Iron corrosion by novel anaerobic microorganisms (original) (raw)
- Letter
- Published: 26 February 2004
- Jan Kuever1,2,
- Marc Mußmann1,
- Achim W. Hassel3,
- Martin Stratmann3 &
- …
- Friedrich Widdel1
Nature volume 427, pages 829–832 (2004) Cite this article
- 8335 Accesses
- 735 Citations
- 7 Altmetric
- Metrics details
Abstract
Corrosion of iron presents a serious economic problem. Whereas aerobic corrosion is a chemical process1, anaerobic corrosion is frequently linked to the activity of sulphate-reducing bacteria (SRB)2,3,4,5,6. SRB are supposed to act upon iron primarily by produced hydrogen sulphide as a corrosive agent3,5,7 and by consumption of ‘cathodic hydrogen’ formed on iron in contact with water2,3,4,5,6,8. Among SRB, Desulfovibrio species—with their capacity to consume hydrogen effectively—are conventionally regarded as the main culprits of anaerobic corrosion2,3,4,5,6,8,9,10; however, the underlying mechanisms are complex and insufficiently understood. Here we describe novel marine, corrosive types of SRB obtained via an isolation approach with metallic iron as the only electron donor. In particular, a _Desulfobacterium_-like isolate reduced sulphate with metallic iron much faster than conventional hydrogen-scavenging Desulfovibrio species, suggesting that the novel surface-attached cell type obtained electrons from metallic iron in a more direct manner than via free hydrogen. Similarly, a newly isolated _Methanobacterium_-like archaeon produced methane with iron faster than do known hydrogen-using methanogens, again suggesting a more direct access to electrons from iron than via hydrogen consumption.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Additional access options:
Figure 1: Microscopy of cultures.
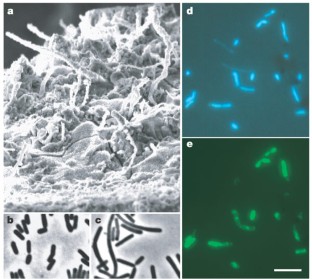
The alternative text for this image may have been generated using AI.
Figure 2: Phylogenetic relationships (based on 16S rRNA gene sequences) of new isolates of sulphate-reducing bacteria from enrichment cultures with metallic iron (strains IS4 and IS5) or hydrogen (strain HS2) and sulphate.
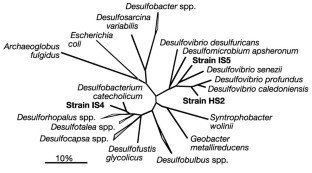
The alternative text for this image may have been generated using AI.
Figure 3: Incubation experiments with iron granules (30 g in 150 ml medium) as sole electron donor.
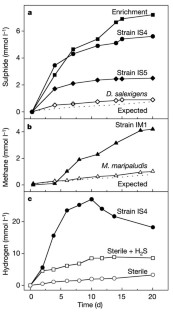
The alternative text for this image may have been generated using AI.
Similar content being viewed by others
References
- Uhlig, H. H. Corrosion and Corrosion Control 3rd edn (Wiley, New York, 1985)
Google Scholar - Hamilton, W. A. Microbially influenced corrosion as a model system for the study of metal microbe interactions: a unifying electron transfer hypothesis. Biofouling 19, 65–76 (2003)
Article CAS Google Scholar - Lee, W., Lewandowski, Z., Nielsen, P. H. & Hamilton, W. A. Role of sulfate-reducing bacteria in corrosion of mild steel: a review. Biofouling 8, 165–194 (1995)
Article CAS Google Scholar - Pankhania, I. P. Hydrogen metabolism in sulphate-reducing bacteria and its role in anaerobic corrosion. Biofouling 1, 27–47 (1988)
Article CAS Google Scholar - Widdel, F. in Biotechnology Focus 3 (eds Finn, R. K. et al.) 277–318 (Hanser, Munich, 1992)
Google Scholar - Cord-Ruwisch, R. in Environmental Microbe-Metal Interaction (ed. Lovley, D. R.) 159–173 (ASM Press, Washington, DC, 2000)
Book Google Scholar - Costello, J. A. Cathodic depolarization by sulphate-reducing bacteria. S. Afr. J. Sci. 70, 202–204 (1974)
CAS Google Scholar - von Wolzogen Kuehr, C. A. H. & van der Vlugt, I. S. The graphitization of cast iron as an electrobiochemical process in anaerobic soil. Water 18, 147–165 (1934)
Google Scholar - Booth, G. H. & Tiller, A. K. Cathodic characteristic of mild steel in suspension of sulphate-reducing bacteria. Corros. Sci. 8, 583–600 (1968)
Article CAS Google Scholar - Pankhania, I. P., Moosavi, A. N. & Hamilton, W. A. Utilization of cathodic hydrogen by Desullfovibrio vulgaris (Hildenborough). J. Gen. Microbiol. 132, 3357–3365 (1986)
CAS Google Scholar - Iverson, W. P. & Olson, G. J. in Current Perspectives in Microbial Ecology (eds Klug, M. J. & Reddy, C. A.) 623–627 (ASM, Washington, DC, 1984)
Google Scholar - Bockris, J. O'M. & Reddy, A. K. N. Modern Electrochemistry Vol. 2 (Plenum, New York, 1970)
Google Scholar - Beech, I. B. et al. Study of parameters implicated in the biodeterioration of mild steel in the presence of different species of sulphate-reducing bacteria. Int. Biodeter. Biodegrad. 34, 289–303 (1994)
Article ADS CAS Google Scholar - Rabus, R., Hansen, T. & Widdel, F. in The Prokaryotes: An Evolving Electronic Resource for the Microbiological Community (eds Dworkin, M., Falkow, S., Rosenberg, E., Schleifer, K.-H. & Stackebrandt, E.) (Springer, New York, 2000)
Google Scholar - Widdel, F. & Bak, F. in The Prokaryotes 2nd edn Vol. 6 (eds Balows, A., Trüper, H. G., Dworkin, M., Harder, W. & Schleifer, K.-H.) 3352–3378 (Springer, New York, 1992)
Book Google Scholar - Hardy, J. A. Utilisation of cathodic hydrogen by sulphate-reducing bacteria. Br. Corros. J. 18, 190–193 (1983)
Article CAS Google Scholar - Laishley, E. J. & Bryant, R. D. in Biochemistry and Physiology of Anaerobic Bacteria (eds Ljungdahl, L. G., Adams, M. W., Barton, L. L., Ferry, J. G. & Johnson, M. K.) 252–260 (Springer, New York, 2003)
Book Google Scholar - Cord-Ruwisch, R. & Widdel, F. Corroding iron as a hydrogen source for sulphate reduction in growing cultures of sulphate-reducing bacteria. Appl. Microbiol. Biotechnol. 25, 169–174 (1986)
Article CAS Google Scholar - Daniels, L., Belay, N., Rajagopal, B. S. & Weimer, P. J. Bacterial methanogenesis and growth from CO2 with elemental iron as the sole source of electrons. Science 237, 509–511 (1987)
Article ADS CAS Google Scholar - Deckena, S. & Blotevogel, K.-H. Fe0-oxidation in the presence of methanogenic and sulphate-reducing bacteria and its possible role in anaerobic corrosion. Biofouling 5, 287–293 (1992)
Article CAS Google Scholar - Schlegel, H. G. General Microbiology 7th edn (Cambridge Univ. Press, Cambridge, 1993)
Google Scholar - Manz, W., Eisenbrecher, M., Neu, T. R. & Szewzyk, U. Abundance and spatial organization of Gram-negative sulfate-reducing bacteria in activated sludge investigated by in situ probing with specific 16S rRNA targeted oligonucleotides. FEMS Microbiol. Ecol. 25, 43–61 (1998)
Article CAS Google Scholar - Appia-Ayme, C., Guiliani, N., Ratouchniak, J. & Bonnefoy, V. Characterization of an operon encoding two _c_-type cytochromes, an _aa_3-type cytochrome oxidase, and rusticyanin in Thiobacillus ferrooxidans ATCC 33020. Appl. Environ. Microbiol. 65, 4781–4787 (1999)
CAS PubMed PubMed Central Google Scholar - Bond, D. R. & Lovley, D. R. Electricity production by Geobacter sulfurreducens attached to electrodes. Appl. Environ. Microbiol. 69, 1548–1555 (2003)
Article CAS Google Scholar - Deppenmeier, U. The unique biochemistry of methanogenesis. Prog. Nucleic Acid Res. Mol. Biol. 71, 223–283 (2002)
Article CAS Google Scholar - Muyzer, G., Teske, A., Wirsen, C. O. & Jannasch, H. W. Phylogenetic relationships of Thiomicrospira species and their identification in deep-sea hydrothermal vent samples by denaturing gradient gel electrophoresis of 16S rDNA fragments. Arch. Microbiol. 164, 165–172 (1995)
Article CAS Google Scholar - Huber, H. et al. A new phylum of Archaea represented by a nanosized hyperthermophilic symbiont. Nature 417, 63–67 (2002)
Article ADS CAS Google Scholar - Ludwig, W. et al. ARB: a software environment for sequence data. (Department of Microbiology, Technical Univ. Munich, 2002); available at 〈http://www.arb-home.de/〉.
- Pernthaler, A., Pernthaler, J. & Amann, R. Fluorescence in situ hybridization and catalyzed reporter deposition for the identification of marine bacteria. Appl. Environ. Microbiol. 68, 3094–3101 (2002)
Article CAS Google Scholar
Acknowledgements
We thank K. Nauhaus, M. Nellesen and G. Herz for technical help. This study was supported by the German Academic Exchange Service, the Fonds der Chemischen Industrie, and the Max Planck Society.
Author information
Authors and Affiliations
- Max Planck Institute for Marine Microbiology, Celsiusstraße 1, 28359, Bremen, Germany
Hang T. Dinh, Jan Kuever, Marc Mußmann & Friedrich Widdel - Institute for Material Testing, Paul-Feller-Straße 1, 28199, Bremen, Germany
Jan Kuever - Max Planck Institute for Iron Research, Max-Planck-Straße 1, 40237, Düsseldorf, Germany
Achim W. Hassel & Martin Stratmann
Authors
- Hang T. Dinh
- Jan Kuever
- Marc Mußmann
- Achim W. Hassel
- Martin Stratmann
- Friedrich Widdel
Corresponding author
Correspondence toFriedrich Widdel.
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Dinh, H., Kuever, J., Mußmann, M. et al. Iron corrosion by novel anaerobic microorganisms.Nature 427, 829–832 (2004). https://doi.org/10.1038/nature02321
- Received: 25 April 2003
- Accepted: 23 December 2003
- Issue date: 26 February 2004
- DOI: https://doi.org/10.1038/nature02321