Novel microbial communities of the Haakon Mosby mud volcano and their role as a methane sink (original) (raw)
- Letter
- Published: 19 October 2006
- Tina Lösekann1 na1,
- Dirk de Beer1,
- Marcus Elvert1 nAff6,
- Thierry Nadalig3 nAff7,
- Katrin Knittel1,
- Rudolf Amann1,
- Eberhard J. Sauter2,
- Michael Schlüter2,
- Michael Klages2,
- Jean Paul Foucher3 &
- …
- Antje Boetius1,2,4
Nature volume 443, pages 854–858 (2006) Cite this article
- 4819 Accesses
- 582 Citations
- 32 Altmetric
- Metrics details
Abstract
Mud volcanism is an important natural source of the greenhouse gas methane to the hydrosphere and atmosphere1,2. Recent investigations show that the number of active submarine mud volcanoes might be much higher than anticipated (for example, see refs 3–5), and that gas emitted from deep-sea seeps might reach the upper mixed ocean6,7,8. Unfortunately, global methane emission from active submarine mud volcanoes cannot be quantified because their number and gas release are unknown9. It is also unclear how efficiently methane-oxidizing microorganisms remove methane. Here we investigate the methane-emitting Haakon Mosby Mud Volcano (HMMV, Barents Sea, 72° N, 14° 44′ E; 1,250 m water depth) to provide quantitative estimates of the in situ composition, distribution and activity of methanotrophs in relation to gas emission. The HMMV hosts three key communities: aerobic methanotrophic bacteria (Methylococcales), anaerobic methanotrophic archaea (ANME-2) thriving below siboglinid tubeworms, and a previously undescribed clade of archaea (ANME-3) associated with bacterial mats. We found that the upward flow of sulphate- and oxygen-free mud volcano fluids restricts the availability of these electron acceptors for methane oxidation, and hence the habitat range of methanotrophs. This mechanism limits the capacity of the microbial methane filter at active marine mud volcanoes to <40% of the total flux.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Additional access options:
Figure 1: The Haakon Mosby Mud Volcano.
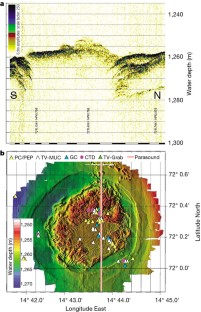
The alternative text for this image may have been generated using AI.
Figure 2: A schematic diagram of the different microbial habitats at the HMMV.
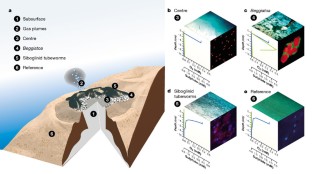
The alternative text for this image may have been generated using AI.
Figure 3: Vertical distribution of methanotrophs in relation to environmental factors.
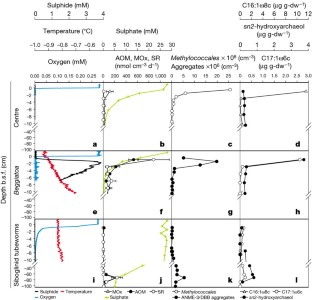
The alternative text for this image may have been generated using AI.
Similar content being viewed by others
References
- Kopf, A. J. Significance of mud volcanism. Rev. Geophys. B 40, 1–49 (2002)
Google Scholar - Milkov, A. V. et al. Global gas flux from mud volcanoes: A significant source of fossil methane in the atmosphere and the ocean. Geophys. Res. Lett. 30, doi:10.1029/2002GL016358. (2003)
- Bohrmann, G. et al. Mud volcanoes and gas hydrates in the Black Sea: new data from Dvurechenskii and Odessa mud volcanoes. Geo-Mar. Lett. 23, 239–249 (2003)
Article ADS CAS Google Scholar - Pinheiro, L. M. et al. Mud volcanism in the Gulf of Cadiz: results from the TTR-10 cruise. Mar. Geol. 195, 131–151 (2003)
Article ADS CAS Google Scholar - Loncke, L. et al. Mud volcanoes, gas chimneys, pockmarks and mounds in the Nile deep-sea fan (Eastern Mediterranean): geophysical evidences. Marine Petrol. Geol. 21, 669–689 (2004)
Article Google Scholar - MacDonald, I. R. et al. Transfer of hydrocarbons from natural seeps to the water column and atmosphere. Geofluids 2, 95–107 (2002)
Article CAS Google Scholar - Rehder, G. et al. Enhanced lifetime of methane bubble streams within the deep ocean. Geophys. Res. Lett. 29, 21–24 (2002)
Article Google Scholar - Sauter, E. J. et al. Methane discharge from a deep-sea submarine mud volcano into the upper water column by gas hydrate-coated methane bubbles. Earth Planet. Sci. Lett. 243, 354–365 (2006)
Article ADS CAS Google Scholar - Vogt, P. R. et al. Haakon Mosby mud volcano: A warm methane seep with seafloor hydrates and chemosynthesis-based Ecosystem in late Quarternary Slide Valley, Bear Island Fan, Barents Sea passive margin. EOS Trans. AGU 78, 187–189 (1997)
Article Google Scholar - Vogt, P. R., Gardner, J. & Crane, K. The Norwegian-Barents-Svalbard (NBS) continental margin: Introducing a natural laboratory of mass wasting, hydrates, and ascent of sediment, pore water, and methane. Geo-Mar. Lett. 19, 2–21 (1999)
Article ADS CAS Google Scholar - Ginsburg, G. D. et al. Gas hydrate accumulation at the Håkon Mosby Mud Volcano. Geo-Mar. Lett. 19, 57–67 (1999)
Article ADS CAS Google Scholar - Damm, E. & Budéus, G. Fate of vent-derived methane in seawater above the Håkon Mosby mud volcano (Norwegian Sea). Mar. Chem. 82, 1–11 (2003)
Article CAS Google Scholar - Milkov, A. V. et al. Geological, geochemical, and microbial processes at the hydrate-bearing Håkon Mosby mud volcano: a review. Chem. Geol. 205, 347–366 (2004)
Article ADS CAS Google Scholar - Kaul, N., Foucher, J. P. & Heesemann, M. Estimating mud expulsion rates from temperature measurements on Håkon Mosby Mud Volcano, SW Barents Sea. Mar. Geol. 229, 1–14 (2006)
Article ADS Google Scholar - de Beer, D. et al. In situ fluxes and zonation of microbial activity in surface sediments of the Håkon Mosby Mud Volcano. Limnol. Oceanogr. 51, 1315–1331 (2006)
Article ADS CAS Google Scholar - Jerosch, K. et al. Spatial distribution of mud flows, chemoautotrophic communities, and biogeochemical habitats at Håkon Mosby Mud Volcano. Mar. Geol. (submitted).
- Distel, D. L., Lee, H. K. W. & Cavanaugh, C. M. Intracellular coexistence of methanotrophic and thioautotrophic bacteria in a hydrothermal vent mussel. Proc. Natl Acad. Sci. USA 92, 9598–9602 (1995)
Article ADS CAS Google Scholar - Knittel, K. et al. Activity, distribution, and diversity of sulfate reducers and other bacteria in sediments above gas hydrate (Cascadia margin, Oregon). Geomicrobiol. J. 20, 269–294 (2003)
Article CAS Google Scholar - Orphan, V. J. et al. Comparative analysis of methane-oxidizing archaea and sulfate- reducing bacteria in anoxic marine sediments. Appl. Environ. Microbiol. 67, 1922–1934 (2001)
Article CAS Google Scholar - Knittel, K. et al. Diversity and distribution of methanotrophic archaea at cold seeps. Appl. Environ. Microbiol. 71, 467–479 (2005)
Article CAS Google Scholar - Boetius, A. et al. A marine microbial consortium apparently mediating anaerobic oxidation of methane. Nature 407, 623–626 (2000)
Article ADS CAS Google Scholar - Niemann, H. et al. Methane emission and consumption at a North Sea gas seep (Tommeliten area). Biogeosciences 2, 335–351 (2005)
Article ADS CAS Google Scholar - Hinrichs, K-U. & Boetius, A. in Ocean Margin Systems (eds Wefer, G., Billett, D. & Hebbeln, D.) 457–477 (Springer, Berlin, 2002)
- Reeburgh, W. S. in Treatise on Geochemistry Vol. 4 (ed. Keeling, R. F.) 65–89 (Elsevier–Pergamon, Oxford, 2003)
- Treude, T. et al. Anaerobic oxidation of methane above gas hydrates at Hydrate Ridge, NE Pacific Ocean. Mar. Ecol. Prog. Ser. 264, 1–14 (2003)
Article ADS CAS Google Scholar - Sibuet, M. & Olu, K. Biogeography, biodiversity and fluid dependence of deep-sea cold-seep communities at active and passive margins. Deep-Sea Res. II 45, 517–567 (1998)
Article ADS Google Scholar - Judd, A. G. et al. The geological methane budget at Continental Margins and its influence on climate change. Geofluids 2, 109–126 (2002)
Article CAS Google Scholar - Luff, R. & Wallmann, K. Fluid flow, methane fluxes, carbonate precipitation and biogeochemical turnover in gas hydrate-bearing sediments at Hydrate Ridge, Cascadia Margin: Numerical modeling and mass balances. Geochim. Cosmochim. Acta 67, 3403–3421 (2003)
Article ADS CAS Google Scholar - Brown, K. M. et al. Correlated transient fluid pulsing and seismic tremor in the Costa Rica subduction zone. Earth Planet. Sci. Lett. 238, 189–203 (2005)
Article ADS CAS Google Scholar - Elvert, M. et al. Characterization of specific membrane fatty acids as chemotaxonomic markers for sulfate-reducing bacteria involved in anaerobic oxidation of methane. Geomicrobiol. J. 20, 403–419 (2003)
Article CAS Google Scholar
Acknowledgements
The expeditions ‘AWI’ on RV L’Atalante in 2001 and ‘ARK XIX3b’ on RV Polarstern in 2003, both with ROV Victor 6000, were jointly planned, coordinated and carried out by the Alfred Wegener Institute for Polar and Marine Research in Bremerhaven, Germany, and the French Research Institute for Exploitation of the Sea (IFREMER) in Brest, France. We thank the captain and crew, the team of the ROV Victor 6000 and the shipboard scientific community of the RV Polarstern and L’Atalante for their help at sea. We thank S. Lüdeling, L. Baumann, V. Beier, I. Busse, F. Heinrich, G. Eickert, A. Nordhausen, J. Rogenhagen and R. Usbeck for technical assistance, C. Edy for help with georeferencing, U. Witte for providing the benthic lander technology, and S. Joye for comments on the manuscript. This is publication no. GEOTECH-235 of the R&D-Programme GEOTECHNOLOGIEN, Project MUMM, funded by the German Ministry of Education and Research (BMBF) and German Research Foundation (DFG). Author Contributions T.L., T.N., K.K. and R.A. carried out the 16S-rRNA-based analyses and microscopy, H.N. and M.E. the lipid biomarker work, A.B. and H.N. the rate measurements, D.B., E.S. and M.S. the microsensor and geochemical measurements, and M.K., M.S., A.B., H.N. and J.P.F. the geo- and video-graphical survey and experimental strategy. A.B., H.N. and D.B. wrote the manuscript text, and H.N., T.L. and K.K. wrote the supplements. All authors discussed the results and commented on the manuscript.
Author information
Author notes
- Marcus Elvert
Present address: DFG Research Center Ocean Margins, University of Bremen, 28334, Bremen, Germany - Thierry Nadalig
Present address: UMR 7156 Université Louis-Pasteur/CNRS, Département Microorganismes, Génomes, Environnement, 67083, Strasbourg Cedex, France - Helge Niemann and Tina Lösekann: These authors contributed equally to this work.
Authors and Affiliations
- Max Planck Institute for Marine Microbiology, 28359, Bremen, Germany
Helge Niemann, Tina Lösekann, Dirk de Beer, Marcus Elvert, Katrin Knittel, Rudolf Amann & Antje Boetius - Alfred Wegener Institute for Polar and Marine Research, 27515, Bremerhaven, Germany
Helge Niemann, Eberhard J. Sauter, Michael Schlüter, Michael Klages & Antje Boetius - Centre Ifremer de Brest, BP70, 29280, Plouzane, France
Thierry Nadalig & Jean Paul Foucher - International University Bremen, 28759, Bremen, Germany
Antje Boetius
Authors
- Helge Niemann
- Tina Lösekann
- Dirk de Beer
- Marcus Elvert
- Thierry Nadalig
- Katrin Knittel
- Rudolf Amann
- Eberhard J. Sauter
- Michael Schlüter
- Michael Klages
- Jean Paul Foucher
- Antje Boetius
Corresponding author
Correspondence toAntje Boetius.
Ethics declarations
Competing interests
The nucleotide sequence data have been deposited in EMBL, GenBank and the DDBJ nucleotide sequence database under accession numbers AJ704650–AJ704653, AJ704631 and AM287206–AM287207. Reprints and permissions information is available at www.nature.com/reprints. The authors declare no competing financial interests.
Supplementary information
Supplementary Notes (download DOC )
This file includes a section about the methane budget, the identity of the different methanotrophic communities as determined by 16S rDNA analysis, fluorescence in situ hybridisation (FISH) and lipid biomarker patterns. This file also contains Supplementary Methods and Supplementary Figure Legends. (DOC 60 kb)
Supplementary Figure 1 (download PDF )
This file contains the phylogenetic tree of archaeal 16S rDNA sequences of Haakon Mosby mud volcano. (PDF 155 kb)
Supplementary Figure 2 (download PDF )
This file contains the phylogenetic tree of bacterial 16S rDNA sequences of Haakon Mosby mud volcano. (PDF 164 kb)
Rights and permissions
About this article
Cite this article
Niemann, H., Lösekann, T., de Beer, D. et al. Novel microbial communities of the Haakon Mosby mud volcano and their role as a methane sink.Nature 443, 854–858 (2006). https://doi.org/10.1038/nature05227
- Received: 16 July 2006
- Accepted: 31 August 2006
- Issue date: 19 October 2006
- DOI: https://doi.org/10.1038/nature05227
This article is cited by
Editorial Summary
Methane in muddy waters
Submarine mud volcanoes may be major players in the emission of the greenhouse gas methane. A select group of microorganisms, called methanotrophs, can consume this gas, but their impact on methane emission in this environment is not well understood. A study of the waters around a mud volcano in the Barents Sea has identified three key methanotrophic communities: aerobic bacteria, anaerobic archaea living beneath tubeworms, and previously undescribed archaea associated with bacterial mats. A natural cap on the capacity of the microbial methane filter was also discovered: the upward flow of sulphate- and oxygen-free volcanic fluids restricts the efficiency of methane oxidation, allowing much of the methane to escape to the hydrosphere and potentially the atmosphere.