Total colourblindness is caused by mutations in the gene encoding the α-subunit of the cone photoreceptor cGMP-gated cation channel (original) (raw)
- Letter
- Published: July 1998
- Tim Marx4,
- Ian Giddings1,3,
- Herbert Jägle2,3,
- Samuel G. Jacobson5,
- Eckhart Apfelstedt-Sylla3,
- Eberhart Zrenner3,
- Lindsay T. Sharpe2,3 &
- …
- Bernd Wissinger1
Nature Genetics volume 19, pages 257–259 (1998) Cite this article
- 1710 Accesses
- 310 Citations
- 7 Altmetric
- Metrics details
Abstract
Total colourblindness (OMIM 216900), also referred to as rod monochromacy (RM) or complete achromatopsia, is a rare, autosomal recessive inherited and congenital disorder characterized by photophobia, reduced visual acuity, nystagmus and the complete inability to discriminate between colours1. Electroretinographic recordings show that in RM, rod photoreceptor function is normal, whereas cone photoreceptor responses are absent. The locus for RM has been mapped to chromosome 2q11 (ref. 2), however the gene underlying RM has not yet been identified. Recently, a suitable candidate gene, CNGA3, encoding the α-subunit of the cone photoreceptor cGMP-gated cation channel, a key component of the phototransduction pathway, has been cloned and assigned to human chromosome 2q11 (Refs 3,4). We report the identification of missense mutations in CNGA3 in five families with RM. Homozygous mutations are present in two families, whereas the remaining families show compound heterozygous mutations. In all cases, the segregation pattern of the mutations is consistent with the autosomal recessive inheritance of the disease and all mutations affect amino acids that are highly conserved among cyclic nucleotide gated channels (CNG) in various species. This is the first report of a colour vision disorder caused by defects other than mutations in the cone pigment genes, and implies at least in this instance a common genetic basis for phototransduction in the three different cone photoreceptors of the human retina.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Additional access options:
Figure 1: Sequences of mutations found in selected RM patients.
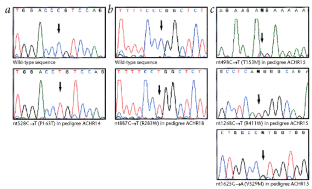
The alternative text for this image may have been generated using AI.
Figure 2: RFLP-based co-segregation analysis of CNGA3 mutations within three selected pedigrees.

The alternative text for this image may have been generated using AI.
Figure 3: Putative topology of the human cone cGMP-gated cation channel α-subunit.

The alternative text for this image may have been generated using AI.
Similar content being viewed by others
Accession codes
Accessions
GenBank/EMBL/DDBJ
References
- Sharpe, L.T. & Nordby, K. Total colour blindness: An introduction . in Night vision: basic, clinical and applied aspects (eds Hess, R.F., Sharpe, L.T. & Nordby, K., Cambridge University Press, Cambridge, 1990).
Google Scholar - Arbour, N. et al. Homozygosity mapping of achromatopsia to chromosome 2 using DNA pooling. Hum. Mol. Genet. 6, 689–694 (1997).
Article CAS Google Scholar - Yu, W.-P., Grunwald, M.E. & Yau, K.-W. Molecular cloning, functional expression and chromosomal localization of a human homolog of the cyclic nucleotide-gated ion channel of retinal cone photoreceptors . FEBS Lett. 393, 211–215 (1996).
Article CAS Google Scholar - Wissinger, B. et al. Cloning, chromosomal localization and functional expression of the gene encoding the α-subunit of the cGMP-gated channel in human cone photoreceptors . Eur. J. Neurosci. 9, 2512– 2521 (1997).
Article CAS Google Scholar - Wissinger, B. et al. Human rod monochromacy: linkage analysis and mapping of a cone photoreceptor expressed candidate gene on chromosome 2q11. Genomics (in press).
- Baumann, A., Frings, S., Godde, M., Seifert, R. & Kaupp, U.B. Primary structure and functional expression of a drosophila cyclic nucleotide-gated channel present in eyes and antennae. EMBO J. 13, 5040–5050 ( 1994).
Article CAS Google Scholar - Coburn, C.M. & Bargmann, C.I. A putative cyclic nucleotide-gated channel is required for sensory development and function in C. elegans . Neuron 17, 695–706 (1996).
Article CAS Google Scholar - Weyand, I. et al. Cloning and functional expression of a cyclic-nucleotide-gated channel from mammalian sperm. Nature 368, 859– 863 (1994).
Article CAS Google Scholar - Bönigk, W. et al. Rod and cone photoreceptor cells express distinct genes for cGMP-gated channels . Neuron 10, 865–877 (1993).
Article Google Scholar - Pittler, S.J. et al. Primary structure and chromosomal localization of human and mouse rod photoreceptor cGMP-gated cation channel. J. Biol. Chem. 267, 6257–6262 (1992).
CAS Google Scholar - Zagotta, W.N. & Siegelbaum, S.A. Structure and function of the cyclic nucleotide-gated channels. Annu. Rev. Neurosci. 19, 235–263 (1996).
Article CAS Google Scholar - Brown, R.L., Gramling, R., Bert, R.J. & Karpen, J.W. Cyclic GMP contact points within the 63kDa subunit and a 240kDa associated protein of retinal rod cGMP-activated channels. Biochemistry 34, 8365–8370 (1995).
Article CAS Google Scholar - Dryja, T.P. et al. Mutations in the gene encoding the α-subunit of the rod cGMP-gated channel in autosomal recessive retinitis pigmentosa. Proc. Natl Acad. Sci. USA 92, 10177–10181 ( 1995).
Article CAS Google Scholar - Dryja, T.P., Berson, E.L., Rao, V.R. & Oprian, D.D. Heterozygous missense mutation in the rhodopsin gene as a cause of congenital stationary night blindness. Nature Genet. 4, 280– 283 (1993).
Article CAS Google Scholar - Fain, G.L. & Lisman, J.E. Photoreceptor degeneration in vitamin A deprivation and retinitis pigmentosa: the equivalent light hypothesis. Exp. Eye Res. 57, 335–340 (1993).
Article CAS Google Scholar - Payne, A.M. et al. A mutation in guanylate cyclase activator 1A (GUCA1A) in an autosomal dominant cone dystrophy pedigree mapping to a new locus on chromosome 6p21.1. Hum. Mol. Genet. 7, 273–277 (1998).
Article CAS Google Scholar - Semple-Dowland, S.L., Lee, N.R., Van Hooser, P., Palczewski, K. & Baehr, W. A null mutation in the photoreceptor guanylate cyclase gene causes the retinal degeneration chicken phenotype. Proc. Natl Acad. Sci. USA 95, 1271– 1276 (1998).
Article Google Scholar - Sharpe, L.T. & Nordby, K. The photoreceptors in the achromat . in Night vision: basic, clinical and applied aspects (eds Hess, R.F., Sharpe, L.T. & Nordby, K., Cambridge University Press, Cambridge, 1990).
Google Scholar
Acknowledgements
This work has been supported by grants of the Deutsche Forschungsgemeinschaft and the Bundesministerium für Bildung, Wissenschaft, Forschung und Technologie to B.W. (SFB430 A5) and to L.T.S. (IKFZ IB3), respectively, and in part from grants of the NIH (EY-05627) and the Foundation Fighting Blindness to S.G.J. We would like to thank D. Hanna and M. Anastasi for providing RM families for investigation.
Author information
Authors and Affiliations
- Molekulargenetisches Labor, Universitäts-Augenklinik Tübingen, Schleichstr. 12-16, Tübingen, 72076, Germany
Susanne Kohl, Ian Giddings & Bernd Wissinger - Psychophysisches Labor, Forschungsstelle für Experimentelle Ophthalmologie, Universitäts-Augenklinik Tübingen, Schleichstr. 12-16, Tübingen, 72076, Germany
Herbert Jägle & Lindsay T. Sharpe - Abteilung für Pathophysiologie des Sehens und Neuroophthalmologie, Universitäts-Augenklinik Tübingen, Schleichstr. 12-16, Tübingen, 72076, Germany
Susanne Kohl, Ian Giddings, Herbert Jägle, Eckhart Apfelstedt-Sylla, Eberhart Zrenner & Lindsay T. Sharpe - Institut für Pathologie, Universität Tübingen, Liebermeisterstr. 8, Tübingen, 72076, Germany
Tim Marx - Department of Ophthalmology, Scheie Eye Institute, University of Pennsylvania, Philadelphia, 19104-2689, Pennsylvania, USA
Samuel G. Jacobson
Authors
- Susanne Kohl
- Tim Marx
- Ian Giddings
- Herbert Jägle
- Samuel G. Jacobson
- Eckhart Apfelstedt-Sylla
- Eberhart Zrenner
- Lindsay T. Sharpe
- Bernd Wissinger
Corresponding author
Correspondence toBernd Wissinger.
Rights and permissions
About this article
Cite this article
Kohl, S., Marx, T., Giddings, I. et al. Total colourblindness is caused by mutations in the gene encoding the α-subunit of the cone photoreceptor cGMP-gated cation channel.Nat Genet 19, 257–259 (1998). https://doi.org/10.1038/935
- Received: 02 April 1998
- Accepted: 28 May 1998
- Issue date: July 1998
- DOI: https://doi.org/10.1038/935