Peptide bond formation by in vitro selected ribozymes (original) (raw)
- Letter
- Published: 06 November 1997
Nature volume 390, pages 96–100 (1997)Cite this article
- 4203 Accesses
- 273 Citations
- 19 Altmetric
- Metrics details
Abstract
An attractive solution to the problem of the origin of protein synthesis in an evolving ‘RNA world’ involves catalysis by nucleic acid without assistance from proteins1,2. Indeed, even the modern ribosome has been considered to be fundamentally an RNA machine3, and the large ribosomal subunit can carry out peptidyl transfer in the absence of most of its protein subunits4. Successive cycles of in vitro selection and amplification5,6,7 have been used to find RNAs that perform many biochemical reactions8,9,10,11,12,13,14,15,16, including transfer of an RNA-linked amino acid to their own 5′-amino-modified terminus15. Here we demonstrate the in vitro selection of ribozymes (196 nucleotides) that perform the same peptidyl transferase reaction as the ribosome: that is, they can join amino acids by a peptide bond. Like ribosome substrates, one amino acid (N-blocked methionine) is esterified to the 3′(2′)-O of adenosine, whereas the acceptor amino acid (phenylalanine) has a free amino group. Our best characterized ribozyme recognizes the amino-acid ester substrate by binding its adenosine moiety, and is therefore capable of utilizing Leu- and Phe- as well as Met-derived substrates. Such lack of specificity with respect to the amino acid is a feature necessary for a generalized protein-synthesizing enzyme.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Additional access options:
Figure 1: Peptide bond formation by a ribosome (left) and by a ribozyme (right).
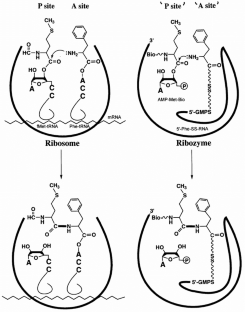
The alternative text for this image may have been generated using AI.
Figure 2: Activity of the selected RNA pool and transcripts from individual clones from generation 19.
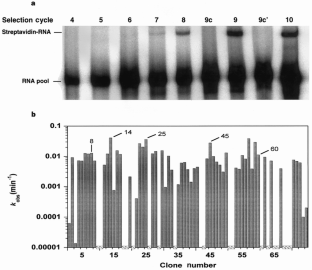
The alternative text for this image may have been generated using AI.
Figure 3: Formation of biotinylated dipeptide catalysed by clone-25 RNA.
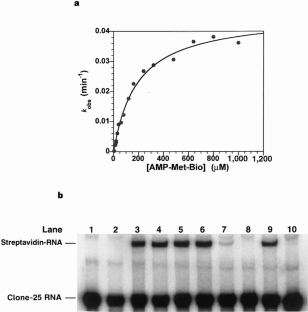
The alternative text for this image may have been generated using AI.
Figure 4: Validation of the peptide bond formed by catalysis of clone-25 RNA by HPLC-ESI/MS analysis of the dipeptide product.
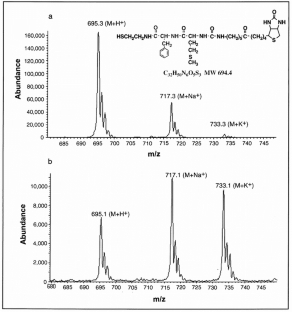
The alternative text for this image may have been generated using AI.
Figure 5: Amino-acid specificity of peptide bond formation catalysed by clone-25 ribozyme.

The alternative text for this image may have been generated using AI.
Similar content being viewed by others
References
- Woese, C. R. in The Genetic Code: The Molecular Basis for Genetic Expression 179 –195 (Harper &Row, New York, ( 1967)).
Google Scholar - Crick, F. H. C. The origin of the genetic code. J. Mol. Biol. 38, 367–379 (1968).
Article CAS Google Scholar - Noller, H. F. in The RNA World (eds Gesteland, R. F. &Atkins, J. F.) 137– 156 (Cold Spring Harbor Laboratory Press, Plainview, NY, ( 1993)).
Google Scholar - Noller, H. F., Hoffarth, V. & Zimniak, L. Unusual resistance of peptidyl transferase to protein extraction procedures. Science 256, 1416 –1419 (1992).
Article ADS CAS Google Scholar - Ellington, A. D. & Szostak, J. W. In vitro selection of RNA molecules that bind specific ligands. Nature 346, 818–822 ( 1990).
Article ADS CAS Google Scholar - Robertson, D. L. & Joyce, G. F. Selection in vitro of an RNA enzyme that specifically cleaves single-stranded DNA. Nature 344, 467–468 (1990).
Article ADS CAS Google Scholar - Tuerk, C. & Gold, L. Systematic evolution of ligands by exponential enrichment: RNA ligands to bacteriophage T4 DNA polymerase. Science 249, 505–510 ( 1990).
Article ADS CAS Google Scholar - Bartel, D. P. & Szostak, J. W. Isolation of new ribozymes from a large pool of random sequences. Science 261, 1411–1418 (1993).
Article ADS CAS Google Scholar - Lorsch, J. R. & Szostak, J. W. In vitro evolution of new ribozymes with polynucleotide kinase activity. Nature 371, 31–36 (1994).
Article ADS CAS Google Scholar - Prudent, J. R., Uno, T. & Schultz, P. G. Expanding the scope of RNA catalysis. Science 264, 1924–1927 ( 1994).
Article ADS CAS Google Scholar - Dai, X.-C., de Mesmaeker, A. & Joyce, G. F. Cleavage of an amide bond by a ribozyme. Science 267, 237–240 ( 1995).
Article ADS CAS Google Scholar - Illangasekare, M., Sanchez, G., Nickles, T. & Yarus, M. Aminoacyl-RNA synthesis catalyzed by an RNA. Science 267, 643– 647 (1995).
Article ADS CAS Google Scholar - Wilson, C. & Szostak, J. W. In vitro evolution of a self-alkylating ribozyme. Nature 374, 777–782 (1995).
Article ADS CAS Google Scholar - Ekland, E. H. & Bartel, D. P. RNA-catalysed RNA polymerization using nucleoside triphosphates. Nature 382, 373–376 (1996).
Article ADS CAS Google Scholar - Lohse, P. A. & Szostak, J. W. Ribozyme-catalysed amino-acid transfer reactions. Nature 381, 442– 444 (1996).
Article ADS CAS Google Scholar - Wecker, M., Smith, D. & Gold, L. In vitro selection of a novel catalytic RNA: characterization of a sulfur alkylation reaction and interaction with a small peptide. RNA 2, 982–994 (1996).
CAS Google Scholar - Nierhaus, K. H., Schultz, H. & Cooperman, B. S. Molecular mechanisms of the ribosomal peptidyltransferase center. Biochem. Int. 1, 185– 192 (1980).
CAS Google Scholar - Pyle, A. M. Ribozymes: a distinct class of metalloenzymes. Science 261, 709–714 (1993).
Article ADS CAS Google Scholar - Maden, B. E. H. & Monro, R. E. Ribosome-catalyzed peptidyl transfer: effects of cations and pH value. Eur. J. Biochem. 6, 309–316 ( 1968).
Article CAS Google Scholar - Piccirilli, J. A., McConnell, T. S., Zaug, A. J., Noller, H. F. & Cech, T. R. Aminoacyl esterase activity of the Tetrahymena ribozyme. Science 256, 1420 –1424 (1992).
Article ADS CAS Google Scholar - Cech, T. R. in The RNA World (eds Gesteland, R. F. &Atkins, J. F.) 239– 269 (Cold Spring Harbor Laboratory Press, Plainview, NY, ( 1993)).
Google Scholar - Bass, B. L. & Cech, T. R. Specific interaction between the self-splicing RNA of Tetrahymena and its guanosine substrate: implications for biological catalysis by RNA. Nature 308, 820–826 (1984).
Article ADS CAS Google Scholar - Sassanfar, M. & Szostak, J. W. An RNA motif that binds ATP. Nature 364, 550–553 (1993).
Article ADS CAS Google Scholar - Monro, R. E., Cerna, J. & Marker, K. A. Ribosome-catalyzed peptidyl transfer: substrate specificity at the P-site. Proc. Natl Acad. Sci. USA 61, 1042–1049 (1968).
Article ADS CAS Google Scholar - Mercer, T. F. B. & Symons, R. H. Peptidyl-donor substrates for ribosomal peptidyl transferase: chemical synthesis and biological activity of N -acetyl aminoacyl di- and trinucleotides. Eur. J. Biochem. 28, 38–45 (1972).
Article CAS Google Scholar - Krayevsky, A. A., Kukhanova, M. K. & Gottikh, B. P. Peptidyl transferase center of bacterial ribosomes: substrate specificity and binding site. Nucleic Acids Res. 2, 2223–2236 (1975).
Article CAS Google Scholar - Azhayev, A. V. et al. Synthesis of amino acid esters of nucleoside 5′-phosphates and their investigation with PMR and CD spectra. Nucleic Acids Res. 4, 2223–2234 ( 1977).
Article CAS Google Scholar
Acknowledgements
We thank K. Goodrich and E. Podell for oligonucleotide synthesis; R. Barkley and O. Averin for HPLC-MS spectra; and B. Golden, R. Gottlieb, G. Joyce, S. Seiwert and O. Uhlenbeck for comments on the manuscript. B.Z. is supported by a postdoctoral fellowship from the National Institute of General Medical Science, NIH. T.R.C. is an investigator of the Howard Hughes Medical Institute and an American Cancer Society Professor.
Author information
Authors and Affiliations
- Department of Chemistry and Biochemistry, Howard Hughes Medical Institute, University of Colorado, Boulder, 80309-0215, Colorado , USA
Biliang Zhang & Thomas R. Cech
Authors
- Biliang Zhang
- Thomas R. Cech
Corresponding author
Correspondence toThomas R. Cech.
Supplementary Information
Rights and permissions
About this article
Cite this article
Zhang, B., Cech, T. Peptide bond formation by in vitro selected ribozymes.Nature 390, 96–100 (1997). https://doi.org/10.1038/36375
- Received: 06 August 1997
- Accepted: 17 September 1997
- Issue date: 06 November 1997
- DOI: https://doi.org/10.1038/36375