Preclinical evaluation of high-resolution CT, 18F-FDG, and 18F-NaF PET imaging for longitudinal monitoring of atherosclerosis (original) (raw)
Abstract
Rationale
Detection of atherosclerosis is essential to the management and prevention of life-threatening cardiovascular events. Although non-invasive imaging modalities, such as 18F-sodium fluoride (18F-NaF), 18F-fluorodeoxyglucose (18F-FDG) PET, and CT, visualize distinct hallmarks of atherosclerosis, there has yet to be a singular multi-cohort interrogation of their strengths and limitations. Thus, we focused on identifying the optimal approach for visualizing atherosclerosis at different stages of disease progression.
Methods
In this study, 6-week-old, male, ApoE deficient mice (Apoe_−/−) were placed on a high cholesterol diet for 12–20 weeks to induce calcific atherosclerotic disease. Age-matched, male, wildtype (WT) C57BL/6 mice fed with regular chow served as the control group. Mice were imaged at 12, 15, 18, and 20 weeks after starting their respective diets. To follow the progression of calcified atherosclerotic lesions, at each time point, in vivo,_ 18F-NaF microPET/CT images were acquired 1 h and 3 h post tracer i.v. injection. In a separate cohort, in vivo 18F-FDG PET/CT images were acquired at 3 and 5 h post tracer i.v. injection to follow inflammation as a result of progressive atherosclerotic lesion formation. High-resolution microCT images were acquired for all mice to visualize aorta calcification. After each imaging session, a subset (n = 3) was euthanized from each group and histological analysis of the aorta was performed to confirm disease progression.
Results
In this comparative study, within the same cohort, 18F-NaF PET detected atherosclerotic calcification earlier than microCT. At both 1 and 3 h post-injection (p.i.), calcified lesions were clearly detected by 18F-NaF with a six-fold higher signal in _Apoe_-/- compared to WT mice. Interestingly, 18F-NaF signal peaked at week 18, whereas aortic CT signal progressively increased with a 13-, 16-, and 29-fold at 15, 18, and 20 weeks, respectively. 18F-FDG arortic accumulation at weeks 12 and 15, were significantly greater in Apoe −/− mice than WT control when images were acquired at 5 h but not at 3 h p.i.. In contrast to histological analysis, at ≥ 16 weeks where inflammation is significantly elevated, 18F-FDG was equivalent in _Apoe_−/− and WT control mice and significantly reduced with disease progression.
Conclusions
Our results show that 18F-NaF PET and 18F-FDG PET are sensitive imaging modalities for the early detection of atherosclerotic lesions. However, both 18F-NaF PET and high-resolution microCT prove to be effective methods for monitoring late-stage and progressive disease.
Similar content being viewed by others
Molecular imaging in atherosclerosis
Article Open access 17 February 2022
Introduction
In the Western world, cardiovascular disease (CVD) is the underlying cause of approximately 50% of all deaths and is the predominant reason for cardiovascular events, including stroke, ischemia, and myocardial infarction [1]. The economic toll is significant worldwide; in the United States, the financial cost of CVD alone is ~ $240 billion annually [[2](/article/10.1007/s00259-025-07276-1#ref-CR2 "Ferrara P, Di Laura D, Cortesi PA, Mantovani LG. The economic impact of hypercholesterolemia and mixed dyslipidemia: a systematic review of cost of illness studies. PLoS ONE. 2021;16:e0254631. https://doi.org/10.1371/journal.pone.0254631
.")\]. Atherosclerosis is the most common contributor to cardiovascular events; with an aging population coupled with the increased prevalence of diabetes and obesity, atherosclerotic disease is predicted to exacerbate the global burden of CVD \[[3](#ref-CR3 "Cocker MS, Mc Ardle B, Spence JD, Lum C, Hammond RR, Ongaro DC, et al. Imaging atherosclerosis with hybrid [18F]fluorodeoxyglucose positron emission tomography/computed tomography imaging: what Leonardo da Vinci could not see. J Nucl Cardiol. 2012;19:1211–25.
https://doi.org/10.1007/s12350-012-9631-9
."),[4](#ref-CR4 "McKenney-Drake ML, Moghbel MC, Paydary K, Alloosh M, Houshmand S, Moe S, et al. (18)F-NaF and (18)F-FDG as molecular probes in the evaluation of atherosclerosis. Eur J Nucl Med Mol Imaging. 2018;45:2190–200.
https://doi.org/10.1007/s00259-018-4078-0
."),[5](#ref-CR5 "Ross R. Atherosclerosis–an inflammatory disease. N Engl J Med. 1999;340:115–26.
https://doi.org/10.1056/NEJM199901143400207
."),[6](/article/10.1007/s00259-025-07276-1#ref-CR6 "Tarkin JM, Joshi FR, Rudd JH. PET imaging of inflammation in atherosclerosis. Nat Rev Cardiol. 2014;11:443–57.
https://doi.org/10.1038/nrcardio.2014.80
.")\].Atherogenesis is a complex, multifaceted, inflammatory process mediated by dynamic molecular interactions between the matrix of the artery, tissue-resident and circulatory cells, and serum constituents that regulate the formation and growth of fibroinflammatory lipid plaques [[7](/article/10.1007/s00259-025-07276-1#ref-CR7 "Bonafiglia QA, Bendeck M, Gotlieb AI. Chapter 7 - vascular pathobiology: atherosclerosis and large vessel disease. In: Maximilian Buja L, Butany J, editors. Cardiovascular pathology. 5th ed. Academic Press; 2022. p. 265–306. https://doi.org/10.1016/B978-0-12-822224-9.00006-2
.")\]. Atherogenesis begins with an increase in vascular permeability that leads to the infiltration, retention, and oxidation of low-density lipoproteins (LDLs) in the subendothelial space \[[8](/article/10.1007/s00259-025-07276-1#ref-CR8 "Clinton SK, Underwood R, Hayes L, Sherman ML, Kufe DW, Libby P. Macrophage colony-stimulating factor gene expression in vascular cells and in experimental and human atherosclerosis. Am J Pathol. 1992;140:301–16.")\]. Lipid buildup in the intimal layers leads to endothelium activation and promotes immune cell infiltration, particularly monocyte-derived macrophages that phagocytose the oxidized LDLs. Inundated with cholesterol faster than they can degrade it, infiltrated and tissue-resident macrophages become lipid-laden foam cells \[[9](/article/10.1007/s00259-025-07276-1#ref-CR9 "Gui Y, Zheng H, Cao RY. Foam cells in atherosclerosis: novel insights into its origins, consequences, and molecular mechanisms. Front Cardiovasc Med. 2022;9:845942.
https://doi.org/10.3389/fcvm.2022.845942
.")\]. Foam cells contribute to the initiation and progression of atherosclerosis, and their accumulation triggers multiple pathways of programmed cell death, which in turn enlarges the necrotic cores and reduces plaque stability \[[9](/article/10.1007/s00259-025-07276-1#ref-CR9 "Gui Y, Zheng H, Cao RY. Foam cells in atherosclerosis: novel insights into its origins, consequences, and molecular mechanisms. Front Cardiovasc Med. 2022;9:845942.
https://doi.org/10.3389/fcvm.2022.845942
.")\]. Accumulation of lipid-containing foam cells beneath the endothelium results in fatty streaks, the first microscopic visible signs of atherosclerosis that over time, evolve into atherosclerotic plaques \[[8](/article/10.1007/s00259-025-07276-1#ref-CR8 "Clinton SK, Underwood R, Hayes L, Sherman ML, Kufe DW, Libby P. Macrophage colony-stimulating factor gene expression in vascular cells and in experimental and human atherosclerosis. Am J Pathol. 1992;140:301–16.")\]. As the fatty streak progresses, foam cell death and necrosis occur, and the resulting breakdown products form a highly thrombogenic lipid core covered by a collagen-rich fibrous cap that expands the plaque within the arterial wall \[[10](/article/10.1007/s00259-025-07276-1#ref-CR10 "Nadra I, Mason JC, Philippidis P, Florey O, Smythe CD, McCarthy GM, et al. Proinflammatory activation of macrophages by basic calcium phosphate crystals via protein kinase C and MAP kinase pathways: a vicious cycle of inflammation and arterial calcification? Circ Res. 2005;96:1248–56.
https://doi.org/10.1161/01.RES.0000171451.88616.c2
.")\].In atherosclerosis, persistent intimal inflammation thickens the vessel walls and triggers vascular calcification [[11](/article/10.1007/s00259-025-07276-1#ref-CR11 "Wu M, Rementer C, Giachelli CM. Vascular calcification: an update on mechanisms and challenges in treatment. Calcif Tissue Int. 2013;93:365–73. https://doi.org/10.1007/s00223-013-9712-z
.")\]. Calcification, characterized by calcium phosphate deposition in the intimal and medial layers, is predictive of atherosclerotic plaque burden \[[10](/article/10.1007/s00259-025-07276-1#ref-CR10 "Nadra I, Mason JC, Philippidis P, Florey O, Smythe CD, McCarthy GM, et al. Proinflammatory activation of macrophages by basic calcium phosphate crystals via protein kinase C and MAP kinase pathways: a vicious cycle of inflammation and arterial calcification? Circ Res. 2005;96:1248–56.
https://doi.org/10.1161/01.RES.0000171451.88616.c2
.")\]. Calcification within the fibrous caps of atherosclerotic plaques may increase local stress, resulting in an increased risk for plaque instability and myocardial infarction \[[12](/article/10.1007/s00259-025-07276-1#ref-CR12 "Shioi A, Ikari Y. Plaque calcification during atherosclerosis progression and regression. J Atheroscler Thromb. 2018;25:294–303.
https://doi.org/10.5551/jat.RV17020
.")\]. Multiple imaging modalities are being developed to interrogate the compositional features of atherosclerosis. The fluorinated glucose analog, 18F-FDG, remains the most widely used PET tracer and has been reported for measuring plaque inflammation via the enhanced glucose consumption of activated macrophages within the aortic vessel walls \[[13](/article/10.1007/s00259-025-07276-1#ref-CR13 "Schillaci O, Danieli R, Padovano F, Testa A, Simonetti G. Molecular imaging of atheroslerotic plaque with nuclear medicine techniques. Int J Mol Med. 2008;22:3–7.")\]. Ex vivo histological assessment confirms a strong correlation between macrophage density and 18F-FDG uptake \[[4](/article/10.1007/s00259-025-07276-1#ref-CR4 "McKenney-Drake ML, Moghbel MC, Paydary K, Alloosh M, Houshmand S, Moe S, et al. (18)F-NaF and (18)F-FDG as molecular probes in the evaluation of atherosclerosis. Eur J Nucl Med Mol Imaging. 2018;45:2190–200.
https://doi.org/10.1007/s00259-018-4078-0
."), [14](/article/10.1007/s00259-025-07276-1#ref-CR14 "Rudd JH, Warburton EA, Fryer TD, Jones HA, Clark JC, Antoun N, et al. Imaging atherosclerotic plaque inflammation with [18F]-fluorodeoxyglucose positron emission tomography. Circulation. 2002;105:2708–11.
https://doi.org/10.1161/01.cir.0000020548.60110.76
."), [15](/article/10.1007/s00259-025-07276-1#ref-CR15 "Sadeghi MM. (18)F-FDG PET and vascular inflammation: time to refine the paradigm? J Nucl Cardiol. 2015;22:319–24.
https://doi.org/10.1007/s12350-014-9917-1
.")\]. While 18F-FDG can assess plaque inflammation, its signal intensity is likely most pronounced in the early stages of the disease, particularly during macrophage differentiation and foam cell formation \[[16](/article/10.1007/s00259-025-07276-1#ref-CR16 "Ogawa M, Nakamura S, Saito Y, Kosugi M, Magata Y. What can be seen by 18F-FDG PET in atherosclerosis imaging? The effect of foam cell formation on 18F-FDG uptake to macrophages in vitro. J Nucl Med. 2012;53:55–8.
https://doi.org/10.2967/jnumed.111.092866
.")\].Detection and quantification of calcification surface area that is indicative of increased plaque instability can be achieved with 18F-NaF PET [[4](/article/10.1007/s00259-025-07276-1#ref-CR4 "McKenney-Drake ML, Moghbel MC, Paydary K, Alloosh M, Houshmand S, Moe S, et al. (18)F-NaF and (18)F-FDG as molecular probes in the evaluation of atherosclerosis. Eur J Nucl Med Mol Imaging. 2018;45:2190–200. https://doi.org/10.1007/s00259-018-4078-0
.")\]. 18F-NaF PET provides an in vivo quantitative visualization of active vascular calcification (< 50 μm). This molecular imaging assay measures changes in the chemical composition (calcium phosphate) that precede the high-density mature calcified plaques (> 100 μm) detectable by anatomical CT imaging \[[4](/article/10.1007/s00259-025-07276-1#ref-CR4 "McKenney-Drake ML, Moghbel MC, Paydary K, Alloosh M, Houshmand S, Moe S, et al. (18)F-NaF and (18)F-FDG as molecular probes in the evaluation of atherosclerosis. Eur J Nucl Med Mol Imaging. 2018;45:2190–200.
https://doi.org/10.1007/s00259-018-4078-0
."), [17](/article/10.1007/s00259-025-07276-1#ref-CR17 "Tzolos E, Dweck MR. (18)F-sodium fluoride ((18)F-NaF) for imaging microcalcification activity in the cardiovascular system. Arterioscler Thromb Vasc Biol. 2020;40:1620–6.
https://doi.org/10.1161/ATVBAHA.120.313785
.")\]. Calcium hydroxyapatite (Ca-HAP) deposition is a key feature of vascular calcification and is the primary site of 18F-NaF binding, a process achieved via the displacement of exposed hydroxyl ions with radioactive fluoride ions \[[18](/article/10.1007/s00259-025-07276-1#ref-CR18 "Czernin J, Satyamurthy N, Schiepers C. Molecular mechanisms of bone 18F-NaF deposition. J Nucl Med. 2010;51:1826–9.
https://doi.org/10.2967/jnumed.110.077933
.")\]. This site for 18F-NaF binding enables quantitative assessment of the calcification surface area. While 18F-NaF PET imaging can be employed to detect areas of active calcification, CT can be utilized to measure buildup of calcified mass/density associated with the disease \[[19](/article/10.1007/s00259-025-07276-1#ref-CR19 "Lloyd DJ, Helmering J, Kaufman SA, Turk J, Silva M, Vasquez S, et al. A volumetric method for quantifying atherosclerosis in mice by using microCT: comparison to en face. PLoS ONE. 2011;6:e18800.
https://doi.org/10.1371/journal.pone.0018800
.")\].Using genetically engineered mouse models of atherosclerosis, preclinical investigations have reported the utility of anatomical microCT, and 18F-NaF or 18F-FDG molecular PET imaging to visualize atherosclerotic disease [[4](/article/10.1007/s00259-025-07276-1#ref-CR4 "McKenney-Drake ML, Moghbel MC, Paydary K, Alloosh M, Houshmand S, Moe S, et al. (18)F-NaF and (18)F-FDG as molecular probes in the evaluation of atherosclerosis. Eur J Nucl Med Mol Imaging. 2018;45:2190–200. https://doi.org/10.1007/s00259-018-4078-0
.")\]. However, there has yet to be a holistic multi-cohort comparative analysis of the advantages and limitations of each imaging assay as a consequence of progressive disease. In this study, we comprehensively compared these translational molecular imaging methods in a mouse model of atherosclerosis and identified the imaging method optimal for each stage of the disease.Materials and methods
Animals
Male apolipoprotein E deficient (_Apoe_−/−) 6–8 weeks old mice (Jackson Laboratory, Cat#002052) were placed on a high-fat diet (HFD) (1.25% w/w cholesterol, Research Diets #D12108 C) for 20 weeks to induce atherosclerosis disease (n = 40). Age-matched male wildtype (WT) C57BL/6 mice were placed on a regular chow diet and served as the experimental control (n = 40). All experimental protocols in this study were reviewed and approved by the Institutional Animal Care and Use Committee of the University of California, Los Angeles.
Histological analysis
Microdissections of the mouse aorta were performed for en face Sudan IV staining of lipid to determine atherosclerotic lesion area and confirm the presence and severity of atherosclerosis. The aorta arch was dissected and cut in the coronal plane to expose the intimal surface. The dorsal descending thoracic region of the aorta was spared, the lesser curvature cut, and the aorta was folded open to reveal the intimal surface of the aortic arch. The specimen was rinsed in 70% ethanol for five minutes by positioning the aorta face-down. The specimen was then transferred to the Sudan IV working solution (1 g Sudan IV powder dissolved in a 1:1 mixture of 70% ethanol and acetone) and stained for 10 min. Followed by two washes in 80% ethanol for three minutes each. The aortic arch was then rinsed in PBS and images were acquired using a stereomicroscope connected to a digital camera at 10 times magnification [[20](/article/10.1007/s00259-025-07276-1#ref-CR20 "Centa M, Ketelhuth DFJ, Malin S, Gistera A. Quantification of atherosclerosis in mice. J Vis Exp. 2019. https://doi.org/10.3791/59828
.")\].Mouse aortic root sectioning and histology
Following sacrifice, mice were perfused with 4% paraformaldehyde (PFA) solution. The mouse aortic root and proximal ascending aorta, along with the base of the heart, was excised and immersed in 4% PFA at 4 °C for 24 h. After passing through a sucrose gradient, the tissue was frozen in OCT blocks and cut into 8 µm-thick sections for immunohistochemical assessment. Immunofluorescence (IF) staining was performed following standard protocols to evaluate aortic root plaque composition [[21](/article/10.1007/s00259-025-07276-1#ref-CR21 "Kim HJ, Cheng P, Travisano S, Weldy C, Monteiro JP, Kundu R, et al. Molecular mechanisms of coronary artery disease risk at the PDGFD locus. Nat Commun. 2023;14:847. https://doi.org/10.1038/s41467-023-36518-9
.")\]. Macrophage infiltrates were stained with an anti-mouse CD68 rabbit polyclonal antibody (1:300 dilution; ab125212, Abcam), followed by incubation with an AF488-conjugated secondary antibody (1:50 dilution; A21206, Invitrogen). Ferangi Blue staining was used to assess calcification, following the manufacturer’s protocol (Ferangi Blue Chromogen Kit 2, Biocare Medical, FB813). Lesion size was defined as the plaque area from the intimal edge to the border of the intima-media junction of all three cusps. Processed sections were imaged using an Echo Revolve microscope at 4 × and 10 × magnifications. Image analysis was performed using ImageJ (National Institutes of Health), using length information embedded in exported files. Statistical significance of total plaque area, CD68-positive area, and Ferangi Blue-positive area was determined using a two-sided t-test. All biological replicates were processed and stained on position-matched aortic root sections to minimize intra-experimental variance.18F-NaF microPET/CT imaging
18F-NaF PET images were acquired in _Apoe_−/− and WT mice at 12 (n = 20/group), 15 (n = 17/group), 18 (n = 14/group) and 20 (n = 11/group) weeks after the start of HFD or regular chow diet, respectively. Mice were anesthetized with 1.5% vaporized isoflurane followed by intravenous (i.v.) tail vein injection of 4.07 ± 0.15 MBq of 18F-NaF. After 60 min, mice received a 6-min PET scan (energy window 350–650 keV) followed by a microCT scan (voltage 80 kVP, current 150 µA, 720 projections, 200 µm resolution) on the GNEXT PET/CT scanner (Sofie Biosciences, Dulles, VA). Mice received a second 9-min PET scan at 3 h post tracer injection followed by a high-resolution microCT scan (voltage 80 kVP, current 150 µA, 720 projections, 100 µm resolution).
18F-FDG microPET/CT imaging
18F-FDG PET images were acquired on a separate cohort after 12 (n = 20/group), 15 (n = 17/group), 18 (n = 14/group), and 20 weeks (n = 11/group) on high-fat (_Apoe_−/−) or regular (WT) diet. Mice were injected 8.33 ± 0.09 MBq of 18F-FDG followed by 60 min of unconscious uptake. Each mouse was scanned at 3 h post tracer injection (static PET, 6 min) followed by a microCT scan (voltage 80 kVP, current 150 µA, 720 projections, 200 µm resolution) scan. Mice received a second PET scan (9 min) 5 h post tracer injection followed by a high-resolution CT scan (voltage 80 kVP, current 150 µA, 720 projections, 100 µm resolution).
MicroCT imaging
To visualize calcification mass, _Apoe_−/− mice and WT mice received a bin-1 high-resolution microCT scan (voltage 80 kVP, current 150 µA, 720 projections, 100 µm resolution) after 12, 15, 18, and 20 weeks on their respective diets. MicroCT data was acquired at the end of PET imaging sessions, resulting in n = 40/group at week 12, n = 34/group at week 15, n = 28/group at week 18, and n = 22/group at week 20.
MicroPET and microCT image quantification
Acquired PET images were reconstructed using a 3D-Ordered Subset Expectation Maximization (OSEM) algorithm (24 subsets and 3 iterations), with random, attenuation, and decay corrections. MicroCT images were reconstructed using a Modified Feldkamp Algorithm. Amide software was used to analyze co-registered microPET/CT images [[22](/article/10.1007/s00259-025-07276-1#ref-CR22 "Loening AM, Gambhir SS. AMIDE: a free software tool for multimodality medical image analysis. Mol Imaging. 2003;2:131–7. https://doi.org/10.1162/15353500200303133
.")\]. PET data were normalized to animal body weight, and expressed in the unit of mean standardized uptake value (SUVmean). For 18F-NaF, a 5×4×2 mm region of interest (ROI) was automatically segmented above the heart using a 1-mm boundary from rib bones, and a signal intensity threshold of 0.6 SUV. For microCT, a 6×6×4 mm fixed-volume ROI and a Hounsfield Unit (HU) threshold of 350 (HU water = 0) was placed in the thoracic cavity. The volumetric calcium content of the aorta (vHU) was defined as the product of mean ROI (HU) and the ROI volume (mm3) summed over all ROIs for each mouse \[[23](/article/10.1007/s00259-025-07276-1#ref-CR23 "Hsu JJ, Lu J, Umar S, Lee JT, Kulkarni RP, Ding Y, et al. Effects of teriparatide on morphology of aortic calcification in aged hyperlipidemic mice. Am J Physiol Heart Circ Physiol. 2018;314:H1203–13.
https://doi.org/10.1152/ajpheart.00718.2017
.")\]. For 18F-FDG images, ROIs for the aorta were obtained by drawing the ROI over the aorta through each slice of the scan.Statistical analysis
Statistical analyses were performed using GraphPad Prism (Version 10.4.1). Two-way ANOVA analysis was performed to determine differences between the two cohorts (WT control vs. Apoe −/− on HFD) in the longitudinal study, combining the effect of group and time on the biomarkers. Tukey’s multiple comparison was used for between-group comparisons, and statistical significance was set at a _p_-value ≤ 0.05. One-way ANOVA was used for within-group analysis with Šídák's multiple comparisons test to determine statistical significance. Correlative analysis was conducted using the Pearson’s rank correlation coefficient and the r2 and _p_-values reported.
Results
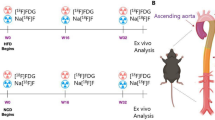
Establishing an atherosclerosis mouse model and confirming disease progression
To induce atherosclerotic disease 6–8-week-old _Apoe_−/− mice (n = 40) were placed on a high fat diet (HFD) for up to 20 weeks and age-matched wildtype (WT) mice (n = 40) were placed on a regular chow diet. _Apoe_−/− and WT mice underwent 18F-NaF or 18F-FDG PET imaging, followed by high-resolution microCT as illustrated in Fig. 1A at weeks 12, 15, 18, and 20. Following each imaging session (Fig. 1B), a subset (n = 3 per group) was euthanized, and the aorta was dissected postmortem. Disease progression was monitored by weekly body weight measurements (Fig. 2A) and disease presentation and severity were confirmed with postmortem Sudan IV staining (Fig. 2B). No significant difference in body weight was observed at early time-points. However, at 20 weeks on HFD, _Apoe_−/− mice showed significantly higher body weight compared to WT control mice (p = 0.01) (Fig. 2A). Longitudinal ANOVA analysis combining the effect of group and time revealed a significant difference (p < 0.0001). Sudan IV staining of the intimal surface of the aorta revealed significant lipid accumulation in _Apoe_−/− mice after 12 weeks on HFD (p = 0.004) compared to regular chow-fed WT mice (Fig. 2C). This trend of increased aortic Sudan IV staining continued into weeks 15 and 18 in _Apoe_−/− mice (p < 0.0005) (Fig. 2C). Longitudinal ANOVA analysis revealed a significant difference (p = 0.0018).
Fig. 1

The alternative text for this image may have been generated using AI.
Experimental study design and timeline of longitudinal in vivo imaging assays. A Two groups of mice, _Apoe_−/− on a high fat diet (HFD) and C57BL/6 wildtype control mice on a regular diet, were imaged with two microPET tracers: 18F-NaF (n = 20/group) and a separate cohort imaged by 18F-FDG (n = 20/group). MicroCT was performed on both cohorts combined (n = 40/group). B Schematic of the imaging protocol. Each cohort of mice was imaged at weeks 12, 15, 18, and 20 after starting their respective diets
Fig. 2

The alternative text for this image may have been generated using AI.
Establishment of atherosclerotic disease in an _Apoe_−/− mouse model. A Longitudinal monitoring of the body weight of _Apoe_−/− mice on a high-fat diet (HFD) compared with C57BL/6 control mice fed a regular chow diet for up to 20 weeks. B Representative Sudan IV red staining and (C) quantification of isolated aorta of _Apoe_−/− and C57BL/6 mice after 12, 15, 18, and 20 weeks on respective diets confirms lipid accumulation in _Apoe_−/−/HFD group. Statistical significance was determined by two-way ANOVA, using Tukey’s multiple comparisons test. *p < 0.05; **p < 0.01; *** p < 0.001; ****p < 0.0001
To investigate the abundance of the targets associated with calcification, which can be imaged with 18F-NaF PET and microCT, and macrophages in inflammatory atherosclerotic plaques assessed with 18F-FDG PET, we performed histology analyses of the aorta roots for CD68 expression and calcification using Ferangi Blue staining. As disease progressed, the total CD68-positive and calcified areas increased 79.7% (p = 0.030) (Fig. 3A and B) and 58.4-fold (p = 0.0357) (Fig. 3C and D) respectively. Additionally, the total lesion area (Fig. 3E), defined as the plaque area from the intimal edge to the border of the intima-media junction of all three cusps, increased 75.5% (p = 0.0088). These results further confirm disease progression in our Apoe −/−/HFD mouse model of atherosclerosis.
Fig. 3

The alternative text for this image may have been generated using AI.
Apoe −/−/HFD mouse aortic root plaque histology analysis. A Representative fluorescent images of mouse aortic root plaque histology stained for CD68 in Apoe −/−/HFD mice at 9 and 16 weeks. Higher magnification images highlight CD68+ macrophage infiltration. B Quantification of total CD68+ area (µm2) at 9 and 16 weeks. C Representative images of aortic root sections stained with Ferangi Blue to assess calcification in Apoe −/−/HFD mice at 9 and 16 weeks. D Quantification of total Ferangi Blue-stained area (µm2) at 9 and 16 weeks. E Quantification of total lesion area (µm2) at 9 and 16 weeks of Ferangi Blue stained aortic root sections. Data are mean ± SEM, N = 3–5. Statistical significance was determined by unpaired two-tailed t-test. *, p < 0.05. **, p < 0.01
Detection of atherosclerosis by in vivo 18F-NaF PET imaging
After 12 weeks on their respective diets, _Apoe_−/− and WT mice (n = 20/group) were imaged with 18F-NaF PET to detect the surface area of calcification. Mice were imaged 1 and 3 h post radiotracer injection (Fig. 4A). Quantitative analysis revealed a trend of increased 18F-NaF uptake in the aortas of _Apoe_−/− mice compared to the control group after each imaging session (Fig. 4B-C). At the early 1 h timepoint, high amounts of circulating 18F-NaF contributed to the elevated background PET signal, with 11 of 20 mice presenting with hyperintense aortic 18F-NaF signals. This phenomenon was resolved when images were acquired at 3 h post tracer injection. As a result, 3 h post-injection was determined to be the optimal time point for 18F-NaF PET image acquisition. _Apoe_−/− aortic calcification peaked at 18 weeks post initiation of HFD, evident by the substantive increase in aorta bound 18F-NaF compared to weeks 12 and 15. Significantly higher aortic 18F-NaF uptake was observed in the _Apoe_−/− mice compared to the WT mice at all time points (p ≤ 0.0005; t-test). Longitudinal ANOVA analysis combining the effect of group and time also revealed a significant difference (p = 0.009).
Fig. 4

The alternative text for this image may have been generated using AI.
Detection of atherosclerosis by in vivo 18F-NaF microPET. A Representative coronal plane of the co-registered microPET/CT images of the _Apoe_−/− and control mice at week 12, 15, 18, and 20 on their respective diets, 1 h and 3 h post injection of 18F-NaF. White arrows indicate 18F-NaF uptake in the aorta. B Quantitative PET of aortic 18F-NaF uptake in the _Apoe_−/−/HFD and WT control mice at week 12 (n = 20/group), 15 (n = 17/group), 18 (n = 14/group), and week 20 (n = 11/group), 1 h and (C) 3 h post injection of 18F-NaF. Statistical significance was determined by mixed-effect two-way ANOVA using Tukey’s multiple comparisons test. *p < 0.05; **p < 0.01; *** p < 0.001; ****p < 0.0001
Detection of atherosclerosis in mice by 18F-FDG PET imaging
A separate cohort of _Apoe_−/− (n = 20) and WT (n = 20) mice were imaged with 18F-FDG PET/CT. Increased 18F-FDG uptake is a marker of atherosclerotic inflammatory disease through the enhanced glycolytic demand of activated immune cells [[4](/article/10.1007/s00259-025-07276-1#ref-CR4 "McKenney-Drake ML, Moghbel MC, Paydary K, Alloosh M, Houshmand S, Moe S, et al. (18)F-NaF and (18)F-FDG as molecular probes in the evaluation of atherosclerosis. Eur J Nucl Med Mol Imaging. 2018;45:2190–200. https://doi.org/10.1007/s00259-018-4078-0
.")\]. Mice were imaged 3 h and 5 h post i.v. injection of 18F-FDG (Fig. [5](/article/10.1007/s00259-025-07276-1#Fig5)A). While no significant difference was detected at 3 h post-injection (Fig. [5](/article/10.1007/s00259-025-07276-1#Fig5)B), quantitative PET analysis revealed optimal aortic signal-to-noise was achieved at 5 h post-18F-FDG injection (Fig. [5](/article/10.1007/s00259-025-07276-1#Fig5)C); significantly higher aortic uptake of 18F-FDG was observed in _Apoe_−/− compared to WT mice. Peak aortic inflammation was observed at week 12 and decreased in weeks 15, 18, and 20 (Fig. [7](/article/10.1007/s00259-025-07276-1#Fig7)). Beyond week 15, no statistically significant difference in aortic 18F-FDG PET signal was observed between _Apoe_−/−/HFD and WT mice (Fig. [5](/article/10.1007/s00259-025-07276-1#Fig5)C). However, longitudinal ANOVA analysis combining the effect of group and time revealed a significant difference (_p_ \= 0.0006). Taken together, our data indicate that 18F-FDG PET imaging is only suitable for the diagnosis of atherosclerotic disease at the early stages.Fig. 5

The alternative text for this image may have been generated using AI.
Detection of atherosclerosis by in vivo 18F-FDG microPET. A Representative transverse plane of the co-registered microPET/CT images of the _Apoe_−/− and control mice at week 12, 15, 18, and 20 on their respective diets, 3 h and 5 h post injection of 18F-FDG. A white arrow indicates 18F-FDG uptake in the aorta. B Quantitative PET analysis obtained 3 h and (C) 5 h post-injection of 18F-FDG in _Apoe_−/−/HFD and WT control mice at week 12 (n = 20/group), 15 (n = 17/group), 18 (n = 14/group), and 20 (n = 11/group). Statistical significance was determined by mixed-effect two-way ANOVA, Tukey’s multiple comparisons test. *p < 0.05; **p < 0.01; *** p < 0.001; ****p < 0.0001; ns, not significant
Detection of atherosclerosis in mice by microCT imaging
Following 18F-NaF or 18F-FDG PET, mice received a high-resolution (100 μm) microCT scan to measure calcification (Fig. 6A). Based on previous studies, the CT HU value of > 300 HU can distinguish coronary artery plaques from surrounding myocardium (HU < 100) as such CT analysis and HU quantification was determined using ROI thresholding of > 350 HUs. Distinct from 18F-NaF, no statistical difference in dense calcification was observed at week 12 between _Apoe_−/− and WT control mice (Fig. 6B), an observation that may be attributed to the presence of calcification mass (< 100 μm) that is below the CT limit of detection. However, by week 15 and up to week 20 (n = 22/group), significantly higher calcified mass was measured in _Apoe_−/− mice. A significant increase in visible and measured calcification density was observed as the disease progressed (p < 0.001). Our results indicate that while CT lacks the sensitivity of PET to detect early-stage atherosclerotic disease, high-resolution CT can effectively monitor disease progression once the calcification reaches or exceeds the CT 100 μm detection limit.
Fig. 6

The alternative text for this image may have been generated using AI.
Detection of atherosclerosis by in vivo microCT. A Representative coronal view with maximum-intensity projection of the _Apoe_−/− and control mice at week 12, 15, 18, and 20 on their respective diets. A dashed red circle indicates calcification observed by the microCT. B Quantitative analysis of aortic calcium content in the _Apoe_−/−/HFD and control mice at week 12 (n = 40/group), 15 (n = 34/group), 18 (n = 28/group), and 20 (n = 22/group). Statistical significance was determined by mixed-effect two-way ANOVA, using Tukey’s multiple comparisons test. *p < 0.05; **p < 0.01; *** p < 0.001; ****p < 0.0001; ns, not significant
Discussion
Leveraging multiple imaging modalities, we longitudinally monitored metabolic glucose demand as a surrogate for inflammation with 18F-FDG PET, intimal calcification activity with 18F-NaF, and calcification density with CT in a preclinical model of atherosclerotic disease. Our goal in conducting this multi-cohort assessment was to identify the modalities that facilitate early diagnosis and enable monitoring of disease progression. While previous studies have reported these three imaging modalities as capable of detecting and/or monitoring disease progression, there has yet to be a systematic multi-cohort longitudinal assessment of how best to leverage each assay in the context of atherosclerosis progression stages [[4](/article/10.1007/s00259-025-07276-1#ref-CR4 "McKenney-Drake ML, Moghbel MC, Paydary K, Alloosh M, Houshmand S, Moe S, et al. (18)F-NaF and (18)F-FDG as molecular probes in the evaluation of atherosclerosis. Eur J Nucl Med Mol Imaging. 2018;45:2190–200. https://doi.org/10.1007/s00259-018-4078-0
.")\].In this study, we observed high accumulation of 18F-NaF and 18F-FDG in different regions, with 18F-NaF signal most pronounced in aortic arch and 18F-FDG accumulated in thoracic aorta. In WT mice, the absence of active calcification resulted in very low arortic 18F-NaF signal. ROIs were drawn by selecting the aortic arch region furthest from skeletal bones to minimize spillover. For 18F-FDG, uptake is high in the myocardium and the brown adipose tissue; the proximity to the aortic arch contributes to substantive signal spillovers that make accurate quantification challenging. Our findings are consistent with previous publications, where the aortic arch is the site of microcalcification or 18F-NaF PET accumulation while 18F-FDG PET is predominantly within the thoracic aorta of Apoe −/−/HFD mice [[24](/article/10.1007/s00259-025-07276-1#ref-CR24 "Aikawa E, Nahrendorf M, Figueiredo JL, Swirski FK, Shtatland T, Kohler RH, et al. Osteogenesis associates with inflammation in early-stage atherosclerosis evaluated by molecular imaging in vivo. Circulation. 2007;116:2841–50. https://doi.org/10.1161/CIRCULATIONAHA.107.732867
."), [25](/article/10.1007/s00259-025-07276-1#ref-CR25 "Khare HA, Binderup T, Hag AMF, Kjaer A. Longitudinal imaging of murine atherosclerosis with 2-deoxy-2-[(18)F]fluoro-D-glucose and [(18)F]-sodium fluoride in genetically modified Apolipoprotein E knock-out and wild type mice. Sci Rep. 2023;13:22983.
https://doi.org/10.1038/s41598-023-49585-1
.")\].A defining characteristic of atherosclerosis is inflammation, and 18F-FDG PET imaging has been successfully utilized to detect inflammatory diseases and shows promise as a non-invasive tool for visualizing inflamed plaques in patients [[5](/article/10.1007/s00259-025-07276-1#ref-CR5 "Ross R. Atherosclerosis–an inflammatory disease. N Engl J Med. 1999;340:115–26. https://doi.org/10.1056/NEJM199901143400207
.")\]. Similarly, in this preclinical study, we observed significantly elevated aortic 18F-FDG signal in the _Apoe_−/− mice compared to WT mice. Pathologically, microscopic inflammation precedes plaque buildup in the arterial walls and is a hallmark of early disease and because of their elevated metabolic activities, macrophages consume glucose at a high rate \[[26](/article/10.1007/s00259-025-07276-1#ref-CR26 "Alie N, Eldib M, Fayad ZA, Mani V. Inflammation, atherosclerosis, and coronary artery disease: PET/CT for the evaluation of atherosclerosis and inflammation. Clin Med Insights Cardiol. 2014;8:13–21.
https://doi.org/10.4137/CMC.S17063
.")\]. Additionally, previous studies have found atherosclerotic 18F-FDG uptake strongly correlates with macrophage density infiltrated within the fibrous cap \[[4](/article/10.1007/s00259-025-07276-1#ref-CR4 "McKenney-Drake ML, Moghbel MC, Paydary K, Alloosh M, Houshmand S, Moe S, et al. (18)F-NaF and (18)F-FDG as molecular probes in the evaluation of atherosclerosis. Eur J Nucl Med Mol Imaging. 2018;45:2190–200.
https://doi.org/10.1007/s00259-018-4078-0
."), [27](/article/10.1007/s00259-025-07276-1#ref-CR27 "Rudd JH, Myers KS, Bansilal S, Machac J, Woodward M, Fuster V, et al. Relationships among regional arterial inflammation, calcification, risk factors, and biomarkers: a prospective fluorodeoxyglucose positron-emission tomography/computed tomography imaging study. Circ Cardiovasc Imaging. 2009;2:107–15.
https://doi.org/10.1161/CIRCIMAGING.108.811752
."), [28](/article/10.1007/s00259-025-07276-1#ref-CR28 "Zhao Y, Zhao S, Kuge Y, Strauss WH, Blankenberg FG, Tamaki N. Localization of deoxyglucose and annexin A5 in experimental atheroma correlates with macrophage infiltration but not lipid deposition in the lesion. Mol Imaging Biol. 2011;13:712–20.
https://doi.org/10.1007/s11307-010-0389-7
.")\]. Interestingly, 18F-FDG PET signal peaked at week 12 (Fig. [7](/article/10.1007/s00259-025-07276-1#Fig7)A) when the plaque burden (Fig. [2](/article/10.1007/s00259-025-07276-1#Fig2)) was lowest (Fig. [7](/article/10.1007/s00259-025-07276-1#Fig7)D). Additionally, we observed a 30% decrease in 18F-FDG PET signal intensity over time, and by week 18 there was no significant difference between diseased and control mice. Our findings highlight the transitory nature of atherosclerotic inflammation and further support reported findings where early-stage aortic accumulation of 18F-FDG is attributed to macrophage foam cell formation and at later stages of disease 18F-FDG PET signal decreased as a consequence of their differentiation \[[26](/article/10.1007/s00259-025-07276-1#ref-CR26 "Alie N, Eldib M, Fayad ZA, Mani V. Inflammation, atherosclerosis, and coronary artery disease: PET/CT for the evaluation of atherosclerosis and inflammation. Clin Med Insights Cardiol. 2014;8:13–21.
https://doi.org/10.4137/CMC.S17063
."), [29](/article/10.1007/s00259-025-07276-1#ref-CR29 "Ishino S, Ogawa M, Mori I, Nishimura S, Ikeda S, Sugita T, et al. 18F-FDG PET and intravascular ultrasonography (IVUS) images compared with histology of atherosclerotic plaques: 18F-FDG accumulates in foamy macrophages. Eur J Nucl Med Mol Imaging. 2014;41:624–33.
https://doi.org/10.1007/s00259-013-2635-0
.")\]. This transitory nature may explain why some patients with known risk factors may not exhibit abnormal tracer uptake, as atheromas may only show transient uptake patterns that are not amenable to imaging over extended periods in human subjects.Fig. 7

The alternative text for this image may have been generated using AI.
Multi-cohort molecular PET and anatomical CT imaging in an _Apoe_−/−/HFD model of progressive atherosclerosis. A Quantitative 18F-FDG, B 18F-NaF PET SUV, and C microCT in the _Apoe_−/− mice fed a high-fat diet to induce atherosclerosis and monitored during early, mid, late, and terminal disease after 12, 15, 18, and 20 weeks of HFD, respectively. D, E, F Correlative analysis in matched subjects comparing 18F-FDG or 18F-NaF PET or anatomical high-resolution CT with terminal Sudan IV staining. A strong correlation between lipid accumulation (Sudan IV) and 18F-NaF PET or anatomical high-resolution CT was observed, while 18F-FDG did not correlate with %Sudan IV-positive area. G, H, I No correlation was observed with 18F-FDG or 18F-NaF SUVmean, while there was a strong correlation between microCT and 18F-FDG or 18F-NaF. Correlative analysis was conducted using the Pearson’s rank correlation coefficient. Statistical significance was determined by one-way ANOVA, Šídák's multiple comparisons test. *p < 0.05; **p < 0.01; *** p < 0.001; ****p < 0.0001
The use of 18F-FDG to visualize atherosclerotic plaques located on the aortic arch and thoracic aorta in live mice is challenging and further confounded by high uptake in the heart and brown adipose tissue in close proximity to diseased regions. Consistent with clinical reports, we identified the thoracic aorta as the primary site of inflammatory 18F-FDG PET signal [[30](/article/10.1007/s00259-025-07276-1#ref-CR30 "Blomberg BA, de Jong PA, Thomassen A, Lam MGE, Vach W, Olsen MH, et al. Thoracic aorta calcification but not inflammation is associated with increased cardiovascular disease risk: results of the CAMONA study. Eur J Nucl Med Mol Imaging. 2017;44:249–58. https://doi.org/10.1007/s00259-016-3552-9
.")\]. The high spillover from the heart and adipose tissue may account for the modest or lack of differences observed between the _Apoe_ _\-/-_/HFD group and the WT control. Lastly, 5 h post-injection imaging was superior to 3 h post-injection, allowing for decreased 18F-FDG blood activity and improved our ability to detect early atherosclerosis. This observation is consistent with clinical observations where blood 18F-FDG activity has been reported to mask artery wall signals and hamper the detection of vascular inflammation \[[31](/article/10.1007/s00259-025-07276-1#ref-CR31 "Ahlman MA, Vigneault DM, Sandfort V, Maass-Moreno R, Dave J, Sadek A, et al. Internal tissue references for 18Fluorodeoxyglucose vascular inflammation imaging: implications for cardiovascular risk stratification and clinical trials. PLoS ONE. 2017;12:e0187995.
https://doi.org/10.1371/journal.pone.0187995
.")\]. Our study suggests delaying PET scan acquisition to ≥ 5 h after 18F-FDG injection may be warranted for more accurate delineation of vascular inflammation.While 18F-FDG is taken up by metabolically active cells and serves as a surrogate for inflammation, 18F-NaF PET monitors a distinctly different hallmark of atherosclerosis. 18F-NaF identifies sites of active calcium deposition within vulnerable atherosclerotic lesions via the physicochemical exchange of hydroxyl groups of Ca-HAP with radioactive 18F− ions [[32](/article/10.1007/s00259-025-07276-1#ref-CR32 "Hu Y, Hu P, Hu B, Chen W, Cheng D, Shi H. Dynamic monitoring of active calcification in atherosclerosis by (18)F-NaF PET imaging. Int J Cardiovasc Imaging. 2021;37:731–9. https://doi.org/10.1007/s10554-020-02019-9
."), [33](/article/10.1007/s00259-025-07276-1#ref-CR33 "Joshi NV, Vesey A, Newby DE, Dweck MR. Will 18F-sodium fluoride PET-CT imaging be the magic bullet for identifying vulnerable coronary atherosclerotic plaques? Curr Cardiol Rep. 2014;16:521.
https://doi.org/10.1007/s11886-014-0521-4
.")\]. Our study revealed 3 h 18F-NaF uptake is required to achieve optimal signal-to-noise and reduce the rate of false positives due to incomplete clearance of the 18F-NaF tracer in circulation. Similar to atherosclerotic minipig studies where 18F-NaF signal was detected in regions of the vessel wall devoid of histological calcification \[[34](/article/10.1007/s00259-025-07276-1#ref-CR34 "Nogales P, Velasco C, Mota-Cobian A, Gonzalez-Cintado L, Mota RA, Espana S, et al. Analysis of (18)F-sodium fluoride positron emission tomography signal sources in atherosclerotic minipigs shows specific binding of (18)F-sodium fluoride to plaque calcifications. Arterioscler Thromb Vasc Biol. 2021;41:e480–90.
https://doi.org/10.1161/ATVBAHA.121.316075
.")\], we attribute these false positives at the early 1 h post-injection time point to blood volume retention within the small vessel wall \[[35](/article/10.1007/s00259-025-07276-1#ref-CR35 "Fayad ZA, Robson PM. Bringing color to atherosclerotic plaque calcification with (18)F-sodium fluoride positron emission tomography imaging. Arterioscler Thromb Vasc Biol. 2021;41:2585–7.
https://doi.org/10.1161/ATVBAHA.121.316773
.")\]. Images acquired at 1 h and 3 h post injection of 18F-NaF detected increased Ca-HAP presence at weeks 12 and 15 in _Apoe_−/−/HFD mice. Compared to control mice, _Apoe_−/−/HFD aortic 18F-NaF signal was 6- and ten-fold higher at weeks 12 and 15 respectively. This trend of increased 18F-NaF uptake persisted and peaked at week 18 (Fig. [7](/article/10.1007/s00259-025-07276-1#Fig7)B). Lastly, our preclinical results are consistent with a recently published clinical 18F-FDG/18F-NaF dual-tracer PET/CT study in patients with carotid atheroma (NCT01724749); similarly, we observed no correlation between 18F-NaF and 18F-FDG PET (Fig. [7](/article/10.1007/s00259-025-07276-1#Fig7)G). Additionally, we observed a strong correlation with increased 18F-NaF with disease progression/severity (_p_ \= 0.02, Fig. [7](/article/10.1007/s00259-025-07276-1#Fig7)E), while clinically, 18F-NaF increased with elevated 10-year CVD risk (_p_ < 0.01), but no significant trend was observed with 18F-FDG \[[36](/article/10.1007/s00259-025-07276-1#ref-CR36 "Patil S, Kata R, Teichner E, Subtirelu R, Ghonim M, Ghonim M, et al. Associations of subclinical microcalcification and inflammation with carotid atheroma development: a dual-tracer PET/CT study. Eur J Nucl Med Mol Imaging. 2025.
https://doi.org/10.1007/s00259-025-07127-z
.")\].In addition to 18F-FDG and 18F-NaF, newer PET tracers are under investigation for better imaging atherosclerosis, such as CXCR4-targeted 68 Ga-pentixafor, CCR2-targeted 64Cu-DOTA-ECL1i, and CCR5-targeted 64Cu-DOTA-DAPTA [[37](/article/10.1007/s00259-025-07276-1#ref-CR37 "Heo GS, Sultan D, Liu Y. Current and novel radiopharmaceuticals for imaging cardiovascular inflammation. Q J Nucl Med Mol Imaging. 2020;64:4–20. https://doi.org/10.23736/S1824-4785.20.03230-6
.")\]. Recently, an 89Zr-CD45 nanobody was reported as a robust PET tracer for imaging inflammation in multiple preclinical models \[[38](/article/10.1007/s00259-025-07276-1#ref-CR38 "SalehiFarid A, Rowley JE, Allen HH, Kruger IG, Tavakolpour S, Neeley K, et al. CD45-PET is a robust, non-invasive tool for imaging inflammation. Nature. 2025.
https://doi.org/10.1038/s41586-024-08441-6
.")\]. While these tracers show promise, further robust comparative preclinical and clinical imaging studies are needed to inform how best these tools can be implemented for detecting and monitoring atherosclerosis.Our preclinical evaluation of translational imaging methods for diagnosis and longitudinal monitoring of atherosclerosis shows that molecular PET imaging enables visualization of microscopic processes such as inflammation and Ca-HAP activity that precede morphological anatomical changes. Anatomical CT is efficient for the visualization of dense calcified deposits. In our preclinical studies, calcified lesions were not detectable by microCT until mid-late stages of the disease (Fig. 7C). In fact, no statistical differences in CT HU values were observed between _Apoe_−/− and control mice at week 12. However, from week 15 onwards, the CT HU values steadily increased over time. Similar to 18F-NaF, we observed a strong correlation between CT HU and disease severity (Fig. 7F). Furthermore a strong correlation was observed between CT HU and both 18F-FDG and 18F-NaF PET (Fig. 7H,I).
Conclusion
The results of this multi-cohort preclinical study reveal 18F-FDG PET and 18F-NaF PET are more effective at detecting early atherosclerotic lesions. Of the two PET assays, only 18F-NaF PET could monitor progressive disease. Importantly, 18F-NaF PET was capable of detecting disease earlier (week 12) than was achieved with anatomical microCT. However, as the disease progressed into later stages (weeks 18–20) microCT proved more effective than 18F-NaF PET. In summary, we have demonstrated the pairing of molecular and anatomical imaging provides a comprehensive overview of atherosclerotic pathobiology, whereby 18F-FDG PET is best used to measure inflammatory disease prevalent in early disease while 18F-NaF PET serves as an early readout of Ca-HAP activity that results in dense calcified lesions which are best visualized by CT, especially during late-stage disease. Importantly, we observed a strong correlation between 18F-NaF (r2 = 0.83, p = 0.0298) and anatomical CT (r2 = 0.60, p = 0.0015) with disease severity. Given the prevalence of integrated PET/CT instruments for the comprehensive detection of disease or monitoring of response to therapeutic interventions, molecular imaging of atherosclerotic inflammation (18F-FDG) and Ca-HAP activity (18F-NaF) should be coupled with high-resolution anatomical CT.
Data availability
The datasets generated during the study are available from the corresponding author on reasonable request.
Abbreviations
microCT:
Micro-computed tomography
microPET:
Micro-positron emission tomography
18F-NaF:
Sodium fluoride radiolabeled with fluorine-18
18F-FDG:
Fluorodeoxyglucose radiolabeled with fluorine-18
Apoe − / − :
Apolipoprotein E deficient
CVD:
Cardiovascular disease
Ca-HAP:
Calcium hydroxyapatite
HFD:
High fat diet
LDL:
Low-density lipoprotein
References
- Pahwa R, Jialal I. Atherosclerosis. Treasure Island: StatPearls; 2024.
Google Scholar - Ferrara P, Di Laura D, Cortesi PA, Mantovani LG. The economic impact of hypercholesterolemia and mixed dyslipidemia: a systematic review of cost of illness studies. PLoS ONE. 2021;16:e0254631. https://doi.org/10.1371/journal.pone.0254631.
Article PubMed PubMed Central CAS Google Scholar - Cocker MS, Mc Ardle B, Spence JD, Lum C, Hammond RR, Ongaro DC, et al. Imaging atherosclerosis with hybrid [18F]fluorodeoxyglucose positron emission tomography/computed tomography imaging: what Leonardo da Vinci could not see. J Nucl Cardiol. 2012;19:1211–25. https://doi.org/10.1007/s12350-012-9631-9.
Article PubMed PubMed Central Google Scholar - McKenney-Drake ML, Moghbel MC, Paydary K, Alloosh M, Houshmand S, Moe S, et al. (18)F-NaF and (18)F-FDG as molecular probes in the evaluation of atherosclerosis. Eur J Nucl Med Mol Imaging. 2018;45:2190–200. https://doi.org/10.1007/s00259-018-4078-0.
Article PubMed PubMed Central Google Scholar - Ross R. Atherosclerosis–an inflammatory disease. N Engl J Med. 1999;340:115–26. https://doi.org/10.1056/NEJM199901143400207.
Article PubMed CAS Google Scholar - Tarkin JM, Joshi FR, Rudd JH. PET imaging of inflammation in atherosclerosis. Nat Rev Cardiol. 2014;11:443–57. https://doi.org/10.1038/nrcardio.2014.80.
Article PubMed CAS Google Scholar - Bonafiglia QA, Bendeck M, Gotlieb AI. Chapter 7 - vascular pathobiology: atherosclerosis and large vessel disease. In: Maximilian Buja L, Butany J, editors. Cardiovascular pathology. 5th ed. Academic Press; 2022. p. 265–306. https://doi.org/10.1016/B978-0-12-822224-9.00006-2.
Chapter Google Scholar - Clinton SK, Underwood R, Hayes L, Sherman ML, Kufe DW, Libby P. Macrophage colony-stimulating factor gene expression in vascular cells and in experimental and human atherosclerosis. Am J Pathol. 1992;140:301–16.
PubMed PubMed Central CAS Google Scholar - Gui Y, Zheng H, Cao RY. Foam cells in atherosclerosis: novel insights into its origins, consequences, and molecular mechanisms. Front Cardiovasc Med. 2022;9:845942. https://doi.org/10.3389/fcvm.2022.845942.
Article PubMed PubMed Central CAS Google Scholar - Nadra I, Mason JC, Philippidis P, Florey O, Smythe CD, McCarthy GM, et al. Proinflammatory activation of macrophages by basic calcium phosphate crystals via protein kinase C and MAP kinase pathways: a vicious cycle of inflammation and arterial calcification? Circ Res. 2005;96:1248–56. https://doi.org/10.1161/01.RES.0000171451.88616.c2.
Article PubMed CAS Google Scholar - Wu M, Rementer C, Giachelli CM. Vascular calcification: an update on mechanisms and challenges in treatment. Calcif Tissue Int. 2013;93:365–73. https://doi.org/10.1007/s00223-013-9712-z.
Article PubMed PubMed Central CAS Google Scholar - Shioi A, Ikari Y. Plaque calcification during atherosclerosis progression and regression. J Atheroscler Thromb. 2018;25:294–303. https://doi.org/10.5551/jat.RV17020.
Article PubMed PubMed Central CAS Google Scholar - Schillaci O, Danieli R, Padovano F, Testa A, Simonetti G. Molecular imaging of atheroslerotic plaque with nuclear medicine techniques. Int J Mol Med. 2008;22:3–7.
PubMed Google Scholar - Rudd JH, Warburton EA, Fryer TD, Jones HA, Clark JC, Antoun N, et al. Imaging atherosclerotic plaque inflammation with [18F]-fluorodeoxyglucose positron emission tomography. Circulation. 2002;105:2708–11. https://doi.org/10.1161/01.cir.0000020548.60110.76.
Article PubMed CAS Google Scholar - Sadeghi MM. (18)F-FDG PET and vascular inflammation: time to refine the paradigm? J Nucl Cardiol. 2015;22:319–24. https://doi.org/10.1007/s12350-014-9917-1.
Article PubMed PubMed Central Google Scholar - Ogawa M, Nakamura S, Saito Y, Kosugi M, Magata Y. What can be seen by 18F-FDG PET in atherosclerosis imaging? The effect of foam cell formation on 18F-FDG uptake to macrophages in vitro. J Nucl Med. 2012;53:55–8. https://doi.org/10.2967/jnumed.111.092866.
Article PubMed CAS Google Scholar - Tzolos E, Dweck MR. (18)F-sodium fluoride ((18)F-NaF) for imaging microcalcification activity in the cardiovascular system. Arterioscler Thromb Vasc Biol. 2020;40:1620–6. https://doi.org/10.1161/ATVBAHA.120.313785.
Article PubMed PubMed Central CAS Google Scholar - Czernin J, Satyamurthy N, Schiepers C. Molecular mechanisms of bone 18F-NaF deposition. J Nucl Med. 2010;51:1826–9. https://doi.org/10.2967/jnumed.110.077933.
Article PubMed CAS Google Scholar - Lloyd DJ, Helmering J, Kaufman SA, Turk J, Silva M, Vasquez S, et al. A volumetric method for quantifying atherosclerosis in mice by using microCT: comparison to en face. PLoS ONE. 2011;6:e18800. https://doi.org/10.1371/journal.pone.0018800.
Article PubMed PubMed Central CAS Google Scholar - Centa M, Ketelhuth DFJ, Malin S, Gistera A. Quantification of atherosclerosis in mice. J Vis Exp. 2019. https://doi.org/10.3791/59828.
Article PubMed Google Scholar - Kim HJ, Cheng P, Travisano S, Weldy C, Monteiro JP, Kundu R, et al. Molecular mechanisms of coronary artery disease risk at the PDGFD locus. Nat Commun. 2023;14:847. https://doi.org/10.1038/s41467-023-36518-9.
Article PubMed PubMed Central CAS Google Scholar - Loening AM, Gambhir SS. AMIDE: a free software tool for multimodality medical image analysis. Mol Imaging. 2003;2:131–7. https://doi.org/10.1162/15353500200303133.
Article PubMed Google Scholar - Hsu JJ, Lu J, Umar S, Lee JT, Kulkarni RP, Ding Y, et al. Effects of teriparatide on morphology of aortic calcification in aged hyperlipidemic mice. Am J Physiol Heart Circ Physiol. 2018;314:H1203–13. https://doi.org/10.1152/ajpheart.00718.2017.
Article PubMed PubMed Central CAS Google Scholar - Aikawa E, Nahrendorf M, Figueiredo JL, Swirski FK, Shtatland T, Kohler RH, et al. Osteogenesis associates with inflammation in early-stage atherosclerosis evaluated by molecular imaging in vivo. Circulation. 2007;116:2841–50. https://doi.org/10.1161/CIRCULATIONAHA.107.732867.
Article PubMed CAS Google Scholar - Khare HA, Binderup T, Hag AMF, Kjaer A. Longitudinal imaging of murine atherosclerosis with 2-deoxy-2-[(18)F]fluoro-D-glucose and [(18)F]-sodium fluoride in genetically modified Apolipoprotein E knock-out and wild type mice. Sci Rep. 2023;13:22983. https://doi.org/10.1038/s41598-023-49585-1.
Article PubMed PubMed Central CAS Google Scholar - Alie N, Eldib M, Fayad ZA, Mani V. Inflammation, atherosclerosis, and coronary artery disease: PET/CT for the evaluation of atherosclerosis and inflammation. Clin Med Insights Cardiol. 2014;8:13–21. https://doi.org/10.4137/CMC.S17063.
Article PubMed Google Scholar - Rudd JH, Myers KS, Bansilal S, Machac J, Woodward M, Fuster V, et al. Relationships among regional arterial inflammation, calcification, risk factors, and biomarkers: a prospective fluorodeoxyglucose positron-emission tomography/computed tomography imaging study. Circ Cardiovasc Imaging. 2009;2:107–15. https://doi.org/10.1161/CIRCIMAGING.108.811752.
Article PubMed PubMed Central Google Scholar - Zhao Y, Zhao S, Kuge Y, Strauss WH, Blankenberg FG, Tamaki N. Localization of deoxyglucose and annexin A5 in experimental atheroma correlates with macrophage infiltration but not lipid deposition in the lesion. Mol Imaging Biol. 2011;13:712–20. https://doi.org/10.1007/s11307-010-0389-7.
Article PubMed Google Scholar - Ishino S, Ogawa M, Mori I, Nishimura S, Ikeda S, Sugita T, et al. 18F-FDG PET and intravascular ultrasonography (IVUS) images compared with histology of atherosclerotic plaques: 18F-FDG accumulates in foamy macrophages. Eur J Nucl Med Mol Imaging. 2014;41:624–33. https://doi.org/10.1007/s00259-013-2635-0.
Article PubMed CAS Google Scholar - Blomberg BA, de Jong PA, Thomassen A, Lam MGE, Vach W, Olsen MH, et al. Thoracic aorta calcification but not inflammation is associated with increased cardiovascular disease risk: results of the CAMONA study. Eur J Nucl Med Mol Imaging. 2017;44:249–58. https://doi.org/10.1007/s00259-016-3552-9.
Article PubMed Google Scholar - Ahlman MA, Vigneault DM, Sandfort V, Maass-Moreno R, Dave J, Sadek A, et al. Internal tissue references for 18Fluorodeoxyglucose vascular inflammation imaging: implications for cardiovascular risk stratification and clinical trials. PLoS ONE. 2017;12:e0187995. https://doi.org/10.1371/journal.pone.0187995.
Article PubMed PubMed Central CAS Google Scholar - Hu Y, Hu P, Hu B, Chen W, Cheng D, Shi H. Dynamic monitoring of active calcification in atherosclerosis by (18)F-NaF PET imaging. Int J Cardiovasc Imaging. 2021;37:731–9. https://doi.org/10.1007/s10554-020-02019-9.
Article PubMed Google Scholar - Joshi NV, Vesey A, Newby DE, Dweck MR. Will 18F-sodium fluoride PET-CT imaging be the magic bullet for identifying vulnerable coronary atherosclerotic plaques? Curr Cardiol Rep. 2014;16:521. https://doi.org/10.1007/s11886-014-0521-4.
Article PubMed Google Scholar - Nogales P, Velasco C, Mota-Cobian A, Gonzalez-Cintado L, Mota RA, Espana S, et al. Analysis of (18)F-sodium fluoride positron emission tomography signal sources in atherosclerotic minipigs shows specific binding of (18)F-sodium fluoride to plaque calcifications. Arterioscler Thromb Vasc Biol. 2021;41:e480–90. https://doi.org/10.1161/ATVBAHA.121.316075.
Article PubMed CAS Google Scholar - Fayad ZA, Robson PM. Bringing color to atherosclerotic plaque calcification with (18)F-sodium fluoride positron emission tomography imaging. Arterioscler Thromb Vasc Biol. 2021;41:2585–7. https://doi.org/10.1161/ATVBAHA.121.316773.
Article PubMed PubMed Central CAS Google Scholar - Patil S, Kata R, Teichner E, Subtirelu R, Ghonim M, Ghonim M, et al. Associations of subclinical microcalcification and inflammation with carotid atheroma development: a dual-tracer PET/CT study. Eur J Nucl Med Mol Imaging. 2025. https://doi.org/10.1007/s00259-025-07127-z.
Article PubMed PubMed Central Google Scholar - Heo GS, Sultan D, Liu Y. Current and novel radiopharmaceuticals for imaging cardiovascular inflammation. Q J Nucl Med Mol Imaging. 2020;64:4–20. https://doi.org/10.23736/S1824-4785.20.03230-6.
Article PubMed PubMed Central Google Scholar - SalehiFarid A, Rowley JE, Allen HH, Kruger IG, Tavakolpour S, Neeley K, et al. CD45-PET is a robust, non-invasive tool for imaging inflammation. Nature. 2025. https://doi.org/10.1038/s41586-024-08441-6.
Article Google Scholar
Acknowledgements
The authors thank Amgen, the UCLA Johnson Comprehensive Cancer Center, Sofie Biosciences, and the faculty and staff at the UCLA Crump Preclinical Imaging Technology Center. We thank Dr. Yin Tintut (UCLA) for training of the mouse aorta microdissection. We thank Dr. Yin Tintut (UCLA) and Dr. Roland Wu (Amgen Inc) for their thoughtful comments and review of our work. We thank Alexandra M. Klomhaus, PhD, and Keren Chen, MS, from the Department of Medicine Statistics Core at the UCLA David Geffen School of Medicine for their assistance with statistical analysis.
Funding
This work was supported, in part, by funding from Amgen, the National Institute of Health (NIH) Cancer Center Support Grant (2 P30 CA016042 - 44), the NIH S10 Shared Instrumentation for Animal Research Grant (1 S10 OD026917 - 01 A1), and the National Center for Advancing Translational Science (NCATS) of the NIH under the UCLA Clinical and Translational Science Institute Grant (UL1 TR001881).
Author information
Authors and Affiliations
- Crump Institute for Molecular Imaging, University of California, Los Angeles, Los Angeles, CA, 90025, USA
Mikayla Tamboline, Jeffrey Collins, Richard Taschereau, Arion F. Chatziioannou, Shili Xu & Oluwatayo F. Ikotun - Department of Molecular and Medical Pharmacology, University of California, Los Angeles, Los Angeles, CA, 90025, USA
Mikayla Tamboline, Jeffrey Collins, Richard Taschereau, Arion F. Chatziioannou, Shili Xu & Oluwatayo F. Ikotun - Department of Medicine, Division of Cardiovascular Medicine, Stanford University, Stanford, CA, 94305, USA
William Jackson, Wenduo Gu, Matthew Worssam & Paul Cheng - Stanford Cardiovascular Institute, Stanford University, Stanford, CA, 94305, USA
Paul Cheng - Cardiovascular, Amgen Inc., Thousand Oaks, CA, 91320, USA
John David & Simon Jackson - Jonsson Comprehensive Cancer Center, David Geffen School of Medicine, University of California, Los Angeles, Los Angeles, CA, 90025, USA
Arion F. Chatziioannou, Shili Xu & Oluwatayo F. Ikotun - Los Angeles, USA
Shili Xu & Oluwatayo F. Ikotun
Authors
- Mikayla Tamboline
- Jeffrey Collins
- William Jackson
- Wenduo Gu
- Paul Cheng
- John David
- Richard Taschereau
- Arion F. Chatziioannou
- Simon Jackson
- Shili Xu
- Oluwatayo F. Ikotun
Contributions
OFI, SX, and SJ conceptualized, designed, supervised, and acquired funding for this work. Data collection and analysis were performed by MT and SX. Immunohistochemistry and analysis were performed by WJ, WG, MW, and PC. Data interpretation by OFI, SX, SJ, and JD. Manuscript draft by MT. All authors contributed to the editing, reviewing, revising, rewriting, and approval of the final manuscript.
Corresponding authors
Correspondence toShili Xu or Oluwatayo F. Ikotun.
Ethics declarations
Ethics approvals
All experimental protocols in this study were reviewed and approved by the Institutional Animal Care and Use Committee of the University of California, Los Angeles. The study was carried out in compliance with the ARRIVE guidelines. This article does not contain any studies with human participants.
Consent to participate
No patients or human subjects involved in this study.
Consent to publish
No patients or human subjects involved in this study.
Competing interests
No potential conflict of interest relevant to this article was reported.
Clinical trial number
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tamboline, M., Collins, J., Jackson, W. et al. Preclinical evaluation of high-resolution CT, 18F-FDG, and 18F-NaF PET imaging for longitudinal monitoring of atherosclerosis.Eur J Nucl Med Mol Imaging 52, 4256–4267 (2025). https://doi.org/10.1007/s00259-025-07276-1
- Received: 30 December 2024
- Accepted: 07 April 2025
- Published: 28 April 2025
- Version of record: 28 April 2025
- Issue date: September 2025
- DOI: https://doi.org/10.1007/s00259-025-07276-1