The insidious effect of diatoms on copepod reproduction (original) (raw)
- Letter
- Published: 11 November 1999
- G. Barone2,
- G. Romano1,
- S. A. Poulet3,
- A. Ianora1,
- G. L. Russo4,
- I. Buttino1,
- G. Mazzarella4,
- M. Laabir1,
- M. Cabrini5 &
- …
- M. G. Giacobbe6
Nature volume 402, pages 173–176 (1999)Cite this article
- 3106 Accesses
- 573 Citations
- 6 Altmetric
- Metrics details
Abstract
The productive regions of the ocean are characterized by seasonal blooms of phytoplankton which are generally dominated by diatoms. This algal class has, therefore, traditionally been regarded as providing the bulk of the food that sustains the marine food chain to top consumers and important fisheries. However, this beneficial role has recently been questioned on the basis of laboratory studies showing that although dominant zooplankton grazers such as copepods feed extensively on diatoms, the hatching success of eggs thus produced is seriously impaired1. Here we present evidence from the field showing that the hatching success of wild copepods feeding on a diatom-dominated bloom is also heavily compromised, with only 12% of the eggs hatching compared with 90% in post-bloom conditions. We report on the structure of the three aldehydes isolated from diatoms that are responsible for this biological activity, and show that these compounds arrest embryonic development in copepod and sea urchin bioassays and have antiproliferative and apoptotic effects on human carcinoma cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Additional access options:
Figure 1: In situ relationship between diatom densities and copepod reproductive success during three cruises along four transects in the North Adriatic Sea during 1997–1998.
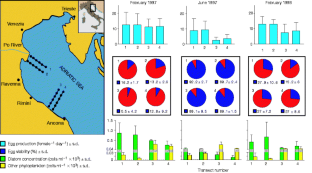
The alternative text for this image may have been generated using AI.
Figure 2: Effect of aldehydes on cell division.
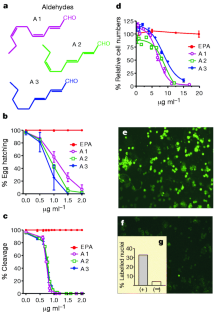
The alternative text for this image may have been generated using AI.
Similar content being viewed by others
References
- Ban,S. et al. The paradox of diatom-copepod interactions. Mar. Ecol. Prog. Ser. 157, 287–293 (1997).
Article ADS Google Scholar - Vollenweider,R. A., Rinaldi,A. & Montanari,G. in Marine Coastal Eutrophication (eds Vollenweider, R. A., Marchetti, R. & Viviani, R.) 63–106 (Elsevier, Amsterdam, 1992).
Book Google Scholar - Peterson,W. T. & Kimmerer,W. J. Processes controlling recruitment of the marine calanoid copepod Temora longicornis in Long Island Sound: egg production, egg mortality, and cohort survival rates. Limnol. Oceanogr. 39, 1594–1605 (1994).
Article ADS Google Scholar - Harkes,P. D. & Begemann,W. J. Identification of some previously unknown aldehydes in cooked chicken. J. Am. Chem. Soc. 51, 356–359 (1974).
CAS Google Scholar - Wendel,T. & Jüttner,F. Lipoxygenase-mediated formation of hydrocarbons and unsaturated aldehydes in freshwater diatoms. Phytochemistry 41, 1445–1449 (1996).
Article CAS Google Scholar - Gerwick,W. H. & Bernart,M. W. in Marine Biotechnology, Vol. 1. Pharmaceutical and Bioactive Natural Products. (eds Attaway, D. H. &Zaborsky, O. R.) 101–152 (Plenum, New York, 1993).
Book Google Scholar - Fogh,J., Wright,W. C. & Loveless,J. D. Absence of HeLa cell contamination in cell lines derived from human tumors. J. Natl Cancer Inst. 58, 209–214 (1977).
Article CAS Google Scholar - Zimmerman,B. T., Crawford,G. D., Dahl,R., Simon,F. T. & Mapoles,J. E. Mechanisms of acetaldehyde-mediated growth inhibition: delayed cell cycle progression and induction of apoptosis. Alcoholism Clin. Exp. Res. 19, 434–440 (1995).
Article CAS Google Scholar - Koski,M., Klein Breteler,W. & Schogt,N. Effect of food quality on rate of growth and development of the pelagic copepod Pseudocalanus elongatus (Copepoda, Calanoida). Mar. Ecol. Prog. Ser. 170, 169–187 (1998).
Article ADS Google Scholar - Harborne,J. B. in Herbivores: Their Interactions with Secondary Plant Metabolites (eds Rosenthal, G. A. & Berenbaum, M. R.) 389–429 (Academic, San Diego, 1991).
Book Google Scholar - Robbins, W. E. et al. Ecdysones and analogs: Effects on development and reproduction of insect. Science 161, 1158–1160 (1968).
Article ADS Google Scholar - Leopold, A. S. et al. Phytoestrogens: Adverse effects on reproduction in California quail. Science 191, 98–100 (1976).
Article ADS Google Scholar - Dutz,J. Repression of fecundity in the meritic copepod Acartia clausi exposed to the toxic dinoflagellate Alexandrium lusitanicum: relationship between feeding and egg production. Mar. Ecol. Prog. Ser. 175, 97–107 (1998).
Article ADS Google Scholar - White,A. W. Marine zooplankton can accumulate and retain dinoflagellate toxins and cause fish kills. Limnol. Oceanogr. 26, 103–109 (1981).
Article ADS Google Scholar - Kleppel,G. S., Holliday,D. V. & Pieper,R. E. Trophic interactions between copepods and microplankton: a question about the role of diatoms. Limnol. Oceanogr. 36, 172–178 (1991).
Article ADS Google Scholar - Runge,J. A. & de Lafontaine,Y. Characterization of the pelagic ecosystem in surface waters of the northern Gulf of St. Lawrence in early summer: the larval redfish-_Calanus_-microplankton interaction. Fish. Oceanogr. 5, 21–37 (1996).
Article Google Scholar - Smetacek,V. S. Role of sinking in diatom life-history cycles: ecological, evolutionary and geological significance. Mar. Biol. 84, 239–251 (1985).
Article Google Scholar - Ianora,A., Poulet,S. A., Miralto,A. & Grottoli,R. The diatom Thalassiosira rotula affects reproductive success in the copepod Acartia clausi. Mar. Biol. 125, 279–286 (1996).
Article Google Scholar - Buttino,I., Miralto,A., Ianora,A., Romano,G. & Poulet,S. A. Water soluble extracts of the diatom Thalassiosira rotula induce aberrations in embryonic tubulin organisation of sea urchin Paracentrotus lividus. Mar. Biol. 134, 147–154 (1999).
Article Google Scholar
Acknowledgements
We thank E. Trivellone and T. Tancredi of the Core Facility, CNR Naples, for NMR assistance, and M. Di Pinto, F. Esposito and V. Mirra for technical help. This work was funded by the Stazione Zoologica and Roscoff Marine Station and an Italian MURST programme PRISMA II, Biogeochemical Cycles.
Author information
Authors and Affiliations
- Stazione Zoologica “A. Dohrn”, Villa Comunale, Naples, 80121, Italy
A. Miralto, G. Romano, A. Ianora, I. Buttino & M. Laabir - Dipartimento di Chimica Organica e Biologica, Università di Napoli “Federico II”, Via Mezzocannone 16, Naples, 80134, Italy
G. Barone - Station Biologique de Roscoff, CNRS, Place Georges Teissier, Roscoff, 29682, France
S. A. Poulet - Istituto di Scienze dell'Alimentazione, CNR, Avellino, 83100, Italy
G. L. Russo & G. Mazzarella - Laboratorio di Biologia Marina, Via August Piccard 54, Trieste, 34040, Italy
M. Cabrini - Istituto Sperimentale Talassografico CNR, Via San Ranieri 86, Messina, 98122, Italy
M. G. Giacobbe
Authors
- A. Miralto
- G. Barone
- G. Romano
- S. A. Poulet
- A. Ianora
- G. L. Russo
- I. Buttino
- G. Mazzarella
- M. Laabir
- M. Cabrini
- M. G. Giacobbe
Corresponding author
Correspondence toA. Miralto.
Supplementary information
Rights and permissions
About this article
Cite this article
Miralto, A., Barone, G., Romano, G. et al. The insidious effect of diatoms on copepod reproduction.Nature 402, 173–176 (1999). https://doi.org/10.1038/46023
- Received: 12 May 1999
- Accepted: 20 August 1999
- Issue date: 11 November 1999
- DOI: https://doi.org/10.1038/46023