RNF12 initiates X-chromosome inactivation by targeting REX1 for degradation (original) (raw)
- Letter
- Published: 29 April 2012
- Eskeatnaf Mulugeta Achame1,
- Jeroen Demmers2,
- Tahsin Stefan Barakat1,
- Eveline Rentmeester1,
- Wilfred van IJcken3,
- J. Anton Grootegoed1 &
- …
- Joost Gribnau1
Nature volume 485, pages 386–390 (2012)Cite this article
- 8830 Accesses
- 209 Citations
- 3 Altmetric
- Metrics details
Subjects
Abstract
Evolution of the mammalian sex chromosomes has resulted in a heterologous X and Y pair, where the Y chromosome has lost most of its genes. Hence, there is a need for X-linked gene dosage compensation between XY males and XX females. In placental mammals, this is achieved by random inactivation of one X chromosome in all female somatic cells1. Upregulation of Xist transcription on the future inactive X chromosome acts against Tsix antisense transcription, and spreading of Xist RNA in cis triggers epigenetic changes leading to X-chromosome inactivation. Previously, we have shown that the X-encoded E3 ubiquitin ligase RNF12 is upregulated in differentiating mouse embryonic stem cells and activates Xist transcription and X-chromosome inactivation2. Here we identify the pluripotency factor REX1 as a key target of RNF12 in the mechanism of X-chromosome inactivation. RNF12 causes ubiquitination and proteasomal degradation of REX1, and Rnf12 knockout embryonic stem cells show an increased level of REX1. Using chromatin immunoprecipitation sequencing, REX1 binding sites were detected in Xist and Tsix regulatory regions. Overexpression of REX1 in female embryonic stem cells was found to inhibit Xist transcription and X-chromosome inactivation, whereas male Rex1+/− embryonic stem cells showed ectopic X-chromosome inactivation. From this, we propose that RNF12 causes REX1 breakdown through dose-dependent catalysis, thereby representing an important pathway to initiate X-chromosome inactivation. Rex1 and Xist are present only in placental mammals, which points to co-evolution of these two genes and X-chromosome inactivation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Additional access options:
Figure 1: RNF12 interacts with REX1 in mouse ESCs.
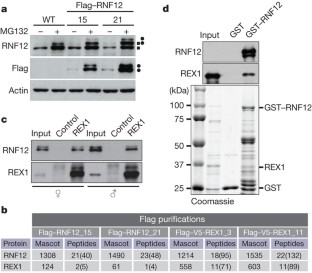
The alternative text for this image may have been generated using AI.
Figure 2: RNF12 polyubiquitinates and targets REX1 for proteasomal degradation.
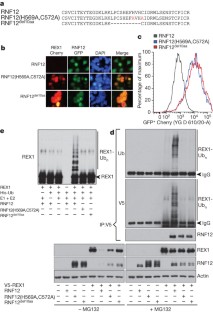
The alternative text for this image may have been generated using AI.
Figure 3: RNF12 is a dose-dependent regulator of REX1 expression.
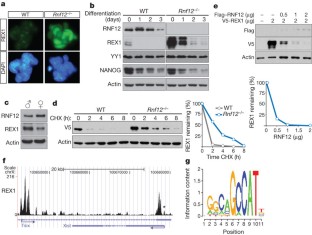
The alternative text for this image may have been generated using AI.
Figure 4: REX1-dependent regulation of XCI.
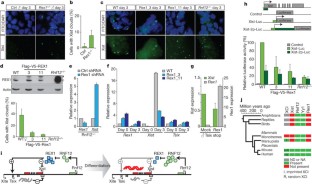
The alternative text for this image may have been generated using AI.
Similar content being viewed by others
References
- Lyon, M. F. Gene action in the X-chromosome of the mouse (Mus musculus L.). Nature 190, 372–373 (1961)
Article ADS CAS Google Scholar - Jonkers, I. et al. RNF12 is an X-encoded dose-dependent activator of X chromosome inactivation. Cell 139, 999–1011 (2009)
Article CAS Google Scholar - Monkhorst, K. et al. The probability to initiate X chromosome inactivation is determined by the X to autosomal ratio and X chromosome specific allelic properties. PLoS ONE 4, e5616 (2009)
Article ADS Google Scholar - Monkhorst, K., Jonkers, I., Rentmeester, E., Grosveld, F. & Gribnau, J. X inactivation counting and choice is a stochastic process: evidence for involvement of an X-linked activator. Cell 132, 410–421 (2008)
Article CAS Google Scholar - Shin, J. et al. Maternal Rnf12/RLIM is required for imprinted X-chromosome inactivation in mice. Nature 467, 977–981 (2010)
Article ADS CAS Google Scholar - Barakat, T. S. et al. RNF12 activates Xist and is essential for X chromosome inactivation. PLoS Genet. 7, e1002001 (2011)
Article CAS Google Scholar - Hosler, B. A., LaRosa, G. J., Grippo, J. F. & Gudas, L. J. Expression of REX-1, a gene containing zinc finger motifs, is rapidly reduced by retinoic acid in F9 teratocarcinoma cells. Mol. Cell. Biol. 9, 5623–5629 (1989)
Article CAS Google Scholar - Scotland, K. B., Chen, S., Sylvester, R. & Gudas, L. J. Analysis of Rex1 (zfp42) function in embryonic stem cell differentiation. Dev. Dyn. 238, 1863–1877 (2009)
Article CAS Google Scholar - Navarro, P. et al. Molecular coupling of Tsix regulation and pluripotency. Nature 468, 457–460 (2010)
Article ADS CAS Google Scholar - Kim, J. D. et al. Rex1/Zfp42 as an epigenetic regulator for genomic imprinting. Hum. Mol. Genet. 20, 1353–1362 (2011)
Article CAS Google Scholar - Masui, S. et al. Rex1/Zfp42 is dispensable for pluripotency in mouse ES cells. BMC Dev. Biol. 8, 45 (2008)
Article Google Scholar - Kim, J. D., Faulk, C. & Kim, J. Retroposition and evolution of the DNA-binding motifs of YY1, YY2 and REX1. Nucleic Acids Res. 35, 3442–3452 (2007)
Article CAS Google Scholar - Donohoe, M. E. et al. Identification of a Ctcf cofactor, Yy1, for the X chromosome binary switch. Mol. Cell 25, 43–56 (2007)
Article CAS Google Scholar - Duret, L. et al. The Xist RNA gene evolved in eutherians by pseudogenization of a protein-coding gene. Science 312, 1653–1655 (2006)
Article ADS CAS Google Scholar - van den Berg, D. L. et al. An Oct4-centered protein interaction network in embryonic stem cells. Cell Stem Cell 6, 369–381 (2010)
Article CAS Google Scholar - Soler, E. et al. A systems approach to analyze transcription factors in mammalian cells. Methods 53, 151–162 (2011)
Article CAS Google Scholar - Dignam, J. D., Lebovitz, R. M. & Roeder, R. G. Accurate transcription initiation by RNA polymerase II in a soluble extract from isolated mammalian nuclei. Nucleic Acids Res. 11, 1475–1489 (1983)
Article CAS Google Scholar
Acknowledgements
We thank R. van der Linden and C. Maduro for their help with some of the experiments, A. Inagaki, C. Andrieu-Soler, D. Warmerdam, L. Zhang and A. Mohd-Sarip for experimental advice, and C. Rougeulle and R. Jaenisch for providing the _Tsix_-stop cells. This work was supported by grants from the Netherlands Organisation for Scientific Research (NWO-TOP and NWO-VICI) and the European Research Council to J.G.
Author information
Authors and Affiliations
- Department of Reproduction and Development, Erasmus MC, University Medical Center, Dr Molewaterplein 50, 3015 GE Rotterdam, The Netherlands,
Cristina Gontan, Eskeatnaf Mulugeta Achame, Tahsin Stefan Barakat, Eveline Rentmeester, J. Anton Grootegoed & Joost Gribnau - Proteomics Center, Erasmus MC, University Medical Center, Dr Molewaterplein 50, 3015 GE Rotterdam, The Netherlands ,
Jeroen Demmers - Biomics Department, Erasmus MC, University Medical Center, Dr Molewaterplein 50, 3015 GE Rotterdam, The Netherlands,
Wilfred van IJcken
Authors
- Cristina Gontan
- Eskeatnaf Mulugeta Achame
- Jeroen Demmers
- Tahsin Stefan Barakat
- Eveline Rentmeester
- Wilfred van IJcken
- J. Anton Grootegoed
- Joost Gribnau
Contributions
C.G. and J.G. designed the experiments. C.G. performed most experiments assisted by E.R. T.S.B., C.G. and J.G. generated the _Rnf12_−/− ES cells line. C.G., J.D., W.v.IJ. and E.M.A. performed the mass spectrometry and sequencing analysis. J.G., J.A.G. and C.G. wrote the manuscript.
Corresponding author
Correspondence toJoost Gribnau.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
PowerPoint slides
Rights and permissions
About this article
Cite this article
Gontan, C., Achame, E., Demmers, J. et al. RNF12 initiates X-chromosome inactivation by targeting REX1 for degradation.Nature 485, 386–390 (2012). https://doi.org/10.1038/nature11070
- Received: 18 November 2010
- Accepted: 09 March 2012
- Published: 29 April 2012
- Issue date: 17 May 2012
- DOI: https://doi.org/10.1038/nature11070
This article is cited by
Editorial Summary
X-chromosome inactivation by RNF12
In placental mammals, X-linked gene dosage compensation between XY males and XX females is achieved by random inactivation of one X chromosome in female somatic cells. The E3 ubiquitin ligase RNF12 is required for initiation of X-chromosome inactivation (XCI) in embryonic stem cells, but its downstream targets have been unclear. Here, Gribnau and colleagues demonstrate that the pluripotency factor REX1 is a key target of RNF12 during XCI. RNF12 degradation of REX1 leads to relief of its inhibitory action on XCI.