Distinguishing benign and malignant thyroid nodules using plasma trimethylamine N-oxide, carnitine, choline and betaine (original) (raw)
Abstract
Purpose
Trimethylamine N-oxide (TMAO), a gut microbiome–derived metabolite, and its precursors (carnitine, choline, betaine) have not been fully examined in relation to thyroid cancer (TC) risk. The aim of this study was to assess the value of TMAO and its precursors in diagnosis of benign and malignant thyroid nodules.
Methods
In this study, high-performance liquid chromatography-tandem mass spectrometry was utilized to measure the levels of plasma TMAO and its precursors (choline, carnitine, and betaine) in 215 TC patients, 63 benign thyroid nodules (BTN) patients and 148 healthy controls (HC). The distribution of levels of TMAO and its precursors among the three groups were compared by the Kruskal–Wallis test. Receiver operating characteristic curve (ROC) analysis was performed to evaluate the sensitivity, specificity, and the predictive accuracy of single and combined biomarkers.
Results
In comparison to HC, TC showed higher levels of TMAO and lower levels of its precursors (carnitine, choline, and betaine) (all P < 0.001). Plasma choline (P < 0.01) and betaine (P < 0.05) were declined in BTN than HC. The levels of carnitine (P < 0.001) and choline (P < 0.05) were significantly higher in BTN than that in TC group. Plasma TMAO showed lower levels in TC with lymph node metastasis (101.5 (73.1–144.5) ng/ml) than those without lymph node metastasis (131 (84.8–201) ng/ml, P < 0.05). Combinations of these four metabolites achieved good performance in the differential diagnosis, with the area under the ROC curve of 0.703, 0.741, 0.793 when discriminating between TC and BTN, BTN and HC, TC and HC, respectively.
Conclusion
Plasma TMAO, along with its precursors could serve as new biomarkers for the diagnosis of benign and malignant thyroid nodules.
Similar content being viewed by others
Introduction
Thyroid cancer (TC), a most common endocrine malignancy, has garnered significant attention due to rapidly increasing global incidence rate (Siegel et al. 2022). Papillary thyroid carcinoma (PTC) remains the most prevalent subtype, accounting for approximately 85% of all cases, followed by follicular, medullary, and anaplastic variants (Fagin and Wells 2016). Although the precise causes of TC remain elusive, extensive research has identified genetic predispositions, radiation exposure (head and neck region), environmental pollutants, high BMI, lifestyle and diet as potential contributors to the disease risk (Bogovic Crncic et al. 2020).
Ultrasonography (US) is the most important diagnostic tool and extremely sensitive for detecting thyroid nodules. Furthermore, US can be used to determine the size, number and characteristics of thyroid nodules, to guide fine-needle aspiration (FNA), and to detect lymph node metastasis (Gharib et al. 2016; Haugen et al. 2016). The US risk stratification system, known as the Thyroid Imaging Reporting and Data System (TIRADS), has been established to classify the malignancy risk of a thyroid nodule and subsequently recommend the requirement for US-guided FNA (Russ et al. 2017; Seo et al. 2015). Although US-guided FNA was a reliable and well accepted method for diagnosing TC (Seiberling et al. 2008), various complications such as bleeding, hoarseness, and infection can occur (Ha et al. 2018). Moreover, the accuracy of the FNA is influenced significantly by factors that are not solely related to the operator and cytologist, but also to the patient (e.g. size, location, homogeneity and vascularity of the nodule) (Blum 2000). Although the overall survival rate of TC is relatively high, early diagnosis is still of paramount importance (Liu et al. [2022b](/article/10.1007/s00432-024-05666-w#ref-CR20 "Liu X, Wong CKH, Chan WWL, Au ICH, Tang EHM, Lang BHH (2022b) Survival after hemithyroidectomy versus total thyroidectomy in non-high-risk differentiated thyroid cancer: population-based analysis. BJS Open. https://doi.org/10.1093/bjsopen/zrac146
")). Identifying effective early diagnostic TC biomarkers can minimize the need for unnecessary biopsies, prevent the misdiagnosis or undertreatment of a curable cancer, and to some extent relieve the patient’s anxiety (Sakorafas et al. [2010](/article/10.1007/s00432-024-05666-w#ref-CR30 "Sakorafas GH, Mastoraki A, Lappas C, Safioleas M (2010) Small (<10 mm) thyroid nodules; how aggressively should they be managed? Onkologie 33:61–64")).In order to meet the high bioenergetic and biosynthetic demands required for vigorous proliferation, cancer cells reprogram their metabolism, leading to the production of abnormal metabolic substances (Mayers and Vander Heiden 2015). Recent metabolomics studies have found that PTC is involved in metabolic abnormalities including glucose, lipid metabolism, amino acids and nucleotide metabolism (Farrokhi Yekta et al. 2018; Zhao et al. 2015). Trimethylamine N-oxide (TMAO) is a gut microbiome–derived metabolite, and is synthesized in multiple steps, with main precursor molecules being carnitine, choline, and betaine (Koeth et al. 2013; Wang et al. 2011). These precursor molecules have been shown to be present in abundance in red meat, eggs, milk, and certain fish products, including salmon (Zeisel and da Costa 2009). Over the past decade, high levels of TMAO have been linked to an increased risk of cardiovascular disease (Heianza et al. [2017](/article/10.1007/s00432-024-05666-w#ref-CR15 "Heianza Y, Ma W, Manson JE, Rexrode KM, Qi L (2017) Gut microbiota metabolites and risk of major adverse cardiovascular disease events and death: a systematic review and meta-analysis of prospective studies. J Am Heart Assoc. https://doi.org/10.1161/JAHA.116.004947
")), stroke (Tu and Xia [2024](/article/10.1007/s00432-024-05666-w#ref-CR38 "Tu R, Xia J (2024) Stroke and vascular cognitive impairment: the role of intestinal microbiota metabolite TMAO. CNS Neurol Disord Drug Targets 23:102–121")), diabetes (Dambrova et al. [2016](/article/10.1007/s00432-024-05666-w#ref-CR6 "Dambrova M, Latkovskis G, Kuka J, Strele I, Konrade I, Grinberga S, Hartmane D, Pugovics O, Erglis A, Liepinsh E (2016) Diabetes is associated with higher trimethylamine N-oxide plasma levels. Exp Clin Endocrinol Diabetes 124:251–256")), and chronic kidney disease (Tang et al. [2015](/article/10.1007/s00432-024-05666-w#ref-CR36 "Tang WH, Wang Z, Kennedy DJ, Wu Y, Buffa JA, Agatisa-Boyle B, Li XS, Levison BS, Hazen SL (2015) Gut microbiota-dependent trimethylamine N-oxide (TMAO) pathway contributes to both development of renal insufficiency and mortality risk in chronic kidney disease. Circ Res 116:448–455")). Furthermore, a growing number of studies have also identified a potential role for TMAO in tumor risk including colorectal cancer (Jalandra et al. [2021](/article/10.1007/s00432-024-05666-w#ref-CR16 "Jalandra R, Dalal N, Yadav AK, Verma D, Sharma M, Singh R, Khosla A, Kumar A, Solanki PR (2021) Emerging role of trimethylamine-N-oxide (TMAO) in colorectal cancer. Appl Microbiol Biotechnol 105:7651–7660")), breast cancer (Wang et al. [2022](/article/10.1007/s00432-024-05666-w#ref-CR40 "Wang H, Rong X, Zhao G, Zhou Y, Xiao Y, Ma D, Jin X, Wu Y, Yan Y, Yang H, Zhou Y, Qian M, Niu C, Hu X, Li DQ, Liu Q, Wen Y, Jiang YZ, Zhao C, Shao ZM (2022) The microbial metabolite trimethylamine N-oxide promotes antitumor immunity in triple-negative breast cancer. Cell Metab 34(581–594):e588")), prostate cancer (Mondul et al. [2015](/article/10.1007/s00432-024-05666-w#ref-CR24 "Mondul AM, Moore SC, Weinstein SJ, Karoly ED, Sampson JN, Albanes D (2015) Metabolomic analysis of prostate cancer risk in a prospective cohort: The alpha-tocolpherol, beta-carotene cancer prevention (ATBC) study. Int J Cancer 137:2124–2132")) and stomach cancer (Stonans et al. [2023](/article/10.1007/s00432-024-05666-w#ref-CR35 "Stonans I, Kuzmina J, Polaka I, Grinberga S, Sevostjanovs E, Liepins E, Aleksandravica I, Santare D, Kirsners A, Skapars R, Pcolkins A, Tolmanis I, Sivins A, Leja M, Dambrova M (2023) The association of circulating L-carnitine, gamma-butyrobetaine and trimethylamine N-oxide levels with gastric cancer. Diagnostics (basel) 13:1341")). Potential mechanisms proposed for the carcinogenesis of TMAO may involve inflammation, oxidative stress, DNA damage, and protein misfolding (Jalandra et al. [2021](/article/10.1007/s00432-024-05666-w#ref-CR16 "Jalandra R, Dalal N, Yadav AK, Verma D, Sharma M, Singh R, Khosla A, Kumar A, Solanki PR (2021) Emerging role of trimethylamine-N-oxide (TMAO) in colorectal cancer. Appl Microbiol Biotechnol 105:7651–7660")).There is evidence suggesting that higher levels of TMAO and its precursors in blood can be indicative of either a higher risk of malignancy or indeed its presence (Gatarek and Kaluzna-Czaplinska 2021). However, to date, no studies have specifically examined the relationship between TMAO and its precursors, as well as their potential impact on thyroid cancer risk. Blood-based biomarker tests may offer a cost-effective and non-invasive method for detecting or predicting the disease, but little is known about the levels of TMAO and its precursors in benign thyroid nodules (BTN) and TC. Herein we conducted a case-control study to reveal differences in the plasma TMAO and its precursors of TC, BTN, and healthy controls (HC) and discover non-invasive biomarkers for diagnosis of malignant and benign thyroid nodules.
Material and methods
Study population and sample collection
Plasma samples obtained from 215 patients diagnosed with TC, 63 patients diagnosed with BTN, and 148 HC were consecutively collected between November 2022 and November 2023 from the Nanjing First Hospital (Nanjing, China). The three subgroups were frequency matched on age and sex. Healthy controls were selected from health management center and they should have no history of cancer, thyroid related diseases, severe liver and kidney diseases, and have normal thyroid ultrasound. Patients with BTN were diagnosed based on ultrasound (nodules in American College of Radiology Thyroid Imaging Reporting and Data System (TI-RADS) category 3) or surgical histopathology. All TC were confirmed by surgical histopathology. TC patients were excluded if they had a history of any other cancers. Additionally, the patients’ tumor stage and lymph node stage were listed based on the Tumor/Node/Metastasis (TNM) classification. We obtained approval from the ethics committee of the Nanjing First Hospital and informed written consents from all participants were acquired.
3 ml of fasting peripheral blood was collected from all subjects and added to EDTA-K2 anticoagulant-containing tubes. After blood collection, the tubes were placed at room temperature (22–26 °C) and immediately centrifuged at 3000 rpm for 10 min, and the upper layer of plasma was collected and stored at − 80 °C until use.
Plasma TMAO and its precursors analysis
Concentrations of plasma TMAO and its precursors (choline, carnitine, and betaine) were determined by high-performance liquid chromatography–tandem mass spectrometry (HPLC/MS–MS). We used trimethylamine-d9 N-Oxide TMAO (d9-TMAO, Cambridge Isotopes, Tewksbury, MA, USA) as an internal standard. Briefly, 50 ul of either the plasma or standards was mixed with 500 μl methanol containing 1 ug/ml internal standard and were subsequently centrifuged at 12000×g for 10 min to precipitate the proteins. Then, the remaining supernatant (5 μl) was injected into a Waters BEH C18 column (2.1 × 50 mm, 1.7 μm; Cat. No. 03433919615148, Massachusetts, USA) at a flow rate of 0.4 ml/min using an HPLC-pump, autosampler, and AB Sciex Triple Quad 4500 mass spectrometer (AB Sciex, FC, USA) with an electrospray ionization source. By mixing solvent A (0.1% formic acid in water) and solvent B (0.1% formic acid in methanol) in different ratios, starting at 10% B, a discontinuous gradient was generated to separate the analytes and then linearly increased to 80% B over 1.0 min, then hold for 1.8 min, then return to 10% B. Quantification of TMAO was performed using multiple reaction monitoring (MRM) transitions at m/z 76.1 → 59.0, d9‐TMAO at m/z 85.1 → 68.1, choline at m/z 104.1 → 60.1, carnitine at m/z 162.0 → 60.2, betaine at m/z 118.1 → 59.2.
Statistical analysis
Shapiro–Wilk normality test was used to assess the normality of continuous variables’ distributions, and when P > 0.05, the distribution is considered to conform to the normal distribution. Continuous variables with normal distribution were described as mean ± standard deviation and compared by Analysis of Variance (ANOVA). Continuous variables with non-normal distribution were presented as median with an interquartile range, which were compared by the Kruskal–Wallis test. If a significant difference was found, a Dunn’s test was further applied to perform pairwise comparisons. Categorical variables were presented as counts and percentages, and tested using Pearson’s Chi-squared test. The correlation between levels of biomarkers was evaluated by Spearman correlation analysis. In addition, receiver operating characteristic curve (ROC) analysis was performed to evaluate the sensitivity, specificity, and the predictive accuracy of single and combined biomarkers. In this study, significance was defined as P < 0.05, and all statistical tests were two-tailed. Statistical analyses and graphing were performed by R software (version 3.6.1) and GraphPad Prism software.
Results
Clinical characteristics and TMAO and precursors expression in HC, BTN and TC groups
Overall, 426 participants including 148 HC, 63 BTN and 215 TC were collected for the analysis. All TC were confirmed by surgical histopathology and were classified as PTC. The distribution of sex and age in the three groups was consistent among all groups and was summarized in Table 1. In the TC group, the rate of lymph node metastasis was 38.14%. Among the TC cases, 96.28% were stage I and 3.72% were stage II.
Table 1 General information of all participants by subgroup
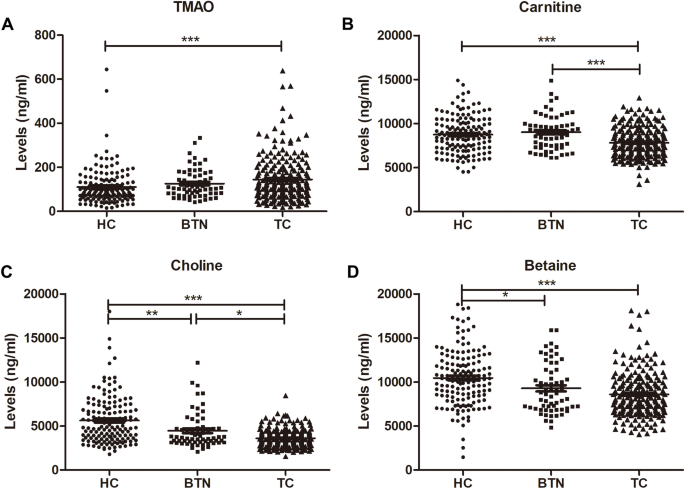
We examined the plasma TMAO and precursors (carnitine, choline, and betaine) expression among the HC group, BTN group and TC group (Table 2, Fig. 1). The results showed that the expression of TMAO in the TC group (median (interquartile range): 31.53 (26.87–37.77) ng/ml) was significantly higher than that in the HC group (118 (77–186.5) ng/ml, P < 0.001) (Fig. 1A). Plasma levels of carnitine were lower in the TC group (7630 (6510–8890) ng/ml) than that in the HC group (8665 (7227.5–10,100) ng/ml, P < 0.001) and BTN group (8970 (7525–9865) ng/ml, P < 0.001) (Fig. 1B). Concentrations of choline were recorded at 4860 (3585–7070) ng/ml in the HC group, 3690 (3195–4740) ng/ml in the BTN group and 3460 (2895–4160) ng/ml in the TC group (HC vs. BTN: P < 0.01; BTN vs. TC: P < 0.05; HC vs. TC: P < 0.001) (Fig. 1C). Plasma levels of betaine were declined in the TC group (8240 (6800–9895) ng/ml, P < 0.001) and BTN group (8720 (7070–11250) ng/ml, P < 0.05) when compared to the HC group (10,350 (8362.5–12,325) ng/ml) (Fig. 1D).
Table 2 Distribution of plasma concentrations of TMAO and precursors in subgroups
Fig. 1
Plasma concentrations of TMAO, carnitine, choline and betaine in the HC group, BTN group and TC group. HC healthy controls, BTN benign thyroid nodules, TC thyroid cancer, TMAO microbiota-mediated trimethylamine N-oxide. *P < 0.05, **P < 0.01, ***P < 0.001
We further evaluated the value of plasma TMAO and precursors in stratifying TC to different lymph node metastasis status and clinical stages. From our results (Table 3), only plasma TMAO showed lower levels in TC with lymph node metastasis (101.5 (73.1–144.5) ng/ml) than those without lymph node metastasis (131 (84.8–201) ng/ml, P < 0.05). No other biomarkers exhibited statistically significant differences in lymph node metastasis and clinical staging of TC.
Table 3 Distribution of plasma concentrations of TMAO and precursors in different TC subgroups
Correlation between TMAO and precursors among different groups
Spearman correlation coefficient values of TMAO and precursors concentrations in the three groups (HC, BTN and TC) were calculated (Table 4). Weak to moderate positive correlation between choline and betaine levels was recorded in both females (R = 0.71, P < 0.001 in HC; R = 0.72, P < 0.001 in BTN, and R = 0.29, P < 0.001 in TC) and males (R = 0.72, P < 0.001 in HC; R = 0.55, P < 0.01 in BTN; R = 0.30, P < 0.01 in TC). Weak positive correlation was also observed between carnitine and betaine in females (R = 0.31, P < 0.01 in HC; R = 0.42, P < 0.01 in BTN, and R = 0.24, P < 0.01 in TC) and males (R = 0.34, P < 0.01 in HC; R = 0.26, P < 0.05 in TC). For carnitine and choline, weak positive correlation was only found in females with BTN (R = 0.49, P < 0.01) and with TC (R = 0.18, P < 0.05), and in healthy males (R = 0.36, P < 0.01) and males with TC (R = 0.26, P < 0.05). No significant or only very weak correlation was found between levels of TMAO and its precursors in the all subgroups.
Table 4 Correlation between TMAO and precursors among different groups
The relationship between TMAO and precursors and the diagnosis of PTC
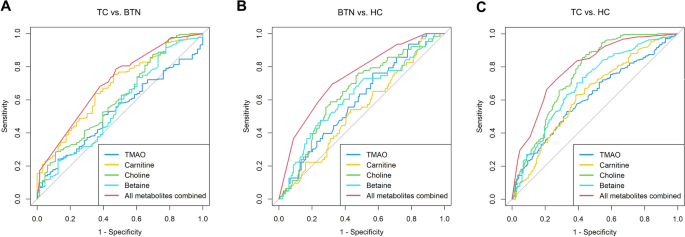
ROC curves were performed for the selected four metabolites. According to our results (Table 5 and Fig. 2), the AUCs of TMAO, carnitine, choline and betaine were 0.532, 0.676, 0.612 and 0.562 when discriminating between TC and BTN (Fig. 2A). Moreover, the AUCs of the four metabolites were 0.596, 0.538, 0.646 and 0.622 in differentiating BTN from HC (Fig. 2B). Furthermore, the AUCs of the four metabolites were 0.615, 0.631, 0.754 and 0.691 in the differential diagnosis of patients with TC and HC (Fig. 2C). Eventually, the multivariate logistic regression model was constructed based on the four metabolites. Combinations of these four metabolites achieved best performance in the differential diagnosis, with AUCs of 0.703, 0.741, 0.793 when discriminating between TC and BTN, BTN and HC, and TC and HC, respectively.
Table 5 Quantitative indexes of different models
Fig. 2
ROC curve analysis of the four single and combined metabolites. (A) TC vs. BTN, (B) BTN vs. HC, (C) TC vs. HC. TC thyroid cancer, BTN benign thyroid nodules, HC healthy controls, TMAO microbiota-mediated trimethylamine N-oxide
Discussion
Metabolic abnormalities are a common occurrence in diseases, leading to the dysfunction of metabolic pathways and abnormal accumulations or deficiencies of metabolites. Metabolite biosignatures from human biofluids, bridging genotype, environment, and phenotype, offer compelling biomarker candidates for clinical diagnosis, classification and prognosis prediction (Qiu et al. 2023). The identification of differential metabolites or metabolic pathway alterations can provide valuable insights into the pathophysiology of diseases, improve the precision and accuracy of patient diagnosis and risk prediction, and aid in the discovery of potential therapeutic targets (Alexander et al. 2023; Liu et al. 2022a; Perea-Gil et al. 2022). The absence of early biomarkers in TC may lead to inadequate diagnosis and unfavorable outcomes. Therefore, it is crucial to develop noninvasive diagnosis and monitoring methods that exhibit high levels of specificity and accuracy.
To the best of our knowledge, this is the first targeted study to investigate the association between plasma TMAO levels and TC risk in humans. We observed a significant increase in plasma TMAO levels in TC patients compared to HC, but no significant difference was observed when compared to BTN patients. In this study, alterations in blood TMAO levels were observed that were in accordance with a recent study conducted by Chinese researchers, who performed untargeted serum metabolomic profiling in 30 TC and 27 HC and found TMAO presented significant higher level in the TC (Zhou et al. 2017). TMAO is a compound produced by the gut microbiota from dietary nutrients. It has been increasingly recognized as a potential risk factor for various diseases, including cancer (Oellgaard et al. 2017). A meta-analysis has examined the associations between TMAO concentrations and cancer risk, and has demonstrated positive correlations between high TMAO levels and increased risks of colorectal cancer, prostate cancer, primary liver cancer, and pancreatic cancer (Li et al. 2022). Our study offers insights into the potential role of TMAO in TC, suggesting that it may serve as a biomarker for TC development. In our study, no significant or only very weak correlation was observed between TMAO and its precursors. Similarly, Zhou et al. supported our findings by demonstrating that there is no significant correlation between carnitine and TMAO (Zhou et al. 2017). These results suggest that the relationship between these compounds may not be as straightforward as previously thought, calling for further investigation into their interactions and potential impact on health.
Furthermore, we observed that only plasma TMAO, rather than its precursor, can distinguish TC patients with different lymph node metastasis statuses. Interestingly, TMAO levels were found to be lower in the lymph node metastasis group compared to the non-metastasis group. In alignment with our study, Seo et al. conducted research to explore the potential role of targeted metabolites, including choline, in predicting lymph node metastasis in patients with PTC. They discovered that none of the metabolites tested could effectively discriminate the presence of lymph node metastasis (Seo et al. 2018). Overall, TMAO can induce tumor growth by promoting cell proliferation, angiogenesis, inflammation or oxidative stress (Chan et al. 2019; Yang et al. 2022). However, recent work strongly supports the idea that TMAO can drive immune activation and promotes antitumor immunity (Mirji et al. 2022; Wang et al. 2022). The observed contrasting roles of TMAO in TC development and tumor progression may be attributed to the complex interactions between TMAO and the tumor microenvironment. In the initial stages of TC development, TMAO promotes tumor growth by modulating cell proliferation, invasion, and angiogenesis. However, as the disease progresses, tumors reduce TMAO levels through specific mechanisms, potentially to evade immune surveillance and facilitate metastasis. This dual effect highlights the complex and dynamic nature of TMAO in tumorigenesis and progression, and underscores the need for further research to fully understand its role in cancer biology.
According to the literatures, choline is related to TC, but the results are controversial. Ryoo et al. utilized nuclear magnetic resonance (NMR) spectroscopy to analyze the metabolome of percutaneous FNA specimens and they discovered that choline levels were elevated in PTC tissue compared to benign nodules (Ryoo et al. 2016). Deja et al. applied NMR-based metabolic profiling and found choline was higher in TC tissues compared to healthy thyroid tissue (Deja et al. 2013). While some studies reported that choline was decreased in TC tissue than benign nodules (Miccoli et al. 2012; Torregrossa et al. 2012). In addition to tissue samples, experts also conduct metabolic profiling research using peripheral blood samples. Consistent with our research, Zhao et al. revealed a decrease in serum choline levels in PTC patients compared to healthy controls (Zhao et al. 2015). However, Yekta et al. discovered that serum choline levels were higher in PTC patients than in healthy controls (Farrokhi Yekta et al. 2018). Carnitine is a derivative of amino acids and an essential nutrient involved in the metabolism of lipids in mammals and our result of low carnitine levels in the serum of TC patients is inconsistent with a previous study (Zhou et al. 2017). Nevertheless, higher blood levels of L-carnitine were demonstrated in male gastric cancer (Stonans et al. 2023). To date, no studies have reported changes of betaine levels in TC. Our study first demonstrated that plasma betaine levels were elevated in TC patients than BTN patients and HC. A recent meta-analysis of cohort studies has provided further support for our findings, indicating that higher levels of betaine may indeed reduce the risk of cancer incidence (Youn et al. 2019).
We believe that the credibility of our results is enhanced because we utilize targeted metabolic techniques and have a larger sample size compared to previous studies. In our study, we observed an overall increasing trend in TMAO levels and a corresponding decreased trend in the levels of its precursors across the three groups of HC, BTN, and TC. This phenomenon may be partly explained by the following possible reason: compared to healthy individuals, the abundance and diversity of intestinal microflora may exhibit significant changes (e.g. increased number and diversity of Clostridium) during the development of benign and malignant nodules, facilitating the conversion of precursors to TMAO (Backhed 2013; Koeth et al. 2013). As our study did not include data on gut microbiota, further research incorporating microbial analysis is warranted to validate our hypotheses. However, there are some limitations in this study. First, as a retrospective case-control design with relatively small sample size, we cannot conclude definitively that there is an association between plasma TMAO and its precursors concentration and TC. Second, dietary information that could influence plasma TMAO levels was not gathered (Hamaya et al. 2020). In the future, larger prospective studies with more comprehensive designs are needed to further investigate the relationship between TMAO and its precursors levels and TC risk. Further studies are also needed to fully understand the mechanisms underlying the effects of TMAO and its precursors on TC and to determine whether it can serve as a potential target for cancer prevention and treatment.
In conclusion, we found that plasma TMAO was elevated in TC patients, whereas its precursors (carnitine, choline and betaine) were decreased. TMAO and its precursors may serve as potential biomarkers for discriminating benign from malignant thyroid nodules.
Data availability
The datasets generated and/or analysed during the current study are available from the corresponding author on reasonable request.
References
- Alexander JL, Mullish BH, Danckert NP, Liu Z, Olbei ML, Saifuddin A, Torkizadeh M, Ibraheim H, Blanco JM, Roberts LA, Bewshea CM, Nice R, Lin S, Prabhudev H, Sands C, Horneffer-van der Sluis V, Lewis M, Sebastian S, Lees CW, Teare JP, Hart A, Goodhand JR, Kennedy NA, Korcsmaros T, Marchesi JR, Ahmad T, Powell N (2023) The gut microbiota and metabolome are associated with diminished COVID-19 vaccine-induced antibody responses in immunosuppressed inflammatory bowel disease patients. EBioMedicine 88:104430
CAS PubMed PubMed Central Google Scholar - Backhed F (2013) Meat-metabolizing bacteria in atherosclerosis. Nat Med 19:533–534
PubMed Google Scholar - Blum M (2000) Ultrasonography of the Thyroid. In: Feingold KR, Anawalt B, Blackman MR, Boyce A, Chrousos G, Corpas E, de Herder WW, Dhatariya K, Dungan K, Hofland J, Kalra S, Kaltsas G, Kapoor N, Koch C, Kopp P, Korbonits M, Kovacs CS, Kuohung W, Laferrere B, Levy M, McGee EA, McLachlan R, New M, Purnell J, Sahay R, Shah AS, Singer F, Sperling MA, Stratakis CA, Trence DL, Wilson DP (eds) Endotext. South Dartmouth (MA)
Google Scholar - Bogovic Crncic T, Ilic Tomas M, Girotto N, Grbac Ivankovic S (2020) Risk factors for thyroid cancer: what do we know so far? Acta Clin Croat 59:66–72
PubMed PubMed Central Google Scholar - Chan CWH, Law BMH, Waye MMY, Chan JYW, So WKW, Chow KM (2019) Trimethylamine-N-oxide as one hypothetical link for the relationship between intestinal microbiota and cancer—where we are and where shall we go? J Cancer 10:5874–5882
CAS PubMed PubMed Central Google Scholar - Dambrova M, Latkovskis G, Kuka J, Strele I, Konrade I, Grinberga S, Hartmane D, Pugovics O, Erglis A, Liepinsh E (2016) Diabetes is associated with higher trimethylamine N-oxide plasma levels. Exp Clin Endocrinol Diabetes 124:251–256
CAS PubMed Google Scholar - Deja S, Dawiskiba T, Balcerzak W, Orczyk-Pawilowicz M, Glod M, Pawelka D, Mlynarz P (2013) Follicular adenomas exhibit a unique metabolic profile. (1)H NMR studies of thyroid lesions. PLoS One 8:84637
Google Scholar - Fagin JA, Wells SA Jr (2016) Biologic and clinical perspectives on thyroid cancer. N Engl J Med 375:1054–1067
CAS PubMed PubMed Central Google Scholar - Farrokhi Yekta R, Rezaei Tavirani M, Arefi Oskouie A, Mohajeri-Tehrani MR, Soroush AR, Akbarzadeh Baghban A (2018) Serum-based metabolic alterations in patients with papillary thyroid carcinoma unveiled by non-targeted 1H-NMR metabolomics approach. Iran J Basic Med Sci 21:1140–1147
PubMed PubMed Central Google Scholar - Gatarek P, Kaluzna-Czaplinska J (2021) Trimethylamine N-oxide (TMAO) in human health. EXCLI J 20:301–319
PubMed PubMed Central Google Scholar - Gharib H, Papini E, Garber JR, Duick DS, Harrell RM, Hegedus L, Paschke R, Valcavi R, Vitti P, Nodules AAATFoT (2016) American association of clinical endocrinologists, American college of endocrinology, and associazione medici endocrinologi medical guidelines for clinical practice for the diagnosis and management of thyroid nodules–2016 update. Endocr Pract 22:622–639
PubMed Google Scholar - Ha EJ, Suh CH, Baek JH (2018) Complications following ultrasound-guided core needle biopsy of thyroid nodules: a systematic review and meta-analysis. Eur Radiol 28:3848–3860
PubMed Google Scholar - Hamaya R, Ivey KL, Lee DH, Wang M, Li J, Franke A, Sun Q, Rimm EB (2020) Association of diet with circulating trimethylamine-N-oxide concentration. Am J Clin Nutr 112:1448–1455
PubMed PubMed Central Google Scholar - Haugen BR, Alexander EK, Bible KC, Doherty GM, Mandel SJ, Nikiforov YE, Pacini F, Randolph GW, Sawka AM, Schlumberger M, Schuff KG, Sherman SI, Sosa JA, Steward DL, Tuttle RM, Wartofsky L (2016) 2015 American thyroid association management guidelines for adult patients with thyroid nodules and differentiated thyroid cancer: the American thyroid association guidelines task force on thyroid nodules and differentiated thyroid cancer. Thyroid 26:1–133
PubMed PubMed Central Google Scholar - Heianza Y, Ma W, Manson JE, Rexrode KM, Qi L (2017) Gut microbiota metabolites and risk of major adverse cardiovascular disease events and death: a systematic review and meta-analysis of prospective studies. J Am Heart Assoc. https://doi.org/10.1161/JAHA.116.004947
Article PubMed PubMed Central Google Scholar - Jalandra R, Dalal N, Yadav AK, Verma D, Sharma M, Singh R, Khosla A, Kumar A, Solanki PR (2021) Emerging role of trimethylamine-N-oxide (TMAO) in colorectal cancer. Appl Microbiol Biotechnol 105:7651–7660
CAS PubMed Google Scholar - Koeth RA, Wang Z, Levison BS, Buffa JA, Org E, Sheehy BT, Britt EB, Fu X, Wu Y, Li L, Smith JD, DiDonato JA, Chen J, Li H, Wu GD, Lewis JD, Warrier M, Brown JM, Krauss RM, Tang WH, Bushman FD, Lusis AJ, Hazen SL (2013) Intestinal microbiota metabolism of L-carnitine, a nutrient in red meat, promotes atherosclerosis. Nat Med 19:576–585
CAS PubMed PubMed Central Google Scholar - Li D, Lu Y, Yuan S, Cai X, He Y, Chen J, Wu Q, He D, Fang A, Bo Y, Song P, Bogaert D, Tsilidis K, Larsson SC, Yu H, Zhu H, Theodoratou E, Zhu Y, Li X (2022) Gut microbiota-derived metabolite trimethylamine-N-oxide and multiple health outcomes: an umbrella review and updated meta-analysis. Am J Clin Nutr 116:230–243
PubMed PubMed Central Google Scholar - Liu Q, Li B, Li Y, Wei Y, Huang B, Liang J, You Z, Li Y, Qian Q, Wang R, Zhang J, Chen R, Lyu Z, Chen Y, Shi M, Xiao X, Wang Q, Miao Q, Fang JY, Gershwin ME, Lian M, Ma X, Tang R (2022a) Altered faecal microbiome and metabolome in IgG4-related sclerosing cholangitis and primary sclerosing cholangitis. Gut 71:899–909
PubMed Google Scholar - Liu X, Wong CKH, Chan WWL, Au ICH, Tang EHM, Lang BHH (2022b) Survival after hemithyroidectomy versus total thyroidectomy in non-high-risk differentiated thyroid cancer: population-based analysis. BJS Open. https://doi.org/10.1093/bjsopen/zrac146
Article PubMed PubMed Central Google Scholar - Mayers JR, Vander Heiden MG (2015) Famine versus feast: understanding the metabolism of tumors in vivo. Trends Biochem Sci 40:130–140
CAS PubMed PubMed Central Google Scholar - Miccoli P, Torregrossa L, Shintu L, Magalhaes A, Chandran J, Tintaru A, Ugolini C, Minuto MN, Miccoli M, Basolo F, Caldarelli S (2012) Metabolomics approach to thyroid nodules: a high-resolution magic-angle spinning nuclear magnetic resonance-based study. Surgery 152:1118–1124
PubMed Google Scholar - Mirji G, Worth A, Bhat SA, El Sayed M, Kannan T, Goldman AR, Tang HY, Liu Q, Auslander N, Dang CV, Abdel-Mohsen M, Kossenkov A, Stanger BZ, Shinde RS (2022) The microbiome-derived metabolite TMAO drives immune activation and boosts responses to immune checkpoint blockade in pancreatic cancer. Sci Immunol 7:0704
Google Scholar - Mondul AM, Moore SC, Weinstein SJ, Karoly ED, Sampson JN, Albanes D (2015) Metabolomic analysis of prostate cancer risk in a prospective cohort: The alpha-tocolpherol, beta-carotene cancer prevention (ATBC) study. Int J Cancer 137:2124–2132
CAS PubMed PubMed Central Google Scholar - Oellgaard J, Winther SA, Hansen TS, Rossing P, von Scholten BJ (2017) Trimethylamine N-oxide (TMAO) as a new potential therapeutic target for insulin resistance and cancer. Curr Pharm Des 23:3699–3712
CAS PubMed Google Scholar - Perea-Gil I, Seeger T, Bruyneel AAN, Termglinchan V, Monte E, Lim EW, Vadgama N, Furihata T, Gavidia AA, Arthur Ataam J, Bharucha N, Martinez-Amador N, Ameen M, Nair P, Serrano R, Kaur B, Feyen DAM, Diecke S, Snyder MP, Metallo CM, Mercola M, Karakikes I (2022) Serine biosynthesis as a novel therapeutic target for dilated cardiomyopathy. Eur Heart J 43:3477–3489
CAS PubMed PubMed Central Google Scholar - Qiu S, Cai Y, Yao H, Lin C, Xie Y, Tang S, Zhang A (2023) Small molecule metabolites: discovery of biomarkers and therapeutic targets. Signal Transduct Target Ther 8:132
PubMed PubMed Central Google Scholar - Russ G, Bonnema SJ, Erdogan MF, Durante C, Ngu R, Leenhardt L (2017) European thyroid association guidelines for ultrasound malignancy risk stratification of thyroid nodules in adults: the EU-TIRADS. Eur Thyroid J 6:225–237
PubMed PubMed Central Google Scholar - Ryoo I, Kwon H, Kim SC, Jung SC, Yeom JA, Shin HS, Cho HR, Yun TJ, Choi SH, Sohn CH, Park S, Kim JH (2016) Metabolomic analysis of percutaneous fine-needle aspiration specimens of thyroid nodules: potential application for the preoperative diagnosis of thyroid cancer. Sci Rep 6:30075
CAS PubMed PubMed Central Google Scholar - Sakorafas GH, Mastoraki A, Lappas C, Safioleas M (2010) Small (<10 mm) thyroid nodules; how aggressively should they be managed? Onkologie 33:61–64
PubMed Google Scholar - Seiberling KA, Dutra JC, Gunn J (2008) Ultrasound-guided fine needle aspiration biopsy of thyroid nodules performed in the office. Laryngoscope 118:228–231
PubMed Google Scholar - Seo H, Na DG, Kim JH, Kim KW, Yoon JW (2015) Ultrasound-based risk stratification for malignancy in thyroid nodules: a four-tier categorization system. Eur Radiol 25:2153–2162
PubMed Google Scholar - Seo JW, Han K, Lee J, Kim EK, Moon HJ, Yoon JH, Park VY, Baek HM, Kwak JY (2018) Application of metabolomics in prediction of lymph node metastasis in papillary thyroid carcinoma. PLoS One 13:e0193883
PubMed PubMed Central Google Scholar - Siegel RL, Miller KD, Fuchs HE, Jemal A (2022) Cancer statistics, 2022. CA Cancer J Clin 72:7–33
PubMed Google Scholar - Stonans I, Kuzmina J, Polaka I, Grinberga S, Sevostjanovs E, Liepins E, Aleksandravica I, Santare D, Kirsners A, Skapars R, Pcolkins A, Tolmanis I, Sivins A, Leja M, Dambrova M (2023) The association of circulating L-carnitine, gamma-butyrobetaine and trimethylamine N-oxide levels with gastric cancer. Diagnostics (basel) 13:1341
CAS PubMed Google Scholar - Tang WH, Wang Z, Kennedy DJ, Wu Y, Buffa JA, Agatisa-Boyle B, Li XS, Levison BS, Hazen SL (2015) Gut microbiota-dependent trimethylamine N-oxide (TMAO) pathway contributes to both development of renal insufficiency and mortality risk in chronic kidney disease. Circ Res 116:448–455
CAS PubMed Google Scholar - Torregrossa L, Shintu L, Nambiath Chandran J, Tintaru A, Ugolini C, Magalhaes A, Basolo F, Miccoli P, Caldarelli S (2012) Toward the reliable diagnosis of indeterminate thyroid lesions: a HRMAS NMR-based metabolomics case of study. J Proteome Res 11:3317–3325
CAS PubMed Google Scholar - Tu R, Xia J (2024) Stroke and vascular cognitive impairment: the role of intestinal microbiota metabolite TMAO. CNS Neurol Disord Drug Targets 23:102–121
CAS PubMed Google Scholar - Wang Z, Klipfell E, Bennett BJ, Koeth R, Levison BS, Dugar B, Feldstein AE, Britt EB, Fu X, Chung YM, Wu Y, Schauer P, Smith JD, Allayee H, Tang WH, DiDonato JA, Lusis AJ, Hazen SL (2011) Gut flora metabolism of phosphatidylcholine promotes cardiovascular disease. Nature 472:57–63
CAS PubMed PubMed Central Google Scholar - Wang H, Rong X, Zhao G, Zhou Y, Xiao Y, Ma D, Jin X, Wu Y, Yan Y, Yang H, Zhou Y, Qian M, Niu C, Hu X, Li DQ, Liu Q, Wen Y, Jiang YZ, Zhao C, Shao ZM (2022) The microbial metabolite trimethylamine N-oxide promotes antitumor immunity in triple-negative breast cancer. Cell Metab 34(581–594):e588
Google Scholar - Yang S, Dai H, Lu Y, Li R, Gao C, Pan S (2022) Trimethylamine N-oxide promotes cell proliferation and angiogenesis in colorectal cancer. J Immunol Res 2022:7043856
PubMed PubMed Central Google Scholar - Youn J, Cho E, Lee JE (2019) Association of choline and betaine levels with cancer incidence and survival: a meta-analysis. Clin Nutr 38:100–109
CAS PubMed Google Scholar - Zeisel SH, da Costa KA (2009) Choline: an essential nutrient for public health. Nutr Rev 67:615–623
PubMed Google Scholar - Zhao WX, Wang B, Zhang LY, Yan SY, Yang YH (2015) Analysis on the metabolite composition of serum samples from patients with papillary thyroid carcinoma using nuclear magnetic resonance. Int J Clin Exp Med 8:18013–18022
CAS PubMed PubMed Central Google Scholar - Zhou J, Li Y, Chen X, Zhong L, Yin Y (2017) Development of data-independent acquisition workflows for metabolomic analysis on a quadrupole-orbitrap platform. Talanta 164:128–136
CAS PubMed Google Scholar
Acknowledgements
We thank all the patients for their invaluable participation and the staff of Nanjing First Hospital for their contribution to the study.
Funding
This work was supported by National Natural Science Foundation of China (No. 82003532, 82272038), The China Postdoctoral Science Foundation (No. 2023M741765), Medical Science and Technology Program of Nanjing (No. JQX22006), Nanjing Health Science and Technology Development Project (No. YKK22114), Jiangsu Provincial Medical Key Discipline Cultivation Unit (No. JSDW202247) and Jiangsu Province Research Hospital Cultivation Unit (No. YJXYYJSDW1).
Author information
Author notes
- Liang Shi, Muhong Guo and Cuixiao Shi contributed equally to this work and share first authorship.
Authors and Affiliations
- Department of Nuclear Medicine, Nanjing First Hospital, Nanjing Medical University, Nanjing, 210006, China
Liang Shi, Cuixiao Shi, Chuan Zhang, Jingjing Fu, Yudan Ni, Feng Wang, Xue Xue & Fei Yu - Department of General Surgery, Nanjing First Hospital, Nanjing Medical University, Nanjing, 210006, China
Muhong Guo - Department of Health Management Center, Nanjing First Hospital, Nanjing Medical University, Nanjing, 210006, China
Gu Gao - Department of Endocrinology, Nanjing First Hospital, Nanjing, 210006, China
Xianghong Xu
Authors
- Liang Shi
- Muhong Guo
- Cuixiao Shi
- Gu Gao
- Xianghong Xu
- Chuan Zhang
- Jingjing Fu
- Yudan Ni
- Feng Wang
- Xue Xue
- Fei Yu
Contributions
FY, XX and FW conceived, designed the study. MG, CS, GG and XX collected the samples and clinical data. XX performed the experiment. FY conducted all statistical analyses. LS and FY drafted the manuscript; CZ, JF, YN played an important role in interpreting the results. All authors revised and approved the final version of the manuscript.
Corresponding authors
Correspondence toFeng Wang, Xue Xue or Fei Yu.
Ethics declarations
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Shi, L., Guo, M., Shi, C. et al. Distinguishing benign and malignant thyroid nodules using plasma trimethylamine N-oxide, carnitine, choline and betaine.J Cancer Res Clin Oncol 150, 142 (2024). https://doi.org/10.1007/s00432-024-05666-w
- Received: 12 January 2024
- Accepted: 23 February 2024
- Published: 20 March 2024
- Version of record: 20 March 2024
- DOI: https://doi.org/10.1007/s00432-024-05666-w