Interaction of glutamic-acid-rich proteins with the cGMP signalling pathway in rod photoreceptors (original) (raw)
- Letter
- Published: 19 August 1999
- Michael Beyermann2,
- Frank Müller1,
- Martin Heck3,
- Marius Vantler1,
- Karl-Wilhelm Koch1,
- Roland Kellner4 nAff4,
- Uwe Wolfrum5 nAff7,
- Christian Bode5 nAff7,
- Klaus Peter Hofmann3 &
- …
- U. Benjamin Kaupp1
Nature volume 400, pages 761–766 (1999) Cite this article
- 469 Accesses
- 142 Citations
- 3 Altmetric
- Metrics details
Abstract
The assembly of signalling molecules into macromolecular complexes (transducisomes) provides specificity, sensitivity and speed in intracellular signalling pathways1,2. Rod photoreceptors in the eye contain an unusual set of glutamic-acid-rich proteins (GARPs) of unknown function3,4,5,6,7. GARPs exist as two soluble forms, GARP1 and GARP2, and as a large cytoplasmic domain (GARP′ part) of the β-subunit of the cyclic GMP-gated channel3,4,5,6,7. Here we identify GARPs as multivalent proteins that interact with the keyplayers of cGMP signalling, phosphodiesterase and guanylate cyclase, and with a retina-specific ATP-binding cassette transporter (ABCR)8,9, through four, short, repetitive sequences. In electron micrographs, GARPs are restricted to the rim region and incisures of discs in close proximity to the guanylate cyclase and ABCR, whereas the phosphodiesterase is randomly distributed. GARP2, the most abundant splice form, associates more strongly with light-activated than with inactive phosphodiesterase, and GARP2 potently inhibits phosphodiesterase activity. Thus, the GARPs organize a dynamic protein complex near the disc rim that may control cGMP turnover and possibly other light-dependent processes. Because there are no similar GARPs in cones, we propose that GARPs may prevent unnecessary cGMP turnover during daylight, when rods are held in saturation by the relatively high light levels.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Additional access options:
Figure 1: Structural features of GARPs and localization to rod photoreceptors.
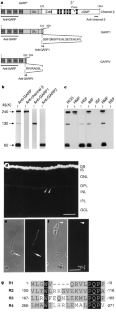
The alternative text for this image may have been generated using AI.
Figure 2: Association of rod outer segment proteins with GARPs.
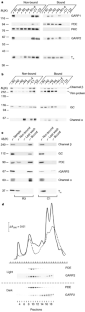
The alternative text for this image may have been generated using AI.
Figure 3: Inhibition of phosphodiesterase (PDE) by rGARP2.
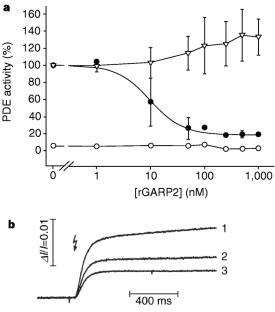
The alternative text for this image may have been generated using AI.
Figure 4: Immunoelectron microscopic localization of GARPs and PDE in photoreceptor cells.
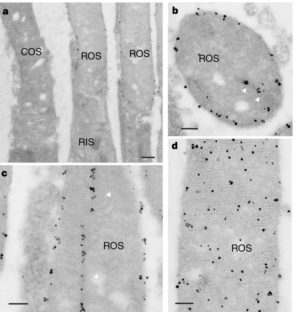
The alternative text for this image may have been generated using AI.
Figure 5: Hypothetical model of protein interactions.
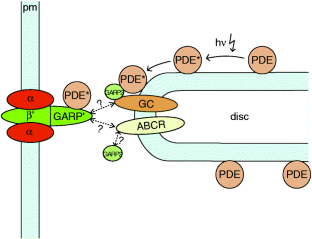
The alternative text for this image may have been generated using AI.
Similar content being viewed by others
References
- Tsunoda, S. et al. Amultivalent PDZ-domain protein assembles signalling complexes in a G-protein-coupled cascade. Nature 388, 243–249 (1997).
Article CAS ADS PubMed Google Scholar - Zuker, C. S. & Ranganathan, R. The path to specificity. Science 283, 650–651 (1999).
Article CAS PubMed Google Scholar - Sugimoto, Y., Yatsunami, K., Tsujimoto, M., Khorana, H. G. & Ichikawa, A. The amino acid sequence of a glutamic acid-rich protein from bovine retina as deduced from the cDNA sequence. Proc. Natl Acad. Sci. USA 88, 3116–3119 (1991).
Article CAS ADS PubMed PubMed Central Google Scholar - Körschen, H. G. et al. A240 kDa protein represents the complete β subunit of the cyclic nucleotide-gated channel from rod photoreceptor. Neuron 15, 627–636 (1995).
Article PubMed Google Scholar - Ardell, M. D. et al. cDNA, gene structure, and chromosomal localization of human GAR1 (CNCG3L), a homolog of the third subunit of bovine photoreceptor cGMP-gated channel. Genomics 28, 32–38 (1995).
Article CAS PubMed Google Scholar - Colville, C. A. & Molday, R. S. Primary structure and expression of the human β-subunit and related proteins of the rod photoreceptor cGMP-gated channel. J. Biol. Chem. 271, 32968–32974 (1996).
Article CAS PubMed Google Scholar - Grunwald, M. E., Yu, W.-P., Yu, H.-H. & Yau, K.-W. Identification of a domain on the β-subunit of the rod cGMP-gated cation channel that mediates inhibition by calcium–calmodulin. J. Biol. Chem. 273, 9148–9157 (1998).
Article CAS PubMed Google Scholar - Illing, M., Molday, L. L. & Molday, R. S. The 220-kDa rim protein of retinal rod outer segment is a member of the ABC transporter superfamily. J. Biol. Chem. 272, 10303–10310 (1997).
Article CAS PubMed Google Scholar - Allikmets, R. et al. Aphotoreceptor cell-specific ATP-binding transporter gene (ABCR) is mutated in recessive Stargardt macular dystrophy. Nature Genet. 15, 236–246 (1997).
Article CAS PubMed Google Scholar - Papermaster, D. S., Schneider, B. G., Zorn, M. A. & Kraehenbuhl, J. P. Immunocytochemical localization of a large intrinsic membrane protein to the incisures and margins of frog rod outer segment disks. J. Cell Biol. 78, 415–425 (1978).
Article CAS PubMed Google Scholar - Montell, C. TRP trapped in fly signaling web. Curr. Opin. Neurobiol. 8, 389–397 (1998).
Article CAS PubMed Google Scholar - Koch, K.-W. & Lambrecht, H.-G. in Signal Transduction in Photoreceptor Cells (eds Hargrave, P. A., Hofmann, K. P. & Kaupp, U. B.) 259–267 (Springer, Berlin, (1992).
Book Google Scholar - Schrem, A., Lange, C., Beyermann, M. & Koch, K.-W. Identification of a domain in guanylyl cyclase-activating protein 1 that interacts with a complex of guanylyl cyclase and tubulin in photoreceptors. J.Biol. Chem. 274, 6244–6249 (1999).
Article CAS PubMed Google Scholar - Heck, M. & Hofmann, K. P. G-protein-effector coupling: A real-time light-scattering assay for transducin-phosphodiesterase interaction. Biochemistry 32, 8220–8227 (1993).
Article CAS PubMed Google Scholar - Liu, X. et al. Ultrastructural localization of retinal guanylate cyclase in human and monkey retinas. Exp. Eye Res. 59, 761–768 (1994).
Article CAS PubMed Google Scholar - Cook, N. J., Molday, L. L., Reid, D., Kaupp, U. B. & Molday, R. S. The cGMP-gated channel of bovine rod photoreceptors is localized exclusively in the plasma membrane. J. Biol. Chem. 264, 6996–6999 (1989).
CAS PubMed Google Scholar - Pedler, C. M. & Tilly, R. The fine structure of photoreceptor discs. Vision Res. 7, 829–836 (1967).
Article CAS PubMed Google Scholar - Eckmiller, M. S. & Toman, A. Association of kinesin with microtubules in diverse cytoskeletal systems in the outer segments of rods and cones. Acta Anat. 162, 133–141 (1998).
Article CAS PubMed Google Scholar - Roof, D. J. & Heuser, J. E. Surfaces of rod photoreceptor disk membranes: Integral membrane components. J. Cell Biol. 95, 487–500 (1982).
Article CAS PubMed Google Scholar - He, W., Cowan, C. W. & Wensel, T. G. RGS9, a GTPase accelerator for phototransduction. Neuron 20, 95–102 (1998).
Article PubMed Google Scholar - Makino, E. R., Handy, J. W., Li, T. & Arshavsky, V. Y. The GTPase activating factor for transducin in rod photoreceptors is the complex between RGS9 and type 5 G protein β subunit. Proc. Natl Acad. Sci. USA 96, 1947–1952 (1999).
Article CAS ADS PubMed PubMed Central Google Scholar - Pugh, E. N. J & Lamb, T. D. Amplification and kinetics of the activation steps in phototransduction. Biochim. Biophys. Acta 1141, 111–149 (1993).
Article CAS PubMed Google Scholar - Bönigk, W. et al. Rod and cone photoreceptor cells express distinct genes for cGMP-gated channels. Neuron 10, 865–877 (1993).
Article PubMed Google Scholar - Zoche, M., Bienert, M., Beyermann, M. & Koch, K.-W. Distinct molecular recognition of calmodulin-binding sites in the neuronal and macrophage nitric oxide synthases: A surface plasmon resonance study. Biochemistry 35, 8742–8747 (1996).
Article CAS PubMed Google Scholar - Hsu, Y.-T. & Molday, R. S. Modulation of the cGMP-gated channel of rod photoreceptor cells by calmodulin. Nature 361, 76–79 (1993).
Article CAS ADS PubMed Google Scholar - Lambrecht, H.-G. & Koch, K.-W. A26 kd calcium binding protein from bovine rod outer segments as modulator of photoreceptor guanylate cyclase. EMBO J. 10, 793–798 (1991).
Article CAS PubMed PubMed Central Google Scholar - Wolfrum, U. Centrin in the photoreceptor cells of mammalian retinae. Cell Motil. Cytoskel. 32, 55–64 (1995).
Article CAS Google Scholar - Wolfrum, U., Liu, X., Schmitt, A., Udovichenko, I. P. & Williams, D. S. Myosin VIIa as a common component of cilia and microvilli. Cell Motil. Cytoskel. 40, 261–271 (1998).
Article CAS Google Scholar - Danscher, G. Localization of gold in biological tissue. A photochemical method for light and electron microscopy. Histochemistry 71, 81–88 (1981).
Article CAS PubMed Google Scholar
Acknowledgements
We thank J. Beavo, J. Nathans, I. Weyand and D. Weitz for antibodies, A. Eckert forpreparing the manuscript, I. Weyand and R. Seifert for critical reading of the manuscript, and E. Pugh and R. Cote for discussions. This work was supported by grants from the EU (U.B.K.), the Deutsche Forschungsgemeinschaft (M.B., K.P.H., K.-W.K., U.B.K., U.W.) and the FAUN-Stiftung (U.W.).
Author information
Author notes
- Roland Kellner
Present address: Biomedizinische Forschung, Merck AG, 64271, Darmstadt, Germany - Uwe Wolfrum & Christian Bode
Present address: Institut für Zoologie, Universitt Mainz, 55099, Mainz, Germany
Authors and Affiliations
- Institut für Biologische Informationsverarbeitung, Forschungszentrum Jülich, 52425, Jülich, Germany
Heinz G. Körschen, Frank Müller, Marius Vantler, Karl-Wilhelm Koch & U. Benjamin Kaupp - Forschungsinstitut für Molekulare Pharmakologie, 10315, Berlin, Germany
Michael Beyermann - Institut für Medizinische Physik und Biophysik, Universitätsklinikum Charité, Humboldt-Universität, 10998, Berlin, Germany
Martin Heck & Klaus Peter Hofmann - Institut für Physiologische Chemie und Pathobiochemie, Universität Mainz, 55099, Mainz, Germany
Roland Kellner - Institut für Zoologie, Universität Karlsruhe, 76128, Karlsruhe, Germany
Uwe Wolfrum & Christian Bode
Authors
- Heinz G. Körschen
- Michael Beyermann
- Frank Müller
- Martin Heck
- Marius Vantler
- Karl-Wilhelm Koch
- Roland Kellner
- Uwe Wolfrum
- Christian Bode
- Klaus Peter Hofmann
- U. Benjamin Kaupp
Corresponding author
Correspondence toU. Benjamin Kaupp.
Rights and permissions
About this article
Cite this article
Körschen, H., Beyermann, M., Müller, F. et al. Interaction of glutamic-acid-rich proteins with the cGMP signalling pathway in rod photoreceptors.Nature 400, 761–766 (1999). https://doi.org/10.1038/23468
- Received: 06 April 1999
- Accepted: 14 June 1999
- Issue date: 19 August 1999
- DOI: https://doi.org/10.1038/23468