Veil architecture in a sulphide-oxidizing bacterium enhances countercurrent flux (original) (raw)
- Letter
- Published: 23 July 1998
Nature volume 394, pages 367–369 (1998) Cite this article
- 628 Accesses
- 57 Citations
- 7 Altmetric
- Metrics details
Abstract
Solute uptake by microorganisms is limited by molecular diffusion through a boundary layer surrounding the cells, and the uptake is not enhanced (or only insignificantly) by convective water transport or by swimming1. It is generally assumed that sediment uptake of oxygen is diffusion-limited, so the steepness ofthe concentration gradient within the 0.5–1-mm-thick diffusive boundary layer is a measure of diffusional flux into thesediment2,3. Here we show that veils, which are formed onsediments by the marine sulphide-oxidizing bacterium Thiovulum majus4, generate convective oxygen transport through the 0.5-mm-thick water layers above the veil at rates that are about 40 times higher than molecular diffusion. Chemosensory behaviour of the cells, combined with their generation of water currents, leads to characteristic, aggregated distribution patterns: areas with high cell densities draw oxygenated water downwards through the veil, whereas areas without cells serve for the upward-directed return flow of deoxygenated water. The microbial community structure thus overcomes the limitations of diffusion and thereby enhances the rates of respiration and sulphide oxidation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Additional access options:
Figure 1: The flow field around Thiovulum cells.

The alternative text for this image may have been generated using AI.
Figure 2: A continuous Thiovulum veil with holes.
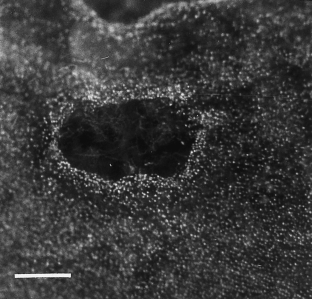
The alternative text for this image may have been generated using AI.
Figure 3
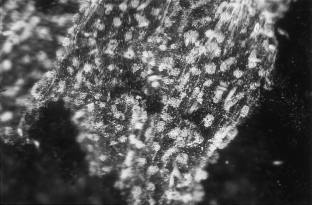
The alternative text for this image may have been generated using AI.
Figure 4
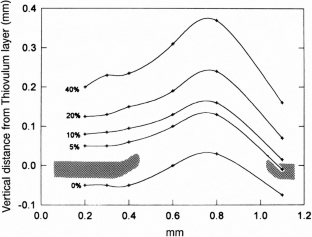
The alternative text for this image may have been generated using AI.
Similar content being viewed by others
References
- Purcell, E. M. Life at low Reynolds number. Am. J. Phys. 45, 3–11 (1977).
Article ADS Google Scholar - Revsbech, N. P., Jørgensen, B. B. & Blackburn, T. H. Oxygen in the sea bottom measured with a microelectrode. Science 207, 1355–1356 (1980).
Article ADS CAS Google Scholar - Revsbech, N. P. & Jøgensen, B. B. Microelectrodes: their use in microbial ecology. Adv. Microbiol. Ecol. 9, 293–352 (1986).
Article Google Scholar - Wirsen, C. O. & Jannasch, H. W. Physiological and morphological observations on Thiovulum sp. J. Bacteriol. 136, 765–774 (1978).
CAS PubMed PubMed Central Google Scholar - Garcia-Pichel, F. Rapid bacterial swimming measured in swarming cells of Thiovulum majus. J. Bacteriol. 171, 3560–3563 (1989).
Article CAS Google Scholar - Fenchel, T. Motility and chemosensory behaviour of the sulphur bacterium Thiovulum majus. Microbiology 140, 3109–3116 (1994).
Article CAS Google Scholar - Jørgensen, B. B. & Revsbech, N. P. Colorless sulfur bacteria, Beggiatoa spp. and Thiovulum spp. in O2and H2S microgradients. Appl. Environ. Microbiol. 45, 1261–1270 (1983).
PubMed PubMed Central Google Scholar - Lighthill, J. Flagellar hydrodynamics. Soc. Indust. Appl. Math. Rev. 18, 161–230 (1976).
MathSciNet MATH Google Scholar - Vogel, S. Life in moving fluids (Grant, Boston, 1981).
Google Scholar - Fenchel, T. & Bernard, C. Mats of colourless sulphur bacteria. I Major microbial processes. Mar. Ecol. Prog. Ser. 128, 161–170 (1995).
Article ADS Google Scholar - Aller, R. C. & Yingst, J. Y. Effects of marine deposit-feeders Heteromastus filiformis (Polychaeta), Macoma balthica (Bivalvia), and Tellina texana (Bivalvia) on averaged solute transport, reaction rates, and microbial distributions. J. Mar. Res. 41, 299–322 (1985).
Article Google Scholar - Dando, P. R. et al. The effects of methane seepage at an intertidal/shallow subtidal site on the shore of Kattegat, Vendsyssel. Bull. Geol. Soc. Denmark 41, 65–79 (1994).
CAS Google Scholar - Gundersen, J. K., Jørgensen, B. B., Larsen, E. & Jannasch, H. W. Mats of giant sulphur bacteria on deep-sea sediments due to fluctuating hydrothermal flow. Nature 360, 454–455 (1992).
Article ADS Google Scholar - Glud, R. N., Gundersen, J. K., Revsbech, N. P. & Jørgensen, B. B. Effects on the benthic diffusive boundary layer imposed by microelectrodes. Limnol. Oceanogr. 39, 462–467 (1994).
Article ADS Google Scholar
Acknowledgements
We thank J. Johansen for technical assistance. This study was supported by grants from the Danish Natural Science Research Council.
Author information
Authors and Affiliations
- Marine Biological Laboratory, University of Copenhagen, 3000 Helsingr, Denmark
Tom Fenchel & Ronnie N. Glud
Authors
- Tom Fenchel
- Ronnie N. Glud
Corresponding author
Correspondence toTom Fenchel.
Rights and permissions
About this article
Cite this article
Fenchel, T., Glud, R. Veil architecture in a sulphide-oxidizing bacterium enhances countercurrent flux.Nature 394, 367–369 (1998). https://doi.org/10.1038/28609
- Received: 27 January 1998
- Accepted: 08 May 1998
- Issue date: 23 July 1998
- DOI: https://doi.org/10.1038/28609