Diabetes mellitus and risk of endometrial cancer: a meta-analysis (original) (raw)
Abstract
Aims/hypothesis
Diabetes has been associated with a statistically significantly increased risk of endometrial cancer in most, but not all studies. To provide a quantitative assessment of the association between diabetes and risk of endometrial cancer, we conducted a meta-analysis of case-control studies and cohort studies.
Subjects and methods
We identified studies by a literature search of PubMed and Embase through to January 2007 and by searching the reference lists of relevant articles. Summary relative risks (RRs) with 95% CIs were calculated using random-effects model.
Results
The analysis of diabetes (largely type 2) and endometrial cancer is based on 16 studies (three cohort and 13 case-control studies), including 96,003 participants and 7,596 cases of endometrial cancer. Twelve of the studies showed a statistically significantly increased risk and four a non-significant increased risk of endometrial cancer. In our meta-analysis we found that diabetes was statistically significantly associated with an increased risk of endometrial cancer (summary RR 2.10, 95% CI 1.75–2.53). The risk estimates were somewhat stronger among case-control (RR 2.22, 95% CI 1.80–2.74) than among cohort studies (RR 1.62, 95% CI 1.21–2.16), stronger among studies adjusting only for age (RR 2.74, 95% CI 1.87–4.00) compared with multivariate adjustment (RR 1.92, 95% CI 1.58–2.33) and slightly lower in studies performed in the USA than in those performed Europe. The analysis of type 1 diabetes and endometrial cancer was based on three studies and found a statistically significant positive association (summary RR 3.15, 95%CI 1.07–9.29).
Conclusions/interpretation
Results from the meta-analysis support a relationship between diabetes and increased risk of endometrial cancer.
Similar content being viewed by others
Introduction
The incidence of type 2 diabetes mellitus and endometrial cancer is increasing in westernised countries [[1](/article/10.1007/s00125-007-0681-5#ref-CR1 "WHO website: http://www.who.int/diabetes/facts/en/
, last accessed 4 May 2006")–[3](/article/10.1007/s00125-007-0681-5#ref-CR3 "Bray F, Dos Santos Silva I, Moller H, Weiderpass E (2005) Endometrial cancer incidence trends in Europe: underlying determinants and prospects for prevention. Cancer Epidemiol Biomarkers Prev 14:1132–1142")\]. The major modifiable determinants of type 2 diabetes, hyperinsulinaemia and insulin resistance are obesity \[[4](/article/10.1007/s00125-007-0681-5#ref-CR4 "Kissebah AH, Vydelingum N, Murray R et al (1982) Relation of body fat distribution to metabolic complications of obesity. J Clin Endocrinol Metab 54:254–260"), [5](/article/10.1007/s00125-007-0681-5#ref-CR5 "Bjorntorp P (1991) Metabolic implications of body fat distribution. Diabetes Care 14:1132–1143")\] and low physical activity \[[6](/article/10.1007/s00125-007-0681-5#ref-CR6 "International Agency for Research on Cancer (2002) Weight control and physical activity. IARC, Lyon")\], both of which have also been shown to be risk factors for endometrial cancer \[[6](/article/10.1007/s00125-007-0681-5#ref-CR6 "International Agency for Research on Cancer (2002) Weight control and physical activity. IARC, Lyon")–[8](/article/10.1007/s00125-007-0681-5#ref-CR8 "Modesitt SC, van Nagell JR Jr (2005) The impact of obesity on the incidence and treatment of gynecologic cancers: a review. Obstet Gynecol Surv 60:683–692")\]. A number of studies have reported a positive association between diabetes and incidence of \[[9](/article/10.1007/s00125-007-0681-5#ref-CR9 "Friberg E, Mantzoros CS, Wolk A (2007) Diabetes and risk of endometrial cancer: a population-based prospective cohort study. Cancer Epidemiol Biomarkers Prev 16:276–280")–[20](/article/10.1007/s00125-007-0681-5#ref-CR20 "O’Mara BA, Byers T, Schoenfeld E (1985) Diabetes mellitus and cancer risk: a multisite case-control study. J Chronic Dis 38:435–441")\] or mortality from \[[21](/article/10.1007/s00125-007-0681-5#ref-CR21 "Folsom AR, Anderson KE, Sweeney C, Jacobs DR Jr (2004) Diabetes as a risk factor for death following endometrial cancer. Gynecol Oncol 94:740–745")\] endometrial cancer. Many studies did not distinguish between type 1 and type 2 diabetes. However, on the basis of the relative prevalence of these two types, the vast majority of cases are likely to be type 2 diabetes. Although epidemiological studies of the relationship of diabetes with risk of endometrial cancer are not entirely consistent \[[22](/article/10.1007/s00125-007-0681-5#ref-CR22 "Rubin GL, Peterson HB, Lee NC, Maes EF, Wingo PA, Becker S (1990) Estrogen replacement therapy and the risk of endometrial cancer: remaining controversies. Am J Obstet Gynecol 162:148–154")–[26](/article/10.1007/s00125-007-0681-5#ref-CR26 "Coughlin SS, Calle EE, Teras LR, Petrelli J, Thun MJ (2004) Diabetes mellitus as a predictor of cancer mortality in a large cohort of US adults. Am J Epidemiol 159:1160–1167")\], most the studies are compatible with a positive association. However, because of the low precision of estimates in studies with a small sample size \[[12](/article/10.1007/s00125-007-0681-5#ref-CR12 "Salazar-Martinez E, Lazcano-Ponce EC, Lira-Lira GG et al (2000) Case-control study of diabetes, obesity, physical activity and risk of endometrial cancer among Mexican women. Cancer Causes Control 11:707–711"), [16](/article/10.1007/s00125-007-0681-5#ref-CR16 "Inoue M, Okayama A, Fujita M, Enomoto T, Tanizawa O, Ueshima H (1994) A case-control study on risk factors for uterine endometrial cancer in Japan. Jpn J Cancer Res 85:346–350"), [25](/article/10.1007/s00125-007-0681-5#ref-CR25 "Terry P, Baron JA, Weiderpass E, Yuen J, Lichtenstein P, Nyren O (1999) Lifestyle and endometrial cancer risk: a cohort study from the Swedish Twin Registry. Int J Cancer 82:38–42")\], reported risk estimates vary from 1.3 \[[23](/article/10.1007/s00125-007-0681-5#ref-CR23 "Kelsey JL, LiVolsi VA, Holford TR et al (1982) A case-control study of cancer of the endometrium. Am J Epidemiol 116:333–342")\] to 7.8 \[[16](/article/10.1007/s00125-007-0681-5#ref-CR16 "Inoue M, Okayama A, Fujita M, Enomoto T, Tanizawa O, Ueshima H (1994) A case-control study on risk factors for uterine endometrial cancer in Japan. Jpn J Cancer Res 85:346–350")\].We performed a meta-analysis to quantitatively summarise results from published cohort and case-control studies and to provide a more precise risk estimate for the association between diabetes and endometrial cancer.
Subjects and methods
Search strategy
We identified studies by a literature search of the PubMed and Embase databases (from January 1966 to January 2007) with the following medical subject heading terms and/or text words: ‘diabetes mellitus’, ‘diabetes’, ‘endometrial cancer’, and ‘corpus uteri’. We also reviewed reference lists of the identified publications for additional pertinent studies. No language restrictions were imposed.
Inclusion and exclusion criteria
The 33 studies considered for inclusion in this meta-analysis consisted of 13 cohort and 20 case-control studies on the association between diabetes and endometrial cancer incidence or mortality [9–41]. Studies were excluded if they provided only an effect estimate with no means to calculate a CI or if the estimates were not adjusted for age. We excluded two studies [28, 30] with unavailable effect estimates and three studies [29, 32, 33] that reported only crude data not adjusted for age. In the event of multiple publications from the same population or cohort, we included only data from the most recent report or, if the reports included exactly the same time frame, the publication with the most control for confounders. We excluded three candidate studies [27, 31, 41] because of overlapping publications.
We included 25 independent studies in this meta-analysis and excluded eight candidate studies.
Data extraction
The data that we extracted included publication data (the first authors’ last names, year of publication and country of population studied), study design, number of exposed and unexposed subjects, follow-up period (for cohort studies), control source (in case-control studies), type of diabetes (type 1 or 2, when available), risk estimates with their corresponding CIs and variables controlled for by matching or in the multivariable model. From each study, we extracted the risk estimates that reflected the greatest degree of control for potential confounders.
Statistical analysis
We divided epidemiological studies of the relationship between diabetes and endometrial cancer risk into three general types according to design: cohort studies (incidence and/or mortality rate ratio), case-control studies (odds ratio) and cohort studies with an external comparison group (standardised incidence and/or mortality ratio). We conducted separate meta-analyses of endometrial cancer incidence and mortality. The measure of effect of interest is the relative risk (RR). Because endometrial cancer is rare, the odds ratio in case-control studies and rate ratios in cohort studies yield similar estimates of RR [42]. Cohort studies that reported standardised incidence: mortality ratio were analysed separately. Studies reporting an estimate for type 1 diabetes were analysed separately.
Summary RR estimates with their corresponding 95% CIs were derived with the method of DerSimonian and Laird [43] using the assumption of a random effects model that incorporated between-study variability. We calculated a pooled RR and its corresponding 95% CI. Statistical heterogeneity between studies was evaluated with Cochran’s Q test and the I 2 statistic [44]. Publication bias was assessed by constructing a funnel plot [45] and by Egger’s regression asymmetry test [46].
For cohort studies that reported incidence rate ratios and for case-control studies, we conducted subgroup meta-analyses to examine potential sources of heterogeneity, including study design, as well as type of control subjects in case-control studies. Statistical analyses were carried out with Stata, version 9.0 (Stata Corp., College Station, TX, USA). We considered p values that were less than 0.05 statistically significant. All statistical tests were two-sided.
Results
Study characteristics
There were 25 independent studies that met the predefined inclusion criteria. Of these 25 studies, five were cohort studies that used incidence and/or mortality rate ratios as the measure of RR [9, 21, 24–26] (Table 1), 13 were case-control studies [10–20, 22, 23] (Table 2) and seven were cohort studies that used standardised incidence and/or mortality ratio as the measure of risk [34–40] (Table 3). Twelve studies were conducted in the USA, ten in Europe, one in South America and one in Asia. In the meta-analysis of diabetes (largely type 2) and endometrial cancer, we included three cohort studies that reported incidence rate ratios [9, 24, 25] and 13 case-control studies [10–20, 22, 23]. These 16 studies had 96,003 participants. The four cohort studies [34, 36–38] that reported standardised incidence ratios were analysed separately. For the meta-analysis of diabetes and endometrial cancer mortality, we included the two cohort studies that reported mortality rate ratio [21, 26]. These two studies enrolled a total of 896 participants. The four cohort studies [34, 36–38] that reported standardised incidence ratios and the three cohort studies [34, 39, 40] that reported standardised mortality ratios were analysed separately.
Table 1 Characteristics of cohort studies of diabetes and endometrial cancer incidence and mortality
Table 2 Characteristics of case-control studies of diabetes and endometrial cancer
Table 3 Characteristics of cohort studies of diabetes and endometrial cancer based on standardised incidence/mortality ratio
Four studies reported on type 1 diabetes and endometrial cancer, one case-control study [11], two studies providing standardised incidence ratio [34, 35] and one study reporting on type 1 diabetes and endometrial cancer mortality providing a standardised mortality ratio [34].
Endometrial cancer
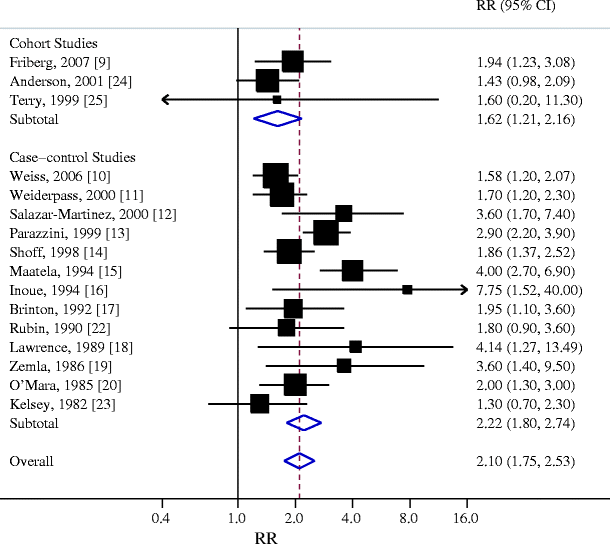
Individual study results and the overall summary result for three cohort and 13 case-control studies of diabetes and endometrial cancer are shown in Fig. 1. Twelve of these 16 studies found a statistically significant positive association between diabetes and endometrial cancer (range of individual RRs 1.3–7.75; summary RR for all 16 studies 2.10, 95% CI 1.75–2.53). The heterogeneity among studies was p = 0.01. In a sensitivity analysis in which one study at a time was excluded and the rest were analysed, we detected a statistically significant positive association between diabetes and endometrial cancer (range of summary RRs 1.98–2.18 with the lower limit of the 95% CI never crossing 1.0).
Fig. 1
Association between diabetes (largely type 2) and endometrial cancer incidence in cohort and case-control studies. Studies are ordered by publication year and stratified on design. Squares, study-specific RR estimate (size of the square reflects the study-specific statistical weight, i.e. the inverse variance); horizontal lines, 95% CI; diamond, summary RR estimate and its corresponding 95% CI. All statistical tests were two-sided. Statistical heterogeneity between studies was assessed with Cochran’s Q test. Test for heterogeneity among cohort studies: Q = 1.01; p = 0.60; I 2 = 0.0%. Test for heterogeneity among case-control studies: Q = 28.34; p = 0.01; I 2 = 57.7%. Test for heterogeneity between cohort and case-control studies: Q = 2.70; p = 0.10
We then conducted subgroup meta-analyses by study design, geographical area, control group (for case-control studies) and adjustments (including all the measured confounders vs adjusted for age only) (Table 4). The association between diabetes and endometrial cancer was somewhat stronger in Europe than in USA and also somewhat stronger among case-control studies. When adjustments are taken into account, the studies adjusting only for age reported a stronger association than those adjusting the RR with a model including all the measured confounders, indicating a presence of confounding.
Table 4 Summary relative risk estimates and 95% CIs for case-control and cohort studies of the association between diabetes and endometrial cancer by study design, geographical area and adjustments
Physical inactivity and body mass index are potentially the most important known confounders of the positive association between diabetes and endometrial cancer risk. When we restricted the meta-analysis to the two studies that controlled for these variables [9, 12], we found a positive association between diabetes and endometrial cancer (summary RR 2.47, 95% CI 1.37–4.45, test for heterogeneity Q = 1.95, p = 0.16, I 2 = 48.8%). A positive association was observed between diabetes and endometrial cancer incidence in the cohort studies that reported standardised incidence ratios [34, 36–38] (Table 3) (summary RR 1.63, 95% CI 1.30–2.05, test for heterogeneity Q = 9.60, p = 0.02, I 2 = 68.8%).
Endometrial cancer mortality
Of the two cohort studies of diabetes and mortality from endometrial cancer [21, 26], one [21] reported a statistically significant positive association and one[26] observed a non-statistically significant positive association. When the studies were pooled, a positive, but non-significant association between diabetes and mortality from endometrial cancer was found (summary RR 1.58, 95% CI 0.94–2.66, test for heterogeneity Q = 1.63, p = 0.20, I 2 = 38.7%). No association was observed between diabetes and endometrial cancer mortality in the cohort studies that reported standardised mortality ratios [34, 39, 40] (summary RR 0.97, 95% CI 0.52–1.81, test for heterogeneity Q = 4.76, p = 0.09, I 2 = 58.0%).
Type 1 diabetes
Two cohorts providing standardised incidence ratios and one case-control study reported on the association between type 1 diabetes and endometrial cancer [11, 34, 35]. When the three studies were meta-analysed, a statistically significant positive association between type 1 diabetes and endometrial cancer was found (summary RR 3.15, 95%CI 1.07–9.29, test for heterogeneity Q = 6.66, p = 0.04, I 2 = 70.0%).
Publication bias
The funnel plot we constructed revealed no evidence for publication bias concerning diabetes and risk of endometrial cancer (data not shown). The p value for Egger’s regression asymmetry test was 0.14, i.e. probability of publication bias was low.
Discussion
Findings from this meta-analysis show that women with diabetes (largely type 2 diabetes) may have an approximately twofold increased risk of developing endometrial cancer compared with non-diabetic individuals. Our analyses on type 1 diabetes suggest that women with type 1 diabetes may have a threefold increased risk of endometrial cancer. However, our analysis does not support the notion that diabetes is associated with increased risk of endometrial cancer mortality.
All previous studies have consistently shown a positive association between diabetes and endometrial cancer incidence. However, heterogeneity exists due to differences in reported strength of the effect estimate. The summary RR was consistent, but slightly higher for case-control than cohort studies, as well as among European studies vs those done in the USA. The latter could reflect a residual confounding from BMI. The studies only adjusting for age also showed a slightly stronger effect estimate, probably due to lack of controlling for BMI. This finding was also consistent in cohort studies reporting a standardised incidence ratio.
Both our findings on endometrial cancer mortality in relation to diabetes (largely type 2), and our data on the risk of endometrial cancer in type 1 diabetes are more limited by uncertainty due to the smaller number of studies and to studies including small numbers of cases.
Our analysis must be interpreted in the context of the limitations of available data. Many studies did not distinguish between type 1 and type 2 diabetes, but given the relative prevalence of these two types, it is likely that the vast majority of cases are type 2 diabetes. Since diabetes is an underdiagnosed disease, some degree of misclassification of exposure to diabetes is probable, but such non-differential misclassification would be expected to have attenuated the true relationship between diabetes and endometrial cancer. Moreover, some subgroup analyses were based on few studies and the results need to be interpreted with caution. As in all meta-analysis, the possibility of publication bias is of concern; however, a formal statistical test did not provide evidence for such bias. Type 2 diabetes and endometrial cancer share some risk factors such as obesity and low physical activity. Thus, the observed increased risk of endometrial cancer associated with diabetes may in part reflect confounding by these factors. Nevertheless, a statistically significant positive association remained when the analysis was limited to the two studies controlling for obesity and physical activity. That said, further residual confounding cannot be ruled out.
Several biological mechanisms have been proposed to potentially underlie the development of endometrial cancer in diabetic women. Hyperinsulinaemia is a common feature of diabetes, obesity and physical inactivity, and insulin has been shown to stimulate the growth of endometrial stromal cells by binding to insulin receptors on endometrial cells [47]. Epidemiological studies have observed an elevated risk of endometrial cancer in relation to high prediagnostic C-peptide concentrations indicating hyperinsulinaemia [48]. Long-term insulin therapy of patients with type 1 diabetes may explain the increased risk of endometrial cancer among diabetic women with type 1 diabetes [35]. Hyperinsulinaemia may also increase levels of bioactive oestrogens by decreasing concentrations of circulating sex hormone binding globulin [49, 50]. Oestrogens have been shown to increase endometrial cancer risk by stimulating proliferation of endometrial cells [51], when unopposed by progesterone [52, 53]. Finally, hyperinsulinaemia leads to decreased levels of insulin-like growth factor binding protein 1. This in turn increases circulating free IGF-1, which stimulates endometrial cell proliferation [54–59]. Finally, the adipocyte-secreted hormone, adiponectin, is an endogenous insulin sensitiser lying upstream of all the above-mentioned hormonal factors and capable of affecting their circulatory levels [60]. Low levels of adiponectin predict not only diabetes but also endometrial cancer incidence [61] and can thus serve as the link between diabetes, hormonal abnormalities and endometrial cancer risk.
Our results have important clinical and public health implications, given the already high and continuously increasing prevalence of type 2 diabetes and endometrial cancer in western societies.
In summary, results from this meta-analysis support an association between diabetes and endometrial cancer. Future studies will need to clearly distinguish between type 1 and type 2 diabetes, as well as different types of treatment for diabetes. It would also be of interest to distinguish between specifically defined subtypes of endometrial cancer.
Abbreviations
References
- WHO website: http://www.who.int/diabetes/facts/en/, last accessed 4 May 2006
- Polednak AP (2003) Trends in incidence rates for obesity-associated cancers in the US. Cancer Detect Prev 27:415–421
Article PubMed Google Scholar - Bray F, Dos Santos Silva I, Moller H, Weiderpass E (2005) Endometrial cancer incidence trends in Europe: underlying determinants and prospects for prevention. Cancer Epidemiol Biomarkers Prev 14:1132–1142
Article PubMed Google Scholar - Kissebah AH, Vydelingum N, Murray R et al (1982) Relation of body fat distribution to metabolic complications of obesity. J Clin Endocrinol Metab 54:254–260
CAS PubMed Google Scholar - Bjorntorp P (1991) Metabolic implications of body fat distribution. Diabetes Care 14:1132–1143
Article CAS PubMed Google Scholar - International Agency for Research on Cancer (2002) Weight control and physical activity. IARC, Lyon
Google Scholar - Schouten LJ, Goldbohm RA, van den Brandt PA (2004) Anthropometry, physical activity, and endometrial cancer risk: results from the Netherlands cohort study. J Natl Cancer Inst 96:1635–1638
Article PubMed Google Scholar - Modesitt SC, van Nagell JR Jr (2005) The impact of obesity on the incidence and treatment of gynecologic cancers: a review. Obstet Gynecol Surv 60:683–692
Article PubMed Google Scholar - Friberg E, Mantzoros CS, Wolk A (2007) Diabetes and risk of endometrial cancer: a population-based prospective cohort study. Cancer Epidemiol Biomarkers Prev 16:276–280
Article PubMed Google Scholar - Weiss JM, Saltzman BS, Doherty JA et al (2006) Risk factors for the incidence of endometrial cancer according to the aggressiveness of disease. Am J Epidemiol 164:56–62
Article PubMed Google Scholar - Weiderpass E, Persson I, Adami HO, Magnusson C, Lindgren A, Baron JA (2000) Body size in different periods of life, diabetes mellitus, hypertension, and risk of postmenopausal endometrial cancer (Sweden). Cancer Causes Control 11:185–192
Article CAS PubMed Google Scholar - Salazar-Martinez E, Lazcano-Ponce EC, Lira-Lira GG et al (2000) Case-control study of diabetes, obesity, physical activity and risk of endometrial cancer among Mexican women. Cancer Causes Control 11:707–711
Article CAS PubMed Google Scholar - Parazzini F, La Vecchia C, Negri E et al (1999) Diabetes and endometrial cancer: an Italian case-control study. Int J Cancer 81:539–542
Article CAS PubMed Google Scholar - Shoff SM, Newcomb PA (1998) Diabetes, body size, and risk of endometrial cancer. Am J Epidemiol 148:234–240
CAS PubMed Google Scholar - Maatela J, Aromaa A, Salmi T, Pohja M, Vuento M, Gronroos M (1994) The risk of endometrial cancer in diabetic and hypertensive patients: a nationwide record-linkage study in Finland. Ann Chir Gynaecol Suppl 208:20–24
CAS PubMed Google Scholar - Inoue M, Okayama A, Fujita M, Enomoto T, Tanizawa O, Ueshima H (1994) A case-control study on risk factors for uterine endometrial cancer in Japan. Jpn J Cancer Res 85:346–350
CAS PubMed Google Scholar - Brinton LA, Berman ML, Mortel R et al (1992) Reproductive, menstrual, and medical risk factors for endometrial cancer: results from a case-control study. Am J Obstet Gynecol 167:1317–1325
CAS PubMed Google Scholar - Lawrence C, Tessaro I, Durgerian S, Caputo T, Richart RM, Greenwald P (1989) Advanced-stage endometrial cancer: contributions of estrogen use, smoking, and other risk factors. Gynecol Oncol 32:41–45
Article CAS PubMed Google Scholar - Zemla B, Guminski S, Banasik R (1986) Study of risk factors in invasive cancer of the corpus uteri. Neoplasma 33:621–629
CAS PubMed Google Scholar - O’Mara BA, Byers T, Schoenfeld E (1985) Diabetes mellitus and cancer risk: a multisite case-control study. J Chronic Dis 38:435–441
Article CAS PubMed Google Scholar - Folsom AR, Anderson KE, Sweeney C, Jacobs DR Jr (2004) Diabetes as a risk factor for death following endometrial cancer. Gynecol Oncol 94:740–745
Article PubMed Google Scholar - Rubin GL, Peterson HB, Lee NC, Maes EF, Wingo PA, Becker S (1990) Estrogen replacement therapy and the risk of endometrial cancer: remaining controversies. Am J Obstet Gynecol 162:148–154
CAS PubMed Google Scholar - Kelsey JL, LiVolsi VA, Holford TR et al (1982) A case-control study of cancer of the endometrium. Am J Epidemiol 116:333–342
CAS PubMed Google Scholar - Anderson KE, Anderson E, Mink PJ et al (2001) Diabetes and endometrial cancer in the Iowa women’s health study. Cancer Epidemiol Biomarkers Prev 10:611–616
CAS PubMed Google Scholar - Terry P, Baron JA, Weiderpass E, Yuen J, Lichtenstein P, Nyren O (1999) Lifestyle and endometrial cancer risk: a cohort study from the Swedish Twin Registry. Int J Cancer 82:38–42
Article CAS PubMed Google Scholar - Coughlin SS, Calle EE, Teras LR, Petrelli J, Thun MJ (2004) Diabetes mellitus as a predictor of cancer mortality in a large cohort of US adults. Am J Epidemiol 159:1160–1167
Article PubMed Google Scholar - Newcomer LM, Newcomb PA, Trentham-Dietz A, Storer BE (2001) Hormonal risk factors for endometrial cancer: modification by cigarette smoking (United States). Cancer Causes Control 12:829–835
Article CAS PubMed Google Scholar - Parslov M, Lidegaard O, Klintorp S et al (2000) Risk factors among young women with endometrial cancer: a Danish case-control study. Am J Obstet Gynecol 182:23–29
Article CAS PubMed Google Scholar - Hagen A, Morack G, Grulich D (1995) Evaluation of epidemiologic risk factors for endometrial carcinoma based on a case-control study. Zentralbl Gynakol 117:368–374 [in German]
CAS PubMed Google Scholar - Feldman S, Cook EF, Harlow BL, Berkowitz RS (1995) Predicting endometrial cancer among older women who present with abnormal vaginal bleeding. Gynecol Oncol 56:376–381
Article CAS PubMed Google Scholar - La Vecchia C, Negri E, Franceschi S, D’Avanzo B, Boyle P (1994) A case-control study of diabetes mellitus and cancer risk. Br J Cancer 70:950–953
PubMed Google Scholar - Geraci P, Mancuso A, Maggio S, Dardanoni G (1988) Risk factors of endometrial cancer in Palermo. Clin Exp Obstet Gynecol 15:129–133
CAS PubMed Google Scholar - Elwood JM, Cole P, Rothman KJ, Kaplan SD (1977) Epidemiology of endometrial cancer. J Natl Cancer Inst 59:1055–1060
CAS PubMed Google Scholar - Swerdlow AJ, Laing SP, Qiao Z et al (2005) Cancer incidence and mortality in patients with insulin-treated diabetes: a UK cohort study. Br J Cancer 92:2070–2075
Article CAS PubMed Google Scholar - Zendehdel K, Nyren O, Ostenson CG, Adami HO, Ekbom A, Ye W (2003) Cancer incidence in patients with type 1 diabetes mellitus: a population-based cohort study in Sweden. J Natl Cancer Inst 95:1797–1800
PubMed Google Scholar - Weiderpass E, Gridley G, Persson I, Nyren O, Ekbom A, Adami HO (1997) Risk of endometrial and breast cancer in patients with diabetes mellitus. Int J Cancer 71:360–363
Article CAS PubMed Google Scholar - Wideroff L, Gridley G, Mellemkjaer L et al (1997) Cancer incidence in a population-based cohort of patients hospitalized with diabetes mellitus in Denmark. J Natl Cancer Inst 89:1360–1365
Article CAS PubMed Google Scholar - Ragozzino M, Melton LJ 3rd, Chu CP, Palumbo PJ (1982) Subsequent cancer risk in the incidence cohort of Rochester, Minnesota, residents with diabetes mellitus. J Chronic Dis 35:13–19
Article CAS PubMed Google Scholar - Verlato G, Zoppini G, Bonora E, Muggeo M (2003) Mortality from site-specific malignancies in type 2 diabetic patients from Verona. Diabetes Care 26:1047–1051
Article PubMed Google Scholar - Kessler II (1970) Cancer mortality among diabetics. J Natl Cancer Inst 44:673–686
CAS PubMed Google Scholar - Adami HO, McLaughlin J, Ekbom A et al (1991) Cancer risk in patients with diabetes mellitus. Cancer Causes Control 2:307–314
Article CAS PubMed Google Scholar - Greenland S (1987) Quantitative methods in the review of epidemiologic literature. Epidemiol Rev 9:1–30
CAS PubMed Google Scholar - DerSimonian R, Laird N (1986) Meta-analysis in clinical trials. Control Clin Trials 7:177–188
Article CAS PubMed Google Scholar - Higgins JP, Thompson SG (2002) Quantifying heterogeneity in a meta-analysis. Stat Med 21:1539–1558
Article PubMed Google Scholar - Egger M, Davey Smith G, Schneider M, Minder C (1997) Bias in meta-analysis detected by a simple, graphical test. BMJ 315:629–634
CAS PubMed Google Scholar - Egger M, Smith GD, Altman DG (2001) Systematic reviews in health care: meta-analysis in context. BMJ, London
Google Scholar - Nagamani M, Stuart CA (1998) Specific binding and growth-promoting activity of insulin in endometrial cancer cells in culture. Am J Obstet Gynecol 179:6–12
Article CAS PubMed Google Scholar - Lukanova A, Zeleniuch-Jacquotte A, Lundin E et al (2004) Prediagnostic levels of C-peptide, IGF-I, IGFBP -1, -2 and -3 and risk of endometrial cancer. Int J Cancer 108:262–268
Article CAS PubMed Google Scholar - Kazer RR (1995) Insulin resistance, insulin-like growth factor I and breast cancer: a hypothesis. Int J Cancer 62:403–406
Article CAS PubMed Google Scholar - Nestler JE, Powers LP, Matt DW et al (1991) A direct effect of hyperinsulinemia on serum sex hormone-binding globulin levels in obese women with the polycystic ovary syndrome. J Clin Endocrinol Metab 72:83–89
Article CAS PubMed Google Scholar - Graham JD, Clarke CL (1997) Physiological action of progesterone in target tissues. Endocr Rev 18:502–519
Article CAS PubMed Google Scholar - Ferenczy A, Bertrand G, Gelfand MM (1979) Proliferation kinetics of human endometrium during the normal menstrual cycle. Am J Obstet Gynecol 133:859–867
CAS PubMed Google Scholar - Key TJ, Pike MC (1988) The dose-effect relationship between ‘unopposed’ oestrogens and endometrial mitotic rate: its central role in explaining and predicting endometrial cancer risk. Br J Cancer 57:205–212
CAS PubMed Google Scholar - Irwin JC, de las Fuentes L, Dsupin BA, Giudice LC (1993) Insulin-like growth factor regulation of human endometrial stromal cell function: coordinate effects on insulin-like growth factor binding protein-1, cell proliferation and prolactin secretion. Regul Pept 48:165–177
Article CAS PubMed Google Scholar - Murphy LJ (1994) Growth factors and steroid hormone action in endometrial cancer. J Steroid Biochem Mol Biol 48:419–423
Article CAS PubMed Google Scholar - Corocleanu M (1993) Hypothesis for endometrial carcinoma carcinogenesis. Preventive prospects. Clin Exp Obstet Gynecol 20:254–258
CAS PubMed Google Scholar - Thiet MP, Osathanondh R, Yeh J (1994) Localization and timing of appearance of insulin, insulin-like growth factor-I, and their receptors in the human fetal mullerian tract. Am J Obstet Gynecol 170:152–156
CAS PubMed Google Scholar - Ordener C, Cypriani B, Vuillermoz C, Adessi GL (1993) Epidermal growth factor and insulin induce the proliferation of guinea pig endometrial stromal cells in serum-free culture, whereas estradiol and progesterone do not. Biol Reprod 49:1032–1044
Article CAS PubMed Google Scholar - Weiderpass E, Brismar K, Bellocco R, Vainio H, Kaaks R (2003) Serum levels of insulin-like growth factor-I, IGF-binding protein 1 and 3, and insulin and endometrial cancer risk. Br J Cancer 89:1697–1704
Article CAS PubMed Google Scholar - Steffes MW, Gross MD, Schreiner PJ et al (2004) Serum adiponectin in young adults-interactions with central adiposity, circulating levels of glucose, and insulin resistance: the CARDIA study. Ann Epidemiol 14:492–498
Article PubMed Google Scholar - Dal Maso L, Augustin LS, Karalis A et al (2004) Circulating adiponectin and endometrial cancer risk. J Clin Endocrinol Metab 89:1160–1163
Article CAS Google Scholar
Acknowledgements
This work was supported by research grants from World Cancer Research Fund International, The Swedish Cancer Foundation, The Swedish Research Council/Longitudinal studies, DK58785 and an award of the American Diabetes Association.
Duality of interest
The authors state that there are no dualities of interest to declare.
Author information
Authors and Affiliations
- Division of Nutritional Epidemiology, The National Institute of Environmental Medicine, Karolinska Institutet, PO Box 210, 171 77, Stockholm, Sweden
E. Friberg, N. Orsini & A. Wolk - Division of Endocrinology, Diabetes and Metabolism,Department of Medicine, Beth Israel Deaconess Medical Center, Harvard Medical School, Boston, MA, USA
C. S. Mantzoros
Authors
- E. Friberg
You can also search for this author inPubMed Google Scholar - N. Orsini
You can also search for this author inPubMed Google Scholar - C. S. Mantzoros
You can also search for this author inPubMed Google Scholar - A. Wolk
You can also search for this author inPubMed Google Scholar
Corresponding author
Correspondence toE. Friberg.
Rights and permissions
About this article
Cite this article
Friberg, E., Orsini, N., Mantzoros, C.S. et al. Diabetes mellitus and risk of endometrial cancer: a meta-analysis.Diabetologia 50, 1365–1374 (2007). https://doi.org/10.1007/s00125-007-0681-5
- Received: 19 December 2006
- Accepted: 15 March 2007
- Published: 03 May 2007
- Issue Date: July 2007
- DOI: https://doi.org/10.1007/s00125-007-0681-5