beta amyloid – NIH Director's Blog (original) (raw)
Changes in Human Microbiome Precede Alzheimer’s Cognitive Declines
Posted on June 27th, 2023 by Lawrence Tabak, D.D.S., Ph.D.
Caption: The human gut teems with bacteria and other microbes. They contribute to our health but also influence our susceptibility to certain diseases, including Alzheimer’s disease. Credit: Donny Bliss, NIH
In people with Alzheimer’s disease, the underlying changes in the brain associated with dementia typically begin many years—or even decades—before a diagnosis. While pinpointing the exact causes of Alzheimer’s remains a major research challenge, they likely involve a combination of genetic, environmental, and lifestyle factors. Now an NIH-funded study elucidates the role of another likely culprit that you may not have considered: the human gut microbiome, the trillions of diverse bacteria and other microbes that live primarily in our intestines [1].
Earlier studies had showed that the gut microbiomes of people with symptomatic Alzheimer’s disease differ from those of healthy people with normal cognition [2]. What this new work advances is that these differences arise early on in people who will develop Alzheimer’s, even before any obvious symptoms appear.
The science still has a ways to go before we’ll know if specific dietary changes can alter the gut microbiome and modify its influence on the brain in the right ways. But what’s exciting about this finding is it raises the possibility that doctors one day could test a patient’s stool sample to determine if what’s present from their gut microbiome correlates with greater early risk for Alzheimer’s dementia. Such a test would help doctors detect Alzheimer’s earlier and intervene sooner to slow or ideally even halt its advance.
The new findings, reported in the journal Science Translational Medicine, come from a research team led by Gautam Dantas and Beau Ances, Washington University School of Medicine, St. Louis. Ances is a clinician who treats and studies people with Alzheimer’s; Dantas is a basic researcher and expert on the gut microbiome.
The pair struck up a conversation one day about the possible connection between the gut microbiome and Alzheimer’s. While they knew about the earlier studies suggesting a link, they were surprised that nobody had looked at the gut microbiomes of people in the earliest, so-called preclinical, stages of the disease. That’s when dementia isn’t detectable, but the brain has formed amyloid-beta plaques, which are associated with Alzheimer’s.
To take a look, they enrolled 164 healthy volunteers, age 68 to 94, who performed normally on standard tests of cognition. They also collected stool samples from each volunteer and thoroughly analyzed them all the microbes from their gut microbiome. Study participants also kept food diaries and underwent extensive testing, including two types of brain scans, to look for signs of amyloid-beta plaques and tau protein accumulation that precede the onset of Alzheimer’s symptoms.
Among the volunteers, about a third (49 individuals) unfortunately had signs of early Alzheimer’s disease. And, as it turned out, their microbiomes showed differences, too.
The researchers found that those with preclinical Alzheimer’s disease had markedly different assemblages of gut bacteria. Their microbiomes differed in many of the bacterial species present. Those species-level differences also point to differences in the way their microbiomes would be expected to function at a metabolic level. These microbiome changes were observed even though the individuals didn’t seem to have any apparent differences in their diets.
The team also found that the microbiome changes correlated with amyloid-beta and tau levels in the brain. But they did not find any relationship to degenerative changes in the brain, which tend to happen later in people with Alzheimer’s.
The team is now conducting a five-year study that will follow volunteers to get a better handle on whether the differences observed in the gut microbiome are a cause or a consequence of the brain changes seen in Alzheimer’s. If it’s a cause, this discovery would raise the tantalizing possibility that specially formulated probiotics or fecal transplants that promote the growth of “good” bacteria over “bad” bacteria in the gut might slow the development of Alzheimer’s and its most devastating symptoms. It’s an exciting area of research and definitely one worth following in the years ahead.
References:
[1] Gut microbiome composition may be an indicator of preclinical Alzheimer’s disease. Ferreiro AL, Choi J, Ryou J, Newcomer EP, Thompson R, Bollinger RM, Hall-Moore C, Ndao IM, Sax L, Benzinger TLS, Stark SL, Holtzman DM, Fagan AM, Schindler SE, Cruchaga C, Butt OH, Morris JC, Tarr PI, Ances BM, Dantas G. Sci Transl Med. 2023 Jun 14;15(700):eabo2984. doi: 10.1126/scitranslmed.abo2984. Epub 2023 Jun 14. PMID: 37315112.
[2] Gut microbiome alterations in Alzheimer’s disease. Vogt NM, Kerby RL, Dill-McFarland KA, Harding SJ, Merluzzi AP, Johnson SC, Carlsson CM, Asthana S, Zetterberg H, Blennow K, Bendlin BB, Rey FE. Sci Rep. 2017 Oct 19;7(1):13537. doi: 10.1038/s41598-017-13601-y. PMID: 29051531; PMCID: PMC5648830.
Links:
Alzheimer’s Disease and Related Dementias (National Institute on Aging/NIH)
Video: How Alzheimer’s Changes the Brain (NIA)
Dantas Lab (Washington University School of Medicine. St. Louis)
Ances Bioimaging Laboratory (Washington University School of Medicine, St. Louis)
NIH Support: National Institute on Aging; National Institute of Diabetes and Digestive and Kidney Diseases
Posted In: News
Tags: aging, Alzheimer’s disease, amyloid plaques, beta amyloid, brain, dementia, diet, fecal transplant, gut, gut bacteria, gut microbiome, microbiome, probiotics, tau
Largest-Ever Alzheimer’s Gene Study Brings New Answers
Posted on March 12th, 2019 by Dr. Francis Collins
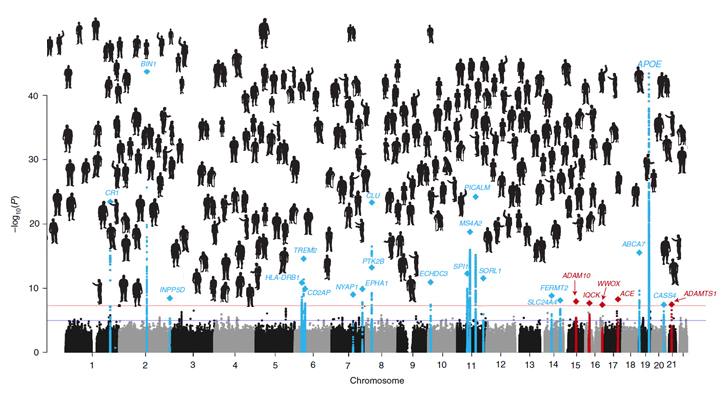
Predicting whether someone will get Alzheimer’s disease (AD) late in life, and how to use that information for prevention, has been an intense focus of biomedical research. The goal of this work is to learn not only about the genes involved in AD, but how they work together and with other complex biological, environmental, and lifestyle factors to drive this devastating neurological disease.
It’s good news to be able to report that an international team of researchers, partly funded by NIH, has made more progress in explaining the genetic component of AD. Their analysis, involving data from more than 35,000 individuals with late-onset AD, has identified variants in five new genes that put people at greater risk of AD [1]. It also points to molecular pathways involved in AD as possible avenues for prevention, and offers further confirmation of 20 other genes that had been implicated previously in AD.
The results of this largest-ever genomic study of AD suggests key roles for genes involved in the processing of beta-amyloid peptides, which form plaques in the brain recognized as an important early indicator of AD. They also offer the first evidence for a genetic link to proteins that bind tau, the protein responsible for telltale tangles in the AD brain that track closely with a person’s cognitive decline.
The new findings are the latest from the International Genomics of Alzheimer’s Project (IGAP) consortium, led by a large, collaborative team including Brian Kunkle and Margaret Pericak-Vance, University of Miami Miller School of Medicine, Miami, FL. The effort, spanning four consortia focused on AD in the United States and Europe, was launched in 2011 with the aim of discovering and mapping all the genes that contribute to AD.
An earlier IGAP study including about 25,500 people with late-onset AD identified 20 common gene variants that influence a person’s risk for developing AD late in life [2]. While that was terrific progress to be sure, the analysis also showed that those gene variants could explain only a third of the genetic component of AD. It was clear more genes with ties to AD were yet to be found.
So, in the study reported in Nature Genetics, the researchers expanded the search. While so-called genome-wide association studies (GWAS) are generally useful in identifying gene variants that turn up often in association with particular diseases or other traits, the ones that arise more rarely require much larger sample sizes to find.
To increase their odds of finding additional variants, the researchers analyzed genomic data for more than 94,000 individuals, including more than 35,000 with a diagnosis of late-onset AD and another 60,000 older people without AD. Their search led them to variants in five additional genes, named IQCK, ACE, ADAM10, ADAMTS1, and WWOX, associated with late-onset AD that hadn’t turned up in the previous study.
Further analysis of those genes supports a view of AD in which groups of genes work together to influence risk and disease progression. In addition to some genes influencing the processing of beta-amyloid peptides and accumulation of tau proteins, others appear to contribute to AD via certain aspects of the immune system and lipid metabolism.
Each of these newly discovered variants contributes only a small amount of increased risk, and therefore probably have limited value in predicting an average person’s risk of developing AD later in life. But they are invaluable when it comes to advancing our understanding of AD’s biological underpinnings and pointing the way to potentially new treatment approaches. For instance, these new data highlight intriguing similarities between early-onset and late-onset AD, suggesting that treatments developed for people with the early-onset form also might prove beneficial for people with the more common late-onset disease.
It’s worth noting that the new findings continue to suggest that the search is not yet over—many more as-yet undiscovered rare variants likely play a role in AD. The search for answers to AD and so many other complex health conditions—assisted through collaborative data sharing efforts such as this one—continues at an accelerating pace.
References:
[1] Genetic meta-analysis of diagnosed Alzheimer’s disease identifies new risk loci and implicates Aβ, tau, immunity and lipid processing. Kunkle BW, Grenier-Boley B, Sims R, Bis JC, et. al. Nat Genet. 2019 Mar;51(3):414-430.
[2] Meta-analysis of 74,046 individuals identifies 11 new susceptibility loci for Alzheimer’s disease. Lambert JC, Ibrahim-Verbaas CA, Harold D, Naj AC, Sims R, Bellenguez C, DeStafano AL, Bis JC, et al. Nat Genet. 2013 Dec;45(12):1452-8.
Links:
Alzheimer’s Disease Genetics Fact Sheet (National Institute on Aging/NIH)
Genome-Wide Association Studies (NIH)
Margaret Pericak-Vance (University of Miami Health System, FL)
NIH Support: National Institute on Aging; National Heart, Lung, and Blood Institute; National Human Genome Research Institute; National Institute of Allergy and Infectious Diseases; Eunice Kennedy Shriver National Institute of Child Health and Human Development; National Institute of Diabetes and Digestive and Kidney Disease; National Institute of Neurological Disorders and Stroke
Posted In: News
Tags: AD, aging, aging brain, Alzheimer’s disease, amyloid plaques, beta amyloid, brain, dementia, early-onset Alzheimer's disease, gene variants, genes, genetics, genome-wide association studies, genomics, GWAS, IGAP, International Genomics of Alzheimer's Project, late-onset Alzheimer's disease, older people, tau
Sleep Loss Encourages Spread of Toxic Alzheimer’s Protein
Posted on February 5th, 2019 by Dr. Francis Collins

Credit: iStock/bowdenimages
In addition to memory loss and confusion, many people with Alzheimer’s disease have trouble sleeping. Now an NIH-funded team of researchers has evidence that the reverse is also true: a chronic lack of sleep may worsen the disease and its associated memory loss.
The new findings center on a protein called tau, which accumulates in abnormal tangles in the brains of people with Alzheimer’s disease. In the healthy brain, active neurons naturally release some tau during waking hours, but it normally gets cleared away during sleep. Essentially, your brain has a system for taking the garbage out while you’re off in dreamland.
The latest findings in studies of mice and people further suggest that sleep deprivation upsets this balance, allowing more tau to be released, accumulate, and spread in toxic tangles within brain areas important for memory. While more study is needed, the findings suggest that regular and substantial sleep may play an unexpectedly important role in helping to delay or slow down Alzheimer’s disease.
It’s long been recognized that Alzheimer’s disease is associated with the gradual accumulation of beta-amyloid peptides and tau proteins, which form plaques and tangles that are considered hallmarks of the disease. It has only more recently become clear that, while beta-amyloid is an early sign of the disease, tau deposits track more closely with disease progression and a person’s cognitive decline.
Such findings have raised hopes among researchers including David Holtzman, Washington University School of Medicine, St. Louis, that tau-targeting treatments might slow this devastating disease. Though much of the hope has focused on developing the right drugs, some has also focused on sleep and its nightly ability to reset the brain’s metabolic harmony.
In the new study published in Science, Holtzman’s team set out to explore whether tau levels in the brain naturally are tied to the sleep-wake cycle [1]. Earlier studies had shown that tau is released in small amounts by active neurons. But when neurons are chronically activated, more tau gets released. So, do tau levels rise when we’re awake and fall during slumber?
The Holtzman team found that they do. The researchers measured tau levels in brain fluid collected from mice during their normal waking and sleeping hours. (Since mice are nocturnal, they sleep primarily during the day.) The researchers found that tau levels in brain fluid nearly double when the animals are awake. They also found that sleep deprivation caused tau levels in brain fluid to double yet again.
These findings were especially interesting because Holtzman’s team had already made a related finding in people. The team found that healthy adults forced to pull an all-nighter had a 30 percent increase on average in levels of unhealthy beta-amyloid in their cerebrospinal fluid (CSF).
The researchers went back and reanalyzed those same human samples for tau. Sure enough, the tau levels were elevated on average by about 50 percent.
Once tau begins to accumulate in brain tissue, the protein can spread from one brain area to the next along neural connections. So, Holtzman’s team wondered whether a lack of sleep over longer periods also might encourage tau to spread.
To find out, mice engineered to produce human tau fibrils in their brains were made to stay up longer than usual and get less quality sleep over several weeks. Those studies showed that, while less sleep didn’t change the original deposition of tau in the brain, it did lead to a significant increase in tau’s spread. Intriguingly, tau tangles in the animals appeared in the same brain areas affected in people with Alzheimer’s disease.
Another report by Holtzman’s team appearing early last month in Science Translational Medicine found yet another link between tau and poor sleep. That study showed that older people who had more tau tangles in their brains by PET scanning had less slow-wave, deep sleep [2].
Together, these new findings suggest that Alzheimer’s disease and sleep loss are even more intimately intertwined than had been realized. The findings suggest that good sleep habits and/or treatments designed to encourage plenty of high quality Zzzz’s might play an important role in slowing Alzheimer’s disease. On the other hand, poor sleep also might worsen the condition and serve as an early warning sign of Alzheimer’s.
For now, the findings come as an important reminder that all of us should do our best to get a good night’s rest on a regular basis. Sleep deprivation really isn’t a good way to deal with overly busy lives (I’m talking to myself here). It isn’t yet clear if better sleep habits will prevent or delay Alzheimer’s disease, but it surely can’t hurt.
References:
[1] The sleep-wake cycle regulates brain interstitial fluid tau in mice and CSF tau in humans. Holth JK, Fritschi SK, Wang C, Pedersen NP, Cirrito JR, Mahan TE, Finn MB, Manis M, Geerling JC, Fuller PM, Lucey BP, Holtzman DM. Science. 2019 Jan 24.
[2] Reduced non-rapid eye movement sleep is associated with tau pathology in early Alzheimer’s disease. Lucey BP, McCullough A, Landsness EC, Toedebusch CD, McLeland JS, Zaza AM, Fagan AM, McCue L, Xiong C, Morris JC, Benzinger TLS, Holtzman DM. Sci Transl Med. 2019 Jan 9;11(474).
Links:
Alzheimer’s Disease and Related Dementias (National Institute on Aging/NIH)
Accelerating Medicines Partnership: Alzheimer’s Disease (NIH)
Holtzman Lab (Washington University School of Medicine, St. Louis)
NIH Support: National Institute on Aging; National Institute of Neurological Disorders and Stroke; National Center for Advancing Translational Sciences; National Cancer Institute; National Institute of Biomedical Imaging and Bioengineering
Posted In: News
Tags: aging, Alzheimer's, Alzheimer’s disease, beta amyloid, brain, cognition, dementia, memory, sleep, sleep loss, sleep-wake cycle, tau, tau protein
Alzheimer’s Disease: Tau Protein Predicts Early Memory Loss
Posted on May 24th, 2016 by Dr. Francis Collins
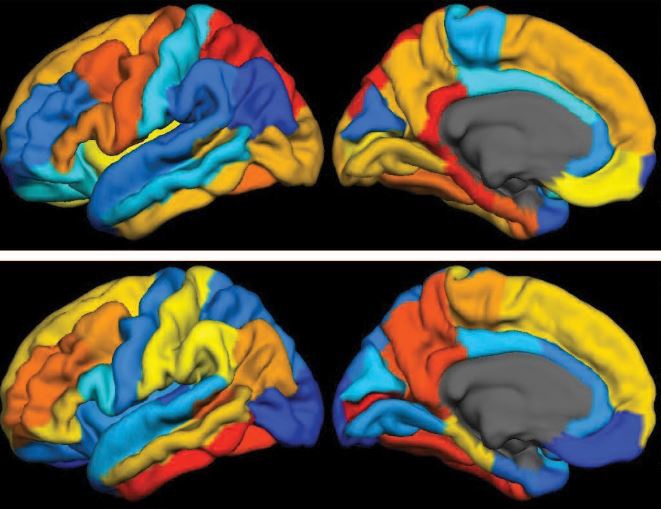
Caption: PET scan images show distribution of tau (top panel) and beta-amyloid (bottom panel) across a brain with early Alzheimer’s disease. Red indicates highest levels of protein binding, dark blue the lowest, yellows and oranges indicate moderate binding.
Credit: Brier et al., Sci Transl Med
In people with Alzheimer’s disease, changes in the brain begin many years before the first sign of memory problems. Those changes include the gradual accumulation of beta-amyloid peptides and tau proteins, which form plaques and tangles that are considered hallmarks of the disease. While amyloid plaques have received much attention as an early indicator of disease, until very recently there hadn’t been any way during life to measure the buildup of tau protein in the brain. As a result, much less is known about the timing and distribution of tau tangles and its relationship to memory loss.
Now, in a study published in Science Translational Medicine, an NIH-supported research team has produced some of the first maps showing where tau proteins build up in the brains of people with early Alzheimer’s disease [1]. The new findings suggest that while beta-amyloid remains a reliable early sign of Alzheimer’s disease, tau may be a more informative predictor of a person’s cognitive decline and potential response to treatment.
Tags: Accelerating Medicines Partnership, age-related memory loss, aging, Alzheimer’s disease, AMP, AMP-AD, ß-amyloid, beta amyloid, brain, brain scan, cerebral cortex, cognitive decline, dementia, early Alzheimer's disease, frontal lobe, imaging, memory, memory loss, neurological disease, neurology, parietal lobe, PET scans, tau, tau protein, temporal lobe, translational medicine
Repurposing an “Old” Drug for Alzheimer’s Disease
Posted on April 7th, 2015 by Dr. Francis Collins
Caption: Here I am with Senator Barbara Mikulski (center) and NCATS Director Chris Austin (right). Credit: NIH
Alzheimer’s disease research is among the many areas of biomedical science that Senator Barbara Mikulski has championed during her nearly 40 years on Capitol Hill. And it’s easy to understand why the Senator is concerned: an estimated 5 million Americans age 65 and older have Alzheimer’s disease, and those numbers are expected to rise exponentially as the U.S. population continues to age.
So, I was thrilled to have some encouraging progress to report last week when Senator Mikulski (D-MD) paid a visit to NIH’s National Center for Advancing Translational Sciences (NCATS) in Gaithersburg, MD. After a whirlwind tour of the cutting-edge robotics facility for high throughput screening of small molecules, she joined me and NCATS Director Dr. Chris Austin in announcing that, thanks to an innovative public-private partnership, an experimental drug originally developed to fight cancer is now showing promise against Alzheimer’s disease.
Tags: aging, Alzheimer’s disease, AMP, AstraZeneca, Barbara Mikulski, beta amyloid, drug development, National Plan to Address Alzheimer's Disease, NCATS, neurology, New Therapeutic Uses program, saracatinib, src kinases, translational science, Yale University School of Medicine
