Fit genotypes and escape variants of subgroup III Neisseria meningitidis during three pandemics of epidemic meningitis (original) (raw)
Abstract
The genetic variability at six polymorphic loci was examined within a global collection of 502 isolates of subgroup III, serogroup A_Neisseria meningitidis._ Nine “genoclouds” were identified, consisting of genotypes that were isolated repeatedly plus 48 descendent genotypes that were isolated rarely. These genoclouds have caused three pandemic waves of disease since the mid-1960s, the most recent of which was imported from East Asia to Europe and Africa in the mid-1990s. Many of the genotypes are escape variants, resulting from positive selection that we attribute to herd immunity. Despite positive selection, most escape variants are less fit than their parents and are lost because of competition and bottlenecks during spread from country to country. Competition between fit genotypes results in dramatic changes in population composition over short time periods.
Keywords: genetic variation, clonal descent, intercontinental spread, population structure, microevolution
Epidemic meningitis has posed a major health problem since it was first recognized in 1805 (1). Epidemics have recurred approximately every 10 years in this century within the African “meningitis belt” of sub-Saharan Africa (2–4) and until the mid-1980s within China (5). Epidemics in other geographical areas are more sporadic, and, in recent decades, only a few industrialized countries have suffered from large epidemics (3), with the notable exceptions of Norway and New Zealand.
Historically, most large epidemics of meningitis were caused by_Neisseria meningitidis_ that express the serogroup A capsular polysaccharide. Most recent epidemics have been caused by two pandemic waves (6) of the clonal grouping of serogroup A bacteria that is called subgroup III (5, 7). The first pandemic wave affected China (mid-1960s), northern Europe (Russia and the Scandinavian countries; 1969-mid 1970s), and Brazil (mid-1970s). A second subgroup III pandemic wave began in China and Nepal in the early 1980s, followed by an epidemic during the annual Hajj pilgrimage to Mecca, Saudi Arabia of 1987. Pilgrims returning from Mecca distributed subgroup III meningococci to most countries of the globe (6, 8). Subgroup III has subsequently caused numerous epidemics throughout Africa (3, 9); in 1996, 150,000 cases of meningococcal disease were reported from Africa (10), most caused by subgroup III (3).
Subgroup III meningococci are so uniform that only a few genetic variants have been revealed by MLEE (multilocus enzyme electrophoresis) (5, 6), RAPD (random amplified polymorphic DNA) (11, 12), or MLST (multilocus sequence typing) (7, 12). Such variants can arise during cocolonization of the nasopharynx with other N. meningitidis and related species because of the import of genes by DNA transformation (13, 14).
The availability of hundreds of subgroup III strains isolated in different countries since the mid-1960s provided an opportunity to elucidate important features of bacterial microevolution. We have used a multilocus approach involving sequence variability at six polymorphic loci to elucidate the molecular epidemiology of subgroup III bacteria. These six loci encode three outer membrane proteins (opaB,opaD, tbpB), one secreted protein (iga), one housekeeping protein (pgm), and one defective insertion sequence (IS) element (IS_1106_A) and were chosen because of their relatively high degree of polymorphism in subgroup III. Previous work has shown that differences at the_opaB_, opaD, and iga loci (which encode opacity adhesins and IgA1 protease) distinguish bacteria isolated during the first pandemic wave or at the beginning of the second pandemic wave from those isolated since 1987 from Mecca pilgrims and in Africa (15). The pgm gene encoding phosphoglucomutase differentiates subgroup III isolates of MLST sequence types 5 through 7 (7). tbpB (transferrin-binding protein B) differed between older and newer isolates from Moscow (12). IS_1106_A is a defective IS element that is inserted upstream of the opcA locus (16) and was shown by preliminary experiments to be polymorphic in subgroup III.
The results show that successive waves of pandemic spread are associated with genoclouds of genetic variants, each containing a particularly fit, dominant genotype. Furthermore, the recent evolution of a new genocloud has now resulted in a third pandemic wave of disease.
Materials and Methods
Bacterial Strains.
Five hundred two subgroup III strains isolated between 1966 and 2000 were tested. The countries and detailed dates of isolation have been described for 314 strains (1966–1996) from diverse global sources (15) and 29 ST7 strains (1995–1999) from Africa (9). One hundred fifty-nine additional isolates were from epidemics in Chad (1988, 9 strains), The Gambia (1997, 54), Ghana (1997–1998, 39; ref. 17), Mongolia (1994–1995, 10; ref. 18), Moscow (1969–1977, 8; 1994–1997, 21; ref.12), and Sudan (1988, 14), and from endemic disease in the United Kingdom (1997–2000, 4). The assignment of these additional isolates to subgroup III is based on multilocus enzyme electrophoresis for isolates from Chad, Sudan, and Mongolia, random amplified polymorphic DNA for isolates from Moscow (12), and by the possession of strongly related pulse-field gel electrophoresis (PFGE) patterns with two restriction endonucleases (17) for isolates from The Gambia, Ghana, and the United Kingdom. Of the 502 isolates, 60 have been shown to belong to the MLST sequence types ST5 to ST7 that are characteristic of subgroup III (9,12). Most of the strains have been serogrouped, serotyped, and serosubtyped by ELISA using monoclonal antibodies and are A:4,21:P1.9,20, as is typical of subgroup III (5).
Sequencing and Accession Numbers.
Independent PCR products from each locus were sequenced from both DNA strands by using automated cycle sequencing (ABI 377) with dRhodamine-labeled terminators (Perkin–Elmer). Sequences of IS_1106_A elements have been deposited under AJ276902-AJ276908 and AJ292239. Sequences of tbpB alleles have been deposited under AJ276909-AJ276937. Other sequences have been described elsewhere (7, 15, 19) (GenBank AJ292235–AJ292238).
Restriction Fragment Length Polymorphism (RFLP) Typing.
All 502 isolates were tested for RFLP at six loci (pgm, iga, opaB, opaD, IS_1106_A, and tbpB) as described (12, 15). In addition, all variant alleles were sequenced from representative strains (pgm, 60 strains; iga, 18;opaB, 43; opaD, 50; IS_1106_A, 21; and_tbpB_ , 35). The sequence variability at iga,opaB, and opaD has been described elsewhere (15). Details of the sequence variability at IS_1106_A are presented in the supplemental data (which are published on the PNAS web site,www.pnas.org). The RFLP analysis of pgm distinguishes_pgm3_ from pgm19, but not from pgm11, which differs by only one nucleotide (7). tbpB alleles that are identical to previously described sequences (tbpB1,10, 11, and 101; ref. 13) were assigned the same numbers, and other alleles were assigned arbitrary numbers from tbpB38 to tbpB66. These allele assignments were based on an aligned stretch of approximately 660 bp that has been sequenced from all isolates. [We note that the_tbpB1_ allele is assigned to both subgroups III and IV-1 although the complete 2057-bp _tbpB_ sequences from subgroups III (accession number AJ276909) and IV-1 (AF058689) differ by two nucleotides outside this aligned region.]
Phylogenetic Analyses_._
tbpB sequences were aligned manually to ensure that codons were not split. The aligned sequences were analyzed by usingsplitstree 3.1 (20), as described (13). χ2 analysis of significance between success and failure was performed by using tests 3 and 4 ofclump (21), which tests the significance of data in sparse matrices by a Monte Carlo method.
Results
Genocloud Structure of Subgroup III.
The genetic fine structure of subgroup III was investigated among 502 subgroup III isolates by using RFLP at opaB,opaD, tbpB , pgm, iga, and IS_1106_A. The isolates tested include 314 subgroup III meningococci from diverse global sources that have been previously described (15) plus 188 recent isolates from Mongolia, Moscow, Africa, and the United Kingdom. Two hundred twenty-seven variant alleles were sequenced from representative strains, and at least two alleles were found for each of the six loci.
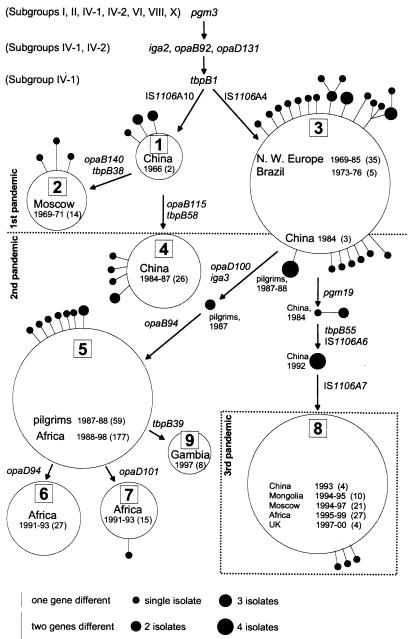
Each unique combination of alleles is referred to as a genotype, and a total of 57 genotypes were found. Most (87%) of the 502 isolates possess one of nine frequent genotypes (Table1). The nine frequent genotypes differed from each other by one to six of the six loci (Fig.1; see Table 5 in supplemental data for details). The other 48 genotypes were isolated only rarely (one to four isolates). They were isolated concurrently with a frequent genotype from the same geographical area and differed from that frequent genotype at only one, or rarely two, of the loci (Fig. 1; Fig. 5). All three criteria (minimal genetic, physical, and temporal distance) were used to assign rare genotypes to groups consisting of one frequent genotype plus its close relatives (Fig. 1). These groups will be referred to as “genoclouds.”
Table 1.
Geographic distribution of frequent and rare genotypes of subgroup III
| Genocloud | Pandemic | Sources | Years | Frequent genotypes, no. of isolates | Rare genotypes | |
|---|---|---|---|---|---|---|
| No. of genotypes | No. of isolates | |||||
| 1 | 1 | China | 1966 | 2 | 3 | 5 |
| 2 | 1 | Moscow | 1969–1971 | 14 | 3 | 3 |
| 3 | 1, 2 | Europe, Brazil, China | 1969–1984 | 43 | 23 | 31 |
| 4 | 2 | China | 1984–1987 | 26 | 4 | 5 |
| 5 | 2 | Pilgrims, Africa | 1987–1998 | 236 | 7 | 8 |
| 6 | 2 | Africa | 1991–1993 | 27 | 0 | |
| 7 | 2 | Africa | 1991–1993 | 15 | 1 | 1 |
| 9 | 2 | The Gambia | 1997 | 8 | 0 | |
| 8 | 3 | China, Mongolia, Moscow, Africa, U.K. | 1993–2000 | 66 | 3 | 3 |
| Intermediate | 4 | 9 | ||||
| Total | 1966–2000 | 437 | 48 | 65 |
Figure 1.
Parsimonious relationships among 57 genotypes in nine genoclouds of subgroup III. Large boxed numbers from 1 to 9 indicate the genocloud designations. Alleles inherited from common ancestors with other serogroup A subgroups are shown at the top, and subsequent changes are shown next to the arrows that indicate lines of descent. Numbers in parentheses indicate numbers of isolates of the frequent genotypes, which are also reflected to a limited extent by the sizes of the circles. Rare genotypes were parsimoniously grouped in genoclouds after consideration of genetic relationships, sources, and dates of isolation. They are indicated by filled dots whose sizes indicate the numbers of isolates. The lengths of the lines connecting the dots to the genocloud circles indicate the numbers of genetic changes from the frequent genotype. Intermediate strains that were not assigned to genoclouds are shown along the arrows leading from genocloud 3.
Six of the genoclouds contain a frequent genotype plus descendent genotypes, but genoclouds 1, 6, and 9 do not fulfill both requirements (Fig. 1). Genocloud 1 contains only a few isolates (Table 1). It was designated as a separate genocloud because it includes all of the available subgroup III strains that were isolated from several million patients with meningococcal disease in China during the mid-1960s, contains distinct genotypes, and is closely related to the deduced ancestral genotype (see below). Genoclouds 6 and 9 possess only one frequent genotype and no rare genotypes. Their designation as genoclouds is somewhat arbitrary, and they might equally well represent the transient expansion of rare genotypes.
Phylogenetic Relationships and Pandemic Spread.
By minimizing genetic and temporal distances between the genoclouds, we constructed a fully parsimonious tree in which each allele arises only once. Four rare genotypes seem to represent intermediates during the evolution from one genocloud to a second and were therefore not assigned to either genocloud (Fig. 1).
The tree is rooted at genoclouds 1 and 3. These genoclouds share_pgm3, iga2_, opaB92, and opaD131 alleles with older strains of the related subgroups IV-1 and IV-2 (15) and the tbpB1 allele with older strains of subgroup IV-1 (ref. 13; Fig. 1). The other genoclouds possess variant alleles at these loci. Genoclouds 1 and 3 differ from subgroup IV-1 at the IS_1106_A locus. Genocloud 1 possesses one IS_1106_A element inserted into a second, defective IS_1106_A element whereas genocloud 3 possesses only the defective IS_1106_A element (see supplemental data). Possibly, genocloud 1 is older and was the direct ancestor of genocloud 3 because an IS_1106_A element inserted into a second, defective IS_1106_A element was also found in subgroup IV-1 (16). Genoclouds 2 and 4 are descended from genocloud 1 because they possess the same double IS_1106_A elements, and genoclouds 5–7 and 9 are derived from genocloud 3 because they possess the same single IS_1106_A element.
The first subgroup III pandemic (mid-1960s to late 1970s) was associated with genoclouds 1 to 3 (Table 1; Fig. 1). The second subgroup III pandemic (early 1980s to present) was associated with genoclouds 3 to 7 and 9. A third pandemic caused by genocloud 8 seems to have started in China in 1993. The progressive evolution of genocloud 8 from genocloud 3 is documented by the isolation in China between 1984 and 1992 of intermediate genotypes with progressive genetic changes. Genocloud 8 has now been isolated from epidemics in Mongolia (1994–1995; ref. 18), Moscow (1994–1997; ref. 12), and Africa (1995–1999; ref. 9), and from endemic disease in the United Kingdom (1997–2000).
Mechanisms of Genetic Change.
Alleles that differed by at least 2 bp from parental alleles were considered to represent import by DNA transformation. This interpretation applies to all novel alleles at tbpB (28 alleles) and to one allele each at iga and pgm (Table 2). Similarly, all eight sequenced IS_1106_A alleles also resulted from import (supplemental data). Eighteen alleles at the opaB and opaD loci are mosaic genes resulting from gene conversion/translocation with segments of orthologous opa genes (most frequently_opaA_ and opaJ) that are present elsewhere in the genome (15). Four alleles differed from their parental alleles by only one nucleotide or by a 12-bp deletion and possibly represent single-step mutations (Table 2).
Table 2.
Sources of alleles in 502 subgroup III isolates
| Locus | Ancestral allele | No. of variant alleles | ||
|---|---|---|---|---|
| Mutation | Import | Translocation | ||
| iga | iga2 | 1 (iga1) | 1 (iga3) | |
| IS_1106_A | Uncertain | 8 (IS_1106_A4-11)* | ||
| pgm | pgm3 | 1 (pgm11) | 1 (pgm19) | |
| opaB | opaB92 | 1 (opaB93)† | 2 (opaB94,5202) | 12 (5× opaA; 4×opaD; 3× opaJ)† |
| opaD | opaD131 | 1 (opaD100) | 8 (7×opaJ; 1× opaB)† | |
| tbpB | tbpB1 | 28 (19× family 1; 6× family 4; 3× family 3) |
Fitness of Individual Genetic Changes.
It is striking that so many novel alleles were observed for the loci investigated here although only 2 novel alleles were detected for six other housekeeping gene fragments among 137 subgroup III isolates (9,12). Possibly sequence polymorphism was particularly high for the loci tested here because some of them encode gene products that are under selection. Indeed, the tbpB, opaB, and_opaD_ loci encode immunogenic outer membrane proteins, and variants at these loci might enable the bacteria to escape herd immunity.
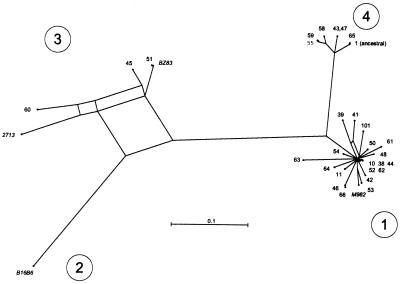
A conservative test for differences in fitness is to compare the number of descendent genotypes associated with different classes of genetic change. Changes at the tbpB locus reflect the import of novel tbpB alleles belonging to one of four very distinct allele families (13). The 28 alleles described here fell into three of the same four families (Fig. 2). The ancestral allele was in family 4, and most of the imported alleles were in family 1. Of the 19 occasions where a tbpB allele in family 1 was imported, only one of these alleles was inherited by descendent genotypes, a significantly lower rate of dissemination than for all other observed changes (Table 3). These results suggest that the fitness of subgroup III is impaired by the acquisition of tbpB alleles of family 1. Acquisition of_tbpB_ of family 3 or translocation of alleles from either the_opaJ_ or opaA loci to the opaB or_opaD_ loci may also have resulted in reduced fitness (Table3), but the numbers of such events were too small to be statistically significant. In contrast, three of the six novel tbpB alleles in family 4, the ancestral family in subgroup III, disseminated to descendent genotypes.
Figure 2.
Splits graph (uncorrected Hamming distances) of relationships of ≈660-bp tbpB fragments. Large circled numbers indicate the allele families. The smaller numbers are tbpB allele numbers. tbpB1 is the ancestral allele in subgroups III and IV-1. tbp10 and tbp11 were previously found in subgroup IV-1 and commensal neisseriae, and_tbpB101_ was found in N. lactamica (13).tbpB38 through tbpB66 have been found only in subgroup III. Sequences from reference strains of N. meningitidis for families 1–3 (B16B6, 2713, BZ83, and M982) are italicized.
Table 3.
Fitness analysis of genotypic changes
| No. of descendent genotypes | No. of genetic events | |
|---|---|---|
| ≥1 (success) | 0 (failure) | |
| tbpB family 1 | 1 | 18 |
| tbpB family 3 | 0 | 3 |
| tbpB family 4 | 3 | 3 |
| pgm | 1 | 1 |
| IS_1106_A | 5 | 4 |
| Translocation of_opaJ_ | 2 | 8 |
| Translocation of opaA | 1 | 4 |
| Other opa changes | 4 | 5 |
| iga | 1 | 1 |
Discussion
We analyzed microevolution within an epidemiological framework for a globally representative collection of subgroup III meningococci. Fifty-seven distinct genotypes in nine genoclouds were detected among 502 strains collected during pandemic spread between 1966 and 2000.
The Genocloud Concept.
We designate a frequent genotype plus its epidemiologically associated descendents as a genocloud. The nine subgroup III genoclouds contained rare genotypes that differed by one or occasionally two of the six polymorphic loci. The frequent genotypes in subgroup III differed from each other by one to six loci (Table 5). All these bacteria are extremely closely related because housekeeping gene fragments were highly uniform among representative isolates (9, 12). Each genocloud persists only transiently (see below), and subgroup III evolution since 1966 has been marked by the successive replacement of individual dominant genoclouds by others.
The genocloud concept may be generally useful for describing sequence variability among clonally related isolates for many bacterial species. Sequence variation is common in most microorganisms, and clonally related bacteria from a common epidemiological source often show some limited genotypic variation (22). Until now, bacteria have been subdivided into clonally related and unrelated groups, with no standard designation for distinct populations within the clonally related groupings. Furthermore, both genetic variability and different frequencies of particular genotypes are integral aspects of the genocloud concept whereas these aspects are lacking in former terminology. Genoclouds are not “quasi-species,” a mathematical concept that describes an equilibrium population of small genomes (23). Unlike quasi-species, the genetic changes in a genocloud are probably not reversible, represent unique events that do not happen repeatedly, and are continuously eliminated during spread from host to host and from country to country.
Immune Selection During Epidemic Spread.
In subgroup III, many of the genetic variants are escape variants that can evade the human immune system (Fig.3). Epidemics and pandemics may begin when particularly fit strains invade a naive human population. Herd immunity develops and selects escape variants for particularly immunogenic proteins such as TbpB. Most TbpB escape variants result from the import of DNA from Neisseria lactamica and_Neisseria spp._ (13) that encode TbpBs of family 1. Family 1 differs extensively from family 4 (Fig. 2), which is ancestral in subgroup III, sufficiently that these two families probably share very few epitopes. We have shown previously (13) that the high rate of import of TbpB alleles is due to positive selection rather than to an elevated recombination rate: the only common feature of numerous independent recombination events with different recombination endpoints was the import of tbpB. Nevertheless, import of TbpB of family 1 (and possibly of family 3) reduces the long-term fitness of the resulting escape variants. Only one of nineteen genotypes that had imported family 1 alleles succeeded in generating descendent genotypes (Table 3) whereas half of the imports of a family 4 (ancestral) allele were successful. Thus, many of the rare genotypes may represent escape variants that have a temporary advantage because of different antigenic composition even though they are less fit than their parent (Fig. 3).
Figure 3.
A model for the formation of rare genotypes and novel genoclouds. Rare genotypes arise by a variety of mechanisms, including import of DNA from unrelated bacteria, translocation of opa alleles, and single step mutations. Those variants that affect antigens and result in immune escape will multiply preferentially because of selection pressures by the host immune system. However, many of these variants also result in lessened fitness and are eliminated by competition and bottlenecks during spread from host to host and country to country. On rare occasions, antigenic variants with increased fitness can form new genoclouds and repeat the cycle.
The term “fitness,” rather than virulence, is appropriate in this context because, during single epidemics, the same serogroup A isolates are isolated from both invasive disease and healthy carriers (24). Furthermore, there is no evidence for frequent healthy carriage of serogroup A meningococci without occasional disease. Thus, for serogroup A, the existence of descendent genotypes among disease isolates reflects efficient transmission between individuals and can therefore be used to calculate measures of fitness.
The data are also suggestive of a similar but less strong pattern of selection at the opa loci. The ancestral subgroup III genotype possessed distinct alleles at all four opa loci (opaA, opaB, opaD, and opaJ). The_opa101_ allele at the opaJ locus is not normally expressed because of a stretch of four CTCTT repeats in the signal peptide region, which results in a nonfunctional translational reading frame. The opa101 allele was repeatedly translocated to the_opaB_ and opaD loci whereas translocation from the other opa loci to opaJ is very rare (15). This asymmetry is suggestive of positive selection for replacement of OpaB or OpaD by the immunologically distinct Opa101 protein. Transposition of the opa101 allele may also be detrimental to long-term fitness (Table 3), but more observations are needed to test whether this pattern is statistically significant. It is also likely that genetic variation does not always reflect selection, either positive or negative. For example, IS_1106_A sequence variants are also frequent although it is hard to imagine how different alleles of IS_1106_A could affect fitness.
Transience of Genoclouds.
The half-life of nasopharyngeal colonization is only a few months (25), and genoclouds disappear from individual countries and areas after several years, even in the absence of vaccination (26–28). Genocloud 1 disappeared from China in the 1960s, and epidemic disease during the 1970s was caused by subgroup V meningococci (5). Genocloud 2 disappeared from Moscow in the 1970s and was replaced by subgroups VI and X (12). Genoclouds 1–4 have not been isolated since 1985. Thus, meningococcal genoclouds survive only if they compete successfully with their parent and other variant genotypes for transmission to other countries and geographic areas.
Genoclouds persist by migrating from country to country or by generating new variants that escape the immune response against previous infections. Our analysis of the tbpB locus suggests that variants that achieve both significant immune escape and high fitness are unusual, possibly because of the rarity of appropriate DNA in the available gene pool. The genocloud structure that we have observed probably reflects how rarely novel genotypes are generated that combine fitness with immune escape at key antigenic sites.
The Third Subgroup III Pandemic.
A third subgroup III pandemic associated with genocloud 8 began in China in 1993 after a decade of slow, progressive microevolution from genocloud 3. Since 1994, genocloud 8 has caused large epidemics in Mongolia (18), Moscow (12), and Africa (9). Genocloud 8 has now reached Western Europe and caused several cases of endemic disease in the United Kingdom between 1997 and 2000.
Based on historical experience (4), it might have been expected that the recent subgroup III epidemics in diverse African countries had surpassed their zenith. In the past, epidemic waves have not extended much longer than 10 years, and most African countries in the “meningitis belt” have now experienced major epidemics caused by genocloud 5 (Table 4 in supplemental data). However, these epidemics do not seem to have generated long-lasting herd immunity against genocloud 8. Genocloud 8 has recently caused epidemics in Sudan, Chad, and Niger (Table 4), where epidemics had been caused by subgroup III bacteria with pgm3 (presumably genocloud 5) only a few years earlier (9). These observations bode poorly for the rapid disappearance of epidemic meningitis from Africa and raise the possibility of renewed epidemic serogroup A disease in Europe and the Americas. Before epidemics in Norway and Finland in the early 1970s, serogroup A disease had long been absent, similarly to the current situation. The availability of vaccines based on the A polysaccharide can help control epidemics, but new generation A-conjugate vaccines are necessary to ensure the prevention of epidemic disease on a global basis.
Supplementary Material
Supplemental Data
Acknowledgments
We gratefully acknowledge the encouragement to present this work by Brian Greenwood, comments on the manuscript by Elisabeth Carniel, Gerd Pluschke, Thierry Wirth, and Sebastian Suerbaum, and the receipt of recent United Kingdom isolates from Ed Kaczmarski. This work was supported by Deutsche Forschungsgemeinschaft Ac 36/9-2 and INTAS-open-97-108.
Abbreviations
IS
insertion sequence
tbpB
transferrin-binding protein B
RFLP
restriction fragment length polymorphism
MLST
multilocus sequence typing
Footnotes
This paper was submitted directly (Track II) to the PNAS office.
Data deposition: The sequences reported in this paper have been deposited in the GenBank database [accession nos. AJ276902–AJ276908 and AJ292239 (IS_1106_A elements) and AJ276909–AJ276937 (tbpB alleles)].
References
- 1.Vieusseux M. J Med Chirurg Pharm. 1806;11:163–182. [Google Scholar]
- 2.Lapeyssonnie L. Bull W H O. 1963;28,Suppl.:53–114. [Google Scholar]
- 3.Caugant D A. Acta Pathol Microbiol Immunol Scand. 1998;106:505–525. [PubMed] [Google Scholar]
- 4.Achtman M. Rev Med Microbiol. 1990;1:29–38. [Google Scholar]
- 5.Wang J-F, Caugant D A, Li X, Hu X, Poolman J T, Crowe B A, Achtman M. Infect Immun. 1992;60:5267–5282. doi: 10.1128/iai.60.12.5267-5282.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Achtman M, Kusecek B, Morelli G, Eickmann K, Wang J, Crowe B, Wall R A, Hassan-King M, Moore P S, Zollinger W. J Infect Dis. 1992;165:53–68. doi: 10.1093/infdis/165.1.53. [DOI] [PubMed] [Google Scholar]
- 7.Maiden M C J, Bygraves J A, Feil E, Morelli G, Russell J E, Urwin R, Zhang Q, Zhou J, Zurth K, Caugant D A, et al. Proc Natl Acad Sci USA. 1998;95:3140–3145. doi: 10.1073/pnas.95.6.3140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Moore P S, Harrison L H, Telzak E E, Ajello G W, Broome C V. J Am Med Assoc. 1988;260:2686–2689. [PubMed] [Google Scholar]
- 9.Nicolas, P., Décousset, L., Riglet, V., Castelli, P., Stor, R. & Blanchet, G. (2001) Emerg. Infect. Dis., in press. [DOI] [PMC free article] [PubMed]
- 10.Tikhomirov E, Santamaria M, Esteves K. World Health Stat Q. 1997;50:170–176. [PubMed] [Google Scholar]
- 11.Bart A, Schuurman I G A, Achtman M, Caugant D A, Dankert J, van der Ende A. J Clin Microbiol. 1998;36:1746–1749. doi: 10.1128/jcm.36.6.1746-1749.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Achtman, M., van der Ende, A., Zhu, P., Koroleva, I. S., Kusecek, B., Morelli, G., Schuurman, I. G. A., Brieske, N., Zurth, K., Kostyukova, N. N. & Platonov, A. E. (2001) Emerg. Infect. Dis., in press. [DOI] [PMC free article] [PubMed]
- 13.Linz B, Schenker M, Zhu P, Achtman M. Mol Microbiol. 2000;36:1049–1058. doi: 10.1046/j.1365-2958.2000.01932.x. [DOI] [PubMed] [Google Scholar]
- 14.Smith J M, Feil E J, Smith N H. BioEssays. 2000;22:1115–1122. doi: 10.1002/1521-1878(200012)22:12<1115::AID-BIES9>3.0.CO;2-R. [DOI] [PubMed] [Google Scholar]
- 15.Morelli G, Malorny B, Müller K, Seiler A, Wang J, del Valle J, Achtman M. Mol Microbiol. 1997;25:1047–1064. doi: 10.1046/j.1365-2958.1997.5211882.x. [DOI] [PubMed] [Google Scholar]
- 16.Zhu P, Morelli G, Achtman M. Mol Microbiol. 1999;33:635–650. doi: 10.1046/j.1365-2958.1999.01514.x. [DOI] [PubMed] [Google Scholar]
- 17.Gagneux S, Hodgson A, Erhard I, Morelli G, Genton B, Smith T, Tanner M, Binka F, Achtman M, Pluschke G. Trop Med Int Health. 2000;5:280–287. [PubMed] [Google Scholar]
- 18.Anonymous. Weekly Epidemiol. Rec. 1995. , October 6, 1995, pp. 281–282. [Google Scholar]
- 19.Malorny B, Morelli G, Kusecek B, Kolberg J, Achtman M. J Bacteriol. 1998;180:1323–1330. doi: 10.1128/jb.180.5.1323-1330.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Huson D H. Bioinformatics. 1998;14:68–73. doi: 10.1093/bioinformatics/14.1.68. [DOI] [PubMed] [Google Scholar]
- 21.Sham P C, Curtis D. Ann Hum Genet. 1995;59:97–105. doi: 10.1111/j.1469-1809.1995.tb01608.x. [DOI] [PubMed] [Google Scholar]
- 22.Day, N. P. J., Moore, C. E., Enright, M. C., Berendt, A. R., Maynard Smith, J., Murphy, M. D., Peacock, S. J., Spratt, B. G. & Feil, E. J. (2001)Science, in press. [DOI] [PubMed]
- 23.Eigen M. Trends Microbiol. 1996;4:216–218. doi: 10.1016/0966-842X(96)20011-3. [DOI] [PubMed] [Google Scholar]
- 24.Crowe B A, Wall R A, Kusecek B, Neumann B, Olyhoek T, Abdillahi H, Hassan-King M, Greenwood B M, Poolman J T, Achtman M. J Infect Dis. 1989;159:686–700. doi: 10.1093/infdis/159.4.686. [DOI] [PubMed] [Google Scholar]
- 25.Blakebrough I S, Greenwood B M, Whittle H C, Bradley A K, Gilles H M. J Infect Dis. 1982;146:626–637. doi: 10.1093/infdis/146.5.626. [DOI] [PubMed] [Google Scholar]
- 26.Scholten R J P M, Bijlmer H A, Poolman J T, Kuipers B, Caugant D A, van Alphen L, Dankert J, Valkenburg H A. J Infect Dis. 1993;16:237–246. doi: 10.1093/clind/16.2.237. [DOI] [PubMed] [Google Scholar]
- 27.Caugant D A, Høiby E A, Magnus P, Scheel O, Hoel T, Bjune G, Wedege E, Eng J, Frøholm L O. J Clin Microbiol. 1994;32:323–330. doi: 10.1128/jcm.32.2.323-330.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kwara A, Adegbola R A, Corrah P T, Weber M, Achtman M, Morelli G, Caugant D A, Greenwood B M. Trop Med Int Health. 1998;3:742–746. doi: 10.1046/j.1365-3156.1998.00300.x. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplemental Data