health disparities – NIH Director's Blog (original) (raw)
NIH’s CARE for Health Primary Care Research Network: Connecting the Lab, the Clinic, and the Community
Posted on October 8th, 2024 by Dr. Monica M. Bertagnolli

Credit: Donny Bliss
Since I became NIH Director last year, one key principle has guided my vision and approach: Our work is not finished when we deliver scientific discoveries; our work is finished when all people are living long and healthy lives.
But unfortunately, we’re seeing some alarming trends in the health of the U.S. population. It’s a bit of a puzzle. On one hand, significant advances in biomedical research over the last several decades have led to lifesaving interventions for a range of diseases and conditions. At the same time, the overall health of the people in this country appears to be stalled and even getting worse.
Looking at one measure of health, U.S. life expectancy is no longer steadily increasing—in fact, we’ve had a decline in life expectancy over the last decade. And though this included a dramatic drop because of the COVID-19 pandemic, the rate was declining before that. Life expectancy in the U.S. is also low compared to peer nations, even though we spend much more money on our health system. Disparities in mortality also persist among certain racial and ethnic groups and geographic regions. Our health is determined not only by the genes we inherit from our parents, but by our environment and social and economic factors. We know that in the U.S. today, your zip code can significantly impact your health.
I believe that biomedical research can play a key role in reversing these trends. In my first blog post, I explained how one of my goals as NIH Director is to ensure that the biomedical research enterprise is more inclusive to people from all walks of life, and I noted we can engage more communities as our research partners by meeting people where they are. Despite having the knowledge and technology to do so, our research and advances are not reaching everyone they should. Many people are not adequately represented in clinical research, and research data is especially lacking for people who are older, uninsured, belong to minority groups, or live in rural locations. Many people also face barriers to participating in clinical research, such as arranging and paying for transportation, getting time off from work and coordinating childcare, or lack of trust in medical institutions. To address these concerning trends in health, we need to do a better job of connecting the lab, the clinic, and the community.
In September, we moved closer to this goal by announcing awards as part of a new NIH primary care clinical research network that aims to improve access to and involve communities in the clinical research that informs medical care. The Communities Advancing Research Equity for Health™ or CARE for Health™ program will actively engage communities historically underrepresented in clinical research. This effort is very close to my heart, as I was born and raised in a rural community, and I’d like to tell you more about how it will work.
In this program, NIH will connect with primary care providers and their patients, giving them access to research and the opportunity to participate in clinical trials. By engaging people on the front lines of health care—in the primary care clinician’s office—we will build an infrastructure that leads to sustained relationships with primary care providers and patients and earns people’s trust. Many of the areas of the country where we want to focus do not have medical specialists, and primary care providers are often the only practitioners available for every health challenge. Through CARE for Health, we want to integrate clinical care with research to support knowledge generation that meets the needs of people in all communities.
The awards we’ve announced, totaling over 5millioninfundingforthefirstyear,willsupportthreeNetworkResearchHubstoestablishtheprogram’sinitialinfrastructure.ThefirstawardeeinstitutionsareOregonHealthandScienceUniversity,theUniversityofWisconsin−Madison,andWestVirginiaUniversity.TheseinstitutionswillparticipateinthreeongoingNIH−fundedclinicaltrialsthatcoverarangeoftopicsimportanttoprimaryhealthcare,includingpainmanagement,opioidandpolysubstanceuse,andgout,withmanymorestudiesonareasimportanttodiversecommunitiestocomeinthefuture.Overall,NIHis[investingapproximately5 million in funding for the first year, will support three Network Research Hubs to establish the program’s initial infrastructure. The first awardee institutions are Oregon Health and Science University, the University of Wisconsin-Madison, and West Virginia University. These institutions will participate in three ongoing NIH-funded clinical trials that cover a range of topics important to primary health care, including pain management, opioid and polysubstance use, and gout, with many more studies on areas important to diverse communities to come in the future. Overall, NIH is [investing approximately 5millioninfundingforthefirstyear,willsupportthreeNetworkResearchHubstoestablishtheprogram’sinitialinfrastructure.ThefirstawardeeinstitutionsareOregonHealthandScienceUniversity,theUniversityofWisconsin−Madison,andWestVirginiaUniversity.TheseinstitutionswillparticipateinthreeongoingNIH−fundedclinicaltrialsthatcoverarangeoftopicsimportanttoprimaryhealthcare,includingpainmanagement,opioidandpolysubstanceuse,andgout,withmanymorestudiesonareasimportanttodiversecommunitiestocomeinthefuture.Overall,NIHisinvestingapproximately30 million in total over fiscal years 2024 and 2025 to pilot the program, which is supported through the NIH Common Fund. After the first year, we will aim to broaden the program so communities throughout the country can participate.
CARE for Health is a new paradigm for biomedical research. NIH has never had a network for the primary care medical environment that works across all 27 Institutes and Centers of NIH. We are starting it with a pilot program because we know that for a program of this scale, we first need to learn from our research teams and from the primary care clinicians who are going to help us bring this kind of research to their communities. We will also ask community members what their health priorities are and allow them to select the research studies that are most meaningful to them. In the future, CARE for Health partners will have a long menu of studies to pick from based on local interests and needs.
Ultimately, we believe that this program will have a meaningful impact on health outcomes, especially among those who have been previously underrepresented and underserved in medical research.
Reference:
Bertagnolli MM. Connecting lab, clinic, and community. Science. DOI:10.1126/science.adq2140 (2024).
NIH Support: NIH Common Fund
Diagnosis and Treatment of Mental Health Conditions During and After Pregnancy is On the Rise, But Disparities Still Exist
Posted on April 18th, 2024 by Dr. Monica M. Bertagnolli

Credit: Marco/Adobe Stock
Pregnancy and childbirth are often thought of as joyful times. Yet, we know that mental health conditions including perinatal depression, anxiety, and post-traumatic stress disorder (PTSD) are common complications during and after pregnancy, and this is contributing to a maternal health crisis in this country.
Now, a trio of NIH-supported studies reported in the journal Health Affairs show that diagnosis and treatment of mental health conditions such as anxiety, depression, and PTSD during pregnancy and in the first year after giving birth rose significantly in Americans with private health insurance from 2008 to 2020.1,2,3 While these are encouraging signs of increasing mental health awareness and service use, these studies also showed that this increase hasn’t happened equally across all demographic groups and states, making it clear there’s more work to do to ensure that people from all walks of life have access to the care they need, regardless of their race, ethnicity, geographic location, financial status, or other factors.
The findings come from a research team including Kara Zivin and Stephanie Hall and their colleagues at the University of Michigan, Ann Arbor. They recognized a worrying crisis in maternal mental illness in the U.S., with serious health risks and many potential long-term negative impacts for new parents and their infants. While earlier studies had looked at the prevalence of mental health conditions in the perinatal period, Zivin and Hall wanted to explore nationwide trends in the diagnosis of PTSD and what they refer to as perinatal mood and anxiety disorders (PMAD) among people giving birth between ages 15 and 44.
In the first study, using a database of administrative medical claims representing insured people across the U.S., the researchers examined PMAD diagnoses (including depressive and anxiety disorders) among those with private health insurance and found that the diagnosis rate had increased by more than 93% over the 12-year period under study. The rates also showed a sharp uptick after 2015, after the Affordable Care Act (ACA) went into effect in 2014. By 2020, 28% of those who were pregnant or in their postpartum period received a diagnosis of PMAD.
The researchers found that rate of suicidality (that is, suicidal ideation or diagnoses of self-harm) among people during pregnancy and just after childbirth more than doubled over the same period, although that rate dipped among those who had received a PMAD diagnosis. The rate at which individuals who were pregnant or in their postpartum period received any form of talk therapy covered by their private insurance increased by 16% from 2008 to 2020.
The second report focused specifically on PTSD diagnoses among privately insured people over the same period. The data show a near quadrupling of PTSD diagnoses in the months surrounding childbirth, with diagnoses in nearly 2% of privately insured people by 2020. Most of the increase in PTSD diagnoses occurred in people who had already been diagnosed with PMAD.
In the third study, the researchers wanted to learn if there were increases in antidepressant prescriptions for those diagnosed with PMAD, especially after new guidelines from several professional organizations were issued in 2015 and 2016. The findings show a decrease of 3% per year from 2008 to 2016, followed by a 32% increase in 2017, with prescription rates continuing to climb each year through 2020. By 2020, slightly less than half of those diagnosed with PMAD received a prescription for an antidepressant, suggesting that the clinical recommendations made a difference in clinical practice. However, there are signs of racial disparities, with White people with PMAD diagnoses receiving antidepressants more often than people in other racial groups.
In fact, all three studies show differences between people in different age, race, ethnicity, and geographic groups. For example, White people were more likely to be diagnosed with PTSD during the perinatal period, followed by Black people. By comparison, people of unknown race, as well as people who identified as Hispanic and Asian, were diagnosed less often. At the same time, the largest increase in PMAD diagnoses among races and ethnicities was among Black people, increasing from 14% of deliveries in 2008 to 22% in 2020.
Looking at age groups, the initial prevalence of PMAD diagnoses was highest among people aged 40 to 44, yet the youngest people (aged 15 to 24) experienced the largest increase in diagnoses. The youngest age group also had the largest increases in antidepressant prescriptions, and those aged 15 to 26 were more likely to be diagnosed with PTSD than those in older age groups. The first study also showed wide variation between states in the rate of PMAD diagnoses before and after implementation of the ACA.
The findings suggest there have been improvements in doctors’ recognition and treatment of PMAD and PTSD in the months surrounding childbirth. Increases in health insurance coverage and laws requiring equal treatment of mental health conditions may also be playing a role, along with greater social awareness and acceptance of mental health conditions generally. The researchers noted, however, that the findings don’t represent people with government-funded health insurance or those who lack health insurance completely.
It will be important to learn in future studies more about those who may still not be receiving the mental health care they need. The researchers report plans to look deeper into changes that have taken place at the state level and the impact of the pandemic and the rise of telehealth since 2020. Other recent NIH-supported research suggests that relatively straightforward interventions to reduce postpartum anxiety and depression can be remarkably effective. The key step will be not only identifying interventions that work, but also figuring out how to deliver effective treatments to the people who need them.
References:
[1] Zivin K, et al. Perinatal Mood and Anxiety Disorders Rose Among Privately Insured People, 2008-20. Health Affairs. DOI: 10.1377/hlthaff.2023.01437 (2024).
[2] Hall SV, et al. Perinatal Posttraumatic Stress Disorder Diagnoses Among Commercially Insured People Increased, 2008-20. Health Affairs. DOI: 10.1377/hlthaff.2023.01447 (2024).
[3] Hall SV, et al. Antidepressant Prescriptions Increased for Privately Insured People With Perinatal Mood And Anxiety Disorder, 2008-20. Health Affairs. DOI: 10.1377/hlthaff.2023.01448 (2024).
NIH Support: National Institute of Mental Health, National Institute on Minority Health and Health Disparities
Words Matter, Actions Have Impact: Updating the NIH Mission Statement
Posted on September 28th, 2023 by Lawrence Tabak, D.D.S., Ph.D.

Credit: Donny Bliss, NIH
I’ve previously written and spoken about how diverse perspectives are essential to innovation and scientific advancement.1 Scientists and experts with different backgrounds and lived experiences can offer diverse and creative solutions to solve complex problems. We’re taking steps to create a culture within the biomedical and behavioral research enterprise of inclusion, equity, and respect for every member of society. We are also working to strengthen our efforts to include populations in research that have not been historically included or equitably treated.
As part of our effort to ensure that all people are included in NIH research, we’re updating our mission statement to reflect better the spirit of the agency’s work to optimize health for all people. The proposed, new statement is as follows:
“To seek fundamental knowledge about the nature and behavior of living systems and to apply that knowledge to optimize health and prevent or reduce illness for all people.”
Recently, we asked a team of subject matter experts to form a subgroup of the Advisory Committee to the Director’s Working Group on Diversity to advise NIH on how we can support the inclusion of people with disabilities in the scientific workforce and in the research enterprise. One of the subgroup’s recommendations was to update the current NIH mission statement to remove “reducing disability.” The subgroup explained that this language could be interpreted as perpetuating ableist beliefs that people with disabilities are flawed and need to be “fixed.”
Disability is often viewed solely as a medical problem requiring a cure or correction. However, this view can be stigmatizing as it focuses only on a perceived flaw in the individual. It does not account for how people identify and view themselves. It also does not account for the ways that society can be unaccommodating for people with disabilities.2,3 It’s important that we recognize the varied, nuanced and complex lived experiences among people with disabilities, many of whom may also face additional barriers as members of racial, ethnic, sexual and gender minority groups, people with lower incomes, and people who live in rural communities that are medically underserved.
Some of you may recall that we updated our mission statement in 2013 to remove phrasing that implied disability was a burden, since many people do not find their disabilities to be burdensome. As we re-examine our mission statement again in 2023, I’m reminded that strengthening diversity, equity, inclusion and accessibility (DEIA) is an ongoing process requiring our sustained engagement.
The input we’ve received has made it clear that words matter—language can perpetuate prejudices and implicit attitudes, which in turn can affect people’s behavior. We also acknowledge that it is time for the agency to review and consider how the words of our mission statement may affect the direction of our science.
In response, we are seeking the public’s input on the proposed, revised statement to ensure that it reflects the NIH mission as accurately as possible. The NIH mission should be inclusive of those who conduct research, those who participate in research, and those we serve—the American public. Anyone interested in providing feedback can send it to this submission website through Nov. 24, 2023.
We are grateful for the subgroup’s work and appreciate their time examining this issue in depth. I also want to recognize the helpful feedback that we’ve received from the disability community within NIH through the years, including recent listening sessions that helped guide the development of NIH’s DEIA Strategic Plan.
Going beyond the scientific workforce, both the Strategic Plan and the subgroup’s report recognize the importance of research on health disparities. People with disabilities often experience health conditions leading to poorer health and face discrimination, inequality and structural barriers that inhibit access to health care, resulting in poorer health outcomes. NIH recently designated people with disabilities as a population with health disparities to encourage research specific to the health issues and unmet health needs of the disability community. NIH also issued a funding opportunity calling for research applications that address the intersecting impact of disability, race, ethnicity, and socioeconomic status on healthcare access and health outcomes.
The subgroup provided additional recommendations that we’re in the process of reviewing. We know one of our key challenges is data gathering that would give us a better snapshot of the workforce and the research we support. According to the CDC, 1 in 4 adults in the United States have a disability. However, in 2022 only 1.3% of principal investigators on NIH research grant applications and awards self-reported a disability. In 2022, 8.6% of the NIH workforce reported having a disability; however, I recognize that this is likely not reflective of the true percentage. We know that some people do not want to self-disclose for numerous reasons, including the fear of discrimination.
We hope that, in part, changing the mission statement would be a step in the right direction of changing the culture at NIH and the larger biomedical and behavioral research enterprise. I know that our efforts have sometimes fallen short, but we will continually work to foster a culture of inclusive excellence where people with disabilities and all people feel like they truly belong and are embraced as an asset to the NIH mission.
References:
[1] MA Bernard et al. The US National Institutes of Health approach to inclusive excellence. Nature Medicine DOI:10.1038/s41591-021-01532-1 (2021)
[2] DS Dunn & EE Andrews. Person-first and identity-first language: Developing psychologists’ cultural competence using disability language The American Psychologist DOI: 10.1037/a0038636 (2015)
[3] International Classification of Functioning, Disability and Health (2002) Towards a Common Language for Functioning, Disability and Health. World Health Organization https://cdn.who.int/media/docs/default-source/classification/icf/icfbeginnersguide.pdf
Links:
ACD Working Group on Diversity, Subgroup on Individuals with Disabilities, NIH
Request for Information: Inviting Comments and Suggestions on Updating the NIH Mission Statement, NIH
NIH designates people with disabilities as a population with health disparities, Sept. 26, 2023, NIH News Releases
NIH-Wide Strategic Plan for Diversity, Equity, Inclusion, and Accessibility (DEIA), NIH
Disability and Health Overview, CDC
Data on Researchers’ Self-Reported Disability Status, NIH Office Of Extramural Research
Total NIH Workforce Demographics for Fiscal Year 2022 Fourth Quarter, NIH Office of Equity, Diversity, and Inclusion
Posted In: Health, News, Science
Tags: DEIA, disabilities, disability, diversity, health disparities, health disparity, health equity, health outcomes, inclusion, NIH Mission Statement
Immune Resilience is Key to a Long and Healthy Life
Posted on June 20th, 2023 by Lawrence Tabak, D.D.S., Ph.D.

Caption: A new measure of immunity called immune resilience is helping researchers find clues as to why some people remain healthier even in the face of varied inflammatory stressors. Credit: Modified from Shutterstock/Ground Picture
Do you feel as if you or perhaps your family members are constantly coming down with illnesses that drag on longer than they should? Or, maybe you’re one of those lucky people who rarely becomes ill and, if you do, recovers faster than others.
It’s clear that some people generally are more susceptible to infectious illnesses, while others manage to stay healthier or bounce back more quickly, sometimes even into old age. Why is this? A new study from an NIH-supported team has an intriguing answer [1]. The difference, they suggest, may be explained in part by a new measure of immunity they call immune resilience—the ability of the immune system to rapidly launch attacks that defend effectively against infectious invaders and respond appropriately to other types of inflammatory stressors, including aging or other health conditions, and then quickly recover, while keeping potentially damaging inflammation under wraps.
The findings in the journal Nature Communications come from an international team led by Sunil Ahuja, University of Texas Health Science Center and the Department of Veterans Affairs Center for Personalized Medicine, both in San Antonio. To understand the role of immune resilience and its effect on longevity and health outcomes, the researchers looked at multiple other studies including healthy individuals and those with a range of health conditions that challenged their immune systems.
By looking at multiple studies in varied infectious and other contexts, they hoped to find clues as to why some people remain healthier even in the face of varied inflammatory stressors, ranging from mild to more severe. But to understand how immune resilience influences health outcomes, they first needed a way to measure or grade this immune attribute.
The researchers developed two methods for measuring immune resilience. The first metric, a laboratory test called immune health grades (IHGs), is a four-tier grading system that calculates the balance between infection-fighting CD8+ and CD4+ T-cells. IHG-I denotes the best balance tracking the highest level of resilience, and IHG-IV denotes the worst balance tracking the lowest level of immune resilience. An imbalance between the levels of these T cell types is observed in many people as they age, when they get sick, and in people with autoimmune diseases and other conditions.
The researchers also developed a second metric that looks for two patterns of expression of a select set of genes. One pattern associated with survival and the other with death. The survival-associated pattern is primarily related to immune competence, or the immune system’s ability to function swiftly and restore activities that encourage disease resistance. The mortality-associated genes are closely related to inflammation, a process through which the immune system eliminates pathogens and begins the healing process but that also underlies many disease states.
Their studies have shown that high expression of the survival-associated genes and lower expression of mortality-associated genes indicate optimal immune resilience, correlating with a longer lifespan. The opposite pattern indicates poor resilience and a greater risk of premature death. When both sets of genes are either low or high at the same time, immune resilience and mortality risks are more moderate.
In the newly reported study initiated in 2014, Ahuja and his colleagues set out to assess immune resilience in a collection of about 48,500 people, with or without various acute, repetitive, or chronic challenges to their immune systems. In an earlier study, the researchers showed that this novel way to measure immune status and resilience predicted hospitalization and mortality during acute COVID-19 across a wide age spectrum [2].
The investigators have analyzed stored blood samples and publicly available data representing people, many of whom were healthy volunteers, who had enrolled in different studies conducted in Africa, Europe, and North America. Volunteers ranged in age from 9 to 103 years. They also evaluated participants in the Framingham Heart Study, a long-term effort to identify common factors and characteristics that contribute to cardiovascular disease.
To examine people with a wide range of health challenges and associated stresses on their immune systems, the team also included participants who had influenza or COVID-19, and people living with HIV. They also included kidney transplant recipients, people with lifestyle factors that put them at high risk for sexually transmitted infections, and people who’d had sepsis, a condition in which the body has an extreme and life-threatening response following an infection.
The question in all these contexts was the same: How well did the two metrics of immune resilience predict an individual’s health outcomes and lifespan? The short answer is that immune resilience, longevity, and better health outcomes tracked together well. Those with metrics indicating optimal immune resilience generally had better health outcomes and lived longer than those who had lower scores on the immunity grading scale. Indeed, those with optimal immune resilience were more likely to:
- Live longer,
- Resist HIV infection or the progression from HIV to AIDS,
- Resist symptomatic influenza,
- Resist a recurrence of skin cancer after a kidney transplant,
- Survive COVID-19, and
- Survive sepsis.
The study also revealed other interesting findings. While immune resilience generally declines with age, some people maintain higher levels of immune resilience as they get older for reasons that aren’t yet known, according to the researchers. Some people also maintain higher levels of immune resilience despite the presence of inflammatory stress to their immune systems such as during HIV infection or acute COVID-19. People of all ages can show high or low immune resilience. The study also found that higher immune resilience is more common in females than it is in males.
The findings suggest that there is a lot more to learn about why people differ in their ability to preserve optimal immune resilience. With further research, it may be possible to develop treatments or other methods to encourage or restore immune resilience as a way of improving general health, according to the study team.
The researchers suggest it’s possible that one day checkups of a person’s immune resilience could help us to understand and predict an individual’s health status and risk for a wide range of health conditions. It could also help to identify those individuals who may be at a higher risk of poor outcomes when they do get sick and may need more aggressive treatment. Researchers may also consider immune resilience when designing vaccine clinical trials.
A more thorough understanding of immune resilience and discovery of ways to improve it may help to address important health disparities linked to differences in race, ethnicity, geography, and other factors. We know that healthy eating, exercising, and taking precautions to avoid getting sick foster good health and longevity; in the future, perhaps we’ll also consider how our immune resilience measures up and take steps to achieve or maintain a healthier, more balanced, immunity status.
References:
[1] Immune resilience despite inflammatory stress promotes longevity and favorable health outcomes including resistance to infection. Ahuja SK, Manoharan MS, Lee GC, McKinnon LR, Meunier JA, Steri M, Harper N, Fiorillo E, Smith AM, Restrepo MI, Branum AP, Bottomley MJ, Orrù V, Jimenez F, Carrillo A, Pandranki L, Winter CA, Winter LA, Gaitan AA, Moreira AG, Walter EA, Silvestri G, King CL, Zheng YT, Zheng HY, Kimani J, Blake Ball T, Plummer FA, Fowke KR, Harden PN, Wood KJ, Ferris MT, Lund JM, Heise MT, Garrett N, Canady KR, Abdool Karim SS, Little SJ, Gianella S, Smith DM, Letendre S, Richman DD, Cucca F, Trinh H, Sanchez-Reilly S, Hecht JM, Cadena Zuluaga JA, Anzueto A, Pugh JA; South Texas Veterans Health Care System COVID-19 team; Agan BK, Root-Bernstein R, Clark RA, Okulicz JF, He W. Nat Commun. 2023 Jun 13;14(1):3286. doi: 10.1038/s41467-023-38238-6. PMID: 37311745.
[2] Immunologic resilience and COVID-19 survival advantage. Lee GC, Restrepo MI, Harper N, Manoharan MS, Smith AM, Meunier JA, Sanchez-Reilly S, Ehsan A, Branum AP, Winter C, Winter L, Jimenez F, Pandranki L, Carrillo A, Perez GL, Anzueto A, Trinh H, Lee M, Hecht JM, Martinez-Vargas C, Sehgal RT, Cadena J, Walter EA, Oakman K, Benavides R, Pugh JA; South Texas Veterans Health Care System COVID-19 Team; Letendre S, Steri M, Orrù V, Fiorillo E, Cucca F, Moreira AG, Zhang N, Leadbetter E, Agan BK, Richman DD, He W, Clark RA, Okulicz JF, Ahuja SK. J Allergy Clin Immunol. 2021 Nov;148(5):1176-1191. doi: 10.1016/j.jaci.2021.08.021. Epub 2021 Sep 8. PMID: 34508765; PMCID: PMC8425719.
Links:
COVID-19 Research (NIH)
HIV Info (NIH)
Sepsis (National Institute of General Medical Sciences/NIH)
Sunil Ahuja (University of Texas Health Science Center, San Antonio)
Framingham Heart Study (National Heart, Lung, and Blood Institute/NIH)
“A Secret to Health and Long Life? Immune Resilience, NIAID Grantees Report,” NIAID Now Blog, June 13, 2023
NIH Support: National Institute of Allergy and Infectious Diseases; National Institute on Aging; National Institute of Mental Health; National Institute of General Medical Sciences; National Heart, Lung, and Blood Institute
Posted In: News
Tags: aging, cardiovascular disease, CD4+, CD8+, COVID-19, Framingham Heart Study, genomics, health disparities, HIV, IHG, immune health grades, immune resilience, immune system, immunity, influenza, kidney transplantation, lifespan, longevity, sepsis, skin cancer, T cells
RECOVER: What Clinical Research Comes Next for Helping People with Long COVID
Posted on March 2nd, 2023 by Gary Gibbons, M.D., National Heart, Lung, and Blood Institute; Walter Koroshetz, M.D., National Institute of Neurological Disorders and Stroke; Hugh Auchincloss, M.D., National Institute of Allergy and Infectious Diseases

One family of RECOVER research participants helping to answer questions about Long COVID. Credit: RECOVER
“I connected with RECOVER to be a part of the answers that I was looking for when I was at my worst.” Long COVID patient and RECOVER representative, Nitza Rochez (Bronx, NY)
People, like Nitza Rochez, who are living with Long COVID—the wide-ranging health issues that can follow an infection with SARS-CoV-2, the coronavirus that causes COVID-19—experience disabling symptoms with significant physical, emotional and financial consequences.
The NIH has been engaging and listening to Nitza and others living with Long COVID even before the start of its Researching COVID to Enhance Recovery (RECOVER) Initiative. But now, with the launch of RECOVER, patients and those with affected family or community members have joined researchers, clinicians, and experts in their efforts to unlock the mysteries of Long COVID. All have come together to understand what causes the condition, identify who is most at risk, and determine how to prevent and treat it.
RECOVER is unprecedented in its size and scope as the most-diverse, deeply characterized cohort of Long COVID patients. We’ve enlisted the help of many patient volunteers, who have enrolled in observational studies designed to help researchers learn as much as possible about people who have Long COVID.
Indeed, thousands of research participants are now providing health information and undergoing in-depth medical evaluations and tests, enabling investigators to look for trends. Additionally, studies of millions of electronic medical records are providing insights about those who have received care during the pandemic. More than 40 studies are being conducted to identify the causes of disease, potential biomarkers of Long COVID, and new therapeutic targets.
In all, RECOVER’s research assets are voluminous. They involve invaluable contributions from many people and communities, including research volunteers, research investigators, and clinical specialists. In addition, millions of health records and numerous related tissues and specimens are being analyzed for possible leads.
At the center of it all is the National Community Engagement Group (NCEG). The NCEG is comprised of people living with Long COVID and those representing others living with the condition, and it is truly instrumental to the initiative’s progress in understanding how and why SARS-CoV-2 impacts people in different ways. It’s also helping researchers learn why some people recover while others do not.
So far, we’ve learned that people hospitalized with COVID-19 are twice as likely to have Long COVID than those who were not hospitalized for infection. We’ve also learned that members of racial and ethnic minority groups with Long COVID were more likely to have been hospitalized with COVID-19.
Similarly, disparities in Long COVID exist within those living in areas with particular environmental exposures [1], and those who were already burdened by other diseases and conditions—such as diabetes and chronic pulmonary disease [2]. We’ve also discovered that the certain types of symptoms of Long COVID are consistent among patients regardless of which SARS-CoV-2 variant caused their initial infection. Yet, people infected with the earlier variants have a higher number of symptoms than those infected with more recent variants.
Patient experiences have guided and will continue to guide the study designs and trajectory of RECOVER. Now, fueled by the knowledge that we have gained, RECOVER is preparing to advance to the next phase of discovery—testing interventions in clinical trials to see if they can help people with Long COVID.
To prepare, we are beginning to identify potential clinical trial sites. This important step will help us to find the right places with the right staff and capabilities for enrolling the appropriate patient populations needed to implement the studies. We’ll ensure that the public knows when these upcoming clinical trials are ready to enroll.
Of course, the design of these RECOVER clinical trials will be critical, and insights gained from patients have been key in this process. Results from RECOVER study questionnaires, surveys, and discussions with people experiencing Long COVID identified symptom clusters considered to be the most significant and burdensome to patients. These include sleep disorders, “brain fog” (trouble thinking clearly), exercise intolerance and fatigue, and nervous system dysfunction affecting people’s ability to regulate normal body functions like heart rate and body temperature.
These patient observations have effectively guided the design of the clinical trials that will evaluate whether certain interventions and therapies can help alleviate symptoms that are part of these specific clusters. We’re excited to be advancing toward this phase of the initiative and, again, are very grateful to patient representatives like Nitza, quoted above, for getting us to this phase.
Effective evaluation of those treatments will be important, too. Early in the pandemic, while many clinical trials were launching, most were not large enough or did not have the appropriate objectives to define effective treatments for acute COVID-19. This left clinicians with few clear options when faced with patients needing help.
Learning from this experience, the RECOVER trials will be harmonized to ensure coordinated and efficient evaluation of interventions—in other words, all potential therapies will be using the same protocols platforms and the same data elements. This consistency accelerates our understanding and strengthens the certainty of findings.
Given the widespread and diverse impact that the virus has on the body, it is highly likely that more than one treatment will be needed for each kind of patient experience. Finding solutions for everyone—people of all races, ethnicities, genders, ages, and geographic locations—is paramount.
RECOVER patient representative, Juan Lewis, of San Antonio shared with us, “In April 2020, I was fighting for my life, and today I fight for my quality of life. COVID impacted me physically, mentally, socially, and financially.”
For people like Juan who are experiencing debilitating Long COVID symptoms, we know that finding answers as quickly as possible is critical. As we look ahead to the next 12 months, we’ll continue the studies evaluating the underlying causes, risk factors, and outcomes of Long Covid, and we anticipate significant scientific progress on research leading to Long COVID treatments.
Keep an eye on the RECOVER website for updates on our progress, and published findings.
References:
[1] Identifying environmental risk factors for post-acute sequelae of SARS-CoV-2 infection: An EHR-based cohort study from the recover program. Zhang Y, Hu H, Fokaidis V, V CL, Xu J, Zang C, Xu Z, Wang F, Koropsak M, Bian J, Hall J, Rothman RL, Shenkman EA, Wei WQ, Weiner MG, Carton TW, Kaushal R. Environ Adv. 2023 Apr;11:100352.
[2] Identifying who has long COVID in the USA: a machine learning approach using N3C data. Pfaff ER, Girvin AT, Bennett TD, Bhatia A, Brooks IM, Deer RR, Dekermanjian JP, Jolley SE, Kahn MG, Kostka K, McMurry JA, Moffitt R, Walden A, Chute CG, Haendel MA; N3C Consortium. Lancet Digit Health. 2022 Jul;4(7):e532-e541.
Links:
RECOVER: Researching COVID to Enhance Recovery
Long COVID: Ask NIH Leader about Latest Research (YouTube)
NIH Builds Large Nationwide Study Population of Tens of Thousands to Support Research on Long-Term Effects of COVID-19, NIH News Release, September 15, 2021
Understanding Long-Term COVID-19 Symptoms and Enhancing Recovery, NIH Director’s Blog, October 4, 2022.
NIH RECOVER Research Identifies Potential Long COVID Disparities. NIH News Release, February 16, 2023.
NIH RECOVER Listening Session, June 2021 (NIH Videocast)
NIH RECOVER Listening Session: Understanding Long COVID Across Communities of Color and Those Hardest Hit by COVID, January 21, 2022 (NIH Videocast)
Note: Dr. Lawrence Tabak, who performs the duties of the NIH Director, has asked the heads of NIH’s Institutes, Centers, and Offices to contribute occasional guest posts to the blog to highlight some of the interesting science that they support and conduct. This is the 25th in the series of NIH guest posts that will run until a new permanent NIH director is in place.
Posted In: Generic
Tags: clinical trials, coronavirus, COVID-19, health disparities, long COVID, Long COVID syndrome, NCEG, novel coronavirus, pandemic, RECOVER Initiative, Researching COVID to Enhance Recovery
A Look Back at Science’s 2022 Breakthroughs
Posted on January 3rd, 2023 by Lawrence Tabak, D.D.S., Ph.D.
Credit: National Institute of Allergy and Infectious Diseases, NIH; Centers for Disease Control and Prevention; Shutterstock/tobe24, Midjourney Inc.
Happy New Year! I hope everyone finished 2022 with plenty to celebrate, whether it was completing a degree or certification, earning a promotion, attaining a physical fitness goal, or publishing a hard-fought scientific discovery.
If the latter, you are in good company. Last year produced some dazzling discoveries, and the news and editorial staff at the journal Science kept a watchful eye on the most high-impact advances of 2022. In December, the journal released its list of the top 10 advances across the sciences, from astronomy to zoology. In case you missed it, Science selected NASA’s James Webb Space Telescope (JWST) as the 2022 Breakthrough of the Year [1].
This unique space telescope took 20 years to complete, but it has turned out to be time well spent. Positioned 1.5-million-kilometers from Earth, the JWST and its unprecedented high-resolution images of space have unveiled the universe anew for astronomers and wowed millions across the globe checking in online. The telescope’s image stream, beyond its sheer beauty, will advance study of the early Universe, allowing astronomers to discover distant galaxies, explore the early formation of stars, and investigate the possibility of life on other planets.
While the biomedical sciences didn’t take home the top prize, they were well represented among _Science_’s runner-up breakthroughs. Some of these biomedical top contenders also have benefited, directly or indirectly, from NIH efforts and support. Let’s take a look:
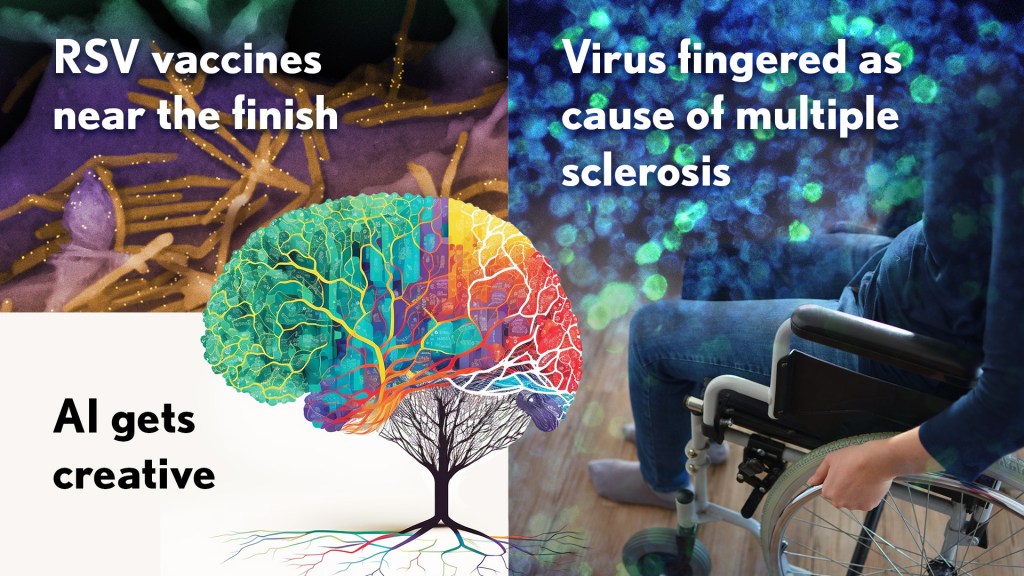
RSV vaccines nearing the finish line: It’s been one of those challenging research marathons. But scientists last year started down the homestretch with the first safe-and-effective vaccine for respiratory syncytial virus (RSV), a leading cause of severe respiratory illness in the very young and the old.
In August, the company Pfizer presented evidence that its experimental RSV vaccine candidate offered protection for those age 60 and up. Later, they showed that the same vaccine, when administered to pregnant women, helped to protect their infants against RSV for six months after birth. Meanwhile, in October, the company GSK announced encouraging results from its late-stage phase III trial of an RSV vaccine in older adults.
As Science noted, the latest clinical progress also shows the power of basic science. For example, researchers have been working with chemically inactivated versions of the virus to develop the vaccine. But these versions have a key viral surface protein that changes its shape after fusing with a cell to start an infection. In this configuration, the protein elicits only weak levels of needed protective antibodies.
Back in 2013, Barney Graham, then with NIH’s National Institute of Allergy and Infectious Diseases (NIAID), and colleagues, solved the problem [2]. Graham’s NIH team discovered a way to lock the protein into its original prefusion state, which the immune system can better detect. This triggers higher levels of potent antibodies, and the discovery kept the science—and the marathon—moving forward.
These latest clinical advances come as RSV and other respiratory viruses, including SARS-CoV-2, the cause of COVID-19, are sending an alarming number of young children to the hospital. The hope is that researchers will cross the finish line this year or next, and we’ll have the first approved RSV vaccine.
Virus fingered as cause of multiple sclerosis: Researchers have long thought that multiple sclerosis, or MS, has a viral cause. Pointing to the right virus with the required high degree of certainty has been the challenge, slowing progress on the treatment front for those in need. As published in Science last January, Alberto Ascherio, Harvard T.H. Chan School of Public Health, Boston, and colleagues produced the strongest evidence yet that MS is caused by the Epstein-Barr virus (EBV), a herpesvirus also known for causing infectious mononucleosis [3].
The link between EBV and MS had long been suspected. But it was difficult to confirm because EBV infections are so widespread, and MS is so disproportionately rare. In the recent study, the NIH-supported researchers collected blood samples every other year from more than 10 million young adults in the U.S. military, including nearly 1,000 who were diagnosed with MS during their service. The evidence showed that the risk of an MS diagnosis increased 32-fold after EBV infection, but it held steady following infection with any other virus. Levels in blood serum of a biomarker for MS neurodegeneration also went up only after an EBV infection, suggesting that the viral illness is a leading cause for MS.
Further evidence came last year from a discovery published in the journal Nature by William Robinson, Stanford University School of Medicine, Stanford, CA, and colleagues. The NIH-supported team found a close resemblance between an EBV protein and one made in the healthy brain and spinal cord [4]. The findings suggest an EBV infection may produce antibodies that mistakenly attack the protective sheath surrounding our nerve cells. Indeed, the study showed that up to one in four people with MS had antibodies that bind both proteins.
This groundbreaking research suggests that an EBV vaccine and/or antiviral drugs that thwart this infection might ultimately prevent or perhaps even cure MS. Of note, NIAID launched last May an early-stage clinical trial for an experimental EBV vaccine at the NIH Clinical Center, Bethesda, MD.
AI Gets Creative: _Science_’s 2021 Breakthrough of the Year was AI-powered predictions of protein structure. In 2022, AI returned to take another well-deserved bow. This time, Science singled out AI’s now rapidly accelerating entry into once uniquely human attributes, such as artistic expression and scientific discovery.
On the scientific discovery side, Science singled out AI’s continued progress in getting creative with the design of novel proteins for vaccines and myriad other uses. One technique, called “hallucination,” generates new proteins from scratch. Researchers input random amino acid sequences into the computer, and it randomly and continuously mutates them into sequences that other AI tools are confident will fold into stable proteins. This greatly simplifies the process of protein design and frees researchers to focus their efforts on creating a protein with a desired function.
AI research now engages scientists around world, including hundreds of NIH grantees. Taking a broader view of AI, NIH recently launched the Artificial Intelligence/Machine Learning Consortium to Advance Health Equity and Researcher Diversity (AIM-AHEAD) Program. It will help to create greater diversity within the field, which is a must. A lack of diversity could perpetuate harmful biases in how AI is used, how algorithms are developed and trained, and how findings are interpreted to avoid health disparities and inequities for underrepresented communities.
And there you have it, some of the 2022 breakthroughs from _Science_‘s news and editorial staff. Of course, the highlighted biomedical breakthroughs don’t capture the full picture of research progress. There were many other milestone papers published in 2022 that researchers worldwide will build upon in the months and years ahead to make further progress in their disciplines and, for some, draw the attention of _Science_’s news and editorial staff. Here’s to another productive year in biomedical research, which the blog will continue to feature and share with you as it unfolds in 2023.
References:
[1] 2022 Breakthrough of the Year. Science. Dec 15, 2022.
[2] Structure of RSV fusion glycoprotein trimer bound to a prefusion-specific neutralizing antibody. McLellan JS, Chen M, Leung S, Kwong PD, Graham BS, et al. Science. 2013 May 31;340(6136):1113-1117.
[3] Longitudinal analysis reveals high prevalence of Epstein-Barr virus associated with multiple sclerosis. Bjornevik K, Cortese M, Healy BC, Kuhle J, Mina MJ, Leng Y, Elledge SJ, Niebuhr DW, Scher AI, Munger KL, Ascherio A. Science. 2022 Jan 21;375(6578):296-301.
[4] Clonally expanded B cells in multiple sclerosis bind EBV EBNA1 and GlialCAM. Lanz TV, Brewer RC, Steinman L, Robinson WH, et al. Nature. 2022 Mar;603(7900):321-327.
Links:
Respiratory Syncytial Virus (RSV) (National Institute of Allergy and Infectious Diseases/NIH)
Multiple Sclerosis (National Institute of Neurological Disorders and Stroke/NIH)
Barney Graham (Morehouse School of Medicine, Atlanta)
Alberto Ascherio (Harvard T.H. Chan School of Public Health, Boston)
Robinson Lab (Stanford Medicine, Stanford, CA)
James Webb Space Telescope (Goddard Space Flight Center/NASA, Greenbelt, MD)
Posted In: News
Tags: 2022 Science Breakthrough of the Year, AI, AI Hallucination, AIM-AHEAD, basic research, Creative AI, EBV, EBV vaccine, Epstein-Barr, GSK, health disparities, James Webb Space Telescope, JWST, machine learning, MS, multiple sclerosis, NIH Clinical Center, Pfizer, respiratory syncytial virus, RSV, RSV vaccine, SARS-CoV-2, vaccine, virus
NIH Collaboration Seeks to Help Understand U.S. Burden of Health Disparities: Why Your County Matters
Posted on September 20th, 2022 by Eliseo J. Pérez-Stable, M.D., National Institute on Minority Health and Health Disparities

Credit: Edgar B. Dews III, National Institute on Minority Health and Health Disparities, NIH
Since the early 1990s, federal support of research has increased to understand minority health and identify and address health disparities. Research in these areas has evolved from a starting point of developing a basic descriptive understanding of health disparities and who is most affected. Now, it is discovering the underlying complexity of factors involved in health outcomes to inform interventions and reduce these disparities.
One of these many factors is where we live, learn, work, and play and how that affects different people. A group of NIH scientists and their colleagues recently published a study in the journal The Lancet that they hope is a step toward better understanding geographic disparities and their role in health equity [1].

Caption: Bottom acronyms are American Indian and Alaska Native (AIAN) and Asian Pacific Islander (API). Credit: GBD US Health Disparities
As Director of NIH’s National Institute on Minority Health and Health Disparities (NIMHD), I worked with NIMHD’s Scientific Director, Anna María Nápoles, to conceive the study and establish the Global Burden of Disease (GBD) U.S. Health Disparities Collaborators at NIH with five NIH Institutes and two Offices. Through this collaboration, NIH funded the Institute for Health Metrics and Evaluation (IHME), University of Washington to conduct the analysis. The IHME has worked for 30 years on the GBD project in over 200 countries.
The Lancet paper offered the first comprehensive U.S. county-level life expectancy estimates to highlight the significant gaps that persist among racial and ethnic populations across the nation. The analysis revealed that despite overall life expectancy gains of 2.3 years from 2000–2019, Black populations experienced shorter life expectancy than White populations.
In addition, American Indian and Alaska Native populations’ life expectancy did not improve and, in fact, decreased in most counties. We found national-level life expectancy advantages for Hispanic/Latino and Asian populations ranging from three to seven years, respectively, compared to White populations. But there were notable exceptions for Hispanic/Latino populations in selected counties in the Southwest.
Certainly the most-alarming trend identified in the paper was that during the study’s last 10 years (2010–2019), life expectancy growth was stagnant across all races and ethnicities. Moreover, 60 percent of U.S. counties experienced a decrease in life expectancy.
While these findings provide an important frame for how disparities exist along many dimensions—by race, ethnicity, and geographic region—they also highlight these differences within our local communities. This level of detail offers an unprecedented opportunity for researchers and public health leaders to focus on where these differences are the most prominent, and possibly give us a clearer picture on what can be done about it.
These data raise many important questions, too. What can we learn from places that are doing well in caring for their most disadvantaged populations? How can these factors be sustained, replicated, and transferred to other places? Are there current policies and/or community services that contribute to or inhibit gaining access to appropriate clinical care, healthy and affordable food, good schools, and/or economic opportunities?
To help answer these questions, the GBD U.S. Health Disparities Collaborators at NIH, in partnership with IHME, have developed a comprehensive database and interactive data visualization tool that provides life expectancy and all-cause mortality by race and ethnicity for 3,110 U.S. counties from 2000-2019. Efforts are underway to expand the database to include causes of death and risk factors by race/ethnicity and education, as well as to disaggregate some of the major racial-ethnic groups.
Using IHME’s established model of comprehensive and replicable data collection, the joint effort aims to improve access to health data resources, bolster analytic approaches, and deliver user-friendly estimates to the wider research and health policy community. The collection’s standardized, comprehensive, historical, and real-time data can be the cornerstone for efforts to address disparities and advance health equity.
It is important to note that the Lancet study only included data from before the COVID-19 pandemic. The pandemic’s disproportionate effect on overall mortality and life expectancy has exacerbated existing health disparities. Disaggregated data are essential in helping to understand the underlying mechanisms of health disparities and guiding the development and implementation of interventions that address local needs.
As a clinician scientist, I have made a personal commitment at NIMHD to foster and encourage data collection with standardized measures, harmonization, and efficient data sharing to help us explore the nuances within all populations and their communities. Without these guiding principles for managing data, inequities remain unseen and unaddressed. Scientists, clinicians, and policymakers can all potentially benefit from this work if we use the data to inform our actions. It is an opportunity to implement real change in our NIH-wide combined efforts to reduce health disparities and improve quality of life and longevity for all populations.
Reference:
[1] Life expectancy by county, race, and ethnicity in the USA, 2000-19: a systematic analysis of health disparities. GBD US Health Disparities Collaborators. Lancet. 2022 Jul 2;400(10345):25-38.
Links:
Understand Health Disparities Series (National Institute on Minority Health and Health Disparities/NIH)
HD Pulse (NIMHD)
PhenX Social Determinants of Health Toolkit (NIMHD)
Institute for Health Metrics (University of Washington, Seattle)
NIH Support: The members of the GBD U.S. Health Disparities Collaborators at NIH include: National Heart, Lung, and Blood Institute; National Cancer Institute; National Institute on Aging; National Institute of Arthritis and Musculoskeletal and Skin Diseases; NIH Office of Disease Prevention; NIH Office of Behavioral and Social Science Research
Note: Dr. Lawrence Tabak, who performs the duties of the NIH Director, has asked the heads of NIH’s Institutes and Centers (ICs) to contribute occasional guest posts to the blog to highlight some of the interesting science that they support and conduct. This is the 17th in the series of NIH IC guest posts that will run until a new permanent NIH director is in place.
Posted In: Generic
Tags: Alaska Native life expectancy, American Indian life expectancy, Asian life expectancy, Black life expectancy, counties, county, ethnicity, GBD U.S. Health Disparities Collaborators at NIH, health disparities, health policy, Hispanic/Latino life expectancy, IHME, Institute for Health Metrics and Evaluation, life expectancy, longevity, minority health, NIMHD, Pacific Islander life expectancy, public health, public health data, quality of life, race, White, White life expectancy, whites
Suicide Prevention Research in a Rapidly Changing World
Posted on September 6th, 2022 by Joshua A. Gordon, M.D., Ph.D., National Institute of Mental Health

Credit: iStock/PeopleImages
As I sit down to write this blog, the COVID-19 pandemic continues to have a widespread impact, and we’re all trying to figure out our “new normal.” For some, figuring out the new normal has been especially difficult, and that’s something for all of us to consider during September, which is National Suicide Prevention Awareness Month. It’s such an important time to share what we know about suicide prevention and consider how we can further this knowledge to those in need.
At NIH’s National Institute of Mental Health (NIMH), we’ve been asking ourselves: What have we learned about suicide risk and prevention during the pandemic? And how should our research evolve to reflect a rapidly changing world?
Addressing Disparities
Over the last few years, people have been concerned about the pandemic’s impact on suicide rates. So far, data suggest that the overall suicide rate in the U.S. has remained steady. But there is concerning evidence that the pandemic has disproportionately affected suicide risk in historically underserved communities.
For example, data suggest that people in minority racial and ethnic groups experienced greater increases in suicidal thoughts during the pandemic [1]. Additional data indicate that suicide rates may be rising among some young adult racial and ethnic minority groups [2].
Structural racism and other social and environmental factors are major drivers of mental health disparities, and NIMH continues to invest in research to understand how these social determinants of health influence suicide risk. This research includes investigations into the effects of long-term and daily discrimination.
To mitigate these effects, it is critical that we identify specific underlying mechanisms so that we can develop targeted interventions. To this end, NIMH is supporting research in underserved communities to identify suicide risk and the protective factors and effective strategies for reducing this risk (e.g., RFA-MH-22-140, RFA-MH-21-188, RFA-MH-21-187). There are important lessons to be learned that we can’t afford to miss.
Building Solid Foundations
The pandemic also underscored the urgent need to support youth mental health. Indeed, in December 2021, U.S. Surgeon General Dr. Vivek Murthy issued the Advisory on Protecting Youth Mental Health, calling attention to increasing rates of depression and suicidal behaviors among young people. Crucially, the advisory highlighted the need to “recognize that mental health is an essential part of overall health.”
At NIMH, we know that establishing a foundation for good mental health early on can support a person’s overall health and well-being over a lifetime. In light of this, we are investing in research to identify effective prevention efforts that can help set kids on positive mental health trajectories early in life.
Additionally, by re-analyzing research investments already made, we are looking to see whether these early prevention efforts have meaningful impacts on later suicide risk and mental health outcomes. These findings may help to improve a range of systems—such as schools, social services, and health care—to better support kids’ mental health needs.
Improving and Expanding Access
The pandemic has also shown us that telehealth can be an effective means of delivering and increasing access to mental health care. The NIMH has supported research examining telehealth as a tool for improving suicide prevention services, including the use of digital tools that can help extend provider reach and support individuals at risk for suicide.
At the same time, NIMH is investing in work to understand the most effective ways to help providers use evidence-based approaches to prevent suicide. This research helps inform federal partners and others about the best ways to support policies and practices that help prevent suicide deaths.
In July, the Substance Abuse and Mental Health Services Administration (SAMHSA) launched the 988 Suicide & Crisis Lifeline, a three-digit suicide prevention and mental health crisis number. This service builds on the existing National Suicide Prevention Lifeline, allowing anyone to call or text 988 to connect with trained counselors and mental health services. Research supported by NIMH helped build the case for such lifelines, and now we’re calling for research aimed at identifying the best ways to help people use this evolving crisis support system.
Looking Ahead
With these and many other efforts, we are hopeful that people who are at risk for suicidal thoughts and behaviors will be able to access the evidence-based support and services they need. This National Suicide Prevention Awareness Month, I’d like to issue a call to action: Help raise awareness by sharing resources on how to recognize the warning signs for suicide and how to get help. By working together, we can prevent suicide and save lives.
References:
[1] Racial and ethnic disparities in the prevalence of stress and worry, mental health conditions, and increased substance use among adults during the COVID-19 pandemic – United States, April and May 2020. McKnight-Eily LR, Okoro CA, Strine TW, Verlenden J, Hollis ND, Njai R, Mitchell EW, Board A, Puddy R, Thomas C. MMWR Morb Mortal Wkly Rep. 2021 Feb 5;70(5):162-166.
[2] One Year In: COVID-19 and Mental Health. National Institute of Mental Health Director’s Message. April 9, 2021.
Links:
988 Suicide & Crisis Lifeline (Substance Abuse and Mental Health Services Administration, Rockville, MD)
Substance Abuse and Mental Health Services Administration Treatment Locator (SAMHSA)
Help for Mental Illnesses (National Institute of Mental Health/NIH)
Suicide Prevention (NIMH)
Digital Shareables on Suicide Prevention (NIMH)
Digital Shareables on Coping with COVID-19 (NIMH)
NIMH Director’s Messages about COVID-19 (NIMH)
NIMH Director’s Messages about Suicide (NIMH)
Note: Dr. Lawrence Tabak, who performs the duties of the NIH Director, has asked the heads of NIH’s Institutes and Centers (ICs) to contribute occasional guest posts to the blog to highlight some of the interesting science that they support and conduct. This is the 16th in the series of NIH IC guest posts that will run until a new permanent NIH director is in place.
Posted In: Generic
Tags: 988, 988 Suicide & Crisis Lifeline, adolescents, COVID-19, crisis support system, depression, evidence-based care, health disparities, mental health, National Suicide Prevention Lifeline, NIMH, pandemic, racial disparities, schools, suicide, suicide prevention, telehealth, underserved communities, Vivek Murthy, youth mental health
Research to Address the Real-Life Challenges of Opioid Crisis
Posted on May 10th, 2022 by Lawrence Tabak, D.D.S., Ph.D.

Caption: NIDA Director Nora Volkow (center), HEAL Initiative Director Rebecca Baker (right), and I discuss NIH’s latest efforts to combat opioid crisis. Credit: Pierce Harman for Rx Drug Abuse & Heroin Summit 2022.
While great progress has been made in controlling the COVID-19 pandemic, America’s opioid crisis continues to evolve in unexpected ways. The opioid crisis, which worsened during the pandemic and now involves the scourge of fentanyl, claims more than 70,000 lives each year in the United States [1]. But throughout the pandemic, NIH has continued its research efforts to help people with a substance use disorder find the help that they so need. These efforts include helping to find relief for the millions of Americans who live with severe and chronic pain.
Recently, I traveled to Atlanta for the Rx and Illicit Drug Summit 2022. While there, I moderated an evening fireside chat with two of NIH’s leaders in combating the opioid crisis: Nora Volkow, director of the National Institute on Drug Abuse (NIDA); and Rebecca Baker, director of Helping to End Addiction Long-term® (HEAL) initiative. What follows is an edited, condensed transcript of our conversation.
Tabak: Let’s start with Nora. When did the opioid crisis begin, and how has it changed over the years
Volkow: It started just before the year 2000 with the over-prescription of opioid medications. People were becoming addicted to them, many from diverted product. By 2010, CDC developed guidelines that decreased the over-prescription. But then, we saw a surge in heroin use. That turned the opioid crisis into two problems: prescription opioids and heroin.
In 2016, we encountered the worst scourge yet. It is fentanyl, an opioid that’s 50 times more potent than heroin. Fentanyl is easily manufactured, and it’s easier than other opioids to hide and transport across the border. That makes this drug very profitable.
What we have seen during the pandemic is the expansion of fentanyl use in the United States. Initially, fentanyl made its way to the Northeast; now it’s everywhere. Initially, it was used to contaminate heroin; now it’s used to contaminate cocaine, methamphetamine, and, most recently, illicit prescription drugs, such as benzodiazepines and stimulants. With fentanyl contaminating all these drugs, we’re also seeing a steep rise in mortality from cocaine and methamphetamine use in African Americans, American Indians, and Alaska natives.
Tabak: What about teens? A recent study in the journal JAMA reported for the first time in a decade that overdose deaths among U.S. teens rose dramatically in 2020 and kept rising through 2021 [2]. Is fentanyl behind this alarming increase?
Volkow: Yes, and it has us very concerned. The increase also surprised us. Over the past decade, we have seen a consistent decrease in adolescent drug use. In fact, there are some drugs that have the lowest usage rates that we’ve ever recorded. To observe this more than doubling of overdose deaths from fentanyl before the COVID pandemic was a major surprise.
Adolescents don’t typically use heroin, nor do they seek out fentanyl. Our fear is adolescents are misusing illicit prescriptions contaminated with fentanyl. Because an estimated 30-40 percent of those tainted pills contain levels of fentanyl that can kill you, it becomes a game of Russian roulette. This dangerous game is being played by adolescents who may just be experimenting with illicit pills.
Tabak: For people with substance use disorders, there are new ways to get help. In fact, one of the very few positive outcomes of the pandemic is the emergence of telehealth. If we can learn to navigate the various regulatory issues, do you see a place for telehealth going forward?
Volkow: When you have a crisis like this one, there’s a real need to accelerate interventions and innovation like telehealth. It certainly existed before the pandemic, and we knew that telehealth was beneficial for the treatment of substance use disorders. But it was very difficult to get reimbursement, making access extremely limited.
When COVID overwhelmed emergency departments, people with substance use disorders could no longer get help there. Other interventions were needed, and telehealth helped fill the void. It also had the advantage of reaching rural populations in states such as Kentucky, West Virginia, Ohio, where easy access to treatment or unique interventions can be challenging. In many prisons and jails, administrators worried about bringing web-based technologies into their facilities. So, in partnership with the Justice Department, we have created networks that now will enable the entry of telehealth into jails and prisons.
Tabak: Rebecca, it’s been four years since the HEAL initiative was announced at this very summit in 2018. How is the initiative addressing this ever-evolving crisis?
Baker: We’ve launched over 600 research projects across the country at institutions, hospitals, and research centers in a broad range of scientific areas. We’re working to come up with new treatment options for pain and addiction. There’s exciting research underway to address the craving and sleep disruption caused by opioid withdrawal. This research has led to over 20 investigational new drug applications to the FDA. Some are for repurposed drugs, compounds that have already been shown to be safe and effective for treating other health conditions that may also have value for treating addiction. Some are completely novel. We have also initiated the first testing of an opioid vaccine, for oxycodone, to prevent relapse and overdose in high-risk individuals.
Tabak: What about clinical research?
Baker: We’re testing multiple different treatments for both pain and addiction. Not everyone with pain is the same, and not every treatment is going to work the same for everyone. We’re conducting clinical trials in real-world settings to find out what works best for patients. We’re also working to implement lifesaving, evidence-based interventions into places where people seek help, including faith, community, and criminal justice settings.
Tabak: The pandemic highlighted inequities in our health-care system. These inequities afflict individuals and populations who are struggling with addiction and overdose. Nora, what needs to be done to address the social determinants of racial disparities?
Volkow: This is an extraordinarily important question. As you noted, certain racial and ethnic groups had disproportionately higher mortality rates from COVID. We have seen the same with overdose deaths. For example, we know that the most important intervention for preventing overdoses is to initiate medications such as methadone, buprenorphine or vivitrol. But Black Americans are initiated on these medications at least five years later than white Americans. Similarly, Black Americans also are less likely to receive the overdose-reversal medication naloxone.
That’s not right. We must ask what are the core causes of limited access to high-quality health care? Low income is a major contributing factor. Helping people get an education is one of the most important factors to address it. Another factor is distrust of the medical system. When racial and ethnic discrimination is compounded by discrimination because a person has a substance use disorder, you can see why it becomes very difficult for some to seek help. As a society, we certainly need to address racial discrimination. But we also need to address discrimination against substance use disorders in people of all races who are vulnerable.
Baker: Our research is tackling these barriers head on with a direct focus on stigma. As Nora alluded to, oftentimes providers may not offer lifesaving medication to some patients, and we’ve developed and are testing research training to help providers recognize and address their own biases and behaviors in caring for different populations.
We have supported research on the drivers of equity. A big part of this is engaging with people with lived experience and making sure that the interventions being designed are feasible in the real world. Not everyone has access to health insurance, transportation, childcare—the support that they may need to sustain treatment and recovery. In short, our research is seeking ways to enhance linkage to treatment.
Nora mentioned the importance of telehealth in improving equity. That’s another research focus, as well as developing tailored, culturally appropriate interventions for addressing pain and addiction. When you have this trust issue, you can’t always go in with a prescription or a recommendation from a physician. So in American and Alaskan native communities, we’re integrating evidence-based prevention approaches with traditional practices like wellness gatherings, cooking together, use of sage and spirituality, along with community support, and seeing if that encourages and increases the uptake of these prevention approaches in communities that need it so much.
Tabak: The most heartbreaking impact of the opioid crisis has been the infants born dependent on opioids. Rebecca, what’s being done to help the very youngest victims of the opioid crisis born with neonatal opioid withdrawal syndrome, or NOWS?
Baker: Thanks for asking about the infants. Babies with NOWS undergo withdrawal at birth and cry inconsolably, often with extreme stomach upset and sometimes even with seizures. Our research found that hospitals across the country vary greatly in how they treat these babies. Our program, ACT NOW, or Advancing Clinical Trials in Neonatal Opioid Withdrawal, aims to provide concrete guidance for nurses in the NICU treating these infants. One of the studies that we call Eat, Sleep, Console focuses on the abilities of the baby. Our researchers are testing if the ability to eat, sleep, or be consoled increases bonding with the mother and if it reduces time in the hospital, as well as other long-term health outcomes.
In addition to that NOWS program, we’ve also launched the HEALthy Brain and Child Development Study, or HBCD, that seeks to understand the long-term consequences of opioid exposure together with all the other environmental and other factors the baby experiences as they grow up. The hope is that together these studies will inform future prevention and treatment efforts for both mental health and also substance use and addiction.
Tabak: As the surge in heroin use and appearance of fentanyl has taught us, the opioid crisis has ever-changing dynamics. It tells us that we need better prevention strategies. Rebecca, could you share what HEAL is doing about prevention?
Baker: Prevention has always been a core component of the HEAL Initiative in a number of ways. The first is by preventing unnecessary opioid exposures through enhanced and evidence-based pain management. HEAL is supporting research on new small molecules, new devices, new biologic therapeutics that could treat pain and distinct pain conditions without opioids. And we’re also researching and providing guidance for clinicians on strategies for managing pain without medication, including acupuncture and physical therapy. They can often be just as effective and more sustainable.
HEAL is also working to address risky opioid use outside of pain management, especially in high-risk groups. That includes teens and young adults who may be experimenting, people lacking stable housing, patients who are on high-dose opioids for pain management, or they maybe have gone off high-dose opioids but still have them in their possession.
Finally, to prevent overdose we have to give naloxone to the people who need it. The HEALing Communities Study has taken some really innovative approaches to providing naloxone in libraries, on the beach, and places where overdoses are actually happening, not just in medical settings. And I think that will be, in our fight against the overdose crisis, a key tool.
Volkow: Larry, I’d like to add a few words on prevention. There are evidence-based interventions that have been shown to be quite effective for preventing substance use among teenagers and young adults. And yet, they are not implemented. We have evidence-based interventions that work for prevention. We have evidence-based interventions that work for treatment. But we don’t provide the resources for their implementation, nor do we train the personnel that can carry it over.
Science can give us tools, but if we do not partner at the next level for their implementation, those tools do not have the impact they should have. That’s why I always bring up the importance of policy in the implementation phase.
Tabak: Rebecca, the opioid crisis got started with a lack of good options for treating pain. Could you share with us how HEAL’s research efforts are addressing the needs of millions of Americans who experience both chronic pain and opioid use disorder?
Baker: It’s so important to remember people with pain. We can’t let our efforts to combat the opioid crisis make us lose sight of the needs of the millions of Americans with pain. One hundred million Americans experience pain; half of them have severe pain, daily pain, and 20 million have such severe pain that they can’t do things that are important to them in their life, family, job, other activities that bring their life meaning.
HEAL recognizes that these individuals need better options. New non-addictive pain treatments. But as you say, there is a special need for people with a substance use disorder who also have pain. They desperately need new and better options. And so we recently, through the HEAL Initiative, launched a new trials network that couples medication-based treatment for opioid use disorders, so that’s methadone or buprenorphine, with new pain-management strategies such as psychotherapy or yoga in the opioid use disorder treatment setting so that you’re not sending them around to lots of different places. And our hope is that this integrated approach will address some of the fragmented healthcare challenges that often results in poor care for these patients.
My last point would be that some patients need opioids to function. We can’t forget as we make sure that we are limiting risky opioid use that we don’t take away necessary opioids for these patients, and so our future research will incorporate ways of making sure that they receive needed treatment while also preventing them from the risks of opioid use disorder.
Tabak: Rebecca, let me ask you one more question. What do you want the folks here to remember about HEAL?
Baker: HEAL stands for Helping to End Addiction Long-term, and nobody knows more than the people in this room how challenging and important that really is. We’ve heard a little bit about the great promise of our research and some of the advances that are coming through our research pipeline, new treatments, new guidance for clinicians and caregivers. I want everyone to know that we want to work with you. By working together, I’m confident that we will tailor these new advances to meet the individual needs of the patients and populations that we serve.
Tabak: Nora, what would you like to add?
Volkow: This afternoon, I met with two parents who told me the story of how they lost their daughter to an overdose. They showed me pictures of this fantastic girl, along with her drawings. Whenever we think about overdose deaths in America, the sheer number—75,000—can make us indifferent. But when you can focus on one person and feel the love surrounding that life, you remember the value of this work.
Like in COVID, substance use disorders are a painful problem that we’re all experiencing in some way. They may have upset our lives. But they may have brought us together and, in many instances, brought out the best that humans can do. The best, to me, is caring for one another and taking the responsibility of helping those that are most vulnerable. I believe that science has a purpose. And here we have a purpose: to use science to bring solutions that can prevent and treat those suffering from substance use disorders.
Tabak: Thanks to both of you for this enlightening conversation.
References:
Links:
Video: Evening Plenary with NIH’s Lawrence Tabak, Nora Volkow, and Rebecca Baker (Rx and Illicit Drug Summit 2022)
SAMHSA’s National Helpline (Substance Abuse and Mental Health Services Administration, Rockville, MD)
Opioids (National Institute on Drug Abuse/NIH)
Fentanyl (NIDA)
Helping to End Addiction Long-term®(HEAL) Initiative (NIH)
Rebecca Baker (HEAL/NIH)
Nora Volkow (NIDA)
Posted In: Generic
Tags: addiction, African American health, benzodiazepine, buprenorphine, cocaine, COVID-19, drugs, fentanyl, HEAL, HEALing Communities Study, health disparities, Helping to End Addiction Long-term, heroin, jails, methadone, methamphetamine, naloxone, neonatal opioid withdrawal syndrome, NIDA, Nora Volkow, NOWS, opioid crisis, opioid medications, opioid overdoses, opioid use disorder, opioids, oxycodone, pain, pain management, pandemic, prevention, prisons, Rebecca Baker, Rx and Illicit Drug Summit, RxDrug Abuse and Heroin Summit, stimulants, substance use disorders, teens, telehealth
All of Us: Release of Nearly 100,000 Whole Genome Sequences Sets Stage for New Discoveries
Posted on March 29th, 2022 by Joshua Denny, M.D., M.S., and Lawrence Tabak, D.D.S., Ph.D.

Nearly four years ago, NIH opened national enrollment for the All of Us Research Program. This historic program is building a vital research community within the United States of at least 1 million participant partners from all backgrounds. Its unifying goal is to advance precision medicine, an emerging form of health care tailored specifically to the individual, not the average patient as is now often the case. As part of this historic effort, many participants have offered DNA samples for whole genome sequencing, which provides information about almost all of an individual’s genetic makeup.
Earlier this month, the All of Us Research Program hit an important milestone. We released the first set of nearly 100,000 whole genome sequences from our participant partners. The sequences are stored in the All of Us Researcher Workbench, a powerful, cloud-based analytics platform that makes these data broadly accessible to registered researchers.
The All of Us Research Program and its many participant partners are leading the way toward more equitable representation in medical research. About half of this new genomic information comes from people who self-identify with a racial or ethnic minority group. That’s extremely important because, until now, over 90 percent of participants in large genomic studies were of European descent. This lack of diversity has had huge impacts—deepening health disparities and hindering scientific discovery from fully benefiting everyone.
The Researcher Workbench also contains information from many of the participants’ electronic health records, Fitbit devices, and survey responses. Another neat feature is that the platform links to data from the U.S. Census Bureau’s American Community Survey to provide more details about the communities where participants live.
This unique and comprehensive combination of data will be key in transforming our understanding of health and disease. For example, given the vast amount of data and diversity in the Researcher Workbench, new diseases are undoubtedly waiting to be uncovered and defined. Many new genetic variants are also waiting to be identified that may better predict disease risk and response to treatment.
To speed up the discovery process, these data are being made available, both widely and wisely. To protect participants’ privacy, the program has removed all direct identifiers from the data and upholds strict requirements for researchers seeking access. Already, more than 1,500 scientists across the United States have gained access to the Researcher Workbench through their institutions after completing training and agreeing to the program’s strict rules for responsible use. Some of these researchers are already making discoveries that promote precision medicine, such as finding ways to predict how to best to prevent vision loss in patients with glaucoma.
Beyond making genomic data available for research, All of Us participants have the opportunity to receive their personal DNA results, at no cost to them. So far, the program has offered genetic ancestry and trait results to more than 100,000 participants. Plans are underway to begin sharing health-related DNA results on hereditary disease risk and medication-gene interactions later this year.
This first release of genomic data is a huge milestone for the program and for health research more broadly, but it’s also just the start. The program’s genome centers continue to generate the genomic data and process about 5,000 additional participant DNA samples every week.
The ultimate goal is to gather health data from at least 1 million or more people living in the United States, and there’s plenty of time to join the effort. Whether you would like to contribute your own DNA and health information, engage in research, or support the All of Us Research Program as a partner, it’s easy to get involved. By taking part in this historic program, you can help to build a better and more equitable future for health research and precision medicine.
Note: Joshua Denny, M.D., M.S., is the Chief Executive Officer of NIH’s All of Us Research Program.
Links:
All of Us Research Program (NIH)
Join All of Us (NIH)
Posted In: News
Tags: All of Us, All of Us Research Program, All of Us Researcher Workbench, big data, cohort, data protection, diversity, DNA, DNA sequencing, EHR, electronic health records, ethnicity, Fitbit, genome, genomics, glaucoma, health disparities, precision medicine, race, research privacy, U.S. Census Bureau, whole genome sequencing