Cytoplasmic viral replication complexes - PubMed (original) (raw)
Review
Cytoplasmic viral replication complexes
Johan A den Boon et al. Cell Host Microbe. 2010.
Abstract
Many viruses that replicate in the cytoplasm compartmentalize their genome replication and transcription in organelle-like structures that enhance replication efficiency and protection from host defenses. In particular, recent studies with diverse positive-strand RNA viruses have further elucidated the ultrastructure of membrane-bound RNA replication complexes and how these complexes function in close coordination with virion assembly and budding. The structure, function, and assembly of some positive-strand RNA virus replication complexes have parallels and potential evolutionary links with the replicative cores of double-strand RNA virus and retrovirus virions and more general similarities with the replication factories of cytoplasmic DNA viruses.
Copyright 2010 Elsevier Inc. All rights reserved.
Figures
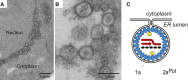
Figure 1
BMV-Induced Perinuclear ER RNA Replication Vesicles and Model of the BMV RNA Replication Complex (A and B) (A) and (B) show low- and high-magnification EM images, respectively, of BMV-induced spherular vesicles invaginated into the perinuclear ER membrane (adapted from Schwartz et al., 2002). (C) Diagram of the vesicular BMV RNA replication complex, illustrating the many copies of the membrane-bound, self-interacting 1a protein (blue) that induce vesicle formation, the ∼20-fold fewer copies of 2a polymerase (yellow), and replicative intermediate RNAs, including positive-strand RNA (red arrows) and negative-strand RNA (black dashed arrow).
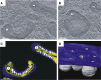
Figure 2
FHV-Induced RNA Replication Vesicles on Outer Mitochondrial Membranes (A and B) (A) and (B) show two of a series of multiple EM tomographic images from different planes of a single thick section of FHV-modified mitochondria in an infected Drosophila cell. Labels 1 and 2 refer to the same mitochondria in both panels. (C) Three-dimensional reconstruction by EM tomography of the mitochondria in (A), showing outer (blue) and inner (yellow) mitochondrial membranes and FHV-induced replication vesicles (white). (D) Angled view of the side and top of RNA replication vesicles showing open neck-like connections with the cytoplasm (adapted from Kopek et al., 2007).
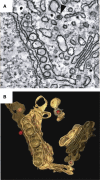
Figure 3
EM Tomographic Three-Dimensional Reconstruction of SARS Coronavirus-Induced, ER-Derived Double-Membrane Vesicles (A and B) Two-dimensional EM sectional view (A) and three-dimensional tomographic reconstruction (B) of SARS coronavirus-induced double-membrane vesicles (yellow/blue) and convoluted membrane structures (brown) (adapted from Knoops et al., 2008).
Figure 4
EM Tomographic Three-Dimensional Reconstruction of Dengue Virus-Induced, ER-Derived Vesicle Packets (A and B) Two-dimensional EM sectional view (A) and three-dimensional tomographic reconstruction (B) of DENV-induced vesicle packets. Virion particles associated with sites of virus assembly and budding are indicated with a black arrowhead in (A) and in red in (B) (adapted from Welsch et al., 2009).
Figure 5
Parallels between Positive-Strand RNA Virus, dsRNA Virus, and Retrovirus Genome Replication (A–C) Schematic representations of (A) the cytoplasmic replicative core of a dsRNA virus, (B) the invaginated, ER membrane-associated RNA replication complex of brome mosaic virus, and (C) a retrovirus virion in the midst of budding from the plasma membrane into the extracellular space. Red arrows represent the positive-strand (i.e., mRNA-sense) viral genomic RNA packaged into each structure to initiate genome replication. Dashed black arrows represent negative-strand RNA in (A) and (B) and negative-strand cDNA in (C), as is initiated prior to virion release for foamy retroviruses.
Similar articles
- DNA virus replication compartments.
Schmid M, Speiseder T, Dobner T, Gonzalez RA. Schmid M, et al. J Virol. 2014 Feb;88(3):1404-20. doi: 10.1128/JVI.02046-13. Epub 2013 Nov 20. J Virol. 2014. PMID: 24257611 Free PMC article. Review. - Virus evolution: fitting lifestyles to a T.
Ahlquist P. Ahlquist P. Curr Biol. 2005 Jun 21;15(12):R465-7. doi: 10.1016/j.cub.2005.06.016. Curr Biol. 2005. PMID: 15964268 Review. - Alternate, virus-induced membrane rearrangements support positive-strand RNA virus genome replication.
Schwartz M, Chen J, Lee WM, Janda M, Ahlquist P. Schwartz M, et al. Proc Natl Acad Sci U S A. 2004 Aug 3;101(31):11263-8. doi: 10.1073/pnas.0404157101. Epub 2004 Jul 27. Proc Natl Acad Sci U S A. 2004. PMID: 15280537 Free PMC article. - Organelle-like membrane compartmentalization of positive-strand RNA virus replication factories.
den Boon JA, Ahlquist P. den Boon JA, et al. Annu Rev Microbiol. 2010;64:241-56. doi: 10.1146/annurev.micro.112408.134012. Annu Rev Microbiol. 2010. PMID: 20825348 Review. - Parallels among positive-strand RNA viruses, reverse-transcribing viruses and double-stranded RNA viruses.
Ahlquist P. Ahlquist P. Nat Rev Microbiol. 2006 May;4(5):371-82. doi: 10.1038/nrmicro1389. Nat Rev Microbiol. 2006. PMID: 16582931 Free PMC article. Review.
Cited by
- Five questions on the cell-to-cell movement of Orthotospoviruses.
Singh P, Raj R, Savithri HS. Singh P, et al. BBA Adv. 2024 Oct 16;6:100124. doi: 10.1016/j.bbadva.2024.100124. eCollection 2024. BBA Adv. 2024. PMID: 39498475 Free PMC article. - The roles and mechanisms of endoplasmic reticulum stress-mediated autophagy in animal viral infections.
Chen L, Wei M, Zhou B, Wang K, Zhu E, Cheng Z. Chen L, et al. Vet Res. 2024 Sep 3;55(1):107. doi: 10.1186/s13567-024-01360-4. Vet Res. 2024. PMID: 39227990 Free PMC article. Review. - Mouse guanylate-binding proteins of the chromosome 3 cluster do not mediate antiviral activity in vitro or in mouse models of infection.
Tessema MB, Feng S, Enosi Tuipulotu D, Farrukee R, Ngo C, Gago da Graça C, Yamomoto M, Utzschneider DT, Brooks AG, Londrigan SL, Man SM, Reading PC. Tessema MB, et al. Commun Biol. 2024 Aug 25;7(1):1050. doi: 10.1038/s42003-024-06748-8. Commun Biol. 2024. PMID: 39183326 Free PMC article. - The astrovirus N-terminal nonstructural protein anchors replication complexes to the perinuclear ER membranes.
Ali H, Noyvert D, Hankinson J, Lindsey G, Lulla A, Lulla V. Ali H, et al. PLoS Pathog. 2024 Jul 15;20(7):e1011959. doi: 10.1371/journal.ppat.1011959. eCollection 2024 Jul. PLoS Pathog. 2024. PMID: 39008516 Free PMC article. - Megataxonomy and global ecology of the virosphere.
Koonin EV, Kuhn JH, Dolja VV, Krupovic M. Koonin EV, et al. ISME J. 2024 Jan 8;18(1):wrad042. doi: 10.1093/ismejo/wrad042. ISME J. 2024. PMID: 38365236 Free PMC article. Review.
References
- Annamalai P., Rao A.L. Replication-independent expression of genome components and capsid protein of brome mosaic virus in planta: a functional role for viral replicase in RNA packaging. Virology. 2005;338:96–111. - PubMed
Publication types
MeSH terms
Grants and funding
- R01 GM035072/GM/NIGMS NIH HHS/United States
- T32 AI078985/AI/NIAID NIH HHS/United States
- R37 GM035072/GM/NIGMS NIH HHS/United States
- HHMI/Howard Hughes Medical Institute/United States
- GM35072/GM/NIGMS NIH HHS/United States
- R01 GM035072-24/GM/NIGMS NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical