Aging is associated with an increase in T cells and inflammatory macrophages in visceral adipose tissue - PubMed (original) (raw)
Comparative Study
. 2011 Dec 15;187(12):6208-16.
doi: 10.4049/jimmunol.1102188. Epub 2011 Nov 9.
Affiliations
- PMID: 22075699
- PMCID: PMC3237772
- DOI: 10.4049/jimmunol.1102188
Comparative Study
Aging is associated with an increase in T cells and inflammatory macrophages in visceral adipose tissue
Carey N Lumeng et al. J Immunol. 2011.
Abstract
Age-related adiposity has been linked to chronic inflammatory diseases in late life. To date, the studies on adipose tissue leukocytes and aging have not taken into account the heterogeneity of adipose tissue macrophages (ATMs), nor have they examined how age impacts other leukocytes such as T cells in fat. Therefore, we have performed a detailed examination of ATM subtypes in young and old mice using state of the art techniques. Our results demonstrate qualitative changes in ATMs with aging that generate a decrease in resident type 2 (M2) ATMs. The profile of ATMs in old fat shifts toward a proinflammatory environment with increased numbers of CD206(-)CD11c(-) (double-negative) ATMs. The mechanism of this aging-induced shift in the phenotypic profile of ATMs was found to be related to a decrease in peroxisome proliferator-activated receptor-γ expression in ATMs and alterations in chemokine/chemokine receptor expression profiles. Furthermore, we have revealed a profound and unexpected expansion of adipose tissue T cells in visceral fat with aging that includes a significant induction of regulatory T cells in fat. Our findings demonstrate a unique inflammatory cell signature in the physiologic context of aging adipose tissue that differs from those induced in setting of diet-induced obesity.
Figures
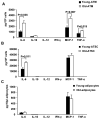
Figure 1. Cytokine production by adipose tissue cells
SVFs and adipocytes from young and old mice were cultured for 24 hours. The conditioned medium was collected to measure cytokine release using the Mouse Inflammatory Cytometric Bead Array (BD Bioscience). Cytokine release from (A) total ATMs (CD11b+), (B) ATSC (CD11b-), and (C) adipocytes. The results represent the mean ± SEM; n=4-6 experiments, representing a total of 15 old and 40 young mice.
Figure 2. Analysis of ATM content in old and young mice
(A) Quantitation of total SVF cells isolated from epididymal fat pads from 6m (black bars)and 22m old (white bars)C57 mice. (B) SVF cells normalized to fat pad mass. (C) ATMs (F4/80+CD11b+) were quantified by flow cytometry. (D) Immunofluorescence localization of resident Type 2 ATMs (MGL1+; green) in young and old mice. Vasculature labeled with isolectin (red). The results represent the mean ± SEM; n=3-5 experiments, representing a total of 12 old and 35 young mice.*_p_-value<0.05.
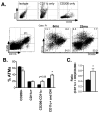
Figure 3. Alterations in ATM subtypes with age
(A) Flow cytometry delineation of ATM subtypes. Definition of control gates is shown (upper panel). After gating for ATMs (F4/80+CD11b+; P1), subtypes were differentiated by CD11c and CD206 staining in young and old mice (representative plots shown in lower panel). (B) ATM subtypes quantified as a percentage of the total ATM population in 6mo (black bars) and 22mo mice (white bars). (C) Ratio of CD11c+ and DN ATMs relative to resident CD206+ ATMs. *_p_-value<0.05.
Figure 4. Paracrine signals from ATM and ATSC
Conditioned medium from young and old ATM (A) and ATSC (CD11b-) (B) was incubated with peritoneal macrophages for 24 hours and the cytokines released were measured by CBA. The results represent the mean ± SEM; n=4 experiments, representing a total of 20 old and 28 young mice.
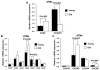
Figure 5. Young and old ATM chemokine receptor expression
(A) Real time PCR was performed to compare the expression of chemokine receptor CCR2,and CX3CR1expression in young and old ATMs. Other CCR (B) and CXCR (C) chemokine receptors were also examined. The results represent the mean ± SEM; n=4-5 experiments, representing a total of 25 old and 40 young mice.
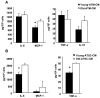
Figure 6. Effects of rosiglitazone on PPARγ gene expression by PCR
Real time RT-PCR analysis of Pparg expression, relative to the control gene guanine nucleotide binding protein (G protein), beta polypeptide 2 like 1 (GNB2L1). (A) Decreased PPARγ expression is seen in old ATMs but not adipocytes. (B) Short term (14 days) treatment of rosiglitazone increased PPARγ expression in old ATMs. (C) Rosiglitazone treatment increased Il10 and reduced Tnfa and Il6 expression in old ATMs. The results represent the mean ± SEM; n=7-10 experiments, representing a total of 30 old and 49 young mice.
Figure 7. – Increase in ATT cells with age
Flow cytometry analysis of ATT cells from SVF of epididymal fat from 6mo (black bars) and 22mo mice (white bars). (A) Percent CD3+ cells in fat. (B) Percentage of T cell subtypes in adipose tissue. Cells were gated on SSClowCD3+ cells in adipose tissue prior to analysis. (C) Quantitation of adipose tissue Tregs. CD3+CD4+FoxP3+ (Treg) and CD3+CD4+FoxP3- (Tconv) were quantitated in young and old mice. (D) Splenic CD3+ cells. (E) T cell subsets and (F)Treg content in spleens of young and old mice. (G) Blood T cell subsets (gated on CD3+ cells). Results represent the mean ± SEM, n=5 per group. *_p_-value <0.05.
Figure 8. – Enlargement of fat associated lymphoid clusters with age
Omental fat pads were dissected and stained with anti-CD4 antibodies to identify FALCs/milky spots. Images from representative samples from young (A) and old (B) mice. Low (left panels) and high power (right) images are shown. Bar=500μm. Right panels are enlargements of the boxed area. Similar results obtained from 5 independent samples. (C) FALC size frequency distribution in omental fat with age. CD4+ FALCs were sized from 4 mice. (D) FALC size represented as mean +/- SEM. *_p_-value<0.001.
Similar articles
- Obesity retunes turnover kinetics of tissue-resident macrophages in fat.
Chen Q, Ruedl C. Chen Q, et al. J Leukoc Biol. 2020 May;107(5):773-782. doi: 10.1002/JLB.1MA1219-275R. Epub 2020 Jan 17. J Leukoc Biol. 2020. PMID: 31951046 - High-fat diet triggers obesity-related early infiltration of macrophages into adipose tissue and transient reduction of blood monocyte count.
Liu Y, Lu X, Li X, Du P, Qin G. Liu Y, et al. Mol Immunol. 2020 Jan;117:139-146. doi: 10.1016/j.molimm.2019.11.002. Epub 2019 Nov 25. Mol Immunol. 2020. PMID: 31778913 - Frontline Science: Rapid adipose tissue expansion triggers unique proliferation and lipid accumulation profiles in adipose tissue macrophages.
Muir LA, Kiridena S, Griffin C, DelProposto JB, Geletka L, Martinez-Santibañez G, Zamarron BF, Lucas H, Singer K, O' Rourke RW, Lumeng CN. Muir LA, et al. J Leukoc Biol. 2018 Apr;103(4):615-628. doi: 10.1002/JLB.3HI1017-422R. Epub 2018 Mar 1. J Leukoc Biol. 2018. PMID: 29493813 Free PMC article. - The "Big Bang" in obese fat: Events initiating obesity-induced adipose tissue inflammation.
Wensveen FM, Valentić S, Šestan M, Turk Wensveen T, Polić B. Wensveen FM, et al. Eur J Immunol. 2015 Sep;45(9):2446-56. doi: 10.1002/eji.201545502. Epub 2015 Aug 19. Eur J Immunol. 2015. PMID: 26220361 Review. - White adipose tissue dysfunction in obesity and aging.
Reyes-Farias M, Fos-Domenech J, Serra D, Herrero L, Sánchez-Infantes D. Reyes-Farias M, et al. Biochem Pharmacol. 2021 Oct;192:114723. doi: 10.1016/j.bcp.2021.114723. Epub 2021 Aug 5. Biochem Pharmacol. 2021. PMID: 34364887 Review.
Cited by
- Similar degrees of obesity induced by diet or aging cause strikingly different immunologic and metabolic outcomes.
Krishna KB, Stefanovic-Racic M, Dedousis N, Sipula I, O'Doherty RM. Krishna KB, et al. Physiol Rep. 2016 Mar;4(6):e12708. doi: 10.14814/phy2.12708. Epub 2016 Mar 31. Physiol Rep. 2016. PMID: 27033445 Free PMC article. - Age-adjusted visceral adiposity index (VAI) is superior to VAI for predicting mortality among US adults: an analysis of the NHANES 2011-2014.
Liu W, Weng S, Chen Y, Cao C, Peng D. Liu W, et al. Aging Clin Exp Res. 2024 Feb 7;36(1):24. doi: 10.1007/s40520-023-02660-z. Aging Clin Exp Res. 2024. PMID: 38321181 Free PMC article. - Dynamics of single-nuclei transcriptomic profiling of adipose tissue from diverse anatomical locations during mouse aging process.
Wu Y, Sun Y, Chen L, Tong X, Liu C, Lu L, Zhang R, Wang S, Chen Z, Zhang J, Han Z, Zeng B, Li M, Jin L. Wu Y, et al. Sci Rep. 2024 Jul 12;14(1):16093. doi: 10.1038/s41598-024-66918-w. Sci Rep. 2024. PMID: 38997312 Free PMC article. - Age, Sex, and Depot-Specific Differences in Adipose-Tissue Estrogen Receptors in Individuals with Obesity.
Porter JW, Barnas JL, Welly R, Spencer N, Pitt J, Vieira-Potter VJ, Kanaley JA. Porter JW, et al. Obesity (Silver Spring). 2020 Sep;28(9):1698-1707. doi: 10.1002/oby.22888. Epub 2020 Jul 31. Obesity (Silver Spring). 2020. PMID: 32734695 Free PMC article. - Accumulation of γδ T cells in visceral fat with aging promotes chronic inflammation.
Bruno MEC, Mukherjee S, Powell WL, Mori SF, Wallace FK, Balasuriya BK, Su LC, Stromberg AJ, Cohen DA, Starr ME. Bruno MEC, et al. Geroscience. 2022 Jun;44(3):1761-1778. doi: 10.1007/s11357-022-00572-w. Epub 2022 Apr 28. Geroscience. 2022. PMID: 35477832 Free PMC article.
References
- Leng SX, Yang H, Walston JD. Decreased cell proliferation and altered cytokine production in frail older adults. Aging Clin Exp Res. 2004;16:249–252. - PubMed
- Horber FF, Gruber B, Thomi F, Jensen EX, Jaeger P. Effect of sex and age on bone mass, body composition and fuel metabolism in humans. Nutrition. 1997;13:524–534. - PubMed
Publication types
MeSH terms
Grants and funding
- P30ES017885/ES/NIEHS NIH HHS/United States
- P30 AG024824-09/AG/NIA NIH HHS/United States
- R01 AG020628/AG/NIA NIH HHS/United States
- R56 AG020628-06A1/AG/NIA NIH HHS/United States
- R01 AG020628-08/AG/NIA NIH HHS/United States
- R01AG020628/AG/NIA NIH HHS/United States
- R01DK090262/DK/NIDDK NIH HHS/United States
- P30 AG024824/AG/NIA NIH HHS/United States
- R01AG028268/AG/NIA NIH HHS/United States
- R01 DK090262/DK/NIDDK NIH HHS/United States
- P30 AR048310-10/AR/NIAMS NIH HHS/United States
- P30 ES017885-02/ES/NIEHS NIH HHS/United States
- P30 AG013283/AG/NIA NIH HHS/United States
- R01 AG028268/AG/NIA NIH HHS/United States
- R01 AR042525/AR/NIAMS NIH HHS/United States
- P30 AG013283-13/AG/NIA NIH HHS/United States
- P30 ES017885/ES/NIEHS NIH HHS/United States
- R01 AG028268-05/AG/NIA NIH HHS/United States
- P30 AR048310/AR/NIAMS NIH HHS/United States
- R56 AG020628/AG/NIA NIH HHS/United States
- P30AG024824/AG/NIA NIH HHS/United States
- R01AR042525/AR/NIAMS NIH HHS/United States
- K08 DK078851/DK/NIDDK NIH HHS/United States
- AG013283/AG/NIA NIH HHS/United States
- K08DK078851/DK/NIDDK NIH HHS/United States
- T32 GM007315/GM/NIGMS NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous