Habitat structure and population persistence in an experimental community (original) (raw)
- Letter
- Published: 01 August 2001
- Edward McCauley2,
- Bruce E. Kendall3,
- Cheryl J. Briggs5,
- Parveiz R. Hosseini4,
- Simon N. Wood6,
- Arne Janssen7,
- Maurice W. Sabelis7,
- Peter Turchin8,
- Roger M. Nisbet4 &
- …
- William W. Murdoch4
Nature volume 412, pages 538–543 (2001)Cite this article
- 1717 Accesses
- 195 Citations
- Metrics details
Abstract
Understanding spatial population dynamics is fundamental for many questions in ecology and conservation1,2,3,4. Many theoretical mechanisms have been proposed whereby spatial structure can promote population persistence, in particular for exploiter–victim systems (host–parasite/pathogen, predator–prey) whose interactions are inherently oscillatory and therefore prone to extinction of local populations5,6,7,8,9,10,11. Experiments have confirmed that spatial structure can extend persistence11,12,13,14,15,16, but it has rarely been possible to identify the specific mechanisms involved. Here we use a model-based approach to identify the effects of spatial population processes in experimental systems of bean plants (Phaseolus lunatus), herbivorous mites (Tetranychus urticae) and predatory mites (Phytoseiulus persimilis). On isolated plants, and in a spatially undivided experimental system of 90 plants, prey and predator populations collapsed; however, introducing habitat structure allowed long-term persistence. Using mechanistic models, we determine that spatial population structure did not contribute to persistence, and spatially explicit models are not needed. Rather, habitat structure reduced the success of predators at locating prey outbreaks, allowing between-plant asynchrony of local population cycles due to random colonization events.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Additional access options:
Figure 1: Experimental layouts and results.
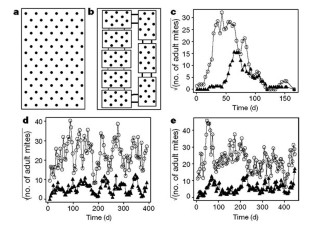
The alternative text for this image may have been generated using AI.
Figure 2: Examples of mite population dynamics on a single plant, from run B of the metapopulation experiment.
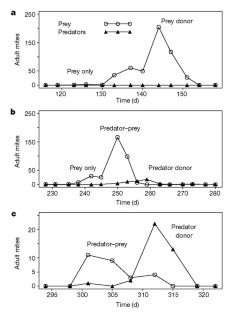
The alternative text for this image may have been generated using AI.
Figure 3: Total prey and predator mite densities in consecutive replicate runs of the simulation models.
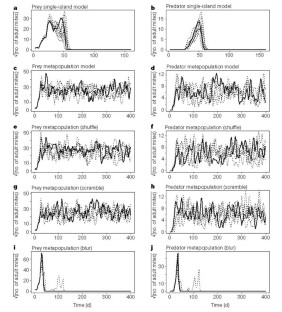
The alternative text for this image may have been generated using AI.
Figure 4: Summary measures of temporal patterns in model output and experimental results.
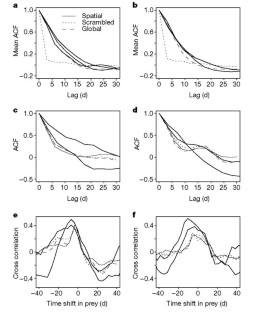
The alternative text for this image may have been generated using AI.
Similar content being viewed by others
References
- Tilman, D. & Kareiva, P. (eds) Spatial Ecology: the Role of Space in Population Dynamics and Interspecific Interactions (Princeton Univ. Press, Princeton, 1997).
Google Scholar - Dieckmann, U., Law, R. & Metz, J. A. J. (eds) The Geometry of Ecological Interactions: Simplifying Spatial Complexity (Cambridge Univ. Press, Cambridge, 2000).
Book Google Scholar - Hanski, I. A. & Gilpin, M. E. (eds) Metapopulation Biology: Ecology, Genetics, and Evolution (Academic, San Diego, 1997).
MATH Google Scholar - Levin, S. A., Grenfell, B., Hastings, A. & Perelson, A. S. Mathematical and computational challenges in population biology and ecosystems science. Science 275, 334–343 (1997).
Article CAS Google Scholar - May, R. M. Host–parasitoid systems in patchy environments: a phenomenological model. J. Anim. Ecol. 47, 833–843 (1978).
Article Google Scholar - Sabelis, M. W. & Diekmann, O. Overall population stability despite local extinction: the stabilizing effect of prey dispersal from predator-invaded patches. Theor. Popul. Biol. 34, 169–176 (1988).
Article Google Scholar - Hassell, M. P. & May, R. M. Spatial heterogeneity and the dynamics of parasitoid–host systems. Ann. Zool. Fenn. 25, 55–61 (1988).
Google Scholar - Hassell, M. P., Comins, H. N. & May, R. M. Spatial structure and chaos in insect population dynamics. Nature 353, 255–258 (1991).
Article ADS Google Scholar - de Roos, A. M., McCauley, E. & Wilson, W. Mobility versus density limited predator–prey dynamics on different spatial scales. Proc. R. Soc. Lond. B 246, 117–122 (1991).
Article ADS Google Scholar - Sabelis, M. W., Diekmann, O. & Jansen, V. A. A. Metapopulation persistence despite local extinction—predator–prey patch models of the Lotka-Volterra type. Biol. J. Linn. Soc. 42, 267–283 (1991).
Article Google Scholar - Murdoch, W. W. Population regulation in theory and practice. Ecology 75, 271–287 (1994).
Article Google Scholar - Huffaker, C. B., Shea, K. P. & Herman, S. G. Experimental studies on predation: complex dispersion and levels of food in an acarine predator–prey interaction. Hilgardia 34, 305–330 (1963).
Article Google Scholar - van de Klashorst, G., Readshaw, G. L., Sabelis, M. W. & Lingeman, R. A demonstration of asynchronous local cycles in an acarine predator–prey system. Exp. Appl. Acarol. 14, 185–199 (1992).
Article Google Scholar - Holyoak, M. & Lawler, S. P. Persistence of an extinction prone predator–prey interaction through metapopulation dynamics. Ecology 77, 1867–1879 (1996).
Article Google Scholar - Janssen, A., van Gool, E., Lingeman, R., Jacas, J. & van de Klashorst, G. Metapopulation dynamics of a persisting predator–prey system in the laboratory: time-series analysis. Exp. Appl. Acarol. 21, 415–430 (1997).
Article Google Scholar - Holyoak, M. Habitat patch arrangement and metapopulation persistence of predators and prey. Am. Nat. 156, 378–389 (2000).
Article Google Scholar - Pels, B. & Sabelis, M. W. Local dynamics, overexploitation and predator dispersal in an acarine predator–prey system. Oikos 86, 573–583 (1999).
Article Google Scholar - McCauley, E. et al. Inferring colonization processes from population dynamics in spatially structured predator–prey systems. Ecology 81, 3350–3361 (2000).
Google Scholar - McCauley, E., de Roos, A. M. & Wilson, W. Dynamics of age- and spatially-structured predator–prey interactions: individual based models and population level formulations. Am. Nat. 142, 412–442 (1993).
Article CAS Google Scholar - de Roos, A. M., McCauley, E. & Wilson, W. Pattern formation and the spatial scale of interactions between predators and their prey. Theor. Popul. Biol. 53, 108–130 (1998).
Article Google Scholar - Nisbet, R. M. & Gurney, W. S. C. Modelling Fluctuating Populations Ch. 10 (Wiley, New York, 1982).
MATH Google Scholar - Helle, W. & Sabelis, M. W. (eds) Spider Mites, their Biology, Natural Enemies, and Control (Elsevier, Amsterdam, 1985).
Google Scholar - Sabelis, M. W. & van der Meer, J. in Dynamics of Physiologically Structured Populations (eds Metz, J. A. J. & Diekmann, O.) 322–344 (Springer, New York, 1986).
Google Scholar - Hastings, A. Spatial heterogeneity and stability of predator–prey systems. Theor. Popul. Biol. 12, 37–48 (1977).
Article MathSciNet CAS Google Scholar - Wright, S. Isolation by distance. Genetics 28, 114–138 (1943).
Article CAS Google Scholar - Gurney, W. S. C., Nisbet, R. M. & Lawton, J. H. The systematic formulation of tractable single-species population models incorporating age structure. J. Anim. Ecol. 52, 479–495 (1983).
Article Google Scholar - Caswell, H. Matrix Population Models: Construction, Analysis, and Interpretation (Sinauer Associates, Sunderland, Massachusetts, 2001).
Google Scholar - Sabelis, M. W. How to analyze prey preference when prey density varies? A new method to discriminate between effects of gut fullness and prey type composition. Oecologia 82, 289–298 (1990).
Article ADS CAS Google Scholar - Sabelis, M. W. & Nagelkerke, C. J. Sex allocation strategies of pseudoarrhenotokous phytoseiid mites. Neth. J. Zool. 37, 117–136 (1987).
Article Google Scholar
Acknowledgements
We thank E. van Gool for assistance with the experiments. This work was undertaken as part of the Working Group on Complex Population Dynamics at the National Center for Ecological Analysis and Synthesis, a centre funded by the US National Science Foundation, University of California–Santa Barbara, and the State of California. A.J. and M.W.S. carried out experiments; E.M., B.E.K., C.J.B. and S.N.W. conceived and fitted colonization models; S.P.E., S.N.W., P.R.H., A.J., P.T., R.M.N. and W.W.N. conceived and fitted structured population models; S.P.E., P.R.H., S.N.W. and C.J.B. implemented the models; S.P.E., E.M. and B.E.K. carried out comparisons of models with data; and S.P.E., E.M., A.J., M.W.S. and P.R.H. prepared the original manuscript.
Author information
Authors and Affiliations
- Department of Ecology and Evolutionary Biology, Cornell University, Ithaca, 14853-2701, New York, USA
Stephen P. Ellner - Ecology Division, Department of Biological Sciences, University of Calgary, Calgary, T2N 1N4, Canada
Edward McCauley - Donald Bren School of Environmental Science and Management,
Bruce E. Kendall - Department of Ecology, Evolution and Marine Biology, University of California, Santa Barbara, 93106, California, USA
Parveiz R. Hosseini, Roger M. Nisbet & William W. Murdoch - Department of Integrative Biology, University of California, Berkeley, 94720, California, USA
Cheryl J. Briggs - School of Mathematical and Computation Sciences, University of St Andrews, Fife, KY16 9SS, Scotland, UK
Simon N. Wood - Institute for Biodiversity and Ecosystem Dynamics, PO Box 94084, Amsterdam, 1090 GB, The Netherlands
Arne Janssen & Maurice W. Sabelis - Department of Ecology and Evolutionary Biology, University of Connecticut, Storrs, 06269, Connecticut, USA
Peter Turchin
Authors
- Stephen P. Ellner
- Edward McCauley
- Bruce E. Kendall
- Cheryl J. Briggs
- Parveiz R. Hosseini
- Simon N. Wood
- Arne Janssen
- Maurice W. Sabelis
- Peter Turchin
- Roger M. Nisbet
- William W. Murdoch
Corresponding author
Correspondence toStephen P. Ellner.
Supplementary information
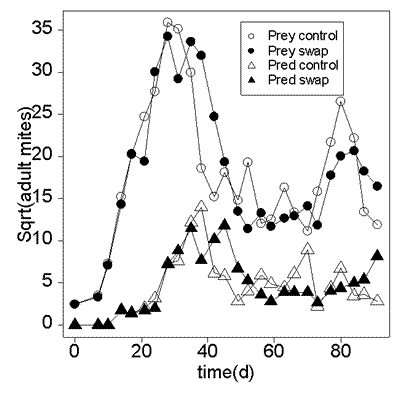
Figure 1 (download GIF )
(GIF 10.6 KB)
Metapopulation swapping experiments
The Metapopulation Swapping experiments followed the same protocols as the two Metapopulation experiments described in the text, again having two replicate systems operating simultaneously in the same environmental chamber and given identical inoculations of mites. In the "swapping" replicate of this experiment, on each census date plants were paired at random and the locations of paired plants were interchanged; in the "control" replicate plants remained in place. All other experimental procedures were the same as in the Metapopulation experiments described in the text. The figure below shows the fluctuations in the total density of prey (circles) and predatory (triangles) mites in the control (open symbols) and swapping (solid symbols) replicates. The experimental results are consistent with our conclusion that spatial pattern did not play an important role in the dynamics of the Metapopulation systems, in that a significant manipulation of the populations' spatial structure had very little effect on short-term dynamics. However the duration of the swapping experiment was only 91d, which is shorter than the extinction time of the SuperIsland experiment and consequently too brief to determine if long-term persistence would occur.
Rights and permissions
About this article
Cite this article
Ellner, S., McCauley, E., Kendall, B. et al. Habitat structure and population persistence in an experimental community.Nature 412, 538–543 (2001). https://doi.org/10.1038/35087580
- Received: 27 March 2001
- Accepted: 20 June 2001
- Published: 01 August 2001
- Issue date: 02 August 2001
- DOI: https://doi.org/10.1038/35087580