay, and reduced apoptosis (Kim et al. 2009) and enhanced clonogenic survival of murine interleukin-3 (IL-3) dependent hematopoietic progenitor 32D cells after irradiation (Epperly et al. 2010). In vivo, tetracycline and ciprofloxacin protected mice from lethal WBI and tetracycline-treated mice had higher capability to form GM-CFU and white cell counts in spleen, blood, and bone marrow after lethal radiation exposure (Kim et al. 2009). In vivo, tetracycline was also shown to be an effective mitigator of lethal WBI with treatment starting 24 hrs after exposure.
Figure 1.
Mitigating and radioprotective in vitro activities of tetracyclines and quinolone analogs correlate. The drugs were added to a murine lymphocyte line either 3 hrs before or 1 hr after 2 Gy radiation and the viability measured at 24 hrs. Among 20 radioprotective antibiotics, 19 were mitigators of radiation damage.
Another drug that showed activity in vitro as a radioprotector and mitigator was the sarco/endoplasmic reticulum Ca2+-ATPase (SERCA) inhibitor cyclopiazonic acid (CPA). Searches of the libraries for SERCA inhibitors identified 6 of them with diverse chemical structures. Further testing showed that all protected cells against radiation but they were often toxic with a small therapeutic window (unpublished).
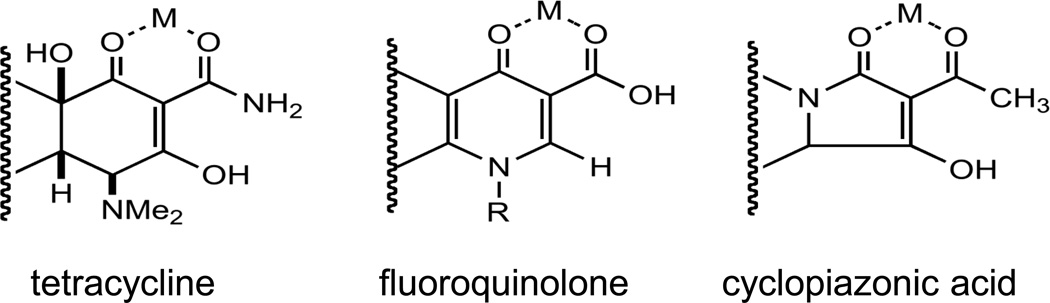
CDD was used to perform SAR using minocycline as the bait. Remarkably, tetracyclines, quinolones, and CPA had a cycloaromatic ring structure in common with a substructure that binds divalent cations, such as Zn2+, Mg2+, Mn2+, and Ca2+ (Kim et al. 2009) (Figure 2). We hypothesize that differences in the type or affinity of metal binding may account for differences in the mitigating abilities of different compounds within these classes. In any event, this pharmacophore seems a promising place to start examining the chemical groups within these classes responsible for activity.
Figure 2.
The core structures of tetracyclines and quinolones and CPA. The tetracyclines should be stronger metal-binders than the fluoroquinolones since they are more potent electron donors due to the OH at the bottom right donating more electron density to the two oxygen atoms at the top while in the fluoroquinolones the nitrogen in the ring donates less electron density to the oxygens. Cyclopiazonic acid is likely to be intermediate in terms of electron-density.
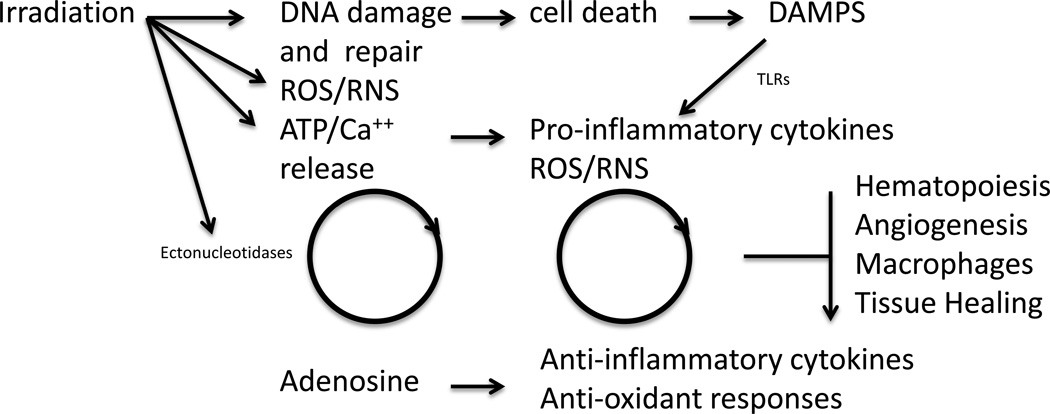
The nature of the common substructure suggested one possible mechanism of action of these agents was intercalation with DNA. In collaboration with Dr. Brendan Price of the Dana-Faber, we showed that all the tested antibiotics activated the Zn-finger-containing histone acetyl transferase (HAT) Tip60 (Kim et al. 2009). HATs, along with Zn-binding histone deacetylases, regulate the acetylation status of the epsilon-amino group of lysine residues of nucleosomal histones in chromatin. Tip60 localizes to the location of DNA double strand breaks (DSB) and is a key component in the remodeling of chromatin structure during their repair (Downey and Durocher 2006, Kusch et al. 2004, van Attikum and Gasser 2009). We hypothesize that these agents relax heterochromatin, allowing the slow component of DNA repair to be completed by non-homologous end joining (Downey and Durocher 2006), in so doing bypassing the requirement for activation of the protein mutated in ataxia telangiectasia (ATM). This would be in keeping with our finding that ATM is not an absolute requirement for tetracycline activity and that the antibiotics do not affect the radiation-induced DNA damage response (Kim et al. 2009). Clearly, DNA repair is normally thought of as being complete by 24 hrs and one might not expect a mitigator that acts on DNA repair to be active if given at this time. However, slow repair involving heterochromatin that might account for 15–20% of lesions and subsequent chromatin remodeling can go on for longer than 24 hrs (Klokov et al. 2006), and persistent DNA damage and genomic instability caused by cycles of oxidative stress continue for days and weeks after radiation exposure (Limoli and Giedzinski 2003, Rugo and Schiestl 2004). Included in this concept is that the DNA damage response and cell death are linked via purinergic and Toll-like receptors (TLR) to anti-inflammatory cytokine production, redox fluctuations, and stem/progenitor cell recovery that combat “waves” of radiation-induced pro-inflammatory damage (Figure 3) (McBride et al. 2004). A possible mechanistic link of DNA repair to inflammation generated through TLR activation comes from the finding that Tip60 activation initiates a cascade of events that represses signal transducer and activator of transcription 3 (STAT 3) and pro-inflammatory cytokine expression (Xiao et al. 2003). Alternatively, these agents might affect radiation-induced changes in calcium flux.
Figure 3.
Links between DNA damage and repair, adenosine, inflammatory cytokines and hematopoiesis showing how agents that affect DNA repair such as tetracyclines and fluoroquinolones, and purine nucleosides and TLR agonists can feed into hematopoiesis through cycles of nucleotide release and cytokine production that are initiated by radiation and perpetuated by homeostatic mechanisms. DAMPS (damage associated molecular pattern ligands), ROS (reactive oxygen species), RNS (reactive nitrogen species).
Initially, we were somewhat surprised to find that the two all-time most successful classes of broad-spectrum antibiotics could have the above activities, and at very low doses. In fact, sub-antimicrobial doses of tetracyclines have been used in dentistry for many years to combat periodontitis (Downey and Durocher 2006). In addition to decreasing pro-inflammatory cytokine production (Ikegaya et al. 2009), they inhibit metalloproteinase action, which is postulated to stem from their ability to bind metals. This has led to the development of tetracycline analogues devoid of anti-microbial action as anti-cancer agents (Zakeri and Wright 2008). In addition, in a randomized placebo-controlled trial, tetracycline was found to reduce inflammation and epithelial damage in gastritis patients at high risk for gastric cancer, independent of bacterial load (Fischbach et al. 2001) and sub-microbicidal doses of minocycline, a second-generation tetracycline have been repeatedly shown to be anti-inflammatory and anti-apoptotic and to be of therapeutic benefit in neurological disease (Orsucci et al. 2009).
Among fluoroquinolones, ciprofloxacin has been shown in a mouse model of chemical colitis to have anti-inflammatory effects distinct from its antibacterial properties (Lahat et al. 2007). Furthermore, certain fluoroquinolones have already been reported to be mitigators of lethality when given after WBI (6.5 Gy), stimulating hematopoiesis and enhancing myeloid progenitor GM-CFU number in the bone marrow (Shalit et al. 1997), which is similar to what we found for tetracyclines. Dalhoff recently reviewed the growing literature on the non-antimicrobial, immunomodulatory effects of quinolones (Dalhoff 2005) and concluded that, as a class, they enhance DNA repair, down-regulate synthesis of pro-inflammatory cytokines, suppress oxidative stress, and enhance production of hematopoietic growth factors.
Other Agents Active in HTS
One agent in our HTS that showed strong radioprotection and moderate mitigating activity was the interferon-α (IFN-α) inducer Tilorone, which had no structurally close relatives in the libraries. Recently IFN-α has been shown to activate dormant hematopoietic stem cells (Essers et al. 2009), suggesting a pathway that could link the sense of “danger” to hematopoiesis. Oral Tilorone can induce IFN-α production in the intestine and systemically (Munakata et al. 2008). In vitro, Tilorone can inhibit DNA repair processes, and in vivo stimulate hematopoiesis and act as a radiation mitigator (Kim, in preparation).
Another group of compounds that was active as mitigators in HTS came from the library of around 200 lipid compounds, where the top five hits were ω-3 or ω-6 polyunsaturated fatty acids (PUFA) or derivatives. Of these, linoleate was the most active agent in vitro and it was active in vivo as a mitigator of lethal WBI exposure. We had already shown that it mitigated radiation-induced DNA, chromosome, and bone marrow damage (Iwamoto et al. 1991, Iwamoto and McBride 1994, Norman et al. 1988a, Norman et al. 1988b). The mechanism of action is unknown but one recent concept is that the beneficial effects of PUFA might come from their antagonistic action on TLR, while saturated fatty acids act as TLR agonists (Lee et al. 2003), in essence modulating cytokine balance and, as a result, inflammatory/immune responses (De Smedt-Peyrusse et al. 2008).
Discussion
Our experience with HTS to identify modulators of radiation responses has been a very positive one. Importantly, we have shown that radioprotector and mitigator in vitro activity can be related to classes of compounds with common structures, for example all tetracyclines and quinolones, some of whom are active in vivo. In spite of our success in identifying such agents, HTS clearly has drawbacks. One is the high frequency of “false negatives”. Because common structures are generally repeatedly expressed within the libraries, the use of powerful chemo-/bio-informatic platforms allows data mining that can identify at least some “misses” so they can be re-evaluated.
A commonly expressed drawback is inability of HTS to predict in vivo efficacy. Not all our “hits” could easily be shown to be effective in vivo, though many were. However, given that the pharmacokinetics and pharmacodynamics of the drugs are not considered in HTS, it is not surprising that translation of findings to in vivo models has been difficult. As a result, the use of 3D cell and fish HTS models is increasing. An additional and related issue for consideration is the relevance of the target. Stem and progenitor cells as targets may identify agents that more directly engage these populations and might be expected to be of greater value for radiation effect modification. For example, the use of cell death by apoptosis may be criticized as being too limited as an endpoint, lacking relevance for the stem cells that must be most critical for tissue recovery. An alternative view is that apoptosis is simply a pathway that some cells use to express damage and that other cells that use different pathways may be similarly affected. Cell-based reporter assays have been created that are simpler and can look more directly for effects on specific signaling pathways by HTS and comparisons of pathway utilization through data mining should be able to evaluate whether effector or effector pathways are more relevant to in vivo efficacy. We believe that a combination of 3D, stem cell, and reporter gene assays, which we are developing, will be particularly informative when compared with our existing data using the CDD platform. By increasing the diversity of biological outcomes available using compound screening libraries in a radiation setting, we hope to improve the success of HTS in identifying mechanisms leading to mitigation of radiation effects without greatly expanding into the use of more and more compound libraries.
In spite of its limitations, we believe that HTS data can produce patterns in data that may illuminate what may be emerging radiobiological principles. One of our findings is that agents that modified radiation-induced apoptosis frequently enhance DNA repair, increase GM-CFU production in bone marrow, and are generally anti-oxidant and anti-inflammatory. How these agents might act to modify the outcome of radiation of normal tissues is outlined in Figure 3. The pathways are clearly complex and have ancient roots in wound healing and homeostasis (Pardo-Martin et al. 2010, Schaue and McBride 2010). Our data suggest that dampening the pro-inflammatory response has a positive effect on hematopoiesis, suggesting that braking mechanisms are potential targets for mitigation, i.e. removal of a mechanism that negatively regulates stem/progenitor cell development may be more effective than enhancing a positive stimulatory pathway. Overall, although the composite response to radiation is obviously time- and dose-dependent, the data indicate that there are several potential targets for intervention after radiation exposure. All seem related to mechanisms that restore homeostatic equilibrium, which may in some cases be a lifetime struggle and hopefully these are amenable to manipulation even at long times after exposure.
In conclusion, in vitro HTS provides a powerful tool, although imperfect, in the identification of radiation modulators or the target pathways. Understanding the key roles of these molecular and cellular targets in multiple tissues will lead to more informed discovery of novel modifiers of radiation responses.
Acknowledgements
This work was supported by University of California at Los Angeles Center for Medical Countermeasures grant U19 AI067769/NIAID.
Footnotes
Declaration of Interest
The authors report no conflicts of interest.
References
- Antonioli L, Fornai M, Colucci R, Ghisu N, Tuccori M, Del Tacca M, Blandizzi C. Regulation of enteric functions by adenosine: pathophysiological and pharmacological implications. Pharmacology & Therapeutics. 2008;120:233–253. doi: 10.1016/j.pharmthera.2008.08.010. [DOI] [PubMed] [Google Scholar]
- Casabon AL, Schmidt BA. Anti-inflammatory and tissue-protectant drug effects: results from a randomized placebo-controlled trial of gastritis patients at high risk for gastric cancer. Alimentary Pharmacology and Therapeutics. 2001;15:831–841. doi: 10.1046/j.1365-2036.2001.00998.x. [DOI] [PubMed] [Google Scholar]
- Corriden R, Insel PA. Basal release of ATP: an autocrine-paracrine mechanism for cell regulation. Science Signaling. 2010;3:re1. doi: 10.1126/scisignal.3104re1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dalhoff A. Immunomodulatory activities of fluoroquinolones. Infection. 2005;33:55–70. doi: 10.1007/s15010-005-8209-8. [DOI] [PubMed] [Google Scholar]
- De Smedt-Peyrusse V, Sargueil F, Moranis A, Harizi H, Mongrand S, Laye S. Docosahexaenoic acid prevents lipopolysaccharide-induced cytokine production in microglial cells by inhibiting lipopolysaccharide receptor presentation but not its membrane subdomain localization. Journal of Neurochemistry. 2008;105:296–307. doi: 10.1111/j.1471-4159.2007.05129.x. [DOI] [PubMed] [Google Scholar]
- Downey M, Durocher D. Chromatin and DNA repair: the benefits of relaxation. Nature Cell Biology. 2006;8:9–10. doi: 10.1038/ncb0106-9. [DOI] [PubMed] [Google Scholar]
- Ehrlich P. Über den jetzigen Stand der Chemotherapie. Dtsch. Chem. Ges. 1909;42:17. [Google Scholar]
- Epperly MW, Franicola D, Shields D, Rwigema JC, Stone B, Zhang X, McBride WH, Georges G, Wipf P, Greenberger JS. Screening of antimicrobial agents for in vitro radiation protection and mitigation capacity, including those used in supportive care regimens for bone marrow transplant recipients. In Vivo. 2010;24:9–19. [PMC free article] [PubMed] [Google Scholar]
- Essers MA, Offner S, Blanco-Bose WE, Waibler Z, Kalinke U, Duchosal MA, Trumpp A. IFNalpha activates dormant haematopoietic stem cells in vivo. Nature. 2009;458:904–908. doi: 10.1038/nature07815. [DOI] [PubMed] [Google Scholar]
- Fischbach LA, Correa P, Ramirez H, Realpe JL, Collazos T, Ruiz B, Bravo LE, Bravo JC, Hennig UG, Wang Q, Gee NH, von Borstel RC. Protection and repair of gamma-radiation-induced lesions in mice with DNA or deoxyribonucleoside treatments. Mutation Research. 1996;350:247–254. doi: 10.1016/0027-5107(95)00109-3. [DOI] [PubMed] [Google Scholar]
- Hofer M, Pospisil M, Znojil V, Vacek A, Weiterova L, Hola J, Vacha J. Drugs elevating extracellular adenosine promote regeneration of haematopoietic progenitor cells in severely myelosuppressed mice: their comparison and joint effects with the granulocyte colony-stimulating factor. European Journal of Haematology. 2002;68:4–11. doi: 10.1034/j.1600-0609.2002.00564.x. [DOI] [PubMed] [Google Scholar]
- Hou B, Xu ZW, Yang CW, Gao Y, Zhao SF, Zhang CG. Protective effects of inosine on mice subjected to lethal total-body ionizing irradiation. Journal of Radiation Research (Tokyo) 2007;48:57–62. doi: 10.1269/jrr.06067. [DOI] [PubMed] [Google Scholar]
- Ikegaya S, Inai K, Iwasaki H, Naiki H, Ueda T. Azithromycin reduces tumor necrosis factor-alpha production in lipopolysaccharide-stimulated THP-1 monocytic cells by modification of stress response and p38 MAPK pathway. Journal of Chemotherapy. 2009;21:396–402. doi: 10.1179/joc.2009.21.4.396. [DOI] [PubMed] [Google Scholar]
- Iwamoto KS, Bennett LR, Villalobos AE, Hutson CA, McBride WH, Norman A. Radiation Protection and Cancer Therapy by Linoleate. In: Honn KV, Marnett LJ, Nigam S, Walden T, editors. Eicosanoids and Other Bioactive Lipids in Cancer and Radiation Injury. 1st ed. New York: Springer; 1991. pp. 103–107. [Google Scholar]
- Iwamoto KS, McBride WH. Production of 13-hydroxyoctadecadienoic acid and tumor necrosis factor-alpha by murine peritoneal macrophages in response to irradiation. Radiation Research. 1994;139:103–108. [PubMed] [Google Scholar]
- Kim K, Pollard JM, Norris AJ, McDonald JT, Sun Y, Micewicz E, Pettijohn K, Damoiseaux R, Iwamoto KS, Sayre JW, Price BD, Gatti RA, McBride WH. High-throughput screening identifies two classes of antibiotics as radioprotectors: tetracyclines and fluoroquinolones. Clinical Cancer Research. 2009;15:7238–7245. doi: 10.1158/1078-0432.CCR-09-1964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klokov D, MacPhail SM, Banath JP, Byrne JP, Olive PL. Phosphorylated histone H2AX in relation to cell survival in tumor cells and xenografts exposed to single and fractionated doses of X-rays. Radiotherapy and Oncology. 2006;80:223–229. doi: 10.1016/j.radonc.2006.07.026. [DOI] [PubMed] [Google Scholar]
- Kusch T, Florens L, Macdonald WH, Swanson SK, Glaser RL, Yates JR, 3rd, Abmayr SM, Washburn MP, Workman JL. Acetylation by Tip60 is required for selective histone variant exchange at DNA lesions. Science. 2004;306:2084–2087. doi: 10.1126/science.1103455. [DOI] [PubMed] [Google Scholar]
- Lahat G, Halperin D, Barazovsky E, Shalit I, Rabau M, Klausner J, Fabian I. Immunomodulatory effects of ciprofloxacin in TNBS-induced colitis in mice. Inflammatory Bowel Diseases. 2007;13:557–565. doi: 10.1002/ibd.20077. [DOI] [PubMed] [Google Scholar]
- Lally BE, Geiger GA, Kridel S, Arcury-Quandt AE, Robbins ME, Kock ND, Wheeler K, Peddi P, Georgakilas A, Kao GD, Koumenis C. Identification and biological evaluation of a novel and potent small molecule radiation sensitizer via an unbiased screen of a chemical library. Cancer Research. 2007;67:8791–8799. doi: 10.1158/0008-5472.CAN-07-0477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee JY, Plakidas A, Lee WH, Heikkinen A, Chanmugam P, Bray G, Hwang DH. Differential modulation of Toll-like receptors by fatty acids: preferential inhibition by n-3 polyunsaturated fatty acids. Journal of Lipid Research. 2003;44:479–486. doi: 10.1194/jlr.M200361-JLR200. [DOI] [PubMed] [Google Scholar]
- Limoli CL, Giedzinski E. Induction of chromosomal instability by chronic oxidative stress. Neoplasia. 2003;5:339–346. doi: 10.1016/S1476-5586(03)80027-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu WC, Chuang WL, Tsai ML, Hong JH, McBride WH, Chiang CS. Cordyceps sinensis health supplement enhances recovery from taxol-induced leukopenia. Exp Biology and Medicine (Maywood) 2008;233:447–455. doi: 10.3181/0708-RM-230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu WC, Wang SC, Tsai ML, Chen MC, Wang YC, Hong JH, McBride WH, Chiang CS. Protection against Radiation-Induced Bone Marrow and Intestinal Injuries by Cordyceps sinensis, a Chinese Herbal Medicine. Radiation Research. 2006;166:900–907. doi: 10.1667/RR0670.1. [DOI] [PubMed] [Google Scholar]
- Mandapathil M, Szczepanski MJ, Szajnik M, Ren J, Lenzner DE, Jackson EK, Gorelik E, Lang S, Johnson JT, Whiteside TL. Increased ectonucleotidase expression and activity in regulatory T cells of patients with head and neck cancer. Clinical Cancer Research. 2009;15:6348–6357. doi: 10.1158/1078-0432.CCR-09-1143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McBride WH, Chiang CS, Olson JL, Wang CC, Hong JH, Pajonk F, Dougherty GJ, Iwamoto KS, Pervan M, Liao YP. A sense of danger from radiation. Radiation Research. 2004;162:1–19. doi: 10.1667/rr3196. [DOI] [PubMed] [Google Scholar]
- Munakata K, Yamamoto M, Anjiki N, Nishiyama M, Imamura S, Iizuka S, Takashima K, Ishige A, Hioki K, Ohnishi Y, Watanabe K. Importance of the interferon-alpha system in murine large intestine indicated by microarray analysis of commensal bacteria-induced immunological changes. BMC Genomics. 2008;9:192. doi: 10.1186/1471-2164-9-192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Norman A, Bennett LR, Mead JF, Iwamoto KS. Antitumor activity of sodium linoleate. Nutrition and Cancer. 1988a;11:107–115. doi: 10.1080/01635588809513977. [DOI] [PubMed] [Google Scholar]
- Norman A, McBride WH, Bennett LR, Santos Mello R, Iwamoto K, Hidmi H. Postirradiation protection of chromosomes by linoleate. International Journal of Radiation Biology. 1988b;54:521–524. doi: 10.1080/09553008814551951. [DOI] [PubMed] [Google Scholar]
- Ohshima Y, Tsukimoto M, Takenouchi T, Harada H, Suzuki A, Sato M, Kitani H, Kojima S. gamma-Irradiation induces P2X(7) receptor-dependent ATP release from B16 melanoma cells. Biochimica et Biophysica Acta. 1800:40–46. doi: 10.1016/j.bbagen.2009.10.008. [DOI] [PubMed] [Google Scholar]
- Orsucci D, Calsolaro V, Mancuso M, Siciliano G. Neuroprotective effects of tetracyclines: molecular targets, animal models and human disease. CNS & Neurological Disorders Drug Targets. 2009;8:222–231. doi: 10.2174/187152709788680689. [DOI] [PubMed] [Google Scholar]
- Pardo-Martin C, Chang TY, Koo BK, Gilleland CL, Wasserman SC, Yanik MF. High-throughput in vivo vertebrate screening. Nature Methods. 2010;7:634–636. doi: 10.1038/nmeth.1481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rugo RE, Schiestl RH. Increases in oxidative stress in the progeny of X-irradiated cells. Radiation Research. 2004;162:416–425. doi: 10.1667/rr3238. [DOI] [PubMed] [Google Scholar]
- Schaue D, McBride WH. Links between innate immunity and normal tissue radiobiology. Radiation Research. 2010;173:406–417. doi: 10.1667/RR1931.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shalit I, Kletter Y, Weiss K, Gruss T, Fabian I. Enhanced hematopoiesis in sublethally irradiated mice treated with various quinolones. Eur J Haematol. 1997;58:92–98. doi: 10.1111/j.1600-0609.1997.tb00930.x. [DOI] [PubMed] [Google Scholar]
- van Attikum H, Gasser SM. Crosstalk between histone modifications during the DNA damage response. Trends in Cell Biology. 2009;19:207–217. doi: 10.1016/j.tcb.2009.03.001. [DOI] [PubMed] [Google Scholar]
- Xiao H, Chung J, Kao HY, Yang YC. Tip60 is a co-repressor for STAT3. The Journal of Biological Chemistry. 2003;278:11197–11204. doi: 10.1074/jbc.M210816200. [DOI] [PubMed] [Google Scholar]
- Zakeri B, Wright GD. Chemical biology of tetracycline antibiotics. Biochemistry and Cell Biology. 2008;86:124–136. doi: 10.1139/O08-002. [DOI] [PubMed] [Google Scholar]
- Zhang JH, Chung TD, Oldenburg KR. A Simple Statistical Parameter for Use in Evaluation and Validation of High Throughput Screening Assays. Journal of Biomolecular Screening. 1999;4:67–73. doi: 10.1177/108705719900400206. [DOI] [PubMed] [Google Scholar]
- Zhou X, Luo L, Dressel W, Shadier G, Krumbiegel D, Schmidtke P, Zepp F, Meyer CU. Cordycepin is an immunoregulatory active ingredient of Cordyceps sinensis. The American Journal of Chinease Medicine. 2008;36:967–980. doi: 10.1142/S0192415X08006387. [DOI] [PubMed] [Google Scholar]