Neocortical neuronal arrangement in Miller Dieker syndrome (original) (raw)
Abstract
Miller Dieker syndrome (MDS, type I lissencephaly) is a neuronal migration disorder, which is caused by deletions along the short arm of chromosome 17 (17p13.3). Recent studies would suggest that the cortical lamination in MDS is inverted, based on morphological criteria. The present neuropathological study examines the cerebral cortex from a 33-week old fetus with MDS using both neuronal and laminar-specific markers. These expression studies demonstrate a relatively preserved cortex and cortical lamination, overlying a layer of immature neurons in MDS brain. The findings are consistent with both a migratory and proliferative defect, giving rise to lissencephaly. Moreover, characterization of such rare human malformations of cortical development by immunohistochemical techniques will provide a greater understanding of the underlying mechanisms.
Access this article
Subscribe and save
- Starting from 10 chapters or articles per month
- Access and download chapters and articles from more than 300k books and 2,500 journals
- Cancel anytime View plans
Buy Now
Price excludes VAT (USA)
Tax calculation will be finalised during checkout.
Instant access to the full article PDF.
Fig. 1

The alternative text for this image may have been generated using AI.
Fig. 2

The alternative text for this image may have been generated using AI.
Fig. 3

The alternative text for this image may have been generated using AI.
Fig. 4
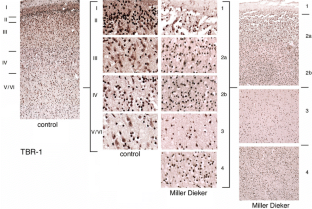
The alternative text for this image may have been generated using AI.
Fig. 5

The alternative text for this image may have been generated using AI.
Fig. 6

The alternative text for this image may have been generated using AI.
Similar content being viewed by others
References
- Banham AH, Beasley N, Campo E, Fernandez PL, Fidler C, Gatter K, Jones M, Mason DY, Prime JE, Trougouboff P, Wood K, Cordell JL (2001) The FOXP1 winged helix transcription factor is a novel candidate tumor suppressor gene on chromosome 3p. Cancer Res 61:8820–8829
PubMed CAS Google Scholar - Barkovich AJ, Koch TK, Carrol CL (1991) The spectrum of lissencephaly: report of ten patients analyzed by magnetic resonance imaging. Ann Neurol 30:139–146
Article PubMed CAS Google Scholar - Caviness VS Jr (1975) Mechanical model of brain convolutional development. Science 189:18–21
Article PubMed Google Scholar - Dobyns WB, Reiner O, Carrozzo R, Ledbetter DH (1993) Lissencephaly. A human brain malformation associated with deletion of the LIS1 gene located at chromosome 17p13. JAMA 270:2838–2842
Article PubMed CAS Google Scholar - Faulkner NE, Dujardin DL, Tai CY, Vaughan KT, O’Connell CB, Wang Y, Vallee RB (2000) A role for the lissencephaly gene LIS1 in mitosis and cytoplasmic dynein function. Nat Cell Biol 2:784–791
Article PubMed CAS Google Scholar - Feng Y, Olson EC, Stukenberg PT, Flanagan LA, Kirschner MW, Walsh CA (2000) LIS1 regulates CNS lamination by interacting with mNudE, a central component of the centrosome. Neuron 28:665–679
Article PubMed CAS Google Scholar - Feng Y, Walsh CA (2004) Mitotic spindle regulation by Nde1 controls cerebral cortical size. Neuron 44:279–293
Article PubMed CAS Google Scholar - Ferrer I, Fernandez-Alvarez E (1977) Lissencephaly: agyria. A study using the Golgi technic. J Neurol Sci 34:109–120
Article PubMed CAS Google Scholar - Gambello MJ, Darling DL, Yingling J, Tanaka T, Gleeson JG, Wynshaw-Boris A (2003) Multiple dose-dependent effects of Lis1 on cerebral cortical development. J Neurosci 23:1719–1729
PubMed CAS Google Scholar - Hirotsune S, Fleck MW, Gambello MJ, Bix GJ, Chen A, Clark GD, Ledbetter DH, McBain CJ, Wynshaw-Boris A (1998) Graded reduction of Pafah1b1 (Lis1) activity results in neuronal migration defects and early embryonic lethality. Nat Genet 19:333–339
Article PubMed CAS Google Scholar - Johnston MV, Haddad R, Carman-Young A, Coyle JT (1982) Neurotransmitter chemistry of lissencephalic cortex induced in ferrets by fetal treatment with methylazoxymethanol acetate. Brain Res 256:285–291
PubMed CAS Google Scholar - Liu Z, Steward R, Luo L (2000) Drosophila Lis1 is required for neuroblast proliferation, dendritic elaboration and axonal transport. Nat Cell Biol 2:776–783
Article PubMed CAS Google Scholar - Miyata H, Chute DJ, Fink J, Villablanca P, Vinters HV (2004) Lissencephaly with agenesis of corpus callosum and rudimentary dysplastic cerebellum: a subtype of lissencephaly with cerebellar hypoplasia. Acta Neuropathol (Berl) 107:69–81
Article Google Scholar - Noctor SC, Palmer SL, McLaughlin DF, Juliano SL (2001) Disruption of layers 3 and 4 during development results in altered thalamocortical projections in ferret somatosensory cortex. J Neurosci 21:3184–3195
PubMed CAS Google Scholar - Pancoast M, Dobyns W, Golden JA (2005) Interneuron deficits in patients with the Miller-Dieker syndrome. Acta Neuropathol (Berl) 109:400–404
Article Google Scholar - Reiner O, Carrozzo R, Shen Y, Wehnert M, Faustinella F, Dobyns WB, Caskey CT, Ledbetter DH (1993) Isolation of a Miller-Dieker lissencephaly gene containing G protein beta-subunit-like repeats. Nature 364:717–721
Article PubMed CAS Google Scholar - Sheen VL, Walsh CA (2003) Developmental genetic malformations of the cerebral cortex. Curr Neurol Neurosci Rep 3:433–441
Article PubMed Google Scholar - Viot G, Sonigo P, Simon I, Simon-Bouy B, Chadeyron F, Beldjord C, Tantau J, Martinovic J, Esculpavit C, Brunelle F, Munnich A, Vekemans M, Encha-Razavi F (2004) Neocortical neuronal arrangement in LIS1 and DCX lissencephaly may be different. Am J Med Genet A 126:123–128
Article Google Scholar
Acknowledgements
This work was supported by grants to CAW from the NINDS (2R37 NS35129 and 1PO1NS40043), the March of Dimes, and the McKnight Foundation. CAW is an Investigator of the Howard Hughes Medical Institute. VLS is supported by grants from the NIMH (1K08MH/NS63886-01), Julian and Carol Cohen, and the Milton Fund. VLS is a Charles A. Dana fellow and a Beckman Young Investigator.
Author information
Authors and Affiliations
- Department of Neurology, Beth Israel Deaconess Medical Center, Division of Neurogenetics and Howard Hughes Medical Institute, Boston, MA, 02115, USA
Volney L. Sheen, Russell J. Ferland, Jason Neal, Megan Harney, Robert S. Hill & Christopher A. Walsh - Nuffield Department of Clinical Laboratory Sciences, University of Oxford, Level 4 Academic Block, John Radcliffe Hospital, Oxford, Oxfordshire, OX3 9DU, UK
Alison Banham & Phillip Brown - Department of Pathology, Feinberg School of Medicine, Northwestern University, Chicago, IL, 60 611-3008, USA
Anjen Chenn - Department of Neuropathology, Brigham and Women’s Hospital, Harvard Medical School, Boston, MA, 02115, USA
Joseph Corbo & Rebecca Folkerth - Department of Neuropathology, Harvard Medical School, Beth Israel Deaconess Medical Center, Boston, MA, 02115, USA
Jonathan Hecht - Program in Biological and Biomedical Sciences, Harvard Medical School, Boston, MA, 02115, USA
Christopher A. Walsh
Authors
- Volney L. Sheen
- Russell J. Ferland
- Jason Neal
- Megan Harney
- Robert S. Hill
- Alison Banham
- Phillip Brown
- Anjen Chenn
- Joseph Corbo
- Jonathan Hecht
- Rebecca Folkerth
- Christopher A. Walsh
Corresponding author
Correspondence toVolney L. Sheen.
Rights and permissions
About this article
Cite this article
Sheen, V.L., Ferland, R.J., Neal, J. et al. Neocortical neuronal arrangement in Miller Dieker syndrome.Acta Neuropathol 111, 489–496 (2006). https://doi.org/10.1007/s00401-005-0010-3
- Received: 21 September 2005
- Revised: 01 November 2005
- Accepted: 02 November 2005
- Published: 03 February 2006
- Issue date: May 2006
- DOI: https://doi.org/10.1007/s00401-005-0010-3