Equilibrium cluster formation in concentrated protein solutions and colloids (original) (raw)
- Letter
- Published: 25 November 2004
- Helen Sedgwick2,
- Frédéric Cardinaux1,
- Wilson C. K. Poon2,
- Stefan U. Egelhaaf2,3 &
- …
- Peter Schurtenberger1
Nature volume 432, pages 492–495 (2004) Cite this article
- 9736 Accesses
- 1046 Citations
- 7 Altmetric
- Metrics details
Abstract
Controlling interparticle interactions, aggregation and cluster formation is of central importance in a number of areas, ranging from cluster formation in various disease processes to protein crystallography and the production of photonic crystals. Recent developments in the description of the interaction of colloidal particles with short-range attractive potentials have led to interesting findings including metastable liquid–liquid phase separation and the formation of dynamically arrested states (such as the existence of attractive and repulsive glasses, and transient gels)1,2,3,4,5,6,7. The emerging glass paradigm has been successfully applied to complex soft-matter systems, such as colloid–polymer systems8 and concentrated protein solutions9. However, intriguing problems like the frequent occurrence of cluster phases remain10,11,12,13. Here we report small-angle scattering and confocal microscopy investigations of two model systems: protein solutions and colloid–polymer mixtures. We demonstrate that in both systems, a combination of short-range attraction and long-range repulsion results in the formation of small equilibrium clusters. We discuss the relevance of this finding for nucleation processes during protein crystallization, protein or DNA self-assembly and the previously observed formation of cluster and gel phases in colloidal suspensions12,13,14,15,16,17.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Additional access options:
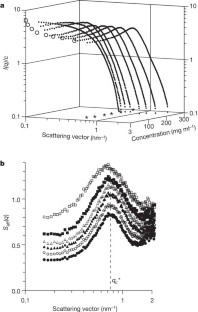
Figure 1: Normalized scattered intensity I(q)/c and corresponding effective structure factors _S_eff(q), as obtained by SAXS from lysozyme solutions of different concentrations c.
The alternative text for this image may have been generated using AI.
Figure 2: Effect of concentration and temperature on the effective structure factor _S_eff(q) as obtained by SANS.
The alternative text for this image may have been generated using AI.
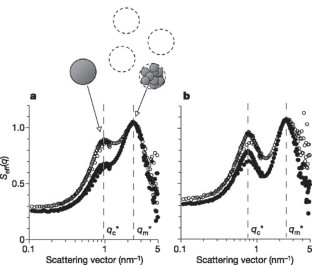
Figure 3: Clusters in protein solutions and colloidal suspensions.

The alternative text for this image may have been generated using AI.
Figure 4: Effect of temperature and ionic strength on the effective structure factor _S_eff(q), obtained by SAXS.
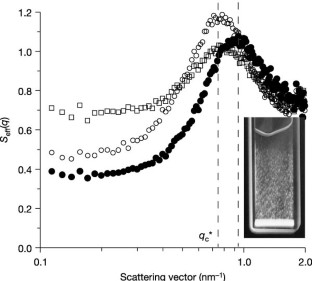
The alternative text for this image may have been generated using AI.
Similar content being viewed by others
References
- Dawson, K. A. The glass paradigm for colloidal glasses, gels, and other arrested states driven by attractive interactions. Curr. Opin. Colloid Interf. Sci. 7, 218–227 (2002)
Article CAS Google Scholar - Trappe, V., Prasad, V., Cipelletti, L., Segre, P. N. & Weitz, D. A. Jamming phase diagram for attractive particles. Nature 411, 772–775 (2001)
Article ADS CAS Google Scholar - Sciortino, F. Disordered materials: one liquid, two glasses. Nature Mater. 1, 145–146 (2002)
Article ADS CAS Google Scholar - Pham, K. N. et al. Multiple glassy states in a simple model system. Science 296, 104–106 (2002)
Article ADS CAS Google Scholar - Eckert, T. & Bartsch, E. Re-entrant glass transition in a colloid-polymer mixture with depletion attractions. Phys. Rev. Lett. 89, 125701–125704 (2002)
Article ADS CAS Google Scholar - Weeks, E. R., Crocker, J. C., Levitt, A. C., Schofield, A. & Weitz, D. A. Three-dimensional direct imaging of structural relaxation near the colloidal glass transition. Science 287, 627–631 (2000)
Article ADS CAS Google Scholar - Foffi, G. et al. Phase equilibria and glass transition in colloidal systems with short-ranged attractive interactions: application to protein crystallization. Phys Rev. E 65, 031407–031417 (2002)
Article ADS Google Scholar - Bergenholtz, J., Poon, W. C. K. & Fuchs, M. Gelation in model colloid-polymer mixtures. Langmuir 19, 4493–4503 (2003)
Article CAS Google Scholar - Kulkarni, A. M., Dixit, N. M. & Zukoski, C. F. Ergodic and non-ergodic phase transitions in globular protein suspensions. Faraday Discuss. 123, 37–50 (2003)
Article ADS CAS Google Scholar - Puertas, A. M., Fuchs, M. & Cates, M. E. Dynamical heterogeneities close to a colloidal gel. J. Chem. Phys. 121, 2813–2822 (2004)
Article ADS CAS Google Scholar - Sciortino, F., Mossa, S., Zaccarelli, E. & Tartaglia, P. Equilibrium cluster phases and low-density arrested disordered states: The role of short-range attraction and long-range repulsion. Phys. Rev. Lett. 93, 055701 (2004)
Article ADS Google Scholar - Groenewold, J. & Kegel, W. K. Anomalously large equilibrium clusters of colloids. J. Phys. Chem. B 105, 11702–11709 (2001)
Article CAS Google Scholar - Segré, P. N., Prasad, V., Schofield, A. B. & Weitz, D. A. Glasslike kinetic arrest at colloidal-gelation transition. Phys. Rev. Lett. 86, 6042–6045 (2001)
Article ADS Google Scholar - Guillot, S., Delsanti, M., Désert, S. & Langevin, D. Surfactant-induced collapse of polymer chains and monodisperse growth of aggregates near the precipitation boundary in carboxymethylcellulose-DTAB aqueous solutions. Langmuir 19, 230–237 (2003)
Article CAS Google Scholar - Muschol, M. & Rosenberger, F. Liquid-liquid phase separation in supersaturated lysozyme solutions and associated precipitate formation/crystallization. J. Chem. Phys. 107, 1953–1962 (1997)
Article ADS CAS Google Scholar - Pedersen, J. S., Hansen, S. & Bauer, R. The aggregation behavior of zinc-free insulin studied by small-angle neutron scattering. Eur. Biophys. J. 22, 379–389 (1994)
Article CAS Google Scholar - Piazza, R. Interactions and phase transitions in protein solutions. Curr. Opin. Colloid Interf. Sci. 5, 38–43 (2000)
Article CAS Google Scholar - Malfois, M., Bonnete, F., Belloni, L. & Tardieu, A. A model of attractive interactions to account for fluid-fluid phase separation of protein solutions. J. Chem. Phys. 105, 3290–3300 (1996)
Article ADS CAS Google Scholar - Broide, M. L., Tomic, T. M. & Saxowsky, M. D. Using phase transitions to investigate the effect of salts on protein interactions. Phys. Rev. E 53, 6325–6335 (1996)
Article ADS CAS Google Scholar - Schurtenberger, P., Chamberlin, R. A., Thurston, G. M., Thomson, J. A. & Benedek, G. B. Observation of critical phenomena in a protein-water solution. Phys. Rev. Lett. 63, 2064–2067 (1989)
Article ADS CAS Google Scholar - Yethiraj, A. & Van Blaaderen, A. A colloidal model system with an interaction tunable from hard sphere to soft and dipolar. Nature 421, 513–517 (2003)
Article ADS CAS Google Scholar - Poon, W. C. K. The physics of a model colloid-polymer mixture. J. Phys. Condens. Matter 14, R859–R880 (2002)
Article ADS CAS Google Scholar - Pham, K. N., Egelhaaf, S. U., Pusey, P. N. & Poon, W. C. K. Glasses in hard spheres with short-range attraction. Phys. Rev. E 69, 11503–11516 (2004)
Article ADS CAS Google Scholar - Rojas, L., Urban, C., Schurtenberger, P., Gisler, T. & Grünberg, H. H. Reappearance of structure in charge-stabilized suspensions. Europhys. Lett. 60, 802–808 (2002)
Article ADS CAS Google Scholar - Tanford, C. & Roxby, R. Interpretation of protein titration curves. Application to lysozyme. Biochemistry 11, 2192–2198 (1972)
Article CAS Google Scholar
Acknowledgements
We thank the Swiss spallation source at the Paul Scherrer Institut (PSI) in Villigen, Switzerland, for the neutron beam time and we acknowledge the help of our local contacts J. Kohlbrecher and S. van Petegem. We thank J. Groenewold, W. Kegel, F. Sciortino, K. Kroy and M. Cates for discussions. We thank A. Schofield for preparing the fluorescent PMMA particles. This work was supported by the Swiss National Science Foundation, the UK Engineering and Physical Sciences Research Council, the Scottish Higher Education Funding Council, and the Marie Curie Network on Dynamical Arrest of Soft Matter and Colloids. A.S. and P.S. conceived and performed the protein experiments; F.C. prepared the pH stabilized protein samples for the control experiments; H.S., W.C.K.P. and S.U.E. carried out and analysed the experiments with the colloid–polymer samples.
Author information
Authors and Affiliations
- Department of Physics, University of Fribourg, Chemin du Musée 3, CH-1700, Fribourg, Switzerland
Anna Stradner, Frédéric Cardinaux & Peter Schurtenberger - School of Physics and COSMIC, The University of Edinburgh, Mayfield Road, EH9 3JZ, Edinburgh, UK
Helen Sedgwick, Wilson C. K. Poon & Stefan U. Egelhaaf - School of Chemistry, The University of Edinburgh, Mayfield Road, EH9 3JZ, Edinburgh, UK
Stefan U. Egelhaaf
Authors
- Anna Stradner
- Helen Sedgwick
- Frédéric Cardinaux
- Wilson C. K. Poon
- Stefan U. Egelhaaf
- Peter Schurtenberger
Corresponding author
Correspondence toPeter Schurtenberger.
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
Supplementary Figure 1 (download DOC )
This figure shows cluster aggregation numbers Nc obtained from samples where the pH is constant at all concentrations compared with those from samples where the pH slightly increases at high concentrations. It demonstrates that there is no measurable influence on Nc upon a small shift in pH. (DOC 38 kb)
Rights and permissions
About this article
Cite this article
Stradner, A., Sedgwick, H., Cardinaux, F. et al. Equilibrium cluster formation in concentrated protein solutions and colloids.Nature 432, 492–495 (2004). https://doi.org/10.1038/nature03109
- Received: 01 September 2004
- Accepted: 06 October 2004
- Issue date: 25 November 2004
- DOI: https://doi.org/10.1038/nature03109