Involvement of the Sigma1 Receptor in Cocaine-induced Conditioned Place Preference: Possible Dependence on Dopamine Uptake Blockade (original) (raw)
Main
Cocaine is a highly addictive substance that is abused by humans worldwide (Warner 1993; Higgins 1997). Cocaine also acts as a potent reinforcer in laboratory animals (Pickens and Thompson 1968; Koob 1992; Stolerman 1992; Woolverton and Johnson 1992). While cocaine inhibits the transport of serotonin (5-HT), norepinephrine and dopamine (DA), it is widely accepted that the addictive and reinforcing actions of cocaine are dependent on the drug's ability to block the dopamine transporter (DAT), thereby increasing dopamine (DA) neurotransmission (Kuhar 1992; Ritz et al. 1987; Parsons et al. 1998; Woolverton 1992). Because of the link between cocaine's addictive liability and the DA reward/reinforcement circuitry of the forebrain (Koob 1992), treatment strategies against cocaine abuse have logically targeted the DA system. However, it has been described that cocaine also interacts with the sigma1 receptor at a similar dose range as observed for the DAT and that the sigma1 receptor is implicated in some of cocaine's effects such as locomotor activation, sensitization, convulsion and lethality (Koe 1976; Reith et al. 1986; Menkel et al. 1991; Ujike et al. 1992; Ritz and George 1993). Therefore, the diverse pharmacological profile of cocaine may contribute to, or modulate, its behavioral effects through a complex mechanism of action affecting the different monoaminergic transporters as well as other targets. Consistent with this hypothesis is the study by Sora et al. (2000) demonstrating that it is possible to establish cocaine-induced place preference in DA or 5-HT transporter knockout mice, leading to the hypothesis that other targets might be involved in producing the rewarding effects of cocaine. An alternative therapeutic approach to treating cocaine addiction, therefore, would be to target the sigma1 receptor, which has recently been demonstrated to be involved in cocaine's rewarding effects measured using a conditioned place preference (CPP) paradigm in mice (Romieu et al. 2000). The sigma1 receptor, localized intracellularly within the neurons, is a 223 amino acid protein, cloned in several animal species and humans (Hanner et al. 1996; Kekuda et al. 1996; Seth et al. 1997, 1998; Pan et al. 1998). The receptor appeared devoid of analogy with any other known mammalian protein and its endogenous ligand has still to be established, but its regional and subcellular distributions have recently been determined by immunohistochemical labeling in rodents (Alonso et al. 2000; Phan et al. 2000). In mice, moderate to intense labeling were observed in most of the dopaminergic structures, including the caudate putamen, nucleus accumbens, amygdala, septum and superficial layers of the cortex. The biological relevance of this receptor is supported by the correlation observed between functional and behavioral effects of selective drugs and affinities at the sigma1 binding sites (for review, see Maurice et al. 1999). The sigma1 receptor mediates a potent modulation of several neurotransmitter systems by affecting intracellular second messengers systems, particularly Ca2+ mobilization (Morin-Surun et al. 1999; Hayashi et al. 2000).
Once cocaine's relative potency to inhibit the binding of sigma1 receptor radiotracers had been established, with a Ki about 1.7 μM (Ritz and George 1993), the involvement of the sigma1 receptor as a possible target of cocaine was suggested. At the behavioral level, several sigma1 receptor antagonists, such as haloperidol, α-(4-fluorophenyl)-4-(5-fluoro-2-pyrimidinyl)-1-piperazine (BMY-14,802), rimcazole, or cis-3-(hexahydroazepin-1-yl)1-(3-chloro-4-cyclohexylphenyl)propene-1 (SR-31742A), attenuated the stimulant effects of cocaine or the development of cocaine-induced locomotor sensitization (Menkel et al. 1991; Ujike et al. 1996). Other selective sigma1 receptor antagonists, N-[2-(3,4-dichlorophenyl)ethyl]-N-methyl-2-(1-pyrrolidinyl)ethylamine (BD1008), 1-[2-(3,4-dichloro-phenyl)ethyl]-4-methylpiperazine (BD1063) and N-[2-(3,4-dichlorophenyl)ethyl]-N-methyl-2-(dimethylamino) ethylamine (BD1047), as well as an antisense oligodeoxynucleotide directed against the sigma1 receptor, attenuated the cocaine-induced convulsions and lethality (McCracken et al. 1999a, b; Matsumoto et al. 2001). Although these data suggest that activation of the sigma1 receptor is involved in several cocaine-induced effects, it remains unclear whether cocaine directly interacts with the receptor at the behaviorally relevant doses. The preliminary report by Romieu et al. (2000) examined the involvement of the sigma1 receptor in cocaine-induced CPP, contributing to the hypothesis that the sigma1 receptor is a critical factor in the acquisition of cocaine-induced CPP. Selective sigma1 receptor antagonists or an antisense oligodeoxynucleotide attenuated or blocked the acquisition of cocaine-induced CPP in the C57BL/6 mouse.
The present study sought to further investigate whether the sigma1 receptor is equally involved in acquisition and expression of the cocaine-induced CPP and whether the sigma1 receptor is sufficient to directly induce CPP or if its activation is subsequent to the inhibition of the DAT. In addition, the effect of the repeated cocaine treatment, which was sufficient to produce locomotor sensitization during CPP conditioning was investigated on the expression of the sigma1 receptor mRNA in different brain dopaminergic structures, using comparative reverse transcriptase-polymerase chain reaction.
METHODS
Animals
Male C57BL/6 mice, one month old and weighing 24–26 g, were purchased from Iffa-Credo (Saint-Germain-sur-l'Arbresle, France). Animals were housed in groups with free access to food and water, except during behavioral experiments. They were kept in a temperature and humidity controlled animal facility on a 12h/12h light:dark cycle (light off at 8 P.M.). Experiments were carried out between 10:00 A.M. and 6:00 P.M., in a soundproof and air-regulated experimental room, to which mice were habituated at least 30 min before each experiment. All animal procedures were conducted in strict adherence to the European Communities Council Directive of 24 November 1986 (86-609/EEC) and the Decree of 20 October 1987 (87-848/EEC).
Drugs
Cocaine hydrochloride was obtained from Cooper (Coopération Pharmaceutique Française, Marseille, France). N-[1-(2-benzo(b)thiophenyl) cyclohexyl]piperidine (BTCP) was kindly provided by Dr. Jean-Marc Kamenka (CNRS, Montpellier, France), 2-(4-morpholinoethyl-1-phenylcyclohexane-1-carboxylate hydrochloride (PRE-084) by Dr. Tsung-Ping Su (NIDA/NIH, Baltimore, MD, USA), (+)-N-cyclopropylmethyl-N-methyl-1,4-diphenyl-1-ethyl-but-3-en-1-ylamine hydrochloride (igmesine) by Dr. François J. Roman (Pfizer GRD, Fresnes, France), N,N-dipropyl-2-(4-methoxy-3-(2-phenylethoxy)phenyl) ethylamine (NE-100) by Dr. Yasuko Karasawa (Taisho Pharmaceutical Co, Tokyo, Japan) and N-[2-(3,4-dichlorophenyl)ethyl]-N-methyl-2-(dimethylamino)ethylamine (BD1047) by Dr. Wayne D. Bowen (NIDDK/NIH, Bethesda, MD). Dizocilpine was from Research Biochemicals Inc. (Natick, MA). Cocaine was dissolved in physiological saline and other drugs were dissolved in distilled water. They were injected intraperitoneally (i.p.) in a volume of 100 μl per 20 g of body weight.
Conditioned Place Preference Procedure
The apparatus consisted of a Plexiglas box divided into two compartments of equal size (15 × 15 × 35 cm high), the first one with black walls and floor, the second one with white walls and floor. Each compartment presented different floor textures, smooth for the black one and covered by a wire mesh grid for the white. A 60 W lamp lit the white compartment during all the experiments. Sliding doors separated the two compartments.
The procedure consisted of three different phases: preconditioning (day 1), conditioning (days 2 to 5) and postconditioning (day 6). For the preconditioning phase, each mouse was placed in the white compartment and after 5 s, the sliding doors were raised. The animal was allowed to freely explore the apparatus for 10 min. The preconditioning phase was repeated after 6 h. The exploration was videotaped and the amount of time spent in each compartment was determined in order to assess the unconditioned preference. Animals showing a strong unconditioned preference (> 570 s), i.e. less than 5%, were discarded. Place preference conditioning was conducted using an unbiased procedure. In each experimental group, half of the animals received either cocaine or other test drugs (BTCP, igmesine or PRE-084) in the spontaneously preferred compartment and the other half in the non-preferred compartment. Immediately after the drug injection, each mouse was confined to the drug-paired compartment for 30 min. In co-administration experiments, mice received selective sigma1 receptor antagonists, NE-100 or BD1047, 10 min before the test drug. After a 6-h washout period, they were administered the vehicle solutions and confined to the other compartment for 30 min. The postconditioning test was performed the last day. Each mouse was again placed in the white compartment and after 5 s, the doors were raised. The animal was allowed to freely explore the apparatus for 10 min. The exploration was videotaped and the amount of time spent in each compartment was determined. In CPP expression experiments, animals received only cocaine during the conditioning period, the sigma1 receptor antagonists being injected only once, 30 min before the post-conditioning session.
Elevated Plus Maze Procedure
The elevated plus-maze was used to evaluate spatial long-term memory, involving the previously described procedure (Itoh et al. 1990, 1991; Maurice et al. 1994). The apparatus consisted of two open arms (23.5 × 8 cm), painted white, and two enclosed arms (23.5 × 8 × 20 cm high), painted black. The arms extended from a central platform (8 × 8 cm), and the maze was elevated to a height of 50 cm above the floor. A fine white line was drawn in the middle of the floor of the enclosed arms. On the first day, 20 min after the drug injection, each mouse was placed at the end of an open arm, facing away from the central platform. The time taken by the mouse to move into one of the enclosed arms, its two front paws crossing the white line, was recorded as the transfer latency for the first day. If the animal did not enter an enclosed arm within 90 s, it was gently pushed on the back into one enclosed arm and the transfer latency was assigned as 90 s. The mouse was allowed to move freely for at least 10 s so that it could remember the presence of the enclosed arms, and it was then gently taken out and returned to its home cage. Retention was examined 24 h later, without drug injection. Each mouse was again placed into the maze and the transfer latency was recorded, up to 90 s.
Locomotor Sensitization
The locomotor sensitization was examined in animals injected with cocaine (0, 10, 20 mg/kg i.p.) once daily for four days, corresponding to the conditioning procedure but in parallel experiments. Mice were placed in a 25 × 40 × 15 cm cage for 40 min. Cocaine or saline was then injected and the animals were again placed in the cage for 40 min. The locomotion was recorded using a CCD camera connected to a microcomputer and analyzed using the Videotrack© II (version 2.65) software (Viewpoint, Champagne-au-Mont-d'Or, France). Locomotion was measured in terms of distance traveled every 10 min, and summed over 30 min, between 10 and 40 min.
Relative Quantification of Sigma1 Receptor mRNA by Reverse Transcriptase-Polymerase Chain Reaction
Immediately after the post-conditioning session, animals treated with 0, 10 or 20 mg/kg cocaine were anesthetized with pentobarbital (2% i.p.). The brains were dissected and the caudate putamen, nucleus accumbens, prefrontal cortex and cerebellum were immediately frozen on dry ice for further RNA preparation. Total RNA was isolated from structures previously frozen, according to the method of Chomczynski and Sacchi (1987). RNA concentration and integrity were estimated using a spectrophotometer and by denaturant gel electrophoresis, respectively. Two micrograms of total RNA was denatured for 10 min before being reverse-transcribed. The cDNA synthesis was carried out at 42°C for 1 h in a reverse transcriptase buffer, containing 1 unit of RNA-guard (Amersham Pharmacia Biotech), 500 mM of each dNTPs, 800 pmol/mg of random primers, and 400 units of Moloney murine leukemia reverse transcriptase in a total volume of 30 μl (Gibco BRL, Life Technologies, Cergy-Pontoise, France).
For polymerase chain reaction assays, sigma1 receptor and β-actin primer sequences were as follows: β-actin: 5′-TGTTACCAACTGGGACGACA (forward) and 3′-TCTCAGCTGTGGTGGTGAAG (reverse), and sigma1 receptor 5′-GCTGCAGTGGGTATTTGTGA (forward) and 3′-ACTTTTCGTGGTGCCCTCTT (reverse). They were checked in the GeneBank database (Bainbridge Island, WA) to avoid inadvertent sequence homologies. In addition, sigma1 receptor primers were chosen to span an intron and thus produce two different size products from DNA and cDNA respectively, so that a possible DNA contamination of the samples was then detectable. The number of polymerase chain reaction cycles, the annealing temperature and the initial cDNA amounts were chosen so that experiments were run in a linear working range for all the amplified polymerase chain reaction products. Polymerase chain reaction amplification was performed from 1 μl of reverse transcriptase products with a final concentration of 1x polymerase chain reaction buffer, 1.5 mM MgCl2, 0.2 mM dNTPs, 0.5 μM of each sigma1 primers, 0.05 μM of each β-actin primers, 2.5 unit of Taq DNA polymerase (Gibco), in a total volume of 50 μl. Co-amplification was performed in a Trio-thermoblock thermal cycler (Biometra, Göttingen, Germany) and began with denaturation at 94°C for 5 min, followed by 30 cycles involving denaturation at 94°C for 1 min, annealing at 58°C for 1 min and extension at 72°C for 1 min 30 s, and a final extension step at 72°C for 5 min. 10 μl of each polymerase chain reaction product were run in a 2% agarose gel electrophoresis. Signal intensity of each polymerase chain reaction product band was determined, using Gel Doc 1000 apparatus and the Molecular Analyst software (Bio-Rad, Hercules, CA, USA) and corrected for local background and normalized against β-actin mRNA levels.
Statistical Analyses
The conditioned score (mean ± S.E.M.) was calculated as the difference of time spent in the drug-paired compartment between the post- and preconditioning phases. Results were analyzed using a parametric analysis of variance (ANOVA), followed by a Newman-Keuls’ test. Elevated plus maze data (mean ± S.E.M.) did not show a normal distribution, since upper cut-off times were set (non-parametric data). They were analyzed by the Mann-Whitney's non-parametric test or the Kruskal-Wallis analysis of variances by ranks. Difference in transfer latencies between days one and two were analyzed using the Wilcoxon's paired non-parametric test. Polymerase chain reaction data are represented as the mean of the normalized values from experiment performed in triplicate and expressed as means ± S.E.M. Statistical analysis of mRNA levels was made using the Mann-Whitney's non-parametric test. All experiments included negative controls, where template RNA, reverse transcriptase or reverse transcriptase products were omitted, resulting in no detectable polymerase chain reaction product. The criterion for statistical significance were p < .05 and p < .01.
RESULTS
Effects of Selective Sigma1 Receptor Antagonists on the Acquisition and Expression of Cocaine-Induced CPP
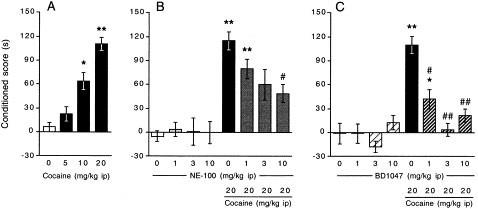
The repetitive administration of cocaine (5–20 mg/kg i.p.) during four days resulted in the development of a dose-dependent place preference in C57BL/6 mice (F3,69 = 10.47, p < .0001, Figure 1 , panel A). At the highest dose tested, 20 mg/kg, the augmentation of time spent in the drug-paired compartment was about +120 s (Figure 1, panel A). The effect of two selective sigma1 receptor antagonists, NE-100 and BD1047, were examined on the acquisition of cocaine-induced CPP. By itself, the pre-administration of NE-100 (1–10 mg/kg i.p.) did not affect the behavior of mice (Figure 1, panel B). When administered 10 min before cocaine, however, it resulted in a dose-dependent decrease of CPP (F7,116 = 3.08, p < .01, Figure 1, panel B). The pre-administration of BD1047 (1–10 mg/kg i.p.) also failed to induce CPP or aversion by itself (Figure 1, panel C). However, the pre-administration of the antagonist before cocaine resulted in a blockade of CPP, significant at 1 mg/kg and complete at 3 mg/kg (F7,118 = 4.01, p < .001, Figure 1, panel C).
Figure 1
Effect of selective sigma1 receptor antagonists on the acquisition of cocaine-induced CPP: dose-response effect of cocaine (panel A), and co-administration of NE-100 (panel B), or BD1047 (panel C). The sigma1 receptor antagonists were administered 10 min before cocaine, which was given immediately before placement in the compartment during the conditioning test. The conditioned score represents the difference in time spent in the drug-paired compartment between the post- and pre-conditioning sessions. Results are expressed as mean ± S.E.M. of n = 12–25 mice per group in panel A), n = 10–18 in panel B and C. *p < .05, **p < .01 vs. the (vehicle+vehicle)-treated group, # p < .05, ## p < .01 vs. the (cocaine+vehicle)-treated group.
In order to determine whether selective sigma1 receptor antagonists could block not only the acquisition but also the expression of cocaine-induced CPP, the sigma1 receptor antagonists were administered during the post-conditioning test. Animals received cocaine during the four days of conditioning and NE-100 or BD1047 were administered once before the post-conditioning session. Each drug significantly attenuated, but did not block, the expression of cocaine-induced CPP (F3,45 = 6.00, p < .01, Figure 2 , panel A for NE-100; F4,59 = 3.53, p < .05, Figure 2, panel B for BD1047).
Figure 2
Effect of selective sigma1 receptor antagonists on the expression of the cocaine-induced CPP: NE-100 (panel A), BD1047 (panel B). The sigma1 receptor antagonists were administered before the post-conditioning test. Results are expressed as mean ± S.E.M. of n = 11–12 mice per group in panel A, and n = 12 in panel B. *p < .05, **p < .01 vs. the (vehicle+vehicle)-treated group, # p < .05 vs. the (cocaine+vehicle)-treated group.
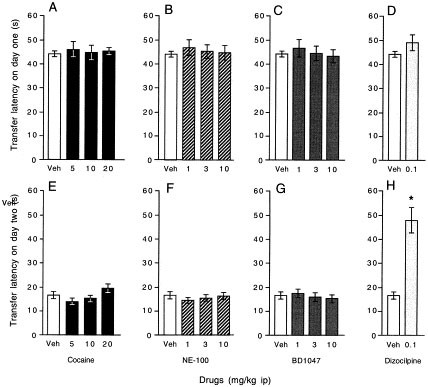
The possible involvement of modulation of learning capacities induced by the drug treatments and interfering with the appetitive behaviors was examined in a parallel experiment. The elevated plus-maze involving a protocol allowing a reliable measure of memory was used, with the benefit that similar instinctive cues as place preference conditioning were involved (Itoh et al. 1990, 1991). On day 1, animals were trained to learn the maze. The latency to move from one open arm to the closed arms was about 44 ± 1 s for vehicle-treated animals (Figure 3 , panel A). On day 2, a significant decrease of the transfer latency was measured, 17 ± 1 s (p < .01, Wilcoxon's paired non-parametric test). This 27 s decrease is regarded as an index of long-term memory (Itoh et al. 1990, 1991; Maurice et al. 1994). None of the drug treatment significantly affected the transfer latency during the first session: KW = 1.07, _p_ > .05 for cocaine (Figure 3, panel A); KW = 0.41, p > .05 for NE-100 (Figure 3, panel B) KW = 2.35, p > .05 for BD1047 (Figure 3, panel C). In addition, none of the treatments affected the learning ability measured during the second session: KW = 1.97, p > .05 for cocaine (Figure 3, panel E); KW = 0.27, p > .05 for NE-100 (Figure 3, panel F) KW = 0.55, p > .05 for BD1047 (Figure 3, panel G). Dizocilpine, the non-competitive N-methyl-D-aspartate (NMDA) receptor antagonist binding to the phencyclidine (PCP) high affinity site of the receptor, was used as a positive control for learning deficits. When administered at 0.1 mg/kg i.p., dizocilpine failed to affect the transfer latency on day 1 (p > .05, Mann-Whitney's test, Figure 3, panel D). On day 2, animals failed to remember the maze and showed no decrease of the transfer latency (p < .05 vs. vehicle-treated group).
Figure 3
Effect of cocaine (panels A, E) or of the selective sigma1 receptor antagonists NE-100 (panels B, F) and BD1047 (panels C, G) in the elevated plus-maze used following a protocol assessing long-term memory. Dizocilpine (panels D, H) was used as a control to show the extent of learning deficits. Results are presented as transfer latencies, mean ± S.E.M., from the open arm to the closed arm during the sessions on day one (panels A–D) and on day two (panels E–H). Drugs were administered i.p. 20 min before the session on day one. Number of animals per group: n = 9–10 in (panels A, E), 10 in (panels B, F), 8–10 in (panels C, G) and 9–10 in (panels D, H). *p < .05 vs. vehicle-treated group; Mann-Whitney's test.
Lack of Direct Involvement of the Sigma1 Receptor in CPP Acquisition
Whether the direct activation of the sigma1 receptor could be sufficient to develop CPP was examined using two selective sigma1 receptor agonists, igmesine and PRE-084. Both drugs, administered in the 10–60 mg/kg i.p. dose-range, failed to induce CPP (F6,75 = 0.17, p > .05, Figure 4 ).
Figure 4
Effect of the selective sigma1 receptor agonists, igmesine and PRE-084, on CPP. The sigma1 receptor agonists were administered immediately before placement in the compartment during the conditioning test. Results are expressed as mean ± S.E.M. of n = 10–14 mice per group.
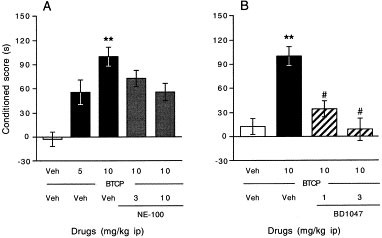
In order to check whether inhibition of the DAT is critical for the activation the sigma1 receptor, the potent DAT inhibitor BTCP was used, because it presents a poor affinity for the sigma1 receptor and a high selectivity for the DAT. After four days of conditioning with BTCP (10 mg/kg i.p.), mice developed a highly significant CPP, as observed with cocaine 20 mg/kg (Figure 5 , panel A). Pre-administration of NE-100 led to a dose-dependent attenuation of the acquisition of CPP (F4,62 = 3.32, p < .05, Figure 5, panel A). Pre-administration of BD1047 led to a blockade of the BTCP-induced CPP (F3,54 = 3.51, p < .05, Figure 5, panel B).
Figure 5
Effect of the selective sigma1 receptor antagonists on BTCP-induced CPP: NE-100 (panel A), BD1047 (panel B). The sigma1 receptor antagonists were administered 10 min before BTCP, which was given immediately before the test. Results are expressed as mean ± S.E.M. of n = 12–15 mice per group in panel A, and n = 12–16 in panel B. **p < .01 vs. the (vehicle+vehicle)-treated group, # p < .05 vs. the (BTCP+vehicle)-treated group.
Effect of the Repeated Treatment with Cocaine on Locomotor Sensitization
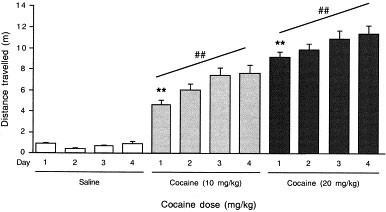
To test whether the repeated treatment with cocaine (10, 20 mg/kg i.p.) during the conditioning period already led to a significant sensitization, the effect of the repeated treatment with cocaine was examined on locomotor activity. A single cocaine administration led to significant locomotor increases, at 10 and at 20 mg/kg, as compared with the saline-treated group (day 1; Figure 6 ). The repeated daily injection of the same dose of cocaine over the four days significantly enhanced the resulting hyperlocomotion (repeated measures ANOVA: F3,27 = 6.11, p < .01; test for linear trend: F = 16.96, _p_ < .001 at 10 mg/kg; repeated measures ANOVA: F3,31 = 5.43, _p_ < .01; test for linear trend: F = 10.63, _p_ < .01 at 20 mg/kg; F3,31 = 0.66, _p_ > .05, for controls; Figure 6), indicating that behavioral sensitization took place during the conditioning procedure.
Figure 6
Effect of the conditioning to cocaine on locomotor sensitization. Animals received either saline solution or cocaine (10, 20 mg/kg i.p.) during 4 days and locomotor activity was recorded during the last 30 min of a 40-min session. **p < .01 vs. the saline-treated group on the same day, ## p < .01 for linear trend (Dunnett's test).
Effect of the Cocaine Treatment on the Sigma1 Receptor mRNA Expression
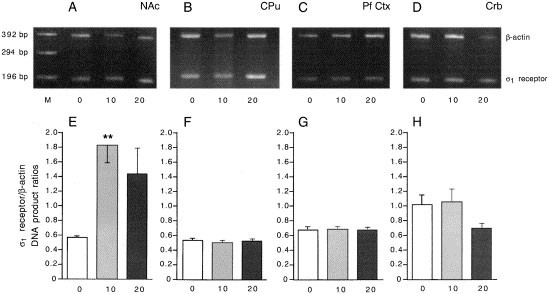
On day 5, immediately after the post-conditioning session and 24 h after the last cocaine injection, animals were sacrificed and structures of interest were dissected out. The caudate putamen was taken as the target area for A10 dopaminergic neurons, the nucleus accumbens and prefrontal cortex as the target areas for A9 dopaminergic neurons, and the cerebellum as a control non-dopaminergic structure. Comparative reverse transcriptase-polymerase chain reaction was performed, and sigma1 receptor and β-actin-related products were measured (Figure 7 , panels A–D). The quantification of sigma1 receptor product/β-actin product ratios indicated that significant augmentations of the sigma1 receptor mRNA occurred after cocaine treatments in the nucleus accumbens (F2,21 = 5.07, p < .05; Figure 7, panel E), but not in the caudate putamen (F2,17 = 0.04, _p_ > .05; Figure 7, panel F), prefrontal cortex (F2,19 = 0.01, p > .05; Figure 7, panel G) or cerebellum (F2,17 = 0.60, p > .05; Figure 7, panel H).
Figure 7
Effect of the conditioning to cocaine on the levels of sigma1 receptor mRNA in different brain regions. (panels A–D) representative RT-PCR blots; (panels E–H) Quantification of the sigma1 receptor/β-actin products ratios. (A–D) Animals received either saline solution or cocaine (10, 20 mg/kg i.p.) during the four days of CPP conditioning. They were sacrificed after the post-conditioning test. The nucleus accumbens (NAc), caudate putamen (CPu), prefrontal cortex (Pf Ctx) and cerebellum (Crb) were dissected out. RNA was extracted and sigma1 receptor and β-actin-related DNA products were amplified using reverse transcriptase-polymerase chain reaction. M: PCR markers lane. Quantifications of the sigma1 receptor/β-actin product ratios are shown in (E–H) for each treatment group. n = 4–8 samples per group. **p < .01 vs. the saline-treated group (Dunnett's test).
DISCUSSION
The present study confirmed the involvement of the sigma1 receptor in the development of cocaine-induced rewarding properties, measured using the CPP procedure in C57Bl/6 mice. Two highly selective sigma1 receptor antagonists, NE-100 and BD1047, were used. Both NE-100 and BD1047 attenuated or blocked not only the acquisition of cocaine-induced CPP when administered before cocaine during the conditioning phase, but also its expression when they were administered alone before the post-conditioning session. These observations thus suggest that activation of the sigma1 receptor is induced following repeated cocaine administration during CPP conditioning. Sigma1 receptor activation seems necessary for the establishment of CPP and, even in the absence of cocaine, when the acquired CPP is expressed in the adequate environment.
Presently, there is no evidence permitting speculation on: (1) the mechanism by which cocaine could directly or indirectly activate the sigma1 receptor; and (2) the similarities or differences of the mechanisms involved in these two modes of sigma1 receptor activation. It may however, involve a complex mechanism of action. Indeed, selective sigma1 receptor agonists, such as igmesine or PRE-084, did not by themselves induce CPP. In addition, the selective DAT inhibitor BTCP was shown to induce CPP at a dose, 10 mg/kg, previously known to produce the same behavioral and neurochemical effects as cocaine at 20 mg/kg (Martin-Fardon et al. 1996; 2000). Cocaine possesses a submicromolar affinity for the DA transporter in the range of 0.2–0.9 μM (Hyttel 1978; Reith et al. 1986; Calligaro and Eldefrawi 1988; Debler et al. 1988) and a micromolar affinity for the sigma1 receptor, in the range of 1.7–6.7 μM (Sharkley et al. 1988; Ritz and George 1993). It is unclear whether the sigma1 receptor activation by cocaine involves a direct interaction of the drug with the receptor, or indirect mechanisms through its primary effect on the DAT. BTCP is a phencyclidine (PCP) derivative, acting as a highly potent DAT inhibitor with an affinity of 14 nM (Vignon et al. 1988). BTCP is however considerably more selective than its parent molecule for the other main PCP targets in the brain. Its affinity for the sigma1 receptor is 3.5 ± 0.4 μM (n = 5), as measured on rat forebrain membranes using [3H](+)-SKF-10,047 as the radiotracer and according to the procedures described by McCann et al. (1994) (T.-P. Su, personal communication). Its affinity for the PCP site on the NMDA receptor is 6.8 μM (Chaudieu et al. 1989). Thus, BTCP shows in vitro selectivity ratios of 1/250 and 1/736 for the DAT versus the sigma1 receptor and the PCP site, respectively. Furthermore, the in vivo binding parameters of this drug have been measured. On one hand, its ID50 is 6.3 mg/kg s.c. for the striatal DAT, labeled in vivo using [3H]BTCP (Maurice et al. 1991). On the other hand, BTCP fails to significantly inhibit the in vivo binding of [3H]TCP to the PCP site in the mouse hippocampus, cortex and cerebellum, at 50 mg/kg (Maurice and Vignon 1990) and the in vivo binding of [3H]SKF-10,047 to the sigma1 site in the mouse hippocampus, at 60 mg/kg (T. Maurice, unpublished result). BTCP thus appears suitable as a highly selective indirect dopaminergic agonist for comparison with cocaine. The observation that sigma1 receptor antagonists blocked the BTCP-induced CPP confirms the primary role of the DAT in the rewarding properties measured using CPP. This observation demonstrates that the sigma1 receptor activation is consequent to the DAT inhibition. Moreover, electron microscopy studies of the subcellular localization of the sigma1 receptor showed that the protein is mainly located within the post-synaptic elements (Alonso et al. 2000; Phan et al. 2000). The electron dense immunoprecipitates associated with plasmic membranes were localized exclusively with the post-synaptic thickening. It is thus likely that sigma1 receptors are activated within the post-synaptic dopaminergic neurons. The sigma1 receptor can be regarded as one component of the complex biochemical mechanisms occurring within the mesolimbic dopaminergic neurons during cocaine abuse. This conclusion is strengthened by the observation that mRNA expression is increased in the nucleus accumbens, the target structure of mesolimbic pathways extending from the ventral tegmental area of the midbrain and critical structure in the expression of behavioral reinforcement and sensitization (Hyman 1996; Koob 1996).
Selective sigma1 receptor agonists failed to induce CPP alone. This result confirms several previous observations indicating that such agonists are almost devoid of behavioral and physiological effects by themselves (Walker et al. 1990; Su et al. 1991; Maurice and Lockhart 1997; Maurice et al. 1999). Sigma1 receptor agonists showed beneficial effects on several kinds of pathological states, including anti-amnesic, anti-depressant and anti-stress activities (Maurice et al. 1999). Similarly, sigma1 receptor antagonists failed to induce behavioral impairments by themselves. The present study demonstrated that neither NE-100 nor BD1047 affected learning ability in the modified elevated plus-maze test procedure. This result is noteworthy because of the sigma1 receptor involvement in learning and memory processes. Indeed, sigma1 receptor antagonists could prevent the cocaine-induced CPP by provoking amnesia. Cocaine is known to produce a strong mnesic imprint in the brain, sustaining not only the place preference mechanisms, but also the state-dependence (Maurice et al. 2001) and drug discrimination properties of the drug (McKenna and Ho 1980). Amnesic drugs, by blocking the chemical mnesic imprint of cocaine, may allow disruption of the environmental association necessary to develop the CPP behavior and would lead to similar results as observed in this study. Although it is already well-known that selective sigma1 receptor antagonists do not affect mnesic capacities by themselves in several memory tests (Maurice et al. 1994), we performed the elevated plus-maze experiment, preferred to more classical memory tests such as passive avoidance or water-maze learning since it involved exogenous and endogenous cues matching the ones necessary for CPP acquisition. The experiment clearly shows that the sigma1 receptor antagonists did not affect memory processes. In turn, their effect on CPP acquisition or expression could be related to anterograde or retrograde amnesia, respectively.
Another hypothesis could be that the sigma1 receptor is involved in CPP acquisition and expression, in a manner more or less independent from the neuronal system activating the CPP. The sigma1 receptor could thus represent an element related to the key mechanism of CPP response. In the present experiments, CPP was induced by DAT inhibition after cocaine administration, but it remains to be determined whether CPP induced by other drugs would also involve activation of the sigma1 receptor. A preliminary result was obtained in the laboratory, showing that acquisition of morphine (10 mg/kg)-induced CPP (+60 ± 11 s, n = 10), following the same protocol as used in the present study, could be blocked by pre-administration of NE-100 (−16 ± 13 s at 3 mg/kg, n = 10; 15 ± 17 s at 10 mg/kg, n = 10). If this result is confirmed, it will suggest that activation of the sigma1 receptor indeed represents a key component of the acquisition and expression of the CPP process itself, independent of the first neuronal mechanism that initiates the behavioral response. If this is the case, activation of the sigma1 receptor induced by cocaine to develop CPP could be the result of a pathway commonly used by abused drugs.
Interestingly, the sigma1 receptor expression is increased in the nucleus accumbens following the conditioning session. This observation could be surprising, considering that the receptor is activated after cocaine administration and down-regulation would be expected to occur. However, the locomotor data demonstrated that cocaine-induced sensitization occurred during the 4-day procedure. The increase in sigma1 receptor expression within the nucleus accumbens thus appears as an adaptative consequence to cocaine sensitization, particularly considering the importance of this structure in the behavioral activation and reward mechanisms (Kalivas and Nakamura 1999). Enhanced sigma1 receptor expression after only a short duration of treatment with cocaine confirms the importance of this particular target in the acquisition of cocaine-induced behaviors and suggests that treatments allowing the down-regulation of the expression of these receptors (Itzhak and Alerhand 1989; Romieu et al. 2000) may, in parallel to pharmacological antagonism, alleviate the intensity of cocaine's effects. Previous studies have suggested that cocaine administration may lead to sigma1 receptor supersensitivity. In particular, Ujike et al. (1992) reported that the sigma1 receptor agonists (+)-3-[3-hydroxyphenyl]-N-(1-propyl)piperidine ((+)-3-PPP) induced more frequent rearing and stereotypies in rats after a 5-day abstinence period following daily 20 mg/kg cocaine injections for 14 consecutive days. The (+)-3-PPP-induced behaviors were attenuated by BMY-14,802 showing that the sigma1 receptor was involved. It must be noted, however, that the sigma1 receptor involvement in rearing and stereotyped behaviors is far from being established and it is not excluded that the behaviors measured here may not or indirectly involve sigma1 receptor activation. In addition, Itzhak (1993) reported a significant upregulation of the number of sigma1 sites, measured using [3H](+)-pentazocine as radiotracer in the substantia nigra, frontal cortex and cerebellum, but not the nucleus accumbens, of rats treated with methamphetamine, 4 mg/kg once daily over 10 days. Although the study has yet to be repeated with cocaine, it could be considered as a convergent indication of increased sigma1 receptor expression induced by indirect dopaminergic agonists. One must note that the results presented here only show increased sigma1 receptor expression following the particular protocol used during CPP conditioning. The exact effect of cocaine-induced sensitization on the sigma1 receptor expression in the different dopaminergic structures has yet to be performed, using adequate administration protocols with a longer duration of injections following by significant withdrawal periods.
The exact mechanism of the sigma1 receptor involvement in cocaine-induced rewarding properties and plasticity has yet to be determined. Considering the most recent hypotheses regarding its cellular function, the sigma1 receptor plays a significant role in second messenger cascades, mechanisms involved in some effects of cocaine (Hayashi et al. 2000; Morin-Surun et al. 1999; Hyman 1996; Pierce et al. 1998; Valjent et al. 2000). Activation of the sigma1 receptor results in its translocation from the endoplasmic reticulum membrane to the vicinity of the plasma membrane and the regulation of several components implicated in plasma membrane-bound signal transduction, including protein kinase C (Morin-Surun et al. 1999). Such a mechanism as demonstrated in hypoglossal neurons in the brain stem may also occur in dopaminergic neurons and may explain the rapid effects of sigma1 receptor agonists/antagonists. Activation of the sigma1 receptor may also, and initially, result in a facilitation of the intracellular Ca2+ mobilizations, through a unique modulation of inositol 1,4,5-trisphophate receptor activity on the endoplasmic reticulum membrane (Hayashi et al. 2000; Hayashi and Su 2001). Through such action, the sigma1 receptor may play an important role in the cellular plasticity occurring during repeated exposure to cocaine, not only mediating the appetitive conditioning induced by the drug, but also long-term changes involved in the plasticity induced by reinforcement and behavioral sensitization. Pharmacological studies demonstrated that both D1 and D2 dopaminergic receptors contribute to cocaine reinforcement and plasticity (Self and Nestler 1995). Cocaine induces supersensitivity of postsynaptic D1 receptors in the nucleus accumbens (Henry and White 1995). Cocaine, even after acute administration, increases adenylyl cyclase and cAMP-dependent protein kinase activities, since D1 receptors are positively linked to the adenylyl cyclase. In turn, cocaine regulates gene expression by inducing cAMP response element-binding (CREB) protein phosphorylation and expression of both AP-1 and non-AP-1 family immediate early genes (Konradi et al. 1994; Self and Nestler 1995). It could thus be proposed that the sigma1 receptor, through its modulation of cellular Ca2+ fluxes within the post-synaptic neuron, may directly affect these cocaine-induced response pathways. The effects of selective sigma1 ligands on CREB phosphorylation of immediate early gene expression have yet to be examined. However, it must be noted that the sigma1 receptor could also be acting within the presynaptic neurons. In particular, previous studies using superfused rat brain slices reported that sigma1 receptor agonists regulate the NMDA-stimulated [3H]DA release in both the striatum and hippocampus (Gonzalez-Alvear and Werling 1994, 1995; Chaki et al. 1998). Low concentrations of the sigma1 receptor agonists (+)-pentazocine or BD737 inhibited the NMDA-stimulated [3H]DA release in a concentration-dependent manner. The sigma1 receptor antagonist DuP 734, haloperidol, BD1008, NE-100, or XJ448 reversed the inhibition of stimulated [3H]DA release by the sigma1 receptor agonists. Furthermore, BD737 and (+)-pentazocine inhibited stimulated [3H]DA release in the presence of tetrodotoxin, suggesting that sigma1 receptors that regulate DA release are located on dopaminergic nerve terminals (Gonzalez-Alvear and Werling 1995; Chaki et al. 1998). The relevance of such effects in the involvement of the sigma1 receptor in cocaine-induced CPP must be further studied.
In summary, the present study provides strong evidence that activation of the sigma1 receptor, although not sufficient by itself, is a necessary component of cocaine-induced CPP properties, a model of drug reward. The sigma1 receptor may be activated indirectly by cocaine, as a result of its interaction with the DA reuptake system. However, its involvement in conditioned place preference induced by other psychoactive drugs has yet to be examined. Modifications of the sigma1 receptor expression were observed in the nucleus accumbens, in parallel to the acquisition of the cocaine-induced sensitization. Thus, acting at this particular target may offer a new promising therapeutic strategy for cocaine addiction, devoid of reported side-effects.
References
- Alonso G, Phan VL, Guillemain I, Saunier M, Legrand A, Anoal M, Maurice T . (2000): Immunocytochemical localization of the σ1 receptor in the adult rat central nervous system. Neuroscience 97: 155–170
Article CAS Google Scholar - Calligaro DO, Eldefrawi ME . (1988): High affinity stereospecific binding of [3H]cocaine in striatum and its relationship to the dopamine transporter. Membr Biochem 7: 87–106
Article CAS Google Scholar - Chaki S, Okuyama S, Ogawa S, Tomisawa K . (1998): Regulation of NMDA-induced [3H]dopamine release from rat hippocampal slices through sigma-1 binding sites. Neurochem Int 33: 29–34
Article CAS Google Scholar - Chaudieu I, Vignon J, Chicheportiche M, Kamenka JM, Trouiller G, Chicheportetiche R . (1989): Role of the aromatic group in the inhibition of phencyclidine binding and dopamine uptake by PCP analogs. Pharmacol Biochem Behav 32: 699–705
Article CAS Google Scholar - Chomczynski P, Sacchi N . (1987): Single-step method of RNA isolation by acid guanidium thiocyanate-phenol-chloroform extraction. Anal Biochem 162: 156–159
Article CAS Google Scholar - Debler EA, Hashim A, Lajtha A, Sershen H . (1988): Ascorbic acid and striatal transport of [3H]-1-methyl-4-phenylpyridine (MPP+) and [3H]dopamine. Life Sci 42: 2553–2559
Article CAS Google Scholar - Gonzalez-Alvear GM, Werling LL . (1994): Regulation of [3H]dopamine release from rat striatal slices by sigma ligands. J Pharmacol Exp Ther 271: 212–219
CAS PubMed Google Scholar - Gonzalez-Alvear GM, Werling LL . (1995): Sigma1 receptors in rat striatum regulate NMDA-stimulated [3H]dopamine release via a presynaptic mechanism. Eur J Pharmacol 294: 713–719
Article CAS Google Scholar - Hanner M, Moebius FF, Flandorfer A, Knaus HG, Striessnig J, Kempner E, Glossman H . (1996): Purification, molecular cloning, and expression of the mammalian sigma1-binding site. Proc Natl Acad Sci USA 93: 8072–8077
Article CAS Google Scholar - Hayashi T, Maurice T, Su TP . (2000): Ca2+ signaling via sigma1-receptors: Novel regulatory mechanism affecting intracellular Ca2+ concentration. J Pharmacol Exp Ther 293: 788–798
CAS PubMed Google Scholar - Hayashi T, Su TP . (2001): Regulating ankyrin dynamics: Roles of sigma-1 receptors. Proc Natl Acad Sci USA 98: 491–496
Article CAS Google Scholar - Henry DJ, White FJ . (1995): The persistence of behavioral sensitization to cocaine parallels enhanced inhibition of nucleus accumbens neurons. J Neurosci 15: 6287–6299
Article CAS Google Scholar - Higgins ST . (1997): The influence of alternative reinforcers on cocaine use and abuse: A brief review. Pharmacol Biochem Behav 57: 419–427
Article CAS Google Scholar - Hyman SE . (1996): Addiction to cocaine and amphetamine. Neuron 16: 901–904
Article CAS Google Scholar - Hyttel J . (1978): Inhibition of [3H]dopamine accumulation in rat striatal synaptosomes by psychotropic drugs. Biochem Pharmacol 27: 1063–1068
Article CAS Google Scholar - Itoh J, Nabeshima T, Kameyama T . (1990): Utility of an elevated plus-maze for the evaluation of memory in mice: Effect of nootropics, scopolamine and electroconvulsive shock. Psychopharmacology 101: 27–33
Article CAS Google Scholar - Itoh J, Nabeshima T, Kameyama T . (1991): Utility of an elevated plus-maze for dissociation of amnesic and behavioral effects of drugs in mice. Eur J Pharmacol 194: 71–76
Article CAS Google Scholar - Itzhak Y . (1993): Repeated methamphetamine-treatment alters brain σ receptors. Eur J Pharmacol 230: 243–244
Article CAS Google Scholar - Itzhak Y, Alerhand S . (1989): Differential regulation of sigma and PCP receptors after chronic administration of haloperidol and phencyclidine in mice. FASEB J 3: 1868–1872
Article CAS Google Scholar - Kalivas PW, Nakamura M . (1999): Neural systems for behavioral activation and reward. Curr Opinion Neurobiol 9: 223–227
Article CAS Google Scholar - Kekuda R, Prasad PD, Fei YJ, Leibach FH, Ganapathy V . (1996): Cloning and functional expression of the human type 1 sigma receptor (hSigmaR1). Biochem Biophys Res Commun 229: 553–558
Article CAS Google Scholar - Koe BK . (1976): Molecular geometry of inhibitors of the uptake of catecholamines and serotonin in synaptosomal preparations of rat brain. J Pharmacol Exp Ther 199: 649–661
CAS PubMed Google Scholar - Konradi C, Cole RL, Heckers S, Hyman SE . (1994): Amphetamine regulates gene expression in rat striatum via transcription factor CREB. J Neurosci 14: 5623–5634
Article CAS Google Scholar - Koob GF . (1992): Drugs of abuse: anatomy, pharmacology and function of reward pathways. Trends Pharmacol Sci 13: 177–184
Article CAS Google Scholar - Koob GF . (1996): Drug addiction: the yin and yang of hedonic homeostasis. Neuron 16: 893–896
Article CAS Google Scholar - Kuhar MJ . (1992): Molecular pharmacology of cocaine: A dopamine hypothesis and its implications. Ciba Found Symp 166: 81–89
CAS PubMed Google Scholar - Martin-Fardon R, Arnaud M, Rousseau E, Kamenka JM, Privat A, Vignon J . (1996): N-[1-(2-Benzo(b)thiophenyl)cyclohexyl]piperidine (BTCP) and cocaine induce similar effects on striatal dopamine: a microdialysis study in freely moving rats. Neurosci Lett 211: 179–182
Article CAS Google Scholar - Martin-Fardon R, Ben-Shahar O, Weiss F . (2000): Non reciprocal cross-sensitization between cocaine and BTCP on locomotor activity in the rat. Pharmacol Biochem Behav 66: 631–635
Article CAS Google Scholar - Matsumoto RR, McCracken KA, Pouw B, Miller J, Bowen WD, Williams W, De Costa BR . (2001): N-alkyl substituted analogs of the sigma receptor ligand BD1008 and traditional sigma receptor ligands affect cocaine-induced convulsions and lethality in mice. Eur J Pharmacol 411: 261–273
Article CAS Google Scholar - Maurice T, Hiramatsu M, Itoh J, Kameyama T, Hasegawa T, Nabeshima T . (1994): Behavioral evidence for a modulating role of σ ligands in memory processes. I. Attenuation of dizocilpine (MK-801)-induced amnesia. Brain Res 647: 44–56
Article CAS Google Scholar - Maurice T, Lockhart BP . (1997): Neuroprotective and anti-amnesic potentials of sigma (σ) receptor ligands. Prog Neuropsychopharmacol Biol Psychiat 21: 69–102
Article CAS Google Scholar - Maurice T, Phan VL, Urani A, Kamei H, Noda Y, Nabeshima T . (1999): Neuroactive neurosteroids as endogenous effector for the sigma1 (σ1) receptor: Pharmacological evidences and therapeutic opportunities. Jpn J Pharmacol 81: 125–155
Article CAS Google Scholar - Maurice T, Urani A, Taurelle V, Mejean C, Romieu P . (2001): The regulatory role of neuroactive steroids in state-dependent vs. state-independent memory retrieval mechanisms. Soc Neurosci Abstr 27: (in press).
- Maurice T, Vignon J . (1990): In vivo labeling of the phencyclidine (PCP)-receptor with 3H-TCP in the mouse brain. J Neurosci Res 26: 377–385
Article CAS Google Scholar - Maurice T, Vignon J, Kamenka JM, Chicheportiche R . (1991): Differential interaction of phencyclidine-like drugs with the dopamine uptake complex in vivo. J Neurochem 56: 553–559
Article CAS Google Scholar - McCann DJ, Weissman AD, Su TP . (1994): Sigma-1 and sigma-2 sites in rat brain: Comparison of regional, ontogenetic, and subcellular patterns. Synapse 17: 182–189
Article CAS Google Scholar - McCracken KA, Bowen WD, de Costa BR, Matsumoto RR . (1999b): Two novel sigma receptor ligands, BD1047 and LR172, attenuate cocaine-induced toxicity and locomotor activity. Eur J Pharmacol 370: 225–232
Article CAS Google Scholar - McCracken KA, Bowen WD, Matsumoto RR . (1999a): Novel σ receptor ligands attenuate the locomotor stimulatory effects of cocaine. Eur J Pharmacol 365: 35–38
Article CAS Google Scholar - McKenna ML, Ho BT . (1980): The role of dopamine in the discriminative stimulus properties of cocaine. Neuropharmacology 19: 297–303
Article CAS Google Scholar - Menkel M, Terry M, Pontecorvo M, Katz JL, Witkin JM . (1991): Selective σ ligands block stimulant effects of cocaine. Eur J Pharmacol 201: 251–252
Article CAS Google Scholar - Morin-Surun MP, Collin T, Denavit-Saubié M, Baulieu EE, Monnet FP . (1999): Intracellular σ1 receptor modulates phospholipase C and protein kinase C activation in the brain stem. Proc Natl Acad Sci USA 96: 8196–8199
Article CAS Google Scholar - Pan YX, Mei J, Xu J, Wan BL, Zuckerman A, Pasternak GW . (1998): Cloning and characterization of a mouse σ1 receptor. J Neurochem 70: 2279–2285
Article CAS Google Scholar - Parsons LH, Kerr TM, Weiss F . (1998): Simple microbore high-performance liquid chromatographic method for the determination of dopamine and cocaine from a single in vivo brain microdialysis sample. J Chromatogr B Biomed Sci Appl 709: 35–45
Article CAS Google Scholar - Phan VL, Alonso G, Sandillon F, Privat A, Maurice T . (2000): Therapeutic potentials of sigma1 (σ1) receptor ligands against cognitive deficits in aging. Soc Neurosci Abstr 26: 2172
Google Scholar - Pickens R, Thompson T . (1968): Cocaine-reinforced behavior in rats: effects of reinforcement magnitude and fixed-ratio size. J Pharmacol Exp Ther 161: 122–129
CAS PubMed Google Scholar - Pierce RC, Quick EA, Reeder DC, Morgan ZR, Kalivas PW . (1998): Calcium-mediated second messengers modulate the expression of behavioral sensitization to cocaine. J Pharmacol Exp Ther 286: 1171–1176
CAS PubMed Google Scholar - Reith MEA, Meisler BE, Sershen H, Lajtha A . (1986): Structural requirements for cocaine congeners to interact with dopamine and serotonin uptake sites in mouse brain and to induce stereotyped behavior. Biochem Pharmacol 35: 1123–1129
Article CAS Google Scholar - Ritz MC, George FR . (1993): Cocaine-induced seizures and lethality appear to be associated with distinct central nervous system binding sites. J Pharmacol Exp Ther 264: 1333–1343
CAS PubMed Google Scholar - Ritz MC, Lamb RJ, Goldberg SR, Kuhar MJ . (1987): Cocaine receptors on dopamine transporters are related to self-administration of cocaine. Science 237: 1219–1223
Article CAS Google Scholar - Romieu P, Martin-Fardon R, Maurice T . (2000): Involvement of the σ1 receptor in the cocaine-induced conditioned place preference. Neuroreport 11: 2885–2888
Article CAS Google Scholar - Self DW, Nestler EJ . (1995): Molecular mechanisms of drug reinforcement and addiction. Ann Rev Neurosci 18: 463–496
Article CAS Google Scholar - Seth P, Fei YJ, Li HW, Huang W, Leibach FH, Ganapathy V . (1998): Cloning and functional characterization of a sigma receptor from rat brain. J Neurochem 70: 922–931
Article CAS Google Scholar - Seth P, Leibach FH, Ganapathy V . (1997): Cloning and structural analysis of the cDNA and the gene encoding the murine type 1 sigma receptor. Biochem Biophys Res Commun 241: 535–540
Article CAS Google Scholar - Sharkley J, Glen KA, Wolfe S, Kuhar MJ . (1988): Cocaine binding at σ receptors. Eur J Pharmacol 149: 171–174
Article Google Scholar - Sora I, Hall FS, Andrews AM, Itokawa M, Li XF, Wei HB, Wichems C, Lesch KP, Murphy DL, Uhl GR . (2000): Molecular mechanisms of cocaine reward: Combined dopamine and serotonin transporter knockouts eliminate cocaine place preference. Proc Natl Acad Sci USA 98: 5300–5305
Article Google Scholar - Stolerman I . (1992): Drugs of abuse: Behavioural principles, methods and terms. Trends Pharmacol Sci 13: 170–176
Article CAS Google Scholar - Su TP, Wu XZ, Cone EJ, Shukla K, Gund TM, Dodge AL, Parish DW . (1991): Sigma compounds derived from phencyclidine: Identification of PRE-084, a new, selective sigma ligand. J Pharmacol Exp Ther 259: 543–548
CAS PubMed Google Scholar - Ujike H, Kuroda S, Otsuki S . (1996): σ Receptor antagonists block the development of sensitization to cocaine. Eur J Pharmacol 296: 123–128
Article CAS Google Scholar - Ujike H, Tsuchida K, Akiyama K, Otsuki S . (1992): Supersensitivity of σ receptors after repeated administration of cocaine. Life Sci 51: PL31–36
Article CAS Google Scholar - Valjent E, Corvol JC, Pages C, Besson MJ, Maldonado R, Caboche J . (2000): Involvement of the extracellular signal-regulated kinase cascade for cocaine-rewarding properties. J Neurosci 20: 8701–8709
Article CAS Google Scholar - Vignon J, Pinet V, Cerruti C, Kamenka JM, Chicheportiche R . (1988): [3H]N-[1-(2-benzo(b)thiophenyl)cyclohexyl]piperidine ([3H]BTCP): A new phencyclidine analog selective for the dopamine uptake complex. Eur J Pharmacol 148: 427–436
Article CAS Google Scholar - Walker JM, Bowen WD, Walker FO, Matsumoto RR, De Costa B, Rice KC . (1990): Sigma receptors: Biology and function. Pharmacol Rev 42: 355–402
CAS Google Scholar - Warner EA . (1993): Cocaine abuse. Ann Intern Med 119: 226–235
Article CAS Google Scholar - Woolverton WL . (1992): Determinants of cocaine self-administration by laboratory animals. Ciba Found Symp 166: 149–161
CAS PubMed Google Scholar - Woolverton WL, Johnson KM . (1992): Neurobiology of cocaine abuse. Trends Pharmacol Sci 13: 193–200
Article CAS Google Scholar
Acknowledgements
The authors are grateful to Drs Rae R. Matsumoto (Oklahoma University, Oklahoma City, OK, USA) and Tsung-Ping Su (NIDA/NIH, Baltimore, MD, UDA) for their important advise and helpful information, and to Drs Yasuko Karasawa, Jean-Marc Kamenka, François J. Roman and Wayne D. Bowen for their kind gift of drugs. The authors heartily thank Christina V. Lorentz for assistance with preparation of the manuscript. This work was supported by the Institut National de la Santé et de la Recherche Médicale.
Author information
Authors and Affiliations
- Behavioral Neuropharmacology group, INSERM U. 336, Institut de Biologie, Montpellier, France
Pascal Romieu MS, Vân-Ly Phan MS & Tangui Maurice Ph.D - Department of Neuropharmacology, The Scripps Research Institute, La Jolla, CA, USA
Rémi Martin-Fardon Ph.D
Authors
- Pascal Romieu MS
You can also search for this author inPubMed Google Scholar - Vân-Ly Phan MS
You can also search for this author inPubMed Google Scholar - Rémi Martin-Fardon Ph.D
You can also search for this author inPubMed Google Scholar - Tangui Maurice Ph.D
You can also search for this author inPubMed Google Scholar
Corresponding author
Correspondence toTangui Maurice Ph.D.
Rights and permissions
About this article
Cite this article
Romieu, P., Phan, VL., Martin-Fardon, R. et al. Involvement of the Sigma1 Receptor in Cocaine-induced Conditioned Place Preference: Possible Dependence on Dopamine Uptake Blockade.Neuropsychopharmacol 26, 444–455 (2002). https://doi.org/10.1016/S0893-133X(01)00391-8
- Received: 14 May 2001
- Revised: 28 August 2001
- Accepted: 12 September 2001
- Published: 12 October 2001
- Issue Date: 01 April 2002
- DOI: https://doi.org/10.1016/S0893-133X(01)00391-8