aging – NIH Director's Blog (original) (raw)
Maternal Brain Hormone Key to Strengthening Bones Could Help Treat Osteoporosis, Bone Fractures
Posted on August 1st, 2024 by Dr. Monica M. Bertagnolli

Credit: Donny Bliss/NIH
More than 200 million people around the world have osteoporosis, a condition that weakens bones to the point that they break easily. Women are at especially high risk after menopause due to declining levels of the hormone estrogen, which helps keep bones strong. While osteoporosis rarely has noticeable symptoms, it can lead to serious injuries when otherwise minor slips and falls cause broken bones that in turn can lead to further fracture risk and fracture-related mortality. So, I’m pleased to share NIH-supported research suggesting a surprising candidate for strengthening bones: a maternal hormone produced in the brain.
The study in mice reported in Nature shows that this newly discovered hormone maintains and rebuilds bone strength in lactating females, even as estrogen levels dip and calcium is lost to the demands of milk production. 1 The findings suggest this hormone—or a drug that acts similarly—could be key to treating osteoporosis and preventing and healing broken bones.
The findings come from a team led by Holly Ingraham, University of California, San Francisco. The researchers knew from studies in mice and humans that a protein related to parathyroid hormone, which is made in the mammary glands, is the main driver for stripping calcium from maternal bones for milk production. As a result of this process, nursing mothers tend to lose a lot of bone. In humans, this bone loss is 10% on average, compared to nearly 30% in mice. Fortunately, these losses are reversed after lactation ends, suggesting to the researchers there must be some other bone-strengthening factor in play.
Previous work in Ingraham’s lab, also supported by NIH, offered other clues. The researchers found that in female mice, blocking a certain estrogen receptor in select neurons in a small area of the brain led to the development of bones that were exceptionally dense and strong. 2 This was an early hint that an unidentified hormone might have a role. The team’s search in this latest study led them to brain-derived communication network factor 3 (CCN3).
The new findings showed that, in lactating female mice, CCN3 is produced in the same brain area identified in the previous study. When the researchers prevented the brain from making CCN3, lactating female mice rapidly lost bone. The researchers also found that male and female young adult and older mice gained a considerable amount of bone mass and strength when their levels of circulating CCN3 were boosted over a two-week period. In fact, in some female mice that were very old or completely lacked estrogen, the hormone more than doubled their bone mass. Tests showed that the animals’ bones weren’t just denser, but also stronger.
Further studies conducted by co-author Thomas Ambrosi, University of California, Davis, revealed that bone stem cells were responsible for receiving signals and generating the new bone. When those cells were exposed to CCN3, they ramped up bone production even more. When the researchers applied a hydrogel patch containing CCN3 to the sites of bone breaks, this spurred the formation of new bone. As a result, the researchers saw rapid bone healing in older mice comparable to what would be expected in much younger mice.
In future studies, the researchers want to gain insight into the underlying mechanisms of CCN3. They also plan to explore the hormone’s potential for treating bone loss in people at increased risk, including postmenopausal women, breast cancer survivors taking estrogen blockers, and those with other conditions leading to unhealthy bone mass, such as genetic bone disorders, chronic kidney disease, or premature ovarian failure. They suggest that more immediate local uses for CCN3 include fracture repair, cartilage regeneration, and bone improvements for anchoring dental implants. It’s a great example of how finding an answer to a scientific puzzle—like how maternal bones stay strong during breastfeeding—can potentially lead to advances that help many more people.
References:
[1] Babey ME, et al. A maternal brain hormone that builds bone. Nature. DOI: 10.1038/s41586-024-07634-3 (2024).
[2] Herber CB, et al. Estrogen signaling in arcuate Kiss1 neurons suppresses a sex-dependent female circuit promoting dense strong bones. Nature Communications. DOI: 10.1038/s41467-018-08046-4 (2019).
NIH Support: National Institute of Diabetes and Digestive and Kidney Diseases, National Institute on Aging, National Institute of General Medical Sciences, National Institute of Arthritis and Musculoskeletal and Skin Diseases
Posted In: Health, Science, Uncategorized
Tags: aging, basic research, Bone, bone fracture, breastfeeding, calcium, estrogen, hormone, maternal health, menopause, osteoporosis
Study of Protective Gene Variant Provides Insight into Delaying Onset of Alzheimer’s Dementia
Posted on July 18th, 2024 by Dr. Monica M. Bertagnolli

Credit: Donny Bliss/NIH
Alzheimer’s disease is currently the seventh leading cause of death in the U.S. While your likelihood of developing Alzheimer’s-related cognitive impairment increases with age, risk for this disease and age of its onset depend on many factors, including the genes you carry. An intriguing new study suggests that having just one copy of a protective gene variant may be enough to delay cognitive impairment from this devastating disease in individuals who are otherwise genetically predisposed to developing early-onset Alzheimer’s dementia.
The findings, from a study supported in part by NIH and reported in The New England Journal of Medicine, offer important insights into the genetic factors and underlying pathways involved in Alzheimer’s dementia.1 While much more study is needed, the findings have potential implications for treatments that could one day work like this gene variant does to delay or perhaps even prevent Alzheimer’s dementia.
This research comes from an international team including Yakeel Quiroz, Massachusetts General Hospital, Boston; Joseph Arboleda-Velasquez, Mass Eye and Ear, Boston; and Francisco Lopera, University of Antioquia, Colombia. For the last 40 years, Lopera has been studying a Colombian family of about 6,000 blood relatives, 1,200 of whom carry a mutation known as Paisa (or Presenilin-1 E280A) that predisposes them to developing early-onset Alzheimer’s dementia. Those who carry a single copy of this gene variant typically show signs of cognitive decline in their early 40s, progressing to dementia by age 50. They frequently die from dementia-related complications in their 60s.
In 2019, the researchers reported on an extraordinary individual who was an exception to this prognosis.2 Even though she carried the Paisa mutation, she didn’t develop any notable cognitive decline until her late 70s—30 years later than expected. The researchers traced her protection against dementia to two copies of a rare variant of the APOE gene dubbed Christchurch. Further study of her brain after death also found lower levels of inflammation and tau protein, which forms damaging tangles inside neurons in the Alzheimer’s brain.
Christchurch is a rare variant, and it’s far more common for people to carry one copy of the protective variant versus two. Would a single copy of the Christchurch variant offer some protection against Alzheimer’s dementia, too? To find out in the new study, the researchers analyzed data from 27 members of this family carrying a single copy of the Christchurch variant among 1,077 carriers of the Paisa mutation.
The researchers compared Christchurch carriers to those without the protective variant and found the variant did delay the age of onset of Alzheimer’s-related cognitive decline and dementia. The median age at the onset of mild cognitive impairment was 52 in family members with the Christchurch variant, compared to approximately age 47 in a matched group without the variant. Similarly, the median age at the onset of dementia was 54, compared to the median age of 50 in noncarriers.
To learn more, the researchers imaged the brains of two of the individuals who had one copy of Christchurch. The brain scans showed lower levels of tau and more normal metabolic activity in brain areas that are known to play a role in Alzheimer’s. Interestingly, their brains still showed accumulations of amyloid proteins, which form plaques that are another hallmark of Alzheimer’s. The team also analyzed autopsy samples from four deceased individuals with one copy of the Christchurch variant and found that blood vessels in their brains appeared healthier, which may help to explain the protective effects of Christchurch. The findings suggest a significant role for blood vessel health in protecting the brain from cognitive decline, as well as a role for disease of the brain blood vessels in contributing to cognitive decline and dementia.
The researchers note this study is limited to a relatively small number of people with both the Paisa and Christchurch variants in one group of related individuals. Further studies involving larger and more diverse samples are needed to learn more about this protective gene variant and its effects on the brain in the general population. The hope is these findings may one day yield new approaches to delaying the onset of Alzheimer’s or slowing its progression in millions more people around the world at risk of developing this devastating disease.
References:
[1] Quiroz YT, et al. APOE3 Christchurch Heterozygosity and Autosomal Dominant Alzheimer’s Disease. The New England Journal of Medicine. DOI: 10.1056/NEJMoa2308583 (2024).
[2] Arboleda-Velasquez JF, et al. Resistance to autosomal dominant Alzheimer’s disease in an APOE3 Christchurch homozygote: a case report. Nature Medicine. DOI: 10.1038/s41591-019-0611-3 (2019).
NIH Support: National Institute on Aging, National Institute of Neurological Disorders and Stroke
Posted In: Health, News, Science
Tags: aging, Alzheimer’s disease, brain, cognitive decline, dementia, DNA, gene variants, genetics, neurological disease
Sequencing Technique Detects Earliest Signs of Genetic Mutations Underlying Cancer, Aging, and More
Posted on July 11th, 2024 by Dr. Monica M. Bertagnolli

Every day, billions of cells in your body divide, each producing two daughter cells. It’s an essential process for your tissues and organs to renew themselves and remain healthy. To do it, cells must first duplicate their DNA to ensure that each daughter cell gets an accurate copy. In this process, mistakes are inevitably made. Most DNA errors are accurately fixed and do not lead to mutations. But when small errors akin to single-letter typos aren’t corrected, they can become permanent in a cell and multiplied with each subsequent cell division. Even cells that don’t divide, such as neurons in your brain, acquire damage and mutations in their DNA with age. As a result, your tissues contain collections of cells with distinct mutations that accumulate over time.
While many of these small errors will show no obvious consequences, others can lead to cancer and other health conditions. Now, a new DNA sequencing technique, described in Natureand developed through research supported by NIH, promises to detect early DNA changes before they become permanent mutations in a cell’s genome. The method, called Hairpin Duplex Enhanced Fidelity Sequencing (HiDEF-seq), could advance our understanding of how and why mutations arise, with potentially important implications for our health. For example, the ability to identify signs that precede mutations may help predict a person’s health risks based on genetic predispositions, environmental exposures, or other factors.
The HiDEF-seq technique comes from an international team led by Gilad Evrony at NYU Grossman School of Medicine’s Center for Human Genetics and Genomics in New York City. To understand how the method works, it helps to remember that each DNA molecule stores genetic information in the form of two complementary strands made up of four molecular “letters,” or chemical bases. Those bases are adenine (A), thymine (T), guanine (G), and cytosine (C). The sequence of about three billion As, Ts, Gs, and Cs in human DNA’s two strands generally should match up, such that As pair with Ts and Gs with Cs.
The first step in which DNA mutations arise usually involves a change in only one of the two DNA strands. Those single-strand errors only become permanent mutations in both strands when a cell’s copying machinery fails to detect the mistake before the cell divides again, or when the cell’s DNA repair machinery makes a mistake in the correction process. However, because other methods to sequence DNA can’t accurately detect changes that are in only one of the DNA strands, it hasn’t been possible for researchers to study this process in detail. This is where HiDEF-seq comes in.
The researchers wanted to develop an approach for directly sequencing single DNA molecules to detect these early-stage DNA errors. Detecting changes that are in only one of the two DNA strands requires an extremely high degree of sequencing accuracy, with less than one error per one billion bases, so the team devised a method to read DNA with higher precision than was previously possible. To put HiDEF-seq to the test, they profiled 134 samples from various human tissues, including those from people with syndromes that predisposed them to cancer due to an unusually high number of new mutations.
The research team found they could use HiDEF-seq to identify changes present in only one of the two DNA strands that were the precursors to mutational events. For example, they identified places where a C was mistakenly paired with a T instead of a G. As expected, those early changes in DNA turned up more often in people with syndromes that increase their risk of cancer than in those without. The patterns of those single-strand DNA changes also looked a lot like the patterns of double-strand DNA mutations seen in people with these syndromes, suggesting that the HiDEF-seq method was indeed seeing the precursors to mutations.
The method can also detect a common form of DNA damage called cytosine deamination, in which cytosine is converted to a different base called uracil (U), which is another source of mutations. Experiments in human sperm, which rarely pick up mutations compared to other cell types, showed a pattern of cytosine deamination that closely matched damage caused by heating healthy DNA in the lab. This led the researchers to suggest that the damage to DNA happens similarly in both situations.
The researchers have already begun to produce a catalog of the various single-strand DNA errors they’ve uncovered. They suggest that HiDEF-seq may allow new ways to monitor the everyday effects of environmental exposures or other insults on our DNA and shed light on the balance in cells between DNA damage, repair, and replication. Along the way, this new technique will enable the continued study of DNA damage and the origins of mutations in a way that hasn’t been possible before.
Reference:
Liu MH, et al. Single-strand mismatch and damage patterns revealed by single-molecule DNA sequencing. Nature. DOI: 10.1038/s41586-024-07532-8 (2024).
NIH Support: Common Fund, Eunice Kennedy Shriver National Institute of Child Health and Human Development, National Institute on Aging, National Institute of Neurological Disorders and Stroke, National Cancer Institute
Posted In: Health, News, Science
Tags: aging, cancer, cell division, DNA, DNA damage, DNA sequencing, genetics, genomics, mutations
Changes in Human Microbiome Precede Alzheimer’s Cognitive Declines
Posted on June 27th, 2023 by Lawrence Tabak, D.D.S., Ph.D.
Caption: The human gut teems with bacteria and other microbes. They contribute to our health but also influence our susceptibility to certain diseases, including Alzheimer’s disease. Credit: Donny Bliss, NIH
In people with Alzheimer’s disease, the underlying changes in the brain associated with dementia typically begin many years—or even decades—before a diagnosis. While pinpointing the exact causes of Alzheimer’s remains a major research challenge, they likely involve a combination of genetic, environmental, and lifestyle factors. Now an NIH-funded study elucidates the role of another likely culprit that you may not have considered: the human gut microbiome, the trillions of diverse bacteria and other microbes that live primarily in our intestines [1].
Earlier studies had showed that the gut microbiomes of people with symptomatic Alzheimer’s disease differ from those of healthy people with normal cognition [2]. What this new work advances is that these differences arise early on in people who will develop Alzheimer’s, even before any obvious symptoms appear.
The science still has a ways to go before we’ll know if specific dietary changes can alter the gut microbiome and modify its influence on the brain in the right ways. But what’s exciting about this finding is it raises the possibility that doctors one day could test a patient’s stool sample to determine if what’s present from their gut microbiome correlates with greater early risk for Alzheimer’s dementia. Such a test would help doctors detect Alzheimer’s earlier and intervene sooner to slow or ideally even halt its advance.
The new findings, reported in the journal Science Translational Medicine, come from a research team led by Gautam Dantas and Beau Ances, Washington University School of Medicine, St. Louis. Ances is a clinician who treats and studies people with Alzheimer’s; Dantas is a basic researcher and expert on the gut microbiome.
The pair struck up a conversation one day about the possible connection between the gut microbiome and Alzheimer’s. While they knew about the earlier studies suggesting a link, they were surprised that nobody had looked at the gut microbiomes of people in the earliest, so-called preclinical, stages of the disease. That’s when dementia isn’t detectable, but the brain has formed amyloid-beta plaques, which are associated with Alzheimer’s.
To take a look, they enrolled 164 healthy volunteers, age 68 to 94, who performed normally on standard tests of cognition. They also collected stool samples from each volunteer and thoroughly analyzed them all the microbes from their gut microbiome. Study participants also kept food diaries and underwent extensive testing, including two types of brain scans, to look for signs of amyloid-beta plaques and tau protein accumulation that precede the onset of Alzheimer’s symptoms.
Among the volunteers, about a third (49 individuals) unfortunately had signs of early Alzheimer’s disease. And, as it turned out, their microbiomes showed differences, too.
The researchers found that those with preclinical Alzheimer’s disease had markedly different assemblages of gut bacteria. Their microbiomes differed in many of the bacterial species present. Those species-level differences also point to differences in the way their microbiomes would be expected to function at a metabolic level. These microbiome changes were observed even though the individuals didn’t seem to have any apparent differences in their diets.
The team also found that the microbiome changes correlated with amyloid-beta and tau levels in the brain. But they did not find any relationship to degenerative changes in the brain, which tend to happen later in people with Alzheimer’s.
The team is now conducting a five-year study that will follow volunteers to get a better handle on whether the differences observed in the gut microbiome are a cause or a consequence of the brain changes seen in Alzheimer’s. If it’s a cause, this discovery would raise the tantalizing possibility that specially formulated probiotics or fecal transplants that promote the growth of “good” bacteria over “bad” bacteria in the gut might slow the development of Alzheimer’s and its most devastating symptoms. It’s an exciting area of research and definitely one worth following in the years ahead.
References:
[1] Gut microbiome composition may be an indicator of preclinical Alzheimer’s disease. Ferreiro AL, Choi J, Ryou J, Newcomer EP, Thompson R, Bollinger RM, Hall-Moore C, Ndao IM, Sax L, Benzinger TLS, Stark SL, Holtzman DM, Fagan AM, Schindler SE, Cruchaga C, Butt OH, Morris JC, Tarr PI, Ances BM, Dantas G. Sci Transl Med. 2023 Jun 14;15(700):eabo2984. doi: 10.1126/scitranslmed.abo2984. Epub 2023 Jun 14. PMID: 37315112.
[2] Gut microbiome alterations in Alzheimer’s disease. Vogt NM, Kerby RL, Dill-McFarland KA, Harding SJ, Merluzzi AP, Johnson SC, Carlsson CM, Asthana S, Zetterberg H, Blennow K, Bendlin BB, Rey FE. Sci Rep. 2017 Oct 19;7(1):13537. doi: 10.1038/s41598-017-13601-y. PMID: 29051531; PMCID: PMC5648830.
Links:
Alzheimer’s Disease and Related Dementias (National Institute on Aging/NIH)
Video: How Alzheimer’s Changes the Brain (NIA)
Dantas Lab (Washington University School of Medicine. St. Louis)
Ances Bioimaging Laboratory (Washington University School of Medicine, St. Louis)
NIH Support: National Institute on Aging; National Institute of Diabetes and Digestive and Kidney Diseases
Posted In: News
Tags: aging, Alzheimer’s disease, amyloid plaques, beta amyloid, brain, dementia, diet, fecal transplant, gut, gut bacteria, gut microbiome, microbiome, probiotics, tau
Immune Resilience is Key to a Long and Healthy Life
Posted on June 20th, 2023 by Lawrence Tabak, D.D.S., Ph.D.

Caption: A new measure of immunity called immune resilience is helping researchers find clues as to why some people remain healthier even in the face of varied inflammatory stressors. Credit: Modified from Shutterstock/Ground Picture
Do you feel as if you or perhaps your family members are constantly coming down with illnesses that drag on longer than they should? Or, maybe you’re one of those lucky people who rarely becomes ill and, if you do, recovers faster than others.
It’s clear that some people generally are more susceptible to infectious illnesses, while others manage to stay healthier or bounce back more quickly, sometimes even into old age. Why is this? A new study from an NIH-supported team has an intriguing answer [1]. The difference, they suggest, may be explained in part by a new measure of immunity they call immune resilience—the ability of the immune system to rapidly launch attacks that defend effectively against infectious invaders and respond appropriately to other types of inflammatory stressors, including aging or other health conditions, and then quickly recover, while keeping potentially damaging inflammation under wraps.
The findings in the journal Nature Communications come from an international team led by Sunil Ahuja, University of Texas Health Science Center and the Department of Veterans Affairs Center for Personalized Medicine, both in San Antonio. To understand the role of immune resilience and its effect on longevity and health outcomes, the researchers looked at multiple other studies including healthy individuals and those with a range of health conditions that challenged their immune systems.
By looking at multiple studies in varied infectious and other contexts, they hoped to find clues as to why some people remain healthier even in the face of varied inflammatory stressors, ranging from mild to more severe. But to understand how immune resilience influences health outcomes, they first needed a way to measure or grade this immune attribute.
The researchers developed two methods for measuring immune resilience. The first metric, a laboratory test called immune health grades (IHGs), is a four-tier grading system that calculates the balance between infection-fighting CD8+ and CD4+ T-cells. IHG-I denotes the best balance tracking the highest level of resilience, and IHG-IV denotes the worst balance tracking the lowest level of immune resilience. An imbalance between the levels of these T cell types is observed in many people as they age, when they get sick, and in people with autoimmune diseases and other conditions.
The researchers also developed a second metric that looks for two patterns of expression of a select set of genes. One pattern associated with survival and the other with death. The survival-associated pattern is primarily related to immune competence, or the immune system’s ability to function swiftly and restore activities that encourage disease resistance. The mortality-associated genes are closely related to inflammation, a process through which the immune system eliminates pathogens and begins the healing process but that also underlies many disease states.
Their studies have shown that high expression of the survival-associated genes and lower expression of mortality-associated genes indicate optimal immune resilience, correlating with a longer lifespan. The opposite pattern indicates poor resilience and a greater risk of premature death. When both sets of genes are either low or high at the same time, immune resilience and mortality risks are more moderate.
In the newly reported study initiated in 2014, Ahuja and his colleagues set out to assess immune resilience in a collection of about 48,500 people, with or without various acute, repetitive, or chronic challenges to their immune systems. In an earlier study, the researchers showed that this novel way to measure immune status and resilience predicted hospitalization and mortality during acute COVID-19 across a wide age spectrum [2].
The investigators have analyzed stored blood samples and publicly available data representing people, many of whom were healthy volunteers, who had enrolled in different studies conducted in Africa, Europe, and North America. Volunteers ranged in age from 9 to 103 years. They also evaluated participants in the Framingham Heart Study, a long-term effort to identify common factors and characteristics that contribute to cardiovascular disease.
To examine people with a wide range of health challenges and associated stresses on their immune systems, the team also included participants who had influenza or COVID-19, and people living with HIV. They also included kidney transplant recipients, people with lifestyle factors that put them at high risk for sexually transmitted infections, and people who’d had sepsis, a condition in which the body has an extreme and life-threatening response following an infection.
The question in all these contexts was the same: How well did the two metrics of immune resilience predict an individual’s health outcomes and lifespan? The short answer is that immune resilience, longevity, and better health outcomes tracked together well. Those with metrics indicating optimal immune resilience generally had better health outcomes and lived longer than those who had lower scores on the immunity grading scale. Indeed, those with optimal immune resilience were more likely to:
- Live longer,
- Resist HIV infection or the progression from HIV to AIDS,
- Resist symptomatic influenza,
- Resist a recurrence of skin cancer after a kidney transplant,
- Survive COVID-19, and
- Survive sepsis.
The study also revealed other interesting findings. While immune resilience generally declines with age, some people maintain higher levels of immune resilience as they get older for reasons that aren’t yet known, according to the researchers. Some people also maintain higher levels of immune resilience despite the presence of inflammatory stress to their immune systems such as during HIV infection or acute COVID-19. People of all ages can show high or low immune resilience. The study also found that higher immune resilience is more common in females than it is in males.
The findings suggest that there is a lot more to learn about why people differ in their ability to preserve optimal immune resilience. With further research, it may be possible to develop treatments or other methods to encourage or restore immune resilience as a way of improving general health, according to the study team.
The researchers suggest it’s possible that one day checkups of a person’s immune resilience could help us to understand and predict an individual’s health status and risk for a wide range of health conditions. It could also help to identify those individuals who may be at a higher risk of poor outcomes when they do get sick and may need more aggressive treatment. Researchers may also consider immune resilience when designing vaccine clinical trials.
A more thorough understanding of immune resilience and discovery of ways to improve it may help to address important health disparities linked to differences in race, ethnicity, geography, and other factors. We know that healthy eating, exercising, and taking precautions to avoid getting sick foster good health and longevity; in the future, perhaps we’ll also consider how our immune resilience measures up and take steps to achieve or maintain a healthier, more balanced, immunity status.
References:
[1] Immune resilience despite inflammatory stress promotes longevity and favorable health outcomes including resistance to infection. Ahuja SK, Manoharan MS, Lee GC, McKinnon LR, Meunier JA, Steri M, Harper N, Fiorillo E, Smith AM, Restrepo MI, Branum AP, Bottomley MJ, Orrù V, Jimenez F, Carrillo A, Pandranki L, Winter CA, Winter LA, Gaitan AA, Moreira AG, Walter EA, Silvestri G, King CL, Zheng YT, Zheng HY, Kimani J, Blake Ball T, Plummer FA, Fowke KR, Harden PN, Wood KJ, Ferris MT, Lund JM, Heise MT, Garrett N, Canady KR, Abdool Karim SS, Little SJ, Gianella S, Smith DM, Letendre S, Richman DD, Cucca F, Trinh H, Sanchez-Reilly S, Hecht JM, Cadena Zuluaga JA, Anzueto A, Pugh JA; South Texas Veterans Health Care System COVID-19 team; Agan BK, Root-Bernstein R, Clark RA, Okulicz JF, He W. Nat Commun. 2023 Jun 13;14(1):3286. doi: 10.1038/s41467-023-38238-6. PMID: 37311745.
[2] Immunologic resilience and COVID-19 survival advantage. Lee GC, Restrepo MI, Harper N, Manoharan MS, Smith AM, Meunier JA, Sanchez-Reilly S, Ehsan A, Branum AP, Winter C, Winter L, Jimenez F, Pandranki L, Carrillo A, Perez GL, Anzueto A, Trinh H, Lee M, Hecht JM, Martinez-Vargas C, Sehgal RT, Cadena J, Walter EA, Oakman K, Benavides R, Pugh JA; South Texas Veterans Health Care System COVID-19 Team; Letendre S, Steri M, Orrù V, Fiorillo E, Cucca F, Moreira AG, Zhang N, Leadbetter E, Agan BK, Richman DD, He W, Clark RA, Okulicz JF, Ahuja SK. J Allergy Clin Immunol. 2021 Nov;148(5):1176-1191. doi: 10.1016/j.jaci.2021.08.021. Epub 2021 Sep 8. PMID: 34508765; PMCID: PMC8425719.
Links:
COVID-19 Research (NIH)
HIV Info (NIH)
Sepsis (National Institute of General Medical Sciences/NIH)
Sunil Ahuja (University of Texas Health Science Center, San Antonio)
Framingham Heart Study (National Heart, Lung, and Blood Institute/NIH)
“A Secret to Health and Long Life? Immune Resilience, NIAID Grantees Report,” NIAID Now Blog, June 13, 2023
NIH Support: National Institute of Allergy and Infectious Diseases; National Institute on Aging; National Institute of Mental Health; National Institute of General Medical Sciences; National Heart, Lung, and Blood Institute
Posted In: News
Tags: aging, cardiovascular disease, CD4+, CD8+, COVID-19, Framingham Heart Study, genomics, health disparities, HIV, IHG, immune health grades, immune resilience, immune system, immunity, influenza, kidney transplantation, lifespan, longevity, sepsis, skin cancer, T cells
To Prevent a Stroke, Household Chores and Leisurely Strolls May Help
Posted on June 21st, 2022 by Lawrence Tabak, D.D.S., Ph.D.

Credit: Shutterstock/Tartila
As we get older, unfortunately our chances of having a stroke rise. While there’s obviously no way to turn back the clock on our age, fortunately there are ways to lower our risk of a stroke and that includes staying physically active. Take walks, ride a bike, play a favorite sport. According to our current exercise guidelines for American adults, the goal is to get in at least two and a half hours each week of moderate-intensity physical activity as well as two days of muscle-strengthening activity [1].
But a new study, published in the journal JAMA Network Open, shows that reducing the chances of a stroke as we get older doesn’t necessarily require heavy aerobic exercise or a sweat suit [2]. For those who are less mobile or less interested in getting out to exercise, the researchers discovered that just spending time doing light-intensity physical activity—such as tending to household chores—“significantly” protects against stroke.
The study also found you don’t have to dedicate whole afternoons to tidying up around the house to protect your health. It helps to just get up out of your chair for five or 10 minutes at a time throughout the day to straighten up a room, sweep the floor, fold the laundry, step outside to water the garden, or just take a leisurely stroll.
That may sound simple, but consider that the average American adult now spends on average six and a half hours per day just sitting [3]. That comes to nearly two days per week on average, much to the detriment of our health and wellbeing. Indeed, the study found that middle-aged and older people who were sedentary for 13 hours or more hours per day had a 44 percent increased risk of stroke.
These latest findings come from Steven Hooker, San Diego State University, CA, and his colleagues on the NIH-supported Reasons for Geographic and Racial Differences in Stroke (REGARDS) study. Launched in 2003, REGARDS continues to follow over time more than 30,000 Black and white participants aged 45 and older.
Hooker and colleagues wanted to know more about the amount and intensity of exercise required to prevent a stroke. Interestingly, the existing data were relatively weak, in part because prior studies looking at the associations between physical activity and stroke risk relied on self-reported data, which don’t allow for precise measures. What’s more, the relationship between time spent sitting and stroke risk also remained unknown.
To get answers, Hooker and team focused on 7,607 adults enrolled in the REGARDS study. Rather than relying on self-reported physical activity data, team members asked participants to wear a hip-mounted accelerometer—a device that records how fast people move—during waking hours for seven days between May 2009 and January 2013.
The average age of participants was 63. Men and women were represented about equally in the study, while about 70 percent of participants were white and 30 percent were Black.
Over the more than seven years of the study, 286 participants suffered a stroke. The researchers then analyzed all the accelerometer data, including the amount and intensity of their physical activity over the course of a normal week. They then related those data to their risk of having a stroke over the course of the study.
The researchers found, as anticipated, that adults who spent the most time doing moderate-to-vigorous intensity physical activity were less likely to have a stroke than those who spent the least time physically active. But those who spent the most time sitting also were at greater stroke risk, whether they got their weekly exercise in or not.
Those who regularly sat still for longer periods—17 minutes or more at a time—had a 54 percent increase in stroke risk compared to those who more often sat still for less than eight minutes. After adjusting for the time participants spent sitting, those who more often had shorter periods of moderate-to-vigorous activity—less than 10 minutes at a time—still had significantly lower stroke risk. But, once the amount of time spent sitting was taken into account, longer periods of more vigorous activity didn’t make a difference.
While high blood pressure, diabetes, and myriad other factors also contribute to a person’s cumulative risk of stroke, the highlighted paper does bring some good actionable news. For each hour spent doing light-intensity physical activity instead of sitting, a person can reduce his or her stroke risk.
The bad news, of course, is that each extra hour spent sitting per day comes with an increased risk for stroke. This bad news shouldn’t be taken lightly. In the U.S., almost 800,000 people have a stroke each year. That’s one person every 40 seconds with, on average, one death every four minutes. Globally, stroke is the second most common cause of death and third most common cause of disability in people, killing more than 6.5 million each year.
If you’re already meeting the current exercise guidelines for adults, keep up the good work. If not, this paper shows you can still do something to lower your stroke risk. Make a habit throughout the day of getting up out of your chair for a mere five or 10 minutes to straighten up a room, sweep the floor, fold the laundry, step outside to water the garden, or take a leisurely stroll. It could make a big difference to your health as you age.
References:
[1] How much physical activity do adults need? Centers for Disease Control and Prevention. June 2, 2022.
[2] Association of accelerometer-measured sedentary time and physical activity with risk of stroke among US adults. Hooker SP, Diaz KM, Blair SN, Colabianchi N, Hutto B, McDonnell MN, Vena JE, Howard VJ. JAMA Netw Open. 2022 Jun 1;5(6):e2215385.
[3] Trends in sedentary behavior among the US population, 2001-2016. Yang L, Cao C, Kantor ED, Nguyen LH, Zheng X, Park Y, Giovannucci EL, Matthews CE, Colditz GA, Cao Y. JAMA. 2019 Apr 23;321(16):1587-1597.
Links:
Stroke (National Institute of Neurological Disorders and Stroke/NIH)
REGARDS Study (University of Alabama at Birmingham)
NIH Support: National Institute of Neurological Disorders and Stroke; National Institute on Aging
Posted In: News
Tags: accelerometer, aging, exercise, household chores, light-intensity physical activity, older people, physical activity, prevention, Reasons for Geographic and Racial Differences in Stroke Study, REGARDS Study, sedentary, senior health, sitting, stroke, walking
Advancing Access to Hearing Health Care
Posted on May 17th, 2022 by Debara L. Tucci, M.D., M.S., M.B.A., National Institute on Deafness and Other Communication Disorders

Credit: Shutterstock/wavebreakmedia
By 2050, the World Health Organization estimates that more than 700 million people—or one in every 10 people around the globe—will have disabling hearing loss. In the United States alone, hearing loss affects an estimated 30 million people [1]. Hearing loss can be frustrating, isolating, and even dangerous. It is also associated with dementia, depression, anxiety, reduced mobility, and falls.
Although hearing technologies, such as hearing aids, have improved, not everyone has equal access to these advancements. In fact, though hearing aids and other assistive devices can significantly improve quality of life, only one in four U.S. adults who could benefit from these devices has ever used one. Why? People commonly report encountering economic barriers, such as the high cost of hearing aids and limited access to hearing health care. For some, the reasons are more personal. They may not believe that hearing aids are effective, or they may worry about a perceived negative association with aging. [2].
As the lead federal agency supporting research initiatives to prevent, detect, and treat hearing loss, NIH’s National Institute on Deafness and Other Communication Disorders (NIDCD) conducts and funds research that identifies ways to break down barriers to hearing health care. Decades of NIDCD research informed a recent landmark announcement by the Food and Drug Administration (FDA) creating a new category of over-the-counter (OTC) hearing aids. When the regulation takes effect (expected in 2022), millions of people who have trouble hearing will be able to purchase less expensive hearing aids without a medical exam, prescription, or fitting by an audiologist.
This exciting development has been on the horizon at NIDCD for some time. Back in 2009, NIDCD’s Working Group on Accessible and Affordable Hearing Health Care for Adults with Mild to Moderate Hearing Loss created a blueprint for research priorities.
The working group’s blueprint led to NIDCD funding of more than 60 research projects spanning the landscape of accessible and affordable hearing health care issues. One study showed that people with hearing loss can independently adjust the settings [3] on their hearing devices in response to changing acoustic environments and, when given the ability to control their own hearing aid settings, they were generally more satisfied with the sound of the devices than with the audiologist fit [4].
In 2017, the first randomized, double-blind, placebo-controlled clinical trial comparing an over-the-counter delivery model [5] of hearing aids with traditional fitting by an audiologist also found that hearing aid users in both groups reported similar benefits. A 2019 follow-up study [6] confirmed these results, supporting the viability of a direct-to-consumer service delivery model. A small-business research grant funded by NIDCD led to the first FDA-approved self-fitting hearing aid.
Meanwhile, in 2016, NIDCD co-sponsored a consensus report from the National Academies of Sciences, Engineering, and Medicine (NASEM). The report, Hearing Health Care for Adults: Priorities for Improving Access and Affordability, which was developed by an independent expert panel, recommended that the FDA create and regulate a new category of over-the-counter hearing devices to improve access to affordable hearing aids for adults with perceived mild-to-moderate hearing loss. These devices will not be intended for children or for adults with more severe hearing loss.
In sum, this targeted portfolio of NIDCD-funded research—together with the research blueprint and the NASEM consensus report—provided a critical foundation for the 2021 FDA rule creating the new class of OTC hearing aids. As a result of these research and policy efforts, this FDA rule will make some types of hearing aids less expensive and easier to obtain, potentially improving the health, safety, and well-being of millions of Americans.
Transforming hearing health care for adults in the U.S. remains a public health priority. The NIH applauds the scientists who provided critical evidence leading to the new category of hearing aids, and NIDCD encourages them to redouble their efforts. Gaps in hearing health care access remain to be closed.
The NIDCD actively solicits applications for research projects to fill these gaps and continue identifying barriers to care and ways to improve access. The NIDCD will also continue to help the public understand the importance of hearing health care with resources on its website, such as Hearing: A Gateway to Our World video and the Adult Hearing Health Care webpage.
References:
[1] Hearing loss prevalence in the United States. Lin F, Niparko J, Ferrucci L. Arch Intern Med. 2011 Nov 14;171(20):1851-1852.
[2] Research drives more accessible, affordable hearing care. Tucci DL, King K. The Hearing Journal. May 2020.
[3] A “Goldilocks” approach to hearing aid self-fitting: Ear-canal output and speech intelligibility index. Mackersie C, Boothroyd A, Lithgow, A. Ear and Hearing. Jan 2019.
[4] Self-adjusted amplification parameters produce large between-subject variability and preserve speech intelligibility. Nelson PB, Perry TT, Gregan M, VanTasell, D. Trends in Hearing. 7 Sep 2018.
[5] The effects of service-delivery model and purchase price on hearing-aid outcomes in older adults: A randomized double-blind placebo-controlled clinical trial. Humes LE, Rogers SE, Quigley TM, Main AK, Kinney DL, Herring C. American Journal of Audiology. 1 Mar 2017.
[6] A follow-up clinical trial evaluating the consumer-decides service delivery model. Humes LE, Kinney DL, Main AK, Rogers SE. American Journal of Audiology. 15 Mar 2019.
Links:
National Institute on Deafness and Other Communication Disorders (NIDCD) (NIH)
Funded Research Projects on Accessible and Affordable Hearing Health Care (NIDCD)
Adult Hearing Health Care (NIDCD)
[Note: Acting NIH Director Lawrence Tabak has asked the heads of NIH’s Institutes and Centers (ICs) to contribute occasional guest posts to the blog to highlight some of the interesting science that they support and conduct. This is the ninth in the series of NIH IC guest posts that will run until a new permanent NIH director is in place.]
Posted In: Generic
Tags: aging, audiologist, clinical trial, FDA, hearing, hearing aids, hearing health care, hearing impaired, hearing loss, hearing technology, National Academy of Sciences Engineering and Medicine, National Institute on Deafness and Other Communication Disorders, NIDCD, NIDCD Working Group on Accessible and Affordable Hearing Health Care, older people, over-the-counter hearing aids, Small Business Innovation Research
Biology of Aging Study Shows Why Curbing Calories Counts
Posted on March 8th, 2022 by Richard J. Hodes, M.D., National Institute on Aging
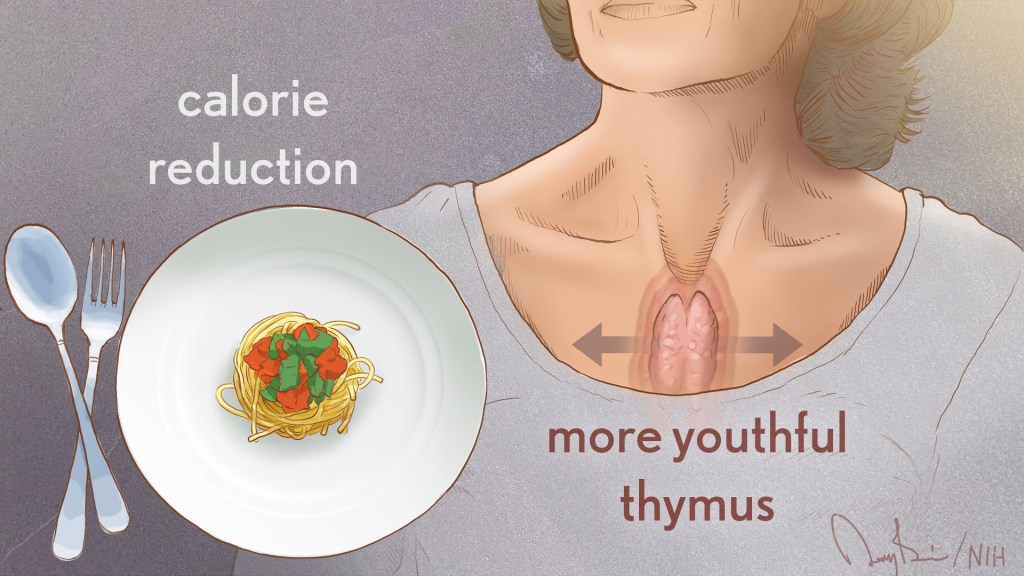
The NIH’s National Institute on Aging (NIA) broadly invests in research to find ways to help people live longer and healthier. As people age, they are more likely to have multiple chronic diseases, and NIA-supported research studies reflect a strong focus on geroscience. This advancing area of science seeks to understand the mechanisms that make aging a major risk factor and driver of common chronic conditions and diseases of older people.
More than 85 years ago, researchers at Cornell University, Ithaca, NY, observed that some lab rodents lived longer when fed a lower calorie diet that otherwise had the appropriate nutrients [1]. Since then, many scientists have studied calorie restriction to shed light on the various biological mechanisms that may explain its benefits and perhaps discover a way to extend healthy years of life, known as our healthspan.
Although there have been many studies of calorie restriction since the Cornell findings, the NIA-supported clinical trial CALERIE, which stands for Comprehensive Assessment of Long-term Effects of Reducing Intake of Energy, provided critical data on the impact of this intervention in people. Completed in 2012, CALERIE was the first carefully controlled study to test whether study participants undergoing moderate calorie restriction would display any of the benefits observed in animal studies.
Volunteers for the CALERIE study were healthy, non-obese adults ages 25 to 45. People in one group were randomly assigned to continue their customary dietary choices, and those in the second group were trained by an expert team of psychologists and dietitians to restrict calories through specific strategies, such as eating smaller servings of food.
In addition to demonstrating that people could sustain moderate calorie restriction for two years, the CALERIE study also showed that this intervention could diminish risk factors for age-related cardiovascular and metabolic diseases [2]. The CALERIE investigators also made their data and biological samples available for other research teams to study further.
Recently, a team led by Vishwa Dixit, Yale University, New Haven, CT, examined CALERIE data to investigate the effects of calorie restriction on immune function. The findings, published in the journal Science, suggest that calorie restriction may improve immune function and reduce chronic inflammation [3,4].
As people age, the size of the thymus, which is part of the immune system, tends to become smaller. As this organ shrinks, its output of T cells declines, which hampers the ability of the immune system to combat infectious diseases. This deficiency of T cells is one of the reasons people over age 40 are at increased susceptibility for a range of diseases.
Dixit’s team noted that MRI scans showed the thymus volume increased among people who reduced their calories for the two-year CALERIE study but was not significantly different in the control group. The increase in thymus size in the group restricting calories was accompanied by an increase in indicators of new T cell production.
Next, the team analyzed immune system effects in belly fat samples from people in the CALERIE study. The team discovered that the PLA2G7 gene—which codes for a protein involved in fat metabolism that is made by immune cells such as T cells—was suppressed after calorie restriction, with evidence that the suppression occurred in immune cells present in fat. They hypothesized that the PLA2G7 gene could have played a role in the improved thymus function resulting from calorie restriction.
To test this hypothesis, the team suppressed the Pla2g7 gene in lab mice. When these mice were two years old, which is equivalent to a human age of about 70, the thymus had not decreased in volume. In addition, the mice had decreased fat mass and lower levels of certain inflammation-promoting substances. These findings suggest that mice without the Pla2g7 gene might have been protected from age-related chronic inflammation, which has been linked to many conditions of old age.
Taken together, the findings extend our understanding of the power of calorie restriction and suggest that it might also improve immune function and reduce chronic inflammation in people. The results also indicate interventions that influence PLA2G7 gene function might have favorable health effects. Additional research is still needed to assess the health effects and to determine whether calorie restriction extends lifespan or healthspan in humans. The NIA is funding more studies to determine the benefits and risks of calorie restriction, as well as the mechanisms that account for its effects.
References:
[1] The effect of retarded growth upon the length of life span and upon the ultimate body size. McCay CM, Crowell MF, Maynard LA. J. Nutr. 1935 July 10(1): 63–79.
[2] A 2-year randomized controlled trial of human caloric restriction: feasibility and effects on predictors of health span and longevity. Ravussin E, Redman LM, Rochon J, Das SK, Fontana L, Kraus WE, Romashkan S, Williamson DA, Meydani SN, Villareal DT, Smith SR, Stein RI, Scott TM, Stewart TM, Saltzman E, Klein S, Bhapkar M, Martin CK, Gilhooly CH, Holloszy JO, Hadley EC, Roberts SB; CALERIE Study Group. J Gerontol A Biol Sci Med Sci. 2015 Sep;70(9):1097-104.
[3] Caloric restriction in humans reveals immunometabolic regulators of health span. Spadaro O, Youm Y, Shchukina I, Ryu S, Sidorov S, Ravussin A, Nguyen K, Aladyeva E, Predeus AN, Smith SR, Ravussin E, Galban C, Artyomov MN, Dixit VD. Science. 2022 Feb 11;375(6581):671-677.
[4] Caloric restriction has a new player. Rhoads TW and Anderson RM. Science. 2022 Feb 11;375(6581):620-621.
Links:
Dietary Restriction (National Institute on Aging, NIH)
What Do We Know About Healthy Aging? (NIA)
Calorie Restriction and Fasting Diets: What Do We Know? (NIA)
Live Long in Good Health: Could Calorie Restriction Mimetics Hold the Key? (NIA)
Geroscience: The Intersection of Basic Aging Biology, Chronic Disease, and Health (NIA)
Comprehensive Assessment of Long-Term Effects of Reducing Intake of Energy (CALERIE) (NIA)
CALERIE Intensive Intervention Database (NIA)
Research Highlights (NIA)
Vishwa Deep Dixit (Yale University, New Haven, CT)
CALERIE Research Network (Duke University, Durham, N.C.)
[Note: Acting NIH Director Lawrence Tabak has asked the heads of NIH’s Institutes and Centers (ICs) to contribute occasional guest posts to the blog to highlight some of the cool science that they support and conduct. This is the fourth in the series of NIH IC guest posts that will run until a new permanent NIH director is in place.]
Posted In: Generic
Tags: aging, CALERIE, calorie, calorie reduction, calorie restriction, calorie-restricted diet, cardiovascular disease, chronic disease, clinical research, Comprehensive Assessment of Long-term Effects of Reducing Intake of Energy, diet, fat metabolism, geroscience, healthspan, immune system, inflammation, metabolic disease, mice, MRI, National Institute on Aging, NIA, PLA2G7, portion size, T cells, thymus
DNA Base Editing May Treat Progeria, Study in Mice Shows
Posted on January 7th, 2021 by Dr. Francis Collins

Credit: Progeria Research Foundation
My good friend Sam Berns was born with a rare genetic condition that causes rapid premature aging. Though Sam passed away in his teens from complications of this condition, called Hutchinson-Gilford progeria syndrome, he’s remembered today for his truly positive outlook on life. Sam expressed it, in part, by his willingness to make adjustments that allowed him, in his words, to put things that he always wanted to do in the “can do” category.
In this same spirit on behalf of the several hundred kids worldwide with progeria and their families, a research collaboration, including my NIH lab, has now achieved a key technical advance to move non-heritable gene editing another step closer to the “can do” category to treat progeria. As published in the journal Nature, our team took advantage of new gene-editing tools to correct for the first time a single genetic misspelling responsible for progeria in a mouse model, with dramatically beneficial effects [1, 2]. This work also has implications for correcting similar single-base typos that cause other inherited genetic disorders.
The outcome of this work is incredibly gratifying for me. In 2003, my NIH lab discovered the DNA mutation that causes progeria. One seemingly small glitch—swapping a “T” in place of a “C” in a gene called lamin A (LMNA)—leads to the production of a toxic protein now known as progerin. Without treatment, children with progeria develop normally intellectually but age at an exceedingly rapid pace, usually dying prematurely from heart attacks or strokes in their early teens.
The discovery raised the possibility that correcting this single-letter typo might one day help or even cure children with progeria. But back then, we lacked the needed tools to edit DNA safely and precisely. To be honest, I didn’t think that would be possible in my lifetime. Now, thanks to advances in basic genomic research, including work that led to the 2020 Nobel Prize in Chemistry, that’s changed. In fact, there’s been substantial progress toward using gene-editing technologies, such as the CRISPR editing system, for treating or even curing a wide range of devastating genetic conditions, such as sickle cell disease and muscular dystrophy
It turns out that the original CRISPR system, as powerful as it is, works better at knocking out genes than correcting them. That’s what makes some more recently developed DNA editing agents and approaches so important. One of them, which was developed by David R. Liu, Broad Institute of MIT and Harvard, Cambridge, MA, and his lab members, is key to these latest findings on progeria, reported by a team including my lab in NIH’s National Human Genome Research Institute and Jonathan Brown, Vanderbilt University Medical Center, Nashville, TN.
The relatively new gene-editing system moves beyond knock-outs to knock-ins [3,4]. Here’s how it works: Instead of cutting DNA as CRISPR does, base editors directly convert one DNA letter to another by enzymatically changing one DNA base to become a different base. The result is much like the find-and-replace function used to fix a typo in a word processor. What’s more, the gene editor does this without cutting the DNA.
Our three labs (Liu, Brown, and Collins) first teamed up with the Progeria Research Foundation, Peabody, MA, to obtain skin cells from kids with progeria. In lab studies, we found that base editors, targeted by an appropriate RNA guide, could successfully correct the LMNA gene in those connective tissue cells. The treatment converted the mutation back to the normal gene sequence in an impressive 90 percent of the cells.
But would it work in a living animal? To get the answer, we delivered a single injection of the DNA-editing apparatus into nearly a dozen mice either three or 14 days after birth, which corresponds in maturation level roughly to a 1-year-old or 5-year-old human. To ensure the findings in mice would be as relevant as possible to a future treatment for use in humans, we took advantage of a mouse model of progeria developed in my NIH lab in which the mice carry two copies of the human LMNA gene variant that causes the condition. Those mice develop nearly all of the features of the human illness
In the live mice, the base-editing treatment successfully edited in the gene’s healthy DNA sequence in 20 to 60 percent of cells across many organs. Many cell types maintained the corrected DNA sequence for at least six months—in fact, the most vulnerable cells in large arteries actually showed an almost 100 percent correction at 6 months, apparently because the corrected cells had compensated for the uncorrected cells that had died out. What’s more, the lifespan of the treated animals increased from seven to almost 18 months. In healthy mice, that’s approximately the beginning of old age.
This is the second notable advance in therapeutics for progeria in just three months. Last November, based on preclinical work from my lab and clinical trials conducted by the Progeria Research Foundation in Boston, the Food and Drug Administration (FDA) approved the first treatment for the condition. It is a drug called Zokinvy, and works by reducing the accumulation of progerin [5]. With long-term treatment, the drug is capable of extending the life of kids with progeria by 2.5 years and sometimes more. But it is not a cure.
We are hopeful this gene editing work might eventually lead to a cure for progeria. But mice certainly aren’t humans, and there are still important steps that need to be completed before such a gene-editing treatment could be tried safely in people. In the meantime, base editors and other gene editing approaches keep getting better—with potential application to thousands of genetic diseases where we know the exact gene misspelling. As we look ahead to 2021, the dream envisioned all those years ago about fixing the tiny DNA typo responsible for progeria is now within our grasp and getting closer to landing in the “can do” category.
References:
[1] In vivo base editing rescues Hutchinson-Gilford Progeria Syndrome in mice. Koblan LW et al. Nature. 2021 Jan 6.
[2] Base editor repairs mutation found in the premature-ageing syndrome progeria. Vermeij WP, Hoeijmakers JHJ. Nature. 6 Jan 2021.
[3] Programmable editing of a target base in genomic DNA without double-stranded DNA cleavage. Komor AC, Kim YB, Packer MS, Zuris JA, Liu DR. Nature. 2016 May 19;533(7603):420-424.
[4] Programmable base editing of A•T to G•C in genomic DNA without DNA cleavage. Gaudelli NM, Komor AC, Rees HA, Packer MS, Badran AH, Bryson DI, Liu DR. Nature. 2017 Nov 23;551(7681):464-471.
[5] FDA approves first treatment for Hutchinson-Gilford progeria syndrome and some progeroid laminopathies. Food and Drug Administration. 2020 Nov 20.
Links:
Progeria (Genetic and Rare Diseases Information Center/NIH)
What are Genome Editing and CRISPR-Cas9? (National Library of Medicine/NIH)
Somatic Cell Genome Editing Program (Common Fund/NIH)
David R. Liu (Harvard University, Cambridge, MA)
Collins Group (National Human Genome Research Institute/NIH)
Jonathan Brown (Vanderbilt University Medical Center, Nashville, TN)
NIH Support: National Human Genome Research Institute; National Center for Advancing Translational Sciences; National Institute of Biomedical Imaging and Bioengineering; National Institute of Allergy and Infectious Diseases; National Institute of General Medical Sciences; Common Fund
Posted In: News
Tags: aging, base editors, CRISPR, DNA, gene editing, genetic diseases, genomics, Hutchinson-Gilford progeria syndrome, inherited disease, lamin-A, LMNA, LMNA gene variant, mouse model, mouse study, progeria, Progeria Research Foundation, progerin, rare disease, sam berns
The People’s Picks for Best Posts
Posted on January 5th, 2021 by Dr. Francis Collins
It’s 2021—Happy New Year! Time sure flies in the blogosphere. It seems like just yesterday that I started the NIH Director’s Blog to highlight recent advances in biology and medicine, many supported by NIH. Yet it turns out that more than eight years have passed since this blog got rolling and we are fast approaching my 1,000th post!
I’m pleased that millions of you have clicked on these posts to check out some very cool science and learn more about NIH and its mission. Thanks to the wonders of social media software, we’ve been able to tally up those views to determine each year’s most-popular post. So, I thought it would be fun to ring in the New Year by looking back at a few of your favorites, sort of a geeky version of a top 10 countdown or the People’s Choice Awards. It was interesting to see what topics generated the greatest interest. Spoiler alert: diet and exercise seemed to matter a lot! So, without further ado, I present the winners:
2013: Fighting Obesity: New Hopes from Brown Fat. Brown fat, one of several types of fat made by our bodies, was long thought to produce body heat rather than store energy. But Shingo Kajimura and his team at the University of California, San Francisco, showed in a study published in the journal Nature, that brown fat does more than that. They discovered a gene that acts as a molecular switch to produce brown fat, then linked mutations in this gene to obesity in humans.
What was also nice about this blog post is that it appeared just after Kajimura had started his own lab. In fact, this was one of the lab’s first publications. One of my goals when starting the blog was to feature young researchers, and this work certainly deserved the attention it got from blog readers. Since highlighting this work, research on brown fat has continued to progress, with new evidence in humans suggesting that brown fat is an effective target to improve glucose homeostasis.
2014: In Memory of Sam Berns. I wrote this blog post as a tribute to someone who will always be very near and dear to me. Sam Berns was born with Hutchinson-Gilford progeria syndrome, one of the rarest of rare diseases. After receiving the sad news that this brave young man had passed away, I wrote: “Sam may have only lived 17 years, but in his short life he taught the rest of us a lot about how to live.”
Affecting approximately 400 people worldwide, progeria causes premature aging. Without treatment, children with progeria, who have completely normal intellectual development, die of atherosclerotic cardiovascular disease, on average in their early teens.
From interactions with Sam and his parents in the early 2000s, I started to study progeria in my NIH lab, eventually identifying the gene responsible for the disorder. My group and others have learned a lot since then. So, it was heartening last November when the Food and Drug Administration approved the first treatment for progeria. It’s an oral medication called Zokinvy (lonafarnib) that helps prevent the buildup of defective protein that has deadly consequences. In clinical trials, the drug increased the average survival time of those with progeria by more than two years. It’s a good beginning, but we have much more work to do in the memory of Sam and to help others with progeria. Watch for more about new developments in applying gene editing to progeria in the next few days.
2015: Cytotoxic T Cells on Patrol. Readers absolutely loved this post. When the American Society of Cell Biology held its first annual video competition, called CellDance, my blog featured some of the winners. Among them was this captivating video from Alex Ritter, then working with cell biologist Jennifer Lippincott-Schwartz of NIH’s Eunice Kennedy Shriver National Institute of Child Health and Human Development. The video stars a roving, specialized component of our immune system called cytotoxic T cells. Their job is to seek out and destroy any foreign or detrimental cells. Here, these T cells literally convince a problem cell to commit suicide, a process that takes about 10 minutes from detection to death.
These cytotoxic T cells are critical players in cancer immunotherapy, in which a patient’s own immune system is enlisted to control and, in some cases, even cure the cancer. Cancer immunotherapy remains a promising area of research that continues to progress, with a lot of attention now being focused on developing immunotherapies for common, solid tumors like breast cancer. Ritter is currently completing a postdoctoral fellowship in the laboratory of Ira Mellman, Genentech, South San Francisco. His focus has shifted to how cancer cells protect themselves from T cells. And video buffs—get this—Ritter says he’s now created even cooler videos that than the one in this post.
2016: Exercise Releases Brain-Healthy Protein. The research literature is pretty clear: exercise is good for the brain. In this very popular post, researchers led by Hyo Youl Moon and Henriette van Praag of NIH’s National Institute on Aging identified a protein secreted by skeletal muscle cells to help explore the muscle-brain connection. In a study in Cell Metabolism, Moon and his team showed that this protein called cathepsin B makes its way into the brain and after a good workout influences the development of new neural connections. This post is also memorable to me for the photo collage that accompanied the original post. Why? If you look closely at the bottom right, you’ll see me exercising—part of my regular morning routine!
2017: Muscle Enzyme Explains Weight Gain in Middle Age. The struggle to maintain a healthy weight is a lifelong challenge for many of us. While several risk factors for weight gain, such as counting calories, are within our control, there’s a major one that isn’t: age. Jay Chung, a researcher with NIH’s National Heart, Lung, and Blood Institute, and his team discovered that the normal aging process causes levels of an enzyme called DNA-PK to rise in animals as they approach middle age. While the enzyme is known for its role in DNA repair, their studies showed it also slows down metabolism, making it more difficult to burn fat.
Since publishing this paper in Cell Metabolism, Chung has been busy trying to understand how aging increases the activity of DNA-PK and its ability to suppress renewal of the cell’s energy-producing mitochondria. Without renewal of damaged mitochondria, excess oxidants accumulate in cells that then activate DNA-PK, which contributed to the damage in the first place. Chung calls it a “vicious cycle” of aging and one that we’ll be learning more about in the future.
2018: Has an Alternative to Table Sugar Contributed to the C. Diff. Epidemic? This impressive bit of microbial detective work had blog readers clicking and commenting for several weeks. So, it’s no surprise that it was the runaway People’s Choice of 2018.
Clostridium difficile (C. diff) is a common bacterium that lives harmlessly in the gut of most people. But taking antibiotics can upset the normal balance of healthy gut microbes, allowing C. diff. to multiply and produce toxins that cause inflammation and diarrhea.
In the 2000s, C. diff. infections became far more serious and common in American hospitals, and Robert Britton, a researcher at Baylor College of Medicine, Houston, wanted to know why. He and his team discovered that two subtypes of C. diff have adapted to feed on the sugar trehalose, which was approved as a food additive in the United States during the early 2000s. The team’s findings, published in the journal Nature, suggested that hospitals and nursing homes battling C. diff. outbreaks may want to take a closer look at the effect of trehalose in the diet of their patients.
2019: Study Finds No Benefit for Dietary Supplements. This post that was another one that sparked a firestorm of comments from readers. A team of NIH-supported researchers, led by Fang Fang Zhang, Tufts University, Boston, found that people who reported taking dietary supplements had about the same risk of dying as those who got their nutrients through food. What’s more, the mortality benefits associated with adequate intake of vitamin A, vitamin K, magnesium, zinc, and copper were limited to amounts that are available from food consumption. The researchers based their conclusion on an analysis of the well-known National Health and Nutrition Examination Survey (NHANES) between 1999-2000 and 2009-2010 survey data. The team, which reported its data in the Annals of Internal Medicine, also uncovered some evidence suggesting that certain supplements might even be harmful to health when taken in excess.
2020: Genes, Blood Type Tied to Risk of Severe COVID-19. Typically, my blog focuses on research involving many different diseases. That changed in 2020 due to the emergence of a formidable public health challenge: the coronavirus disease 2019 (COVID-19) pandemic. Since last March, the blog has featured 85 posts on COVID-19, covering all aspects of the research response and attracting more visitors than ever. And which post got the most views? It was one that highlighted a study, published last June in the New England Journal of Medicine, that suggested the clues to people’s variable responses to COVID-19 may be found in our genes and our blood types.
The researchers found that gene variants in two regions of the human genome are associated with severe COVID-19 and correspondingly carry a greater risk of COVID-19-related death. The two stretches of DNA implicated as harboring risks for severe COVID-19 are known to carry some intriguing genes, including one that determines blood type and others that play various roles in the immune system.
In fact, the findings suggest that people with blood type A face a 50 percent greater risk of needing oxygen support or a ventilator should they become infected with the novel coronavirus. In contrast, people with blood type O appear to have about a 50 percent reduced risk of severe COVID-19.
That’s it for the blog’s year-by-year Top Hits. But wait! I’d also like to give shout outs to the People’s Choice winners in two other important categories—history and cool science images.
Top History Post: HeLa Cells: A New Chapter in An Enduring Story. Published in August 2013, this post remains one of the blog’s greatest hits with readers. The post highlights science’s use of cancer cells taken in the 1950s from a young Black woman named Henrietta Lacks. These “HeLa” cells had an amazing property not seen before: they could be grown continuously in laboratory conditions. The “new chapter” featured in this post is an agreement with the Lacks family that gives researchers access to the HeLa genome data, while still protecting the family’s privacy and recognizing their enormous contribution to medical research. And the acknowledgments rightfully keep coming from those who know this remarkable story, which has been chronicled in both book and film. Recently, the U.S. Senate and House of Representatives passed the Henrietta Lacks Enhancing Cancer Research Act to honor her extraordinary life and examine access to government-funded cancer clinical trials for traditionally underrepresented groups.
Top Snapshots of Life: A Close-up of COVID-19 in Lung Cells. My blog posts come in several categories. One that you may have noticed is “Snapshots of Life,” which provides a showcase for cool images that appear in scientific journals and often dominate Science as Art contests. My blog has published dozens of these eye-catching images, representing a broad spectrum of the biomedical sciences. But the blog People’s Choice goes to a very recent addition that reveals exactly what happens to cells in the human airway when they are infected with the coronavirus responsible for COVID-19. This vivid image, published in the New England Journal of Medicine, comes from the lab of pediatric pulmonologist Camille Ehre, University of North Carolina at Chapel Hill. This image squeezed in just ahead of another highly popular post from Steve Ramirez, Boston University, in 2019 that showed “What a Memory Looks Like.”
As we look ahead to 2021, I want to thank each of my blog’s readers for your views and comments over the last eight years. I love to hear from you, so keep on clicking! I’m confident that 2021 will generate a lot more amazing and bloggable science, including even more progress toward ending the COVID-19 pandemic that made our past year so very challenging.
Posted In: Generic
Tags: aging, blood type, brown fat, C. diff, cancer, cancer immunotherapy, cathepsin, cathepsin B, Clostridium difficile, coronavirus, COVID-19, cytotoxic T cells, diet, dietary supplements, DNA-PK, exercise, FDA, genes, HeLa cells, Henrietta Lacks, Henrietta Lacks Enhancing Cancer Research Act, Hutchinson-Gilford progeria syndrome, Ira Mellman, Jennifer Lippincott-Schwartz, lonafarnib, lung cells, memory, mitochondria, muscle, obesity, progeria, sam berns, T cells, trehalose, weight gain, Zokinvy