Theaflavin-3, 3’-digallate inhibits ovarian cancer stem cells via suppressing Wnt/β-Catenin signaling pathway (original) (raw)
. Author manuscript; available in PMC: 2019 Nov 1.
Abstract
Recent evidence indicates that ovarian cancer stem cells (CSCs) are responsible for ovarian cancer recurrence and drug resistance, resulting in the low long-term survival rate of patients with advanced ovarian cancer. We aimed to study the inhibitory effect of theaflavin-3, 3’-digallate (TF3), a black tea polyphenol on ovarian CSCs. Here, we showed that TF3 inhibited the proliferation of A2780/CP70 and OVCAR3 tumorshpere cells by suppressing their cell viability and colony formation capacity. TF3 inhibited the tumorsphere formation capacity of A2780/CP70 and OVCAR3 CSCs in serum-free and non-adherent conditions. TF3 inhibited A2780/CP70 and OVCAR3 CSCs isolated from tumorspheres by decreasing their cell viability and upregulating the protein expression of caspase-3 and −7 in the cells. We also revealed that TF3 inhibited ovarian CSCs through Wnt/β-catenin signaling pathway. Our results suggested that TF3 could inhibit ovarian CSCs and might be a potential agent for eradicating ovarian cancer.
Keywords: theaflavin-3, 3’-digallate, ovarian cancer stem cell, anti-proliferation, ALDH, β-Catenin
1. Introduction
Ovarian cancer ranks fifth in cancer related deaths among women and has the highest death rate among uterine, cervical, and ovarian cancers(Siegel, Miller, & Jemal, 2016). The combination of surgery and chemotherapy based on taxane and platinum is the standard therapy for advanced ovarian cancer(Zhan, Wang, & Ngai, 2013). Although the deeper understanding of ovarian cancer has rendered first-line chemotherapy effective, 70% of the patients relapse within 18 months of the chemotherapy(Sharma, Tandon, & Semwal, 2014). The long-term survival rate of patients with advanced ovarian cancer remains only less than 40%(Al Rawahi, et al., 2012). Recent evidences indicate that CSCs, a small subset of cancer cells with stem cell properties drive cancer relapse(Ahmed, Abubaker, Findlay, & Quinn, 2013; Reya, Morrison, Clarke, & Weissman, 2001). CSCs possess the capabilities to self-renew, differentiate, resist conventional chemotherapy and metastasis. Conventional chemotherapy is able to kill most cancer cells, but CSCs survive for their chemotherapy-resistance which further results in tumor recurrence in local and distant organs. CSCs in ovarian cancer are characterized by several markers including SOX2, Nanog, CD24, OCT4, CD133, CD44, CD117, ABCG2 and aldehyde dehydrogenase isoform 1 (ALDH1)(Bapat, 2010; Scarpa & Ninfali, 2015; Zeimet, et al., 2012). Nowadays, ALDH1 is widely used as the maker to identify ovarian cancer stem cells by using flow cytometry and fluorescent substrates in the Aldefluor kit(Ma & Allan, 2011). Several signaling pathway have been demonstrated to determine and regulate the self-renewal property of CSCs, including Hedgehog, Notch, and Wnt/β-catenin pathway(Li Song & Miele, 2005; Liu, et al., 2006; Smalley & Dale, 1999).
Considering the importance of CSCs in tumor initiation, progression, recurrence, and metastasis, it’s urgent to explore new drugs and novel therapies to target CSCs in ovarian cancer. Phytochemicals have already played an important and beneficial part in cancer treatment for over 50 years(Butler, Robertson, & Cooper, 2014; Cragg, Kingston, & Newman, 2011). About 49% of clinically used chemotherapy agents in Western countries over approximate 70 years were either obtained or derived from phytochemicals(Newman & Cragg, 2016). They are a proven resource for further exploring new anticancer drugs. Recent evidences indicate that some of the phytochemicals exhibiting inhibitory effects against a variety of cancer cells are also able to kill CSCs(Pistollato, et al., 2017; Singh, Sharma, Ghosh, Park, & Jeong, 2017), such as curcumin(Kakarala, et al., 2010; Tsai, et al., 2015), sulforaphane(Li, et al., 2010; Srivastava, Tang, Zhu, Meeker, & Shankar, 2011), β-carotene(Lee, Park, & Kim, 2013), piperine(Kakarala, et al., 2010), genistein(Dandawate, Padhye, Ahmad, & Sarkar, 2013) and (−)-epigallocatechin gallate (EGCG)(Chung & Vadgama, 2015).
Theaflavin-3, 3’-digallate (TF3), a polyphenolic compound extracted from black tea has exhibited potent anticancer properties. TF3 has been demonstrated to inhibit human liver, lung, gastric(K. Wang, et al., 2011), and breast cancer(Lin, Chen, & Lin-Shiau, 2006) as well as ovarian cancer(Gao, Rankin, Tu, & Chen, 2016; Tu, et al., 2016). TF3 was selective and cytotoxic to ovarian cancer cells and less cytotoxic to normal ovarian cells(Tu, et al., 2016). An ideal chemotherapy agent is expected to selectively target CSCs together with the non-CSCs in tumor bulk. Besides, TF3 is formed from the co-oxidation of (−)-epicatechin gallate (ECG) and EGCG during black tea production which possesses some functional groups of EGCG. Therefore, we investigated whether TF3 exposure inhibited the proliferation of ovarian CSCs and elucidated the underlying signaling pathways. The present study provides the first evidence that TF3 inhibited ovarian CSCs by suppressing Wnt/β-catenin pathway.
2. Materials and Methods
2.1. Cell culture and reagents
Cisplatin-resistant human ovarian cancer cell lines A2780/CP70 and OVCAR-3 were obtained from Dr. Βinghua Jiang (West Virginia University). All cells were cultured in RPMI-1640 medium (Sigma) supplemented with 10% fetal bovine serum (Invitrogen) and 1% penicillin-streptomycin (Sigma). The cells were cultured in a humidified incubator at 37˚C with 5% CO2. At about 80% confluence, the cells were detached from culture dishes with accutase solution (Stem cell Technologies) for passaging and further experiments. TF3 monomers were isolated and purified using a previously established method(Xu, Jin, Wu, & Tu, 2010). 10 mM TF3 stock solutions were prepared in DMSO (Sigma) and stored at −20°C.
The detached cells were suspended at 1×104 cells/mL in MammoCult medium (MammoCult basal medium (Stem cell Technologies) supplemented with 10% MammoCult proliferation supplement (Stem cell Technologies) and 1% penicillin-streptomycin). The suspended cells were seeded with 2 mL in 6-well ultra-low attachment plates (Corning) and cultured for 6 days to form 1st generation tumor spheres. The tumor spheres were detached with accutase solution and passaged twice. Then the 3rd generation tumor spheres were detached for further experiments.
2.2. Cell viability assay
Cells were seeded in 96-well ultra-low attachment plates (Corning) at 4×104 cells/well. Then the cells were treated with designated concentrations of TF3 or an equal amount of DMSO (as vehicle) for 24 h. After incubation, cell viability was assessed by MTS assay (Promega), according to the manufacturer’s instructions. Cell viability was expressed as a percentage compared to that of control cells.
2.3. Colony formation assay
Cells were added into 6-well ultra-low attachment plates at 4×105 cells/well and treated with different concentrations of TF3 (5, 10, and 20 μM) or an equal amount of DMSO (as vehicle) for 24 h. After incubation, the cells were further cultured for 7 days in 6-well plates with drug-free medium at 2×103 cells per well. Colonies were fixed with ice-cold methanol for 15 min and then stained with 0.5% crystal violet solution in 25% methanol for 10 min. The stained 6-well plates were carefully rinsed with distilled water several times and photographed directly.
2.4. Tumorsphere formation assay
Cells were added into 6-well ultra-low attachment plates at 4×105 cells/well and treated with designated concentrations of TF3 for 24 h. After incubation, the cells were collected and cultured in drug-free MammoCult medium in 6-well ultra-low attachment plates at 2×103 cells per well for 6 days. The spheroids were photographed under a microscope.
2.5. ALDH assay and ALDH based sorting
Cells were added into 6-well ultra-low attachment plates at 4×105 cells/well and treated with different concentrations of TF3 (0, 5, 10, and 20 μM) for 24 h. After incubation, ALDH assay was done using ALDEFLUOR kit according to the protocol instructions (Stem cell Technologies). The stained cells were analyzed by flow cytometry (FACSCalibur system, BD Biosciences).
For ALDH based sorting, ALDH assay was carried out. Then ALDH-positive (ALDH+) and –negative (ALDH−) cells were sorted using flow cytometry (FACSCalibur system, BD Biosciences).
2.6. Transfect ALDH+ cells with pcDNA3-S33Y β-Catenin plasmid
ALDH+ cells were seeded in 6-well ultra-low attachment plates at 4×105 cells/well. The cells were transfected with pcDNA3-S33Y β-Catenin plasmid (Addgene) using jetPrime transfection reagent (VWR International) following manufacturer’s protocol. After incubation for 4h, medium was replaced by MammoCult medium. The transfected cells were cultured for 24h and then used for further experiments.
2.7. Western blotting
After treatments, cells were lysed using M-PER Mammalian Protein Extraction Reagent (Pierce) supplemented with Halt™ Protease and Phosphatase Inhibitor Single-Use Cocktail (Life Technologies). The protein levels were determined using BCA Protein assay kit (Pierce). After boiling for 6 min in loading buffer (Bio-Rad), equal amounts of protein were subjected to sodium dodecyl sulfate polyacrylamide gel electrophoresis. The separated proteins were transferred onto a nitrocellulose blotting membrane. The membrane was blocked with 5% nonfat dried milk in Tris Buffered Saline (Bio-Rad) containing 0.1% Tween-20 (TBST) at room temperature for 1 h and subsequently incubated with specific primary antibodies overnight at 4 °C. The membrane was washed three times (10 min each) with TBST and then incubated with an appropriate secondary antibody conjugated with horseradish peroxide for 1 h at room temperature. After three 10 min washes with TBST, antigen-antibody complex in each blot was visualized with Super Signal West Dura Extended Duration Substrate (Life technologies) and ChemiDoc™ MP System (Bio-Rad). Protein bands were quantitated with NIH Image J software and normalized by GAPDH bands for analysis.
2.8. Statistical analysis
Unless otherwise noted, data are presented as mean ± SD from three-independent experiments. Student’s t test was used for comparisons of more than two independent groups. p< 0.05 was considered statistically significant, and p<0.01 was considered statistically highly significant.
3. Results
3.1. TF3 suppressed the proliferation of cells in ovarian tumorspheres
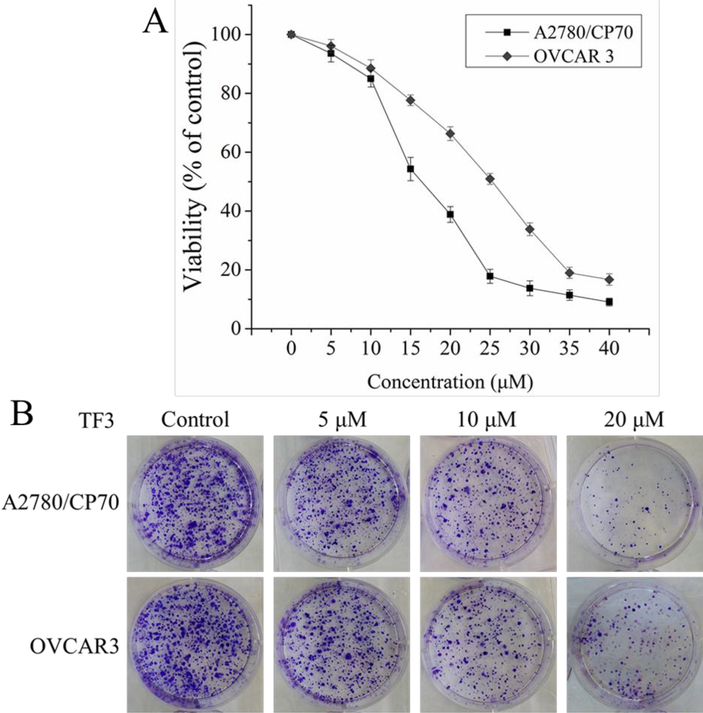
To evaluate the anti-proliferative effect of TF3 on the cells in tumorspheres from A2780/CP70 and OVCAR3 cells, we detected whether treatment with TF3 at the concentration of 5, 10, 15, 20, 25, 30, 35, and 40 μM affected the viability of tumorsphere cells using the MTS assay. As showed in Figure 1A, treatment with TF3 decreased the viability of both cells in a dose-dependent manner. The IC50 values of TF3 against A2780/CP70 and OVCAR3 tumorsphere cells were 16.29 and 21.20 μM which were evaluated by Statistical Analysis System. The concentration of 20 μM is close to both IC50 values. The concentrations of TF3 (5, 10, and 20 μM) were selected for further study.
Figure 1.
TF3 suppressed the proliferation of A2780/CP70 and OVCAR3 tumorsphere cells. (A) Treatment with TF3 at various concentrations inhibited the viability of both cells. All measurements were done in triplicates. Data represent means ± SD from three independent experiments. (B) Treatment with TF3 at designated concentrations inhibited the colony formation of both cells. The cells were treated with TF3 for 24h and cultured at a cell density of 2×103 per well for 7 days to form colony.
Colony formation assay is an effective method to evaluate the proliferation capacity of single cells. Therefore, we performed colony formation assay to further determine the anti-proliferative effect of TF3 on A2780/CP70 and OVCAR3 tumorsphere cells. As showed in Figure 1B, treatment of TF3 decreased the number of both cell colonies in a dose-dependent manner.
Thus, the results of cell viability assay and colony formation assay indicated that treatment with TF3 showed potent anti-proliferative effect on A2780/CP70 and OVCAR3 tumorsphere cells.
3.2. TF3 inhibited tumorsphere formation of ovarian CSCs
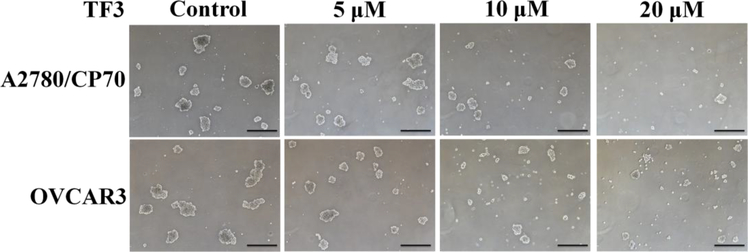
To study whether TF3 inhibited ovarian CSCs resulting in the anti-proliferative effect, tumorsphere formation assay was carried out to evaluate the inhibitory effect of TF3 against CSCs. As showed in Figure 2, treatment with TF3 decreased the size and number of both A2780/CP70 and OVCAR3 tumorspheres in a dose-dependent manner. The result indicated that treatment with TF3 exhibited potent inhibitory effect against ovarian CSCs.
Figure 2.
Treatment with TF3 at designated concentration inhibited tumorsphere formation of A2780/CP70 and OVCAR3 CSCs. The cells were treated with TF3 for 24h and cultured in drug-free medium in 6-well ultra-low attachment plates at 2×103 cells/well for 6 days to form spheroids. The tumorspheres were detected and photographed by Zeiss microscopy (magnification, ×40). Scale bar size is 100 μm.
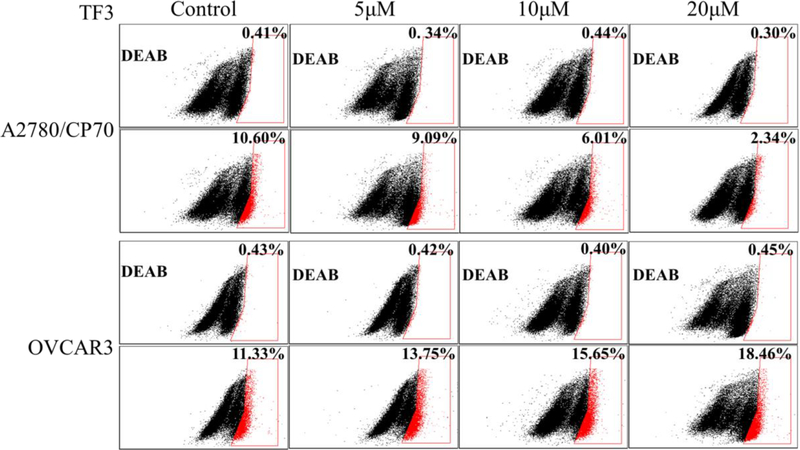
3.3. TF3 affected the percentage of ovarian CSCs in tumorspheres
Enhanced ALDH activity is a hallmark of CSCs. To verify whether TF3 targeted ovarian CSCs, ALDH assay was performed to determine the effect of TF3 on the percentage of ALDH+ cells in A2780/CP70 and OVCAR3 tumorspheres. As showed in Figure 3, treatment with TF3 decreased the percentage of ALDH+ cells in A2780/CP70 tumorspheres and increased the percentage of ALDH+ cells in OVCAR3 tumorspheres in a dose-dependent manner. Treatment with TF3 at the concentration of 5, 10, and 20 μM decreased the percentage of A2780/CP70 ALDH+ cells from 10.60% to 9.09, 6.01, and 2.34%, respectively. Treatment with TF3 at the concentration of 5, 10, and 20 μM increased the percentage of OVCAR3 ALDH+ cells from 11.33% to 13.75, 16.65, and 18.46%, respectively. The result indicated that treatment with TF3 exhibited stronger inhibitory effect against A2780/CP70 CSCs than non-CSCs and lower inhibitory effect against OVCAR3 CSCs than non-CSCs.
Figure 3.
Treatment with TF3 at designated concentration decreased the percentage of ALDH+ cells in A2780/CP70 tumorspheres and increased the percentage of ALDH+ cells in OVCAR3 tumorspheres. The tumorsphere cells were treated with TF3 for 24h and subjected to ALDH assay. The percentage of ALDH+ cells in each group was normalized by respective control (DEAB).
3.4. TF3 inhibited ovarian CSCs and non-CSCs
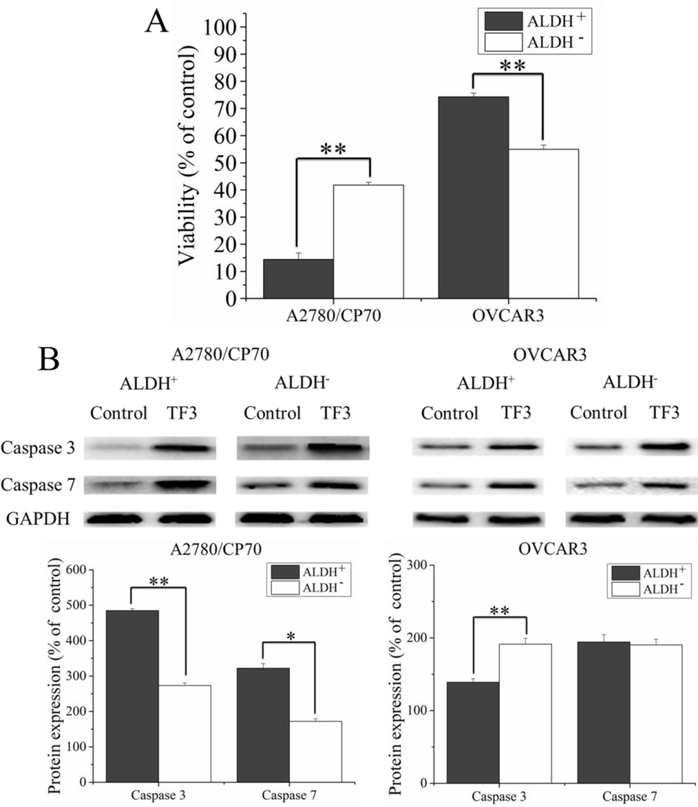
To confirm the inhibitory effect of TF3 against ovarian CSCs and non-CSCs, ALDH+ and ALDH− cells were sorted by flow cytometry. Then the sorted cells were treated with 20 μM TF3 and subjected to cell viability assay and western blot analysis. As showed in Figure 2A, treatment with TF3 inhibited the viability of ALDH+ and ALDH− cells at different degrees. Treatment with TF3 decreased the viability of ALDH+ and ALDH− cells sorted from A2780/CP70 tumorspheres by 85.6% and 58.2% and sorted from OVCAR3 tumorspheres by 25.7% and 35.0%, respectively. Treatment with TF3 showed stronger anti-proliferative effect against ALDH+ cells than ALDH− cells sorted from A2780/CP70 tumorspheres (p< 0.01). Treatment with TF3 showed stronger anti-proliferative effect against ALDH− cells than ALDH+ cells sorted from OVCAR3 tumorspheres (p< 0.01).
Furthermore, the protein levels of apoptosis-related proteins, including caspase −3 and −7 in ALDH+ and ALDH− cells were analyzed by western blot. As showed in Figure 2B, treatment with TF3 induced different upregulating effect on the protein levels of caspase −3 (17/19 kDa) and −7 (18 kDa) in ALDH+ and ALDH− cells. Treatment with TF3 increased the protein levels of caspase-3 by 4.84- and 2.74-fold and the protein levels of caspase-7 by 3.22- and 1.72-fold in ALDH+ cells and ALDH− cells sorted from A2780/CP70 tumorspheres. Treatment with TF3 increased the protein levels of caspase-3 by 1.39- and 1.91-fold and the protein levels of caspase-7 by 1.94- and 1.90-fold in ALDH+ cells and ALDH− cells sorted from OVCAR3 tumorspheres. The result indicated that treatment with TF3 exhibited stronger pro-apoptotic effect against ALDH+ cells than ALDH− cells sorted from A2780/CP70 tumorspheres by upregulating the protein levels of caspase −3 and −7 (p< 0.05). Treatment with TF3 exhibited stronger pro-apoptotic effect against ALDH− cells than ALDH+ cells sorted from OVCAR3 tumorspheres by upregulating the protein levels of caspase −3 (p< 0.01).
Taken together, treatment with TF3 exhibited stronger inhibitory effect against A2780/CP70 CSCs than non-CSCs and against OVCAR3 non-CSCs than CSCs.
3.5. TF3 suppressed ovarian CSCs through Wnt/β-catenin signaling pathway
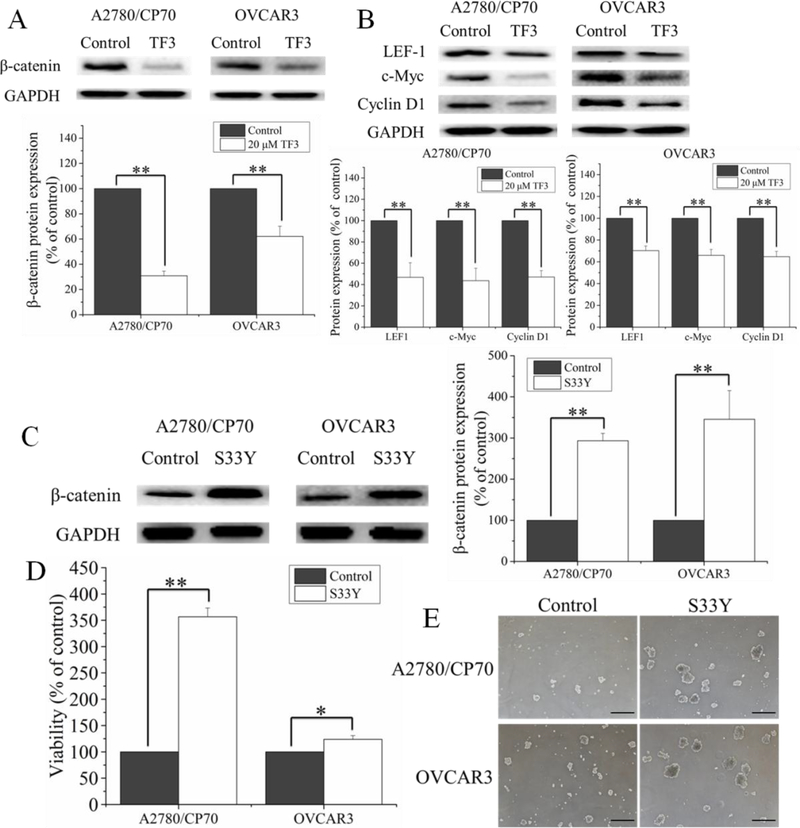
Wnt/β-catenin signaling pathway is identified to be a pivotal regulator of CSC phenotype in many tumors(de Sousa e Melo & Vermeulen, 2016). To clarify the role of Wnt/β-catenin signaling pathway in the inhibitory effect of TF3 on ovarian CSCs, the protein levels of β-catenin in A2780/CP70 and OVCAR3 ALDH+ cells were detected by western blotting. As shown in Figure 5A, treatment with 20 μM TF3 significantly downregulated the protein levels of β-catenin in A2780/CP70 and OVCAR3 ALDH+ cells (p< 0.01). Furthermore, the protein expression levels of β-catenin target, LEF-1 and TCF/LEF targets, c-Myc and cyclin D1 were detected by western blotting. As shown in Figure 5B, treatment with 20 μM TF3 significantly downregulated the protein levels of LEF-1, c-Myc and cyclin D1 (p< 0.01).
Figure 5.
TF3 inhibited ovarian CSCs through Wnt/β-catenin signaling pathway. (A) Treatment with 20 μM TF3 downregulated the protein levels of β-Catenin in ALDH+ cells sorted from A2780/CP70 and OVCAR3 tumorspheres. (B) Treatment with 20 μM TF3 downregulated the protein levels of LEF-1, c-Myc and cyclin D1 in ALDH+ cells sorted from A2780/CP70 and OVCAR3 tumorspheres. (C) S33Y transfection upregulated the protein levels of β-Catenin in ALDH+ cells sorted from A2780/CP70 and OVCAR3 tumorspheres. (D) S33Y transfection attenuated the inhibitory effect of 20 μM TF3 on viability of the ALDH+ cell sorted from A2780/CP70 and OVCAR3 tumorspheres. (E) S33Y transfection attenuated the inhibitory effect of 20 μM TF3 on the sphere formation of ALDH+ cell sorted from A2780/CP70 and OVCAR3 tumorspheres. The tumorspheres were detected and photographed by Zeiss microscopy (magnification, ×40). Scale bar size is 100 μm. Data represent means ± SD of three independent experiments. * p< 0.05, ** p< 0.01, compared with respective controls.
To further study the effect of β-catenin on the suppression of ovarian CSCs induced by TF3, β-catenin in A2780/CP70 and OVCAR3 ALDH+ cells was selectively overexpressed by β-Catenin plasmid (S33Y) transfection. As shown in Figure 5C, S33Y transfection significantly upregulated the protein levels of β-catenin in A2780/CP70 and OVCAR3 ALDH+ cells (p< 0.01). β-Catenin overexpression significantly attenuated the inhibitory effect of 20 μM TF3 on the cell viability (p < 0.05 or 0.01) (Figure 5D) and tumorsphere formation (Figure 5E) of A2780/CP70 and OVCAR3 ALDH+ cells.
Taken together, these results suggested that TF3 suppressed ovarian CSCs through Wnt/β-catenin signaling pathway.
4. Discussion
Ovarian cancer is the most lethal gynecological cancer. Acquired resistance to chemotherapy after the conventional therapy and high recurrence rate render the long-term survival rate of patients with advanced ovarian cancer low(Paclitaxel, 2017). Increasing evidence supports the CSC concept and demonstrates that chemotherapies are effective to eradicate differentiated ovarian cancer cells, but is hard to effectively eliminate CSCs(Ahmed, et al., 2013). CSCs are believed to be the cause of recurrence and drug resistance(Steg, et al., 2012). Thus, targeting CSCs becomes a fundamental treatment strategy for cancers. TF3 is one of the major bioactive polyphenols in black tea which was an important dietary source of flavonols, and whose consumption could lower ovarian cancer risk(Cassidy, Huang, Rice, Rimm, & Tworoger, 2014). Our previous study has shown that TF3 could suppress human ovarian cancer cells by inducing apoptosis and cell cycle arrest and inhibiting angiogenesis(Gao, et al., 2016; Tu, et al., 2016). Whether TF3 could inhibit the proliferation of ovarian CSCs is still not been investigated yet. This study is the first to show that TF3 inhibited ovarian CSCs by suppressing Wnt/β-catenin pathway.
In the current study, we found that TF3 exhibited anti-proliferative effect against A2780/CP70 and OVCAR3 tumorsphere cells using MTS assay and colony formation assay. In vitro tumorsphere formation assay is an effective method to evaluate the proliferative capacity of CSCs. Only CSCs can survive and proliferate in serum-free, non-adherent conditions(Pastrana, Silva-Vargas, & Doetsch, 2011). TF3 inhibited the tumorsphere formation capacity of tumorsphere cells, showing anti-proliferative effect against ovarian CSCs. Enhanced ALDH activity is a hallmark of CSCs(Marcato, Dean, Giacomantonio, & Lee, 2011). The ALDH activity of CSCs is widely believed to be determined by ALDH1A1 which is one of 19 ALDH isoforms expressed in humans. High ALDH expression was involved in the resistance to chemotherapy and the cancer relapse in in ovarian cancer patients(Burgos-Ojeda, Rueda, & Buckanovich, 2012; Y.-C. Wang, et al., 2012). The ALDH activity in tumorsphere cells treated with TF3 was detected using ALDEFLUOR assay followed by flow cytometry analysis. Treatment with TF3 decreased the percentage of ALDH+ cells in A2780/CP70 tumorspheres and increased the percentage of ALDH+ cells in OVCAR3 tumorspheres which indicated that TF3 showed different inhibitory effect against ALDH+/− cells in A2780/CP70 and OVCAR3 tumorspheres. To further study the inhibitory effect of TF3 against ALDH+ and ALDH− cells in the tumorspheres, the ALDH+ and ALDH− cells were sorted, treated with TF3 and subjected to cell viability assay and western blot analysis. Treatment with TF3 exhibited stronger inhibitory effect against A2780/CP70 ALDH+ cells than ALDH− cells and lower inhibitory effect against OVCAR3 ALDH+ cells than ALDH− cells. Taken together, TF3 showed different inhibitory effect against ovarian CSCs and non-CSCs.
Wnt/β-catenin signaling pathway played an important role in the proliferation and self-renew of CSCs(de Sousa e Melo & Vermeulen, 2016). Activated Wnt signaling pathway was able to inhibit GSK-3β, protecting β-catenin from phosphorylation and degradation. The stable β-catenin could bind to the members of the TCF/LEF family of transcription factors in the nucleus, thus modulating the expression of self-renew target genes. It has been demonstrated that EGCG(Kim, et al., 2006), piperine(Kakarala, et al., 2010), sulforaphane(Li, et al., 2010), β-carotene(Lee, et al., 2013), quercetin(Zhou, et al., 2010), resveratrol(Shankar, et al., 2011), and curcumin(Teiten, et al., 2011) was able to kill CSCs through inhibiting the Wnt/β-catenin signaling pathway. Targeting Wnt/β-catenin signaling pathway is an effective strategy for eliminating CSCs by using phytochemicals. Our results showed that TF3 downregulated the protein levels of β-catenin, LEF-1, c-Myc and cyclin D1 in ovarian CSCs. β-Catenin overexpression attenuated the inhibitory effect of TF3 on the viability and tumorsphere formation capacity of ovarian CSCs. The present study showed TF3 was able to suppress the proliferation of ovarian CSCs through inhibiting Wnt/β-catenin signaling pathway.
5. Conclusion
TF3 could inhibit the proliferation of A2780/CP70 and OVCAR3 CSCs and non-CSCs. TF3 exhibited stronger inhibitory effect against A2780/CP70 CSCs than non-CSCs and against OVCAR3 non-CSCs than CSCs. TF3 could inhibit A2780/CP70 and OVCAR3 CSCs through Wnt/β-catenin signaling pathway. Our results suggested that TF3 could inhibit ovarian CSCs and might be a potential agent for eradicating ovarian cancer.
Figure 4.
Treatment with 20 μM TF3 showed different effect on ALDH+ cells and ALDH− cells sorted from A2780/CP70 and OVCAR3 tumorspheres. (A) Treatment with TF3 inhibited the viability of ALDH+ and ALDH− cells sorted from A2780/CP70 and OVCAR3 tumorspheres at different degrees. (B) Treatment with TF3 upregulated the protein levels of caspase-3 (17/19 kDa) and −7 (18 kDa) in ALDH+ and ALDH− cells sorted from A2780/CP70 and OVCAR3 tumorspheres at different degrees. Data were represented as means ± SD of three independent experiments. * p< 0.05, ** p< 0.01, compared with respective controls.
Highlights.
- TF3 inhibited the proliferation of ovarian CSCs and non-CSCs.
- TF3 exhibited different inhibitory effect against ovarian CSCs and non-CSCs.
- TF3 inhibited A2780/CP70 CSCs more strongly than non-CSCs.
- TF3 inhibited OVCAR3 non-CSCs more strongly than CSCs.
- TF3 inhibited ovarian CSCs through Wnt/β-catenin signaling pathway.
Acknowledgments
We thank Dr. Kathy Brundage from the Flow Cytometry Core at the West Virginia University for providing technical help on apoptosis analysis. This research was supported by Collaborative Innovation Center of Chinese Oolong Tea Industry (2015) 75 and Key Research and Development Projects of Industrialization Model Projects on Exploring Functional Components and Related Products from Tea Flowers and Fruit (2018C02012). This research was also supported by NIH grants P20RR016477 from the National Center for Research Resources and P20GM103434 from the National Institute for General Medical Sciences (NIGMS) awarded to the West Virginia IDeA Network of Biomedical Research Excellence. This study was also supported by Grant Number P20GM104932 from NIGMS, a component of the National Institutes of Health (NIH) and its contents are solely the responsibility of the authors and do not necessarily represent the official view of NIGMS or NIH. This study was also supported by COBRE grant GM102488/RR032138, ARIA S10 grant RR020866, FORTESSA S10 grant OD016165 and INBRE grant GM103434.
Footnotes
Conflicts of interest
The authors declare that they have no competing interests.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Ahmed N, Abubaker K, Findlay J, & Quinn M (2013). Cancerous ovarian stem cells: obscure targets for therapy but relevant to chemoresistance. Journal of cellular biochemistry, 114, 21–34. [DOI] [PubMed] [Google Scholar]
- Al Rawahi T, Lopes A, Bristow R, Bryant A, Elattar A, Chattopadhyay S, & Galaal K (2012). Surgical cytoreduction for recurrent epithelial ovarian cancer. Cochrane Database Syst Rev, 2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bapat SA (2010). Human ovarian cancer stem cells. Reproduction, 140, 33–41. [DOI] [PubMed] [Google Scholar]
- Burgos-Ojeda D, Rueda BR, & Buckanovich RJ (2012). Ovarian cancer stem cell markers: prognostic and therapeutic implications. Cancer letters, 322, 1–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Butler MS, Robertson AA, & Cooper MA (2014). Natural product and natural product derived drugs in clinical trials. Natural product reports, 31, 1612–1661. [DOI] [PubMed] [Google Scholar]
- Cassidy A, Huang T, Rice MS, Rimm EB, & Tworoger SS (2014). Intake of dietary flavonoids and risk of epithelial ovarian cancer. The American journal of clinical nutrition, ajcn. 088708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chung SS, & Vadgama JV (2015). Curcumin and epigallocatechin gallate inhibit the cancer stem cell phenotype via down-regulation of STAT3–NFκB signaling. Anticancer research, 35, 39–46. [PMC free article] [PubMed] [Google Scholar]
- Cragg GM, Kingston DG, & Newman DJ (2011). Anticancer agents from natural products: CRC press. [Google Scholar]
- Dandawate P, Padhye S, Ahmad A, & Sarkar FH (2013). Novel strategies targeting cancer stem cells through phytochemicals and their analogs. Drug delivery and translational research, 3, 165–182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Sousa e Melo, F., & Vermeulen L (2016). Wnt signaling in cancer stem cell biology. Cancers, 8, 60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gao Y, Rankin GO, Tu Y, & Chen YC (2016). Theaflavin-3, 3’-digallate decreases human ovarian carcinoma OVCAR-3 cell-induced angiogenesis via Akt and Notch-1 pathways, not via MAPK pathways. International journal of oncology, 48, 281–292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kakarala M, Brenner DE, Korkaya H, Cheng C, Tazi K, Ginestier C, Liu S, Dontu G, & Wicha MS (2010). Targeting breast stem cells with the cancer preventive compounds curcumin and piperine. Breast cancer research and treatment, 122, 777–785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim J, Zhang X, Rieger-Christ KM, Summerhayes IC, Wazer DE, Paulson KE, & Yee AS (2006). Suppression of Wnt signaling by the green tea compound (–)-epigallocatechin 3-gallate (EGCG) in invasive breast cancer cells requirement of the transcriptional repressor HBP1. Journal of Biological Chemistry, 281, 10865–10875. [DOI] [PubMed] [Google Scholar]
- Lee HA, Park S, & Kim Y (2013). Effect of β-carotene on cancer cell stemness and differentiation in SK-N-BE (2) C neuroblastoma cells. Oncology reports, 30, 1869–1877. [DOI] [PubMed] [Google Scholar]
- Li Song L, & Miele L (2005). Role of Notch signaling in cell-fate determination of human mammary stem/progenitor cells. The Women’s Oncology Review, 5, 9–11. [Google Scholar]
- Li Y, Zhang T, Korkaya H, Liu S, Lee H-F, Newman B, Yu Y, Clouthier SG, Schwartz SJ, & Wicha MS (2010). Sulforaphane, a dietary component of broccoli/broccoli sprouts, inhibits breast cancer stem cells. Clinical Cancer Research, 16, 2580–2590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin J-K, Chen Y-W, & Lin-Shiau S-Y (2006). Inhibition of breast cancer cell proliferation by theaflavins from black tea through suppressing proteasomal activities. In: AACR. [Google Scholar]
- Liu S, Dontu G, Mantle ID, Patel S, Ahn N. s., Jackson KW, Suri P, & Wicha MS (2006). Hedgehog signaling and Bmi-1 regulate self-renewal of normal and malignant human mammary stem cells. Cancer research, 66, 6063–6071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma I, & Allan AL (2011). The role of human aldehyde dehydrogenase in normal and cancer stem cells. Stem cell reviews and reports, 7, 292–306. [DOI] [PubMed] [Google Scholar]
- Marcato P, Dean CA, Giacomantonio CA, & Lee PW (2011). Aldehyde dehydrogenase: its role as a cancer stem cell marker comes down to the specific isoform. Cell cycle, 10, 1378–1384. [DOI] [PubMed] [Google Scholar]
- Newman DJ, & Cragg GM (2016). Natural products as sources of new drugs from 1981 to 2014. J. Nat. Prod, 79, 629–661. [DOI] [PubMed] [Google Scholar]
- Paclitaxel TV (2017). Recurrent Epithelial Ovarian Cancer. Clinical Trials in Ovarian Cancer, 200. [Google Scholar]
- Pastrana E, Silva-Vargas V, & Doetsch F (2011). Eyes wide open: a critical review of sphere-formation as an assay for stem cells. Cell stem cell, 8, 486–498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pistollato F, Iglesias RC, Ruiz R, Aparicio S, Crespo J, Lopez LD, Giampieri F, & Battino M (2017). The use of natural compounds for the targeting and chemoprevention of ovarian cancer. Cancer Letters, 411, 191–200. [DOI] [PubMed] [Google Scholar]
- Reya T, Morrison SJ, Clarke MF, & Weissman IL (2001). Stem cells, cancer, and cancer stem cells. nature, 414, 105. [DOI] [PubMed] [Google Scholar]
- Scarpa E-S, & Ninfali P (2015). Phytochemicals as innovative therapeutic tools against cancer stem cells. International journal of molecular sciences, 16, 15727–15742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shankar S, Nall D, Tang S-N, Meeker D, Passarini J, Sharma J, & Srivastava RK (2011). Resveratrol inhibits pancreatic cancer stem cell characteristics in human and KrasG12D transgenic mice by inhibiting pluripotency maintaining factors and epithelial-mesenchymal transition. PloS one, 6, e16530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sharma S, Tandon S, & Semwal B (2014). Momordica charantia Linn.: a comprehensive review on bitter remedy. JOURNAL OF PHARMACEUTICAL RESEARCH & OPINION, 1. [Google Scholar]
- Siegel RL, Miller KD, & Jemal A (2016). Cancer statistics, 2016. CA: a cancer journal for clinicians, 66, 7–30. [DOI] [PubMed] [Google Scholar]
- Singh AK, Sharma N, Ghosh M, Park YH, & Jeong DK (2017). Emerging importance of dietary phytochemicals in fight against cancer: Role in targeting cancer stem cells. Crit Rev Food Sci Nutr, 57, 3449–3463. [DOI] [PubMed] [Google Scholar]
- Smalley MJ, & Dale TC (1999). Wnt signalling in mammalian development and cancer. Cancer and Metastasis Reviews, 18, 215–230. [DOI] [PubMed] [Google Scholar]
- Srivastava RK, Tang S-N, Zhu W, Meeker D, & Shankar S (2011). Sulforaphane synergizes with quercetin to inhibit self-renewal capacity of pancreatic cancer stem cells. Frontiers in bioscience (Elite edition), 3, 515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steg AD, Bevis KS, Katre AA, Ziebarth A, Dobbin ZC, Alvarez RD, Zhang K, Conner M, & Landen CN (2012). Stem cell pathways contribute to clinical chemoresistance in ovarian cancer. Clinical Cancer Research, 18, 869–881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Teiten M-H, Gaascht F, Cronauer M, Henry E, Dicato M, & Diederich M (2011). Anti-proliferative potential of curcumin in androgen-dependent prostate cancer cells occurs through modulation of the Wingless signaling pathway. International journal of oncology, 38, 603–611. [DOI] [PubMed] [Google Scholar]
- Tsai C-F, Hsieh T-H, Lee J-N, Hsu C-Y, Wang Y-C, Kuo K-K, Wu H-L, Chiu C-C, Tsai E-M, & Kuo P-L (2015). Curcumin suppresses phthalate-induced metastasis and the proportion of cancer stem cell (CSC)-like cells via the inhibition of AhR/ERK/SK1 signaling in hepatocellular carcinoma. Journal of agricultural and food chemistry, 63, 10388–10398. [DOI] [PubMed] [Google Scholar]
- Tu Y, Kim E, Gao Y, RANkIN GO, Li B, & ChEN YC (2016). Theaflavin-3, 3’-digallate induces apoptosis and G2 cell cycle arrest through the Akt/MDM2/p53 pathway in cisplatin-resistant ovarian cancer A2780/CP70 cells. International journal of oncology, 48, 2657–2665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang K, Liu Z, Huang J, BEKHIT AED, Liu F, Dong X, Gong Y, & Fu D (2011). The inhibitory effects of pure black tea theaflavins on the growth of four selected human cancer CELLS. Journal of Food Biochemistry, 35, 1561–1567. [Google Scholar]
- Wang Y-C, Yo Y-T, Lee H-Y, Liao Y-P, Chao T-K, Su P-H, & Lai H-C (2012). ALDH1-bright epithelial ovarian cancer cells are associated with CD44 expression, drug resistance, and poor clinical outcome. The American journal of pathology, 180, 1159–1169. [DOI] [PubMed] [Google Scholar]
- Xu Y, Jin Y, Wu Y, & Tu Y (2010). Isolation and purification of four individual theaflavins using semi-preparative high performance liquid chromatography. Journal of Liquid Chromatography & Related Technologies, 33, 1791–1801. [Google Scholar]
- Zeimet A, Reimer D, Sopper S, Boesch M, Martowicz A, Roessler J, Wiedemair A, Rumpold H, Untergasser G, & Concin N (2012). Ovarian cancer stem cells. Neoplasma, 59, 747. [DOI] [PubMed] [Google Scholar]
- Zhan Q, Wang C, & Ngai S (2013). Ovarian cancer stem cells: a new target for cancer therapy. BioMed research international, 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou W, Kallifatidis G, Baumann B, Rausch V, Mattern J, Gladkich J, Giese N, Moldenhauer G, Wirth T, & Büchler MW (2010). Dietary polyphenol quercetin targets pancreatic cancer stem cells. International journal of oncology, 37, 551–561. [DOI] [PubMed] [Google Scholar]