Proteins Structure: Secondary Structure (original) (raw)
Secondary Structure of Protein Organizational assumption points are
- No rotation occurs around the peptide bond (as it is partly double-bonded in nature).
- The chain of amino acids forms a rhythmical structure – forming a repeating pattern.
- That the maximum number of interactions from Hydrogen bonding possible are occurring, independent of the type of residue (amino acid).
- Basic explanation of the secondary structure of protein
- What is the Secondary Structure of Protein?
- Other Secondary Structures
Basic explanation of the secondary structure of protein
1. As mentioned, the C-N bond is partly double bonded and so does not rotate. The bond length of a normal C-N bond is 1.49Å (angstroms), while the length of a normal C=N bond is 1.28Å. The length of the peptide bond is between these, at 1.28Å.
This is due to the C-N bond resonating between single and double bonded forms, as shown above.
2. Two different folding points exist. These are called phi and psi. A perfect helix structure (covered later) needs both phi (Φ) and psi (Ψ) to be at an angle of about -60 degrees.
3. Hydrogen bonds occur between the C=O and H-N of other amino acids. In α helixes, the C=O: would form a hydrogen bond to the N-H of 4th residues ahead in the spiral (directly above).
What is the Secondary Structure of Protein?
The types of secondary protein structure can be classified into 3 types based on the Number of Polypeptide chains present in the polypeptide molecule. They are,
a) The α helix – Having One Polypeptide Chain
b) The β-pleated sheet – Having Two Polypeptide chains
c) The Triple helical structure – Having Three Polypeptide chains
1. The α-Helix
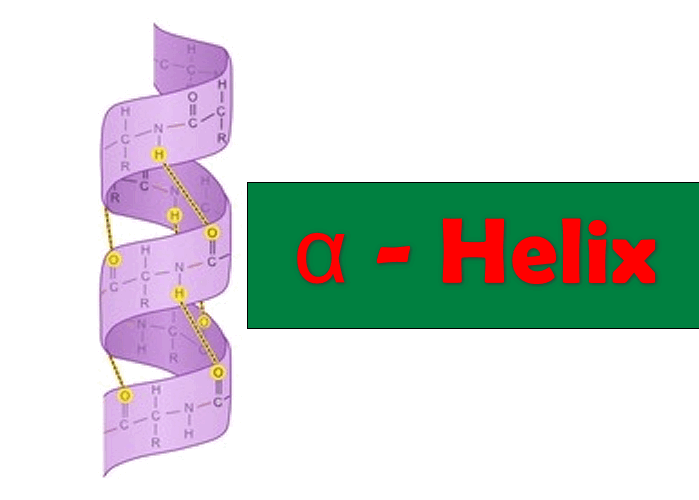
- The alpha helix consists of one polypeptide chain.
- It is present both in the fibrous proteins in globular proteins.
- It characterizes the keratin α as well as fibrinogen and myosin.
- This helical shape is the result of intramolecular chemical bonds leading to the structure of the spiral staircase.
- The right-handed α helix is more stable than the left-handed helix.
- This helical shape has no right of 0.54 nm.
- Certain amino acids (particularly PROLINE) disrupts the α helix. The larger number of acidic (Asp, Glu) or Basic (Lys, Arg and His) amino acids also interfere with α helix structure.
- Stability is ensured by:
- hydrogen bonds which are formed between the groups C = O and NH 4 amino acids apart. these links have an inclination of 30 °.
- the Van der Waals interactions.
- its length plus a helix α is, the more stable it is very strong cooperativity.
- In a turn of the helix is 3.66 amino acids, and travels a distance of 0.54nm. The spacing of each amino acid is o.15nm. 18 amino acids there are 5 rounds.
- In general, an α helix consists of 5 to more than 40 amino acids.
- Amino acids promote the formation of α helix are Ala, Glu, Leu, Met.
- Amino acids are bad trainers Pro, Gly, Tyr, Ser.
- Α helices can be hydrophilic, amphipathic or hydrophobic.
- It depends on the amino acid composition of the propeller.
- Indeed, the amino acid radicals are turning out the axis of the helix, standard terms their response to their environment. Thus, if the α helix contains only hydrophobic amino acids, so it is put in contact with hydrophobic surfaces, such as the lipid bilayer.
- If the hydrophobic residues are positioned on one side and hydrophilic residues on the other side, the α helix is amphipathic (or amphiphilic ).
- That is to say, we will find the interface of the hydrophilic and hydrophobic regions.
- This is the alpha-helix structure of the protein.
2. β-Pleated Sheet Structure

- The β-pleated sheet structure (beta-sheet structure) proposed by Pauling and Corey.
- The β-pleated sheet structure has two Polypeptide chains.
- It consists of the juxtaposition of β strands, chain conformation very stretched.
- Chains are presented in “Pleated sheet “(to take the first topographical sense- a succession of “roofs”).
- Involved in the peptide bonds that cross-linking and there are many bends.
- Fewer hydrogen bonds between the strands,
- The beta-pleated sheet structure can be divided into two types based on the orientation of peptide chains. in a sheet, maybe parallel or antiparallel.
- In Parallel sheet structure, the orientation of the two polypeptide chains is in the same direction. The Amino groups (-NH2) in the two polypeptide chains are in the same direction. Eg: β-Keratin
- In Anti-Parallel sheet structure, the orientation of the two polypeptide chains is in the opposite direction. The Amino groups (-NH2) in the two polypeptide chains are in the opposite direction. Eg: Silk Fibroin
- This type of sheet is more stable because the hydrogen bonds are in perfect alignment.
- After the α helices, they dominate in the secondary structures of proteins are very often present in β sheets: Gly, Val,
Ile
(3 nonpolar amino acids.) - In vivo, the β sheets do not have a really flat 3D structure. They are subjected to torsion in proteins.
- They are represented by a bunch of arrows indicating the direction of strand: the amino terminus to the carboxy terminus, and torsion.
- They are frequently found buried in the tertiary structure of globular proteins.
3. Triple Helical Structure

- The Secondary structure of collagen is the Rod-shaped molecule and most abundant protein of mammals.
- The structure of Collagen is in Triple helical in structure.
- It is the principal structural element of the human body and makes up 25% o 33% of all the body protein.
- It is found in the connective tissues such as tendons, cartilages, the organic matrix of bones and the cornea of the eye.
- Every third amino acid is Glycine in the Collagen.
- In triple helical structure, three polypeptide chains are twisted around each other itself.
Other Secondary Structures
a) Loops and bends (or towers)
Around one-third of the amino acids in a protein are part of loops or elbows to U-turns, or beta turns of the peptide chain. Elbows usually bind two antiparallel β strands. They include 2-4 short amino acids. The shorter they are, the fewer possible spatial conformations. For stabilized, there may be hydrogen bonding between the first and fourth amino acids. Amino acids are good trainers, elbows Gly and Pro.
Loops are longer and therefore comport more than four amino acids. They, therefore, allow more possible conformations. All amino acid loops do not participate in intramolecular hydrogen bonds. It allows for easier interaction with the solvent. Generally, there are loops between helices α, α helices and β strands, strands or parallel β sheets of different transcription factors have an extraordinary reason: helix-loop-helix.
b) Poly-gly, pro-poly left helix collagen
Poly-pro and poly-Gly are synthetic polymers of proline and glycine. aqueous solvent in poly-pro has a left helix conformation, poly-gly oscillates between the left propeller and β sheet. chains naturally containing these two amino acids can, therefore, have a left-handed helix structure as collagen. The left propeller is smaller than α helices, they account for only three amino acids per turn of the helix.
c. Super-secondary structures
- They concern optionally as globular proteins. These are flexible and have a wide variety of activities. The structures (or patterns) super-secondary represent typical associations of frequent flyers and propellers.
- The pattern of ” EF-hand “of calmodulin super secondary is a helix-loop-helix. The loop will accommodate a calcium atom. Some biological properties are related to these structures.
- Quote also: the Greek key motif (4 β strands), the leucine zipper (two α helices), the reason zinc finger (1 loop, a zinc atom, two
Cys
+ 2
His
or 4
Cys
), the strand pattern β – α helix – β strand.
Q. Study this representation of a polypeptide
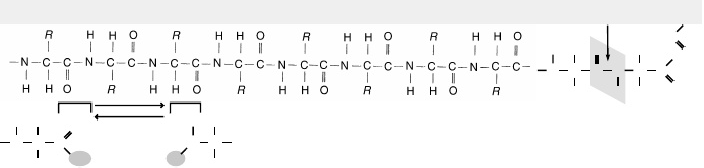
a. This is the______________structure of a protein. What are Groups?
b. ________What shapes do the secondary structure of a protein normally assume?
c. ____________What type of bond between amino acids is necessary to maintain secondary shape?
d. ______________How does the tertiary shape of a globular protein come about?
e. ________What would cause a protein to have a quaternary shape?
Q. Which type of secondary structure is most common in antibodies?
a)alpha-helices
b)beta strands
c)Both alpha helices and beta strands contribute about the same amount (50%/50%) to the structure of an antibody molecule.
d)Antibodies have neither alpha helices or beta strands.
e)hydrogen bonds
Ans: There are a few alpha-helices in antibodies, but mostly beta-strands.
Q. What maintains the secondary structure of a protein?
The secondary structure of Proteins is maintained by hydrogen bonds between amide hydrogens and carbonyl oxygens of the peptide backbone. The major secondary structures are α-helices and β-structures.
Q. What is the function of a secondary protein structure?
The secondary structure of proteins is a local interaction between stretches of a polypeptide chain and includes α-helix and β-pleated sheet structures.
Additional readings:
- Why Peptide bonds are Backbone of the Proteins?
- Why Proteins are Very Important? How to Explain?
- How the Tertiary structure of Protein is Organized closely?
Discover more from Biochemistry Den
Subscribe to get the latest posts sent to your email.