A human 5′-tyrosyl DNA phosphodiesterase that repairs topoisomerase-mediated DNA damage (original) (raw)
- Letter
- Published: 01 October 2009
- Sherif F. El Khamisy1,2 na1,
- Maria C. Zuma1,
- Kay Osborn1 &
- …
- Keith W. Caldecott1
Nature volume 461, pages 674–678 (2009)Cite this article
- 5848 Accesses
- 383 Citations
- 9 Altmetric
- Metrics details
Abstract
Topoisomerases regulate DNA topology and are fundamental to many aspects of chromosome metabolism1,2. Their activity involves the transient cleavage of DNA, which, if it occurs near sites of endogenous DNA damage or in the presence of topoisomerase poisons, can result in abortive topoisomerase-induced DNA strand breaks3,4,5. These breaks feature covalent linkage of the enzyme to the DNA termini by a 3′- or 5′-phosphotyrosyl bond and are implicated in hereditary human disease6,7,8, chromosomal instability and cancer4,9, and underlie the clinical efficacy of an important class of anti-tumour poisons3,9,10. The importance of liberating DNA termini from trapped topoisomerase is illustrated by the progressive neurodegenerative disease observed in individuals containing a mutation in tyrosyl-DNA phosphodiesterase 1 (TDP1), an enzyme that cleaves 3′-phosphotyrosyl bonds6,7,8. However, a complementary human enzyme that cleaves 5′-phosphotyrosyl bonds has not been reported, despite the effect of DNA double-strand breaks containing such termini on chromosome instability and cancer6,7,8. Here we identify such an enzyme in human cells and show that this activity efficiently restores 5′-phosphate termini at DNA double-strand breaks in preparation for DNA ligation. This enzyme, TTRAP, is a member of the Mg2+/Mn2+-dependent family of phosphodiesterases. Cellular depletion of TTRAP results in increased susceptibility and sensitivity to topoisomerase-II-induced DNA double-strand breaks. TTRAP is, to our knowledge, the first human 5′-tyrosyl DNA phosphodiesterase to be identified, and we suggest that this enzyme is denoted tyrosyl DNA phosphodiesterase-2 (TDP2).
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Additional access options:
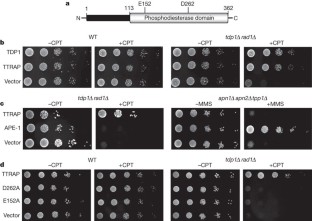
Figure 1: Human TTRAP suppresses the sensitivity of yeast to Top1-induced DNA damage.
The alternative text for this image may have been generated using AI.
Figure 2: TTRAP possesses 3′- and 5′-tyrosyl DNA phosphodiesterase (TDP) activity.

The alternative text for this image may have been generated using AI.
Figure 3: Reduced 5′-TDP activity in TTRAP-deficient human cell extracts.

The alternative text for this image may have been generated using AI.
Figure 4: TTRAP affects etoposide sensitivity and DNA damage accumulation.

The alternative text for this image may have been generated using AI.
Similar content being viewed by others
References
- Champoux, J. J. DNA topoisomerases: structure, function, and mechanism. Annu. Rev. Biochem. 70, 369–413 (2001)
Article CAS Google Scholar - Wang, J. C. Cellular roles of DNA topoisomerases: a molecular perspective. Nature Rev. Mol. Cell Biol. 3, 430–440 (2002)
Article CAS Google Scholar - Li, T. K. & Liu, L. F. Tumor cell death induced by topoisomerase-targeting drugs. Annu. Rev. Pharmacol. Toxicol. 41, 53–77 (2001)
Article Google Scholar - Deweese, J. E. & Osheroff, N. The DNA cleavage reaction of topoisomerase II: wolf in sheep’s clothing. Nucleic Acids Res. 37, 738–748 (2009)
Article CAS Google Scholar - Pourquier, P. & Pommier, Y. Topoisomerase I-mediated DNA damage. Adv. Cancer Res. 80, 189–216 (2001)
Article CAS Google Scholar - El-Khamisy, S. F. et al. Defective DNA single-strand break repair in spinocerebellar ataxia with axonal neuropathy-1. Nature 434, 108–113 (2005)
Article ADS CAS Google Scholar - Takashima, H. et al. Mutation of TDP1, encoding a topoisomerase I-dependent DNA damage repair enzyme, in spinocerebellar ataxia with axonal neuropathy. Nature Genet. 32, 267–272 (2002)
Article CAS Google Scholar - Yang, S. W. et al. A eukaryotic enzyme that can disjoin dead-end covalent complexes between DNA and type I topoisomerases. Proc. Natl Acad. Sci. USA 93, 11534–11539 (1996)
Article ADS CAS Google Scholar - Nitiss, J. L. Targeting DNA topoisomerase II in cancer chemotherapy. Nature Rev. Cancer 9, 338–350 (2009)
Article CAS Google Scholar - Pommier, Y. Topoisomerase I inhibitors: camptothecins and beyond. Nature Rev. Cancer 6, 789–802 (2006)
Article CAS Google Scholar - Vance, J. R. & Wilson, T. E. Yeast Tdp1 and Rad1-Rad10 function as redundant pathways for repairing Top1 replicative damage. Proc. Natl Acad. Sci. USA 99, 13669–13674 (2002)
Article ADS CAS Google Scholar - Liu, C., Pouliot, J. J. & Nash, H. A. Repair of topoisomerase I covalent complexes in the absence of the tyrosyl-DNA phosphodiesterase Tdp1. Proc. Natl Acad. Sci. USA 99, 14970–14975 (2002)
Article ADS CAS Google Scholar - Pype, S. et al. TTRAP, a novel protein that associates with CD40, tumor necrosis factor (TNF) receptor-75 and TNF receptor-associated factors (TRAFs), and that inhibits nuclear factor-κB activation. J. Biol. Chem. 275, 18586–18593 (2000)
Article CAS Google Scholar - Rodrigues-Lima, F., Josephs, M., Katan, M. & Cassinat, B. Sequence analysis identifies TTRAP, a protein that associates with CD40 and TNF receptor-associated factors, as a member of a superfamily of divalent cation-dependent phosphodiesterases. Biochem. Biophys. Res. Commun. 285, 1274–1279 (2001)
Article CAS Google Scholar - Nitiss, J. L. et al. Amsacrine and etoposide hypersensitivity of yeast cells overexpressing DNA topoisomerase II. Cancer Res. 52, 4467–4472 (1992)
CAS PubMed Google Scholar - Xu, G. L. et al. TTRAP is a novel PML nuclear bodies-associated protein. Biochem. Biophys. Res. Commun. 375, 395–398 (2008)
Article CAS Google Scholar - Mielke, C., Christensen, M. O., Barthelmes, H. U. & Boege, F. Enhanced processing of UVA-irradiated DNA by human topoisomerase II in living cells. J. Biol. Chem. 279, 20559–20562 (2004)
Article CAS Google Scholar - Mielke, C., Kalfalah, F. M., Christensen, M. O. & Boege, F. Rapid and prolonged stalling of human DNA topoisomerase I in UVA-irradiated genomic areas. DNA Repair (Amst.) 6, 1757–1763 (2007)
Article CAS Google Scholar - Kingma, P. S. & Osheroff, N. The response of eukaryotic topoisomerases to DNA damage. Biochim. Biophys. Acta 1400, 223–232 (1998)
Article CAS Google Scholar - Connelly, J. C. & Leach, D. R. Repair of DNA covalently linked to protein. Mol. Cell 13, 307–316 (2004)
Article CAS Google Scholar - Hartsuiker, E., Neale, M. J. & Carr, A. M. Distinct requirements for the Rad32(Mre11) nuclease and Ctp1(CtIP) in the removal of covalently bound topoisomerase I and II from DNA. Mol. Cell 33, 117–123 (2009)
Article CAS Google Scholar - Pei, H. et al. EAPII interacts with ETS1 and modulates its transcriptional function. Oncogene 22, 2699–2709 (2003)
Article CAS Google Scholar - Esguerra, C. V. et al. Ttrap is an essential modulator of Smad3-dependent Nodal signaling during zebrafish gastrulation and left-right axis determination. Development 134, 4381–4393 (2007)
Article CAS Google Scholar - Zucchelli, S. et al. Aggresome-forming TTRAP mediates pro-apoptotic properties of Parkinson’s disease-associated DJ-1 missense mutations. Cell Death Differ. 16, 428–438 (2009)
Article CAS Google Scholar - Vance, J. R. & Wilson, T. E. Repair of DNA strand breaks by the overlapping functions of lesion-specific and non-lesion-specific DNA 3′ phosphatases. Mol. Cell. Biol. 21, 7191–7198 (2001)
Article CAS Google Scholar - Nitiss, K. C., Malik, M., He, X., White, S. W. & Nitiss, J. L. Tyrosyl-DNA phosphodiesterase (Tdp1) participates in the repair of Top2-mediated DNA damage. Proc. Natl Acad. Sci. USA 103, 8953–8958 (2006)
Article ADS CAS Google Scholar - Whitehouse, C. J. et al. XRCC1 stimulates human polynucleotide kinase activity at damaged DNA termini and accelerates DNA single-strand break repair. Cell 104, 107–117 (2001)
Article CAS Google Scholar - Parsons, J. L., Dianova, I. I. & Dianov, G. L. APE1 is the major 3′-phosphoglycolate activity in human cell extracts. Nucleic Acids Res. 32, 3531–3536 (2004)
Article CAS Google Scholar - Reynolds, J. J. et al. Defective DNA ligation during short-patch single-strand break repair in ataxia oculomotor apraxia-1. Mol. Cell. Biol. 29, 1354–1362 (2008)
Article Google Scholar - Katyal, S. et al. TDP1 facilitates chromosomal single-strand break repair in neurons and is neuroprotective in vivo . EMBO J. 26, 4720–4731 (2007)
Article CAS Google Scholar
Acknowledgements
This work was funded by the MRC (G0600776), the BBSRC (BB/C516595/1), and CR-UK (C6563/A10192). S.F.E.K. and F.C.L. were also funded by Fellowships from the Wellcome Trust (S.F.E.K.; 085284), Marie Curie (FCL; 2007-2-1-IEF-221222) and EMBO (FCL; ALTF 956-2006). We thank T. Wilson, H. Nash, M. Neale, J. Nitiss and E. Hoffmann for materials.
Author Contributions F.C.L. developed the genetic screen and conducted the mammalian cell experiments. M.C.Z. and F.C.L. conducted the yeast experiments. K.O. and S.F.E.K. prepared the recombinant proteins, and S.F.E.K. conducted the biochemical experiments. K.W.C., F.C.L. and S.F.E.K. designed and interpreted the experiments. K.W.C. coordinated the project and wrote the manuscript.
Author information
Author notes
- Felipe Cortes Ledesma and Sherif F. El Khamisy: These authors contributed equally to this work.
Authors and Affiliations
- Genome Damage and Stability Centre, University of Sussex, Science Park Road, Falmer, Brighton, Sussex BN1 9RQ, UK ,
Felipe Cortes Ledesma, Sherif F. El Khamisy, Maria C. Zuma, Kay Osborn & Keith W. Caldecott - Biochemistry Department, Faculty of Pharmacy, Ain Shams University, Abbassia 11566, Cairo, Egypt,
Sherif F. El Khamisy
Authors
- Felipe Cortes Ledesma
- Sherif F. El Khamisy
- Maria C. Zuma
- Kay Osborn
- Keith W. Caldecott
Corresponding authors
Correspondence toFelipe Cortes Ledesma or Keith W. Caldecott.
Supplementary information
PowerPoint slides
Rights and permissions
About this article
Cite this article
Ledesma, F., El Khamisy, S., Zuma, M. et al. A human 5′-tyrosyl DNA phosphodiesterase that repairs topoisomerase-mediated DNA damage.Nature 461, 674–678 (2009). https://doi.org/10.1038/nature08444
- Received: 29 April 2009
- Accepted: 20 August 2009
- Issue date: 01 October 2009
- DOI: https://doi.org/10.1038/nature08444
This article is cited by
Editorial Summary
DNA repair: the role of TTRAP
The mechanism of topoisomerase action involves making a transient break in DNA. If the topoisomerase makes a break near another DNA lesion, the break can persist, with the topoisomerase attached to either the 3′ or 5′ end via a phosphotyrosyl bond. If these covalent adducts are not removed efficiently, cancer and neurodegenerative disease can result. In humans, a protein, TDP1, that removes adducts using a 3′-phosphotyrosyl bond has been identified. In this work, Ledesma et al. identify a phosphodiesterase, TTRAP, as the protein that removes adducts at the 5′-phosphotyrosyl bond.