Motion-induced spatial conflict (original) (raw)
- Letter
- Published: 11 September 2003
Nature volume 425, pages 181–184 (2003)Cite this article
- 1245 Accesses
- 30 Citations
- 4 Altmetric
- Metrics details
Abstract
Borders defined by small changes in brightness (luminance contrast) or by differences in colour (chromatic contrast) appear to move more slowly than those defined by strong luminance contrast1,2,3,4. As spatial coding is influenced by motion5,6,7, if placed in close proximity, the different types of moving border might appear to drift apart8. Using this configuration, we show here that observers instead report a clear illusory spatial jitter of the low-luminance-contrast boundary. This visible interaction between motion and spatial-position coding occurred at a characteristic rate (∼ 22.3 Hz), although the stimulus motion was continuous and invariant. The jitter rate did not vary with the speed of movement. The jitter was not due to small involuntary movements of the eyes, because it only occurred at a specific point within the stimulus, the low-luminance-contrast boundary. These findings show that the human visual system contains a neural mechanism that periodically resolves the spatial conflict created by adjacent moving borders that have the same physical but different perceptual speeds.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Additional access options:
Figure 1: ‘Fluttering hearts’ illusion.

The alternative text for this image may have been generated using AI.
Figure 2: Schematic diagrams of the stimulus configurations.
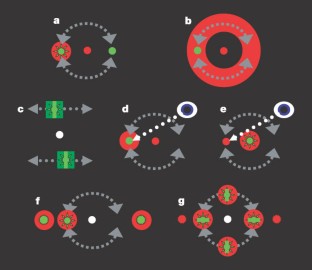
The alternative text for this image may have been generated using AI.
Figure 3: Perceived jitter.
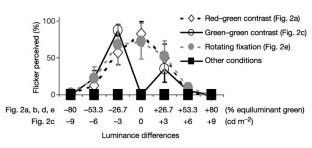
The alternative text for this image may have been generated using AI.
Figure 4: Perceived jitter rate.
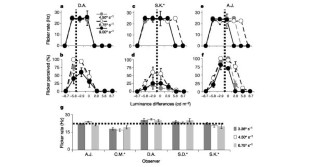
The alternative text for this image may have been generated using AI.
Similar content being viewed by others
References
- Anstis, S. Footsteps and inchworms: Illusions show that contrast affects apparent speed. Perception 30, 785–794 (2001)
Article CAS Google Scholar - Blakemore, M. R. & Snowden, R. J. The effect of contrast upon perceived speed: a general phenomenon? Perception 28, 33–48 (1999)
Article CAS Google Scholar - Cavanagh, P., Tyler, C. W. & Favreau, O. E. Perceived velocity of moving chromatic gratings. J. Opt. Soc. Am. A1, 893–899 (1984)
Article ADS Google Scholar - Thompson, P. Perceived rate of movement depends on contrast. Vision Res. 22, 377–380 (1982)
Article CAS Google Scholar - De Valois, R. L. & De Valois, K. K. Vernier acuity with stationary moving Gabors. Vision Res. 31, 1619–1626 (1991)
Article CAS Google Scholar - Nishida, S. & Johnston, A. Influence of motion signals on the perceived position of spatial pattern. Nature 397, 610–612 (1999)
Article ADS CAS Google Scholar - Whitney, D. & Cavanagh, P. Motion distorts visual space: shifting the perceived position of remote stationary objects. Nature Neurosci. 3, 954–959 (2000)
Article CAS Google Scholar - Nguyen-Tri, D. & Faubert, J. The fluttering-heart illusion: a new hypothesis. Perception 32, 627–634 (2003)
Article Google Scholar - Helmholtz, H. Treatise on Physiological Optics (Dover, New York, 1962)
Google Scholar - von Grunau, M. W. The “fluttering heart” and spatio-temporal characteristics of color processing III. Interactions between the systems of the rods and the long-wavelength cones. Vision Res. 16, 397–401 (1976)
Article CAS Google Scholar - Bridgeman, B. & Stark, L. Ocular proprioception and efference copy in registering visual direction. Vision Res. 31, 1903–1913 (1991)
Article CAS Google Scholar - Sherrington, C. S. Observations on the sensual role of the proprioceptive nerve supply of the extrinsic ocular muscles. Brain 41, 332–343 (1918)
Article Google Scholar - Wiesel, T. N. & Hubel, D. H. Spatial and chromatic interactions in the lateral geniculate body of the rhesus monkey. J. Neurophysiol. 29, 1115–1116 (1966)
Article CAS Google Scholar - Campbell, F. W., Robson, J. G. & Westheimer, G. Fluctuations of accommodation under steady viewing. J. Physiol. (Lond.) 145, 579–594 (1959)
Article CAS Google Scholar - Murakami, I. & Cavanagh, P. A jitter after-effect reveals motion-based stabilization of vision. Nature 395, 798–801 (1998)
Article ADS CAS Google Scholar - Murakami, I. Illusory jitter in a static stimulus surrounded by a synchronously flickering pattern. Vision Res. 43, 957–969 (2003)
Article MathSciNet Google Scholar - Von der Marlsburg, C. & Schneider, W. A neural cocktail-party processor. Biol. Cybern. 54, 29–40 (1986)
Article Google Scholar - Engel, A. K., Konig, P., Kreiter, A. K. & Singer, W. Interhemispheric synchronization of oscillatory neuronal responses in cat visual cortex. Science 252, 1177–1179 (1991)
Article ADS CAS Google Scholar - Engel, A. K. & Singer, W. Temporal binding and the neural correlates of sensory awareness. Trends Cogn. Sci. 5, 16–25 (2001)
Article Google Scholar - Anstis, S. M. & Cavanagh, P. in Color Vision: Physiology and Psychophysics (eds Mollon, J. D. & Sharpe, L. T.) 155–166 (Academic, London, 1983)
Google Scholar
Acknowledgements
We are grateful to C. Clifford, J. Dale, F. Kandil, S. Nishida and Q. Zaidi for their suggestions and comments.
Author information
Authors and Affiliations
- Department of Psychology and Institute of Cognitive Neuroscience, University College London, Gower Street, WC1E 6BT, London, UK
Derek H. Arnold & Alan Johnston
Authors
- Derek H. Arnold
- Alan Johnston
Corresponding author
Correspondence toDerek H. Arnold.
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Arnold, D., Johnston, A. Motion-induced spatial conflict.Nature 425, 181–184 (2003). https://doi.org/10.1038/nature01955
- Received: 03 July 2003
- Accepted: 24 July 2003
- Issue date: 11 September 2003
- DOI: https://doi.org/10.1038/nature01955
This article is cited by
Visual predictions, neural oscillations and naïve physics
- Blake W. Saurels
- Wiremu Hohaia
- Derek H. Arnold
Scientific Reports (2021)
Discretized Theta-Rhythm Perception Revealed by Moving Stimuli
- Ryohei Nakayama
- Isamu Motoyoshi
- Takao Sato
Scientific Reports (2018)