In vivo antigen challenge in celiac disease identifies a single transglutaminase-modified peptide as the dominant A-gliadin T-cell epitope (original) (raw)
- Article
- Published: March 2000
Nature Medicine volume 6, pages 337–342 (2000) Cite this article
- 3131 Accesses
- 505 Citations
- 34 Altmetric
- Metrics details
Abstract
Celiac disease (CD) is an increasingly diagnosed enteropathy (prevalence, 1:200–1:300)1 that is induced by dietary exposure to wheat gliadins2 (as well as related proteins in rye and barley) and is strongly associated with HLA-DQ2 (α1*0501, β1*0201), which is present in over 90% of CD patients3. Because a variety of gliadin peptides have been identified as epitopes for gliadin-specific T-cell clones4,5,6 and as bioactive sequences in feeding studies and in ex vivo CD intestinal biopsy challenge7,8,9, it has been unclear whether a ‘dominant’ T-cell epitope is associated with CD. Here, we used fresh peripheral blood lymphocytes from individual subjects undergoing short-term antigen challenge and tissue transglutaminase-treated, overlapping synthetic peptides spanning A-gliadin to demonstrate a transient, disease-specific, DQ2-restricted, CD4 T-cell response to a single dominant epitope. Optimal gamma interferon release in an ELISPOT assay was elicited by a 17-amino-acid peptide corresponding to the partially deamidated peptide of A-gliadin amino acids 57–73 (Q65E). Consistent with earlier reports indicating that host tissue transglutaminase modification of gliadin enhances gliadin-specific CD T-cell responses10, tissue transglutaminase specifically deamidated Q65 in the peptide of A-gliadin amino acids 56–75. Discovery of this dominant epitope may allow development of antigen-specific immunotherapy for CD.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Additional access options:
Figure 1: Amino-acid sequence of A-gliadin

The alternative text for this image may have been generated using AI.
Figure 2: Ex vivo IFN-γ responses of PBMCs to A-gliaden peptides.

The alternative text for this image may have been generated using AI.
Figure 3: Definition of the minimal epitope in tTG-treated pool 3.
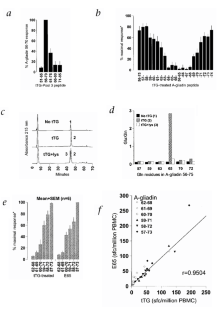
The alternative text for this image may have been generated using AI.
Figure 4: Analysis of dominant A-gliadin epitope.
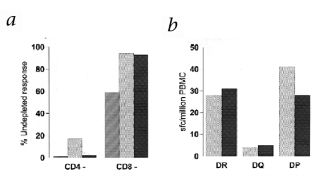
The alternative text for this image may have been generated using AI.
Figure 5: Dynamics of PBMC IFN-γ responses by ELISPOT assay and proliferative responses.
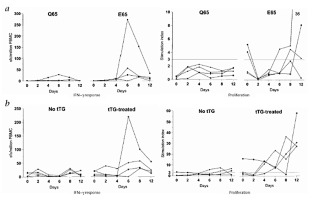
The alternative text for this image may have been generated using AI.
Similar content being viewed by others
References
- Hin, H., Bird, G., Fisher, P., Mahy, N. & Jewell, D. Coeliac disease in primary care: case finding study. Br. Med. J. 318, 164–167 (1999).
Article CAS Google Scholar - Kendall, M., Schneider, R., Cox, P. & Hawkins, C. Gluten subfractionation in coeliac disease. Lancet 2, 1065–1067 (1972).
Article CAS Google Scholar - Sollid, L.M., Thorsby, E. HLA susceptibility genes in celiac disease: genetic mapping and role in pathogenesis. Gastroenterol. 105, 910–922 (1993).
Article CAS Google Scholar - Molberg, O. et al. Gliadin specific, HLA DQ2-restricted T cells are commonly found in small intestinal biopsies from coeliac disease patients, but not from controls. Scand. J. Immunol. 46, 103–109 (1997).
Article CAS Google Scholar - van der Wal, Y. et al. Selective deamidation by tissue transglutaminase strongly enhances gliadin-specific T cell reactivity. J. Immunol. 161, 1585–1588 (1998).
Google Scholar - Sjostrom, H. et al. Identification of a gliadin T-cell epitope in coeliac disease: general importance of gliadin deamidation for intestinal T-cell recognition. Scand. J. Immunol. 48, 111–115 (1998).
Article CAS Google Scholar - Mantzaris, G. & Jewell, D. In vivo toxicity of a synthetic dodecapeptide from A gliadin in patients with coeliac disease. Scand. J. Gastroenterol. 26, 392–398 (1991).
Article CAS Google Scholar - de Ritis, G. et al. In vitro (organ culture) studies of the toxicity of specific A-gliadin peptides in celiac disease. Gastroenterology 94, 41–49 (1988).
Article CAS Google Scholar - Mauri, L. et al. In vitro activities of A-gliadin-related synthetic peptides. Scand. J. Gastroenterol. 31, 247–253 (1996).
Article Google Scholar - Molberg, O. et al. Tissue transglutaminase selectively modifies gliadin peptides that are recognized by gut-derived T cells in celiac disease. Nature Med. 4, 713–717 (1998).
Article CAS Google Scholar - Kasarda, D. et al. Nucleic acid (cDNA) and amino acid sequence of α-type gliadins from wheat (Tricetum aestivum). Proc. Natl. Acad. Sci. USA 81, 4712–4716 (1984).
Article CAS Google Scholar - Gutgemann, I., Fahrer, A., Altman, J., Davis, M. & Chien, Y-h. Induction of rapid T cell activation and tolerance by systemic presentation of orally administered antigen. Immunity 8, 667–673 (1998).
- Greenbergm, C.S., Birckbichlerm, P. & Ricem, R. Transglutaminases: multifunctional cross-linking enzymes that stablize tissues. FASEB J. 5, 3071–3077 (1991).
Article Google Scholar - Lehmann, P.V., Forsthuber, T., Miller, A. & Serzarz, E.E. Spreading of T-cell autoimmunity to cryptic determinants of an autoantigen. Nature 359, 155–157 (1992).
Article Google Scholar - Moskophidis, D., Lechner, F., Pircher, H. & Zinkernagel, R. Virus persistence in acutely infected immunocompetent mice by exhaustion of antiviral cytotoxic effector T cells. Nature 362, 758–761 (1993).
Article CAS Google Scholar - Lahat, N. et al. Cytokine profile in coeliac disease. Scand. J. Immunol. 49, 441–446 (1999).
Article CAS Google Scholar - Vartdal, F. et al. The binding motif of the disease associated HLA-DQ (α1*0501, β1*0201) molecule. Eur. J. Immunol. 26, 2764–2772 (1996).
Article CAS Google Scholar - Ward, R. Codex alimentarius- the proposed gluten-free standard (position paper) (The Coeliac Society of the United Kingdom, High Wycombe, UK, 1999).
- Bunce, M. et al. Phototyping: comprehensive DNA typing for HLA-A, B, C, DRB1, DRB3, DRB4, DRB5 & DQB1 by PCR with 144 primer mixes utilizing sequence-specific primers (PCR-SSP). Tissue Antigens 46, 355–367 (1995).
Article CAS Google Scholar - Olerup, O., Aldener, A. & Fogdell, A. HLA-DQB1 and DQA1 typing by PCR amplification with sequence-specific primers in 2 hours. Tissue Antigens 41, 119–134 (1993).
Article CAS Google Scholar - Mullighan, C.G., Bunce, M. & Welsh, K.I. High-resolution HLA-DQB1 typing using the polymerase chain reaction and sequence-specific primers. Tissue Antigens. 50, 688–692 (1997).
Article CAS Google Scholar - Plebanski, M. et al. Protection from Plasmodium berghei infection by priming and boosting T cells to a single class I restricted epitope with recombinant carriers suitable for human use. Eur. J. Immunol. 28, 4345–4355 (1998).
Article CAS Google Scholar
Acknowledgements
We thank A. Willis for HPLC and amino-acid analysis; G. Bird and H. Griffith for assays of antibody against endomysium; M. Bunce for tissue typing; A. Pathan and A. Lalvani for the gift of antibodies against class II; A. Connell for dietary advice; and J. Simmons and A. Ellis for referring subjects for the study. R.P.A. was supported by a CJ Martin/Menzies Fellowship from the National Health and Medical Research Council of Australia 987001. A.V.S.H. is a Wellcome Trust Principal Research Fellow. The work was supported by the Coeliac Society of the United Kingdom and Isis Innovation (University of Oxford, UK).
Author information
Authors and Affiliations
- Nuffield Department of Medicine, Institute of Molecular Medicine, John Radcliffe Hospital, University of Oxford, Oxford, OX3 9DU, UK
Robert P. Anderson, Pilar Degano, Andrew J. Godkin & Adrian V.S. Hill - Nuffield Department of Medicine, Gastroenterology Unit, John Radcliffe Hospital, University of Oxford, Oxford, OX3 9DU, UK
Robert P. Anderson & Derek P. Jewell
Authors
- Robert P. Anderson
- Pilar Degano
- Andrew J. Godkin
- Derek P. Jewell
- Adrian V.S. Hill
Corresponding author
Correspondence toRobert P. Anderson.
Rights and permissions
About this article
Cite this article
Anderson, R., Degano, P., Godkin, A. et al. In vivo antigen challenge in celiac disease identifies a single transglutaminase-modified peptide as the dominant A-gliadin T-cell epitope.Nat Med 6, 337–342 (2000). https://doi.org/10.1038/73200
- Received: 18 August 1999
- Accepted: 29 December 1999
- Issue date: March 2000
- DOI: https://doi.org/10.1038/73200