Measurement of the first ionization potential of lawrencium, element 103 (original) (raw)
- Letter
- Published: 09 April 2015
- M. Asai1,
- A. Borschevsky2,3,
- T. Stora4,
- N. Sato1,
- Y. Kaneya1,5,
- K. Tsukada1,
- Ch. E. Düllmann3,6,7,
- K. Eberhardt3,7,
- E. Eliav8,
- S. Ichikawa1,9,
- U. Kaldor8,
- J. V. Kratz7,
- S. Miyashita10,
- Y. Nagame1,5,
- K. Ooe11,
- A. Osa1,
- D. Renisch7,
- J. Runke6,
- M. Schädel1,
- P. Thörle-Pospiech3,7,
- A. Toyoshima1 &
- …
- N. Trautmann7
Nature volume 520, pages 209–211 (2015)Cite this article
- 12k Accesses
- 139 Citations
- 191 Altmetric
- Metrics details
Subjects
Abstract
The chemical properties of an element are primarily governed by the configuration of electrons in the valence shell. Relativistic effects influence the electronic structure of heavy elements in the sixth row of the periodic table, and these effects increase dramatically in the seventh row—including the actinides—even affecting ground-state configurations1,2. Atomic s and _p_1/2 orbitals are stabilized by relativistic effects, whereas _p_3/2, d and f orbitals are destabilized, so that ground-state configurations of heavy elements may differ from those of lighter elements in the same group. The first ionization potential (IP1) is a measure of the energy required to remove one valence electron from a neutral atom, and is an atomic property that reflects the outermost electronic configuration. Precise and accurate experimental determination of IP1 gives information on the binding energy of valence electrons, and also, therefore, on the degree of relativistic stabilization. However, such measurements are hampered by the difficulty in obtaining the heaviest elements on scales of more than one atom at a time3,4,5. Here we report that the experimentally obtained IP1 of the heaviest actinide, lawrencium (Lr, atomic number 103), is 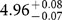 electronvolts. The IP1 of Lr was measured with 256Lr (half-life 27 seconds) using an efficient surface ion-source and a radioisotope detection system coupled to a mass separator. The measured IP1 is in excellent agreement with the value of 4.963(15) electronvolts predicted here by state-of-the-art relativistic calculations. The present work provides a reliable benchmark for theoretical calculations and also opens the way for IP1 measurements of superheavy elements (that is, transactinides) on an atom-at-a-time scale.
electronvolts. The IP1 of Lr was measured with 256Lr (half-life 27 seconds) using an efficient surface ion-source and a radioisotope detection system coupled to a mass separator. The measured IP1 is in excellent agreement with the value of 4.963(15) electronvolts predicted here by state-of-the-art relativistic calculations. The present work provides a reliable benchmark for theoretical calculations and also opens the way for IP1 measurements of superheavy elements (that is, transactinides) on an atom-at-a-time scale.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Additional access options:
Figure 1: Schematic experimental set-up used to measure the IP1 of Lr on an atom-at-a-time scale.
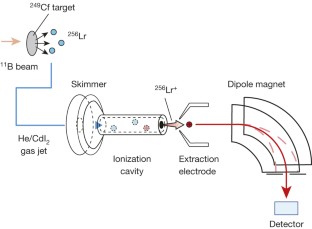
The alternative text for this image may have been generated using AI.
Figure 2: The ionization efficiency (_I_eff) of various short-lived isotopes as a function of the effective IP1 (IP1*) at 2,700 K.
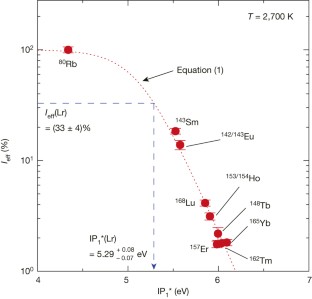
The alternative text for this image may have been generated using AI.
Similar content being viewed by others
References
- Pyykkö, P. Relativistic effects in structural chemistry. Chem. Rev. 88, 563–594 (1988)
Article Google Scholar - Pershina, V. in Relativistic Methods for Chemists (eds Barysz, M. & Ishikawa, Y.) 451–520 (Challenges and Advances in Computational Chemistry and Physics 10, Springer, 2010)
Book Google Scholar - Oganessian Heaviest nuclei from 48Ca-induced reactions. J. Phys. G 34, R165–R242 (2007)
Article CAS ADS Google Scholar - Schädel M., Shaughnessy D., eds. The Chemistry of Superheavy Elements 2nd edn (Springer, 2014)
- Türler, A. & Pershina, V. Advances in the production and chemistry of the heaviest elements. Chem. Rev. 113, 1237–1312 (2013)
Article Google Scholar - Schädel, M. et al. Chemical properties of element 106 (seaborgium). Nature 388, 55–57 (1997)
Article ADS Google Scholar - Eichler, R. et al. Chemical characterization of bohrium (element 107). Nature 407, 63–65 (2000)
Article CAS ADS Google Scholar - Düllmann, E. et al. Chemical investigation of hassium (element 108). Nature 418, 859–862 (2002)
Article ADS Google Scholar - Eichler, R. et al. Chemical characterization of element 112. Nature 447, 72–75 (2007)
Article CAS ADS Google Scholar - Even, J. et al. Synthesis and detection of a seaborgium carbonyl complex. Science 345, 1491–1493 (2014)
Article CAS ADS Google Scholar - Köhler, S. et al. First experimental determination of the ionization potentials of berkelium and californium. Angew. Chem. Int. Edn Engl. 35, 2856–2858 (1996)
Article Google Scholar - Peterson, J. et al. Determination of the first ionization potential of einsteinium by resonance ionization mass spectroscopy (RIMS). J. Alloy. Comp. 271–273, 876–878 (1998)
Article Google Scholar - Sewtz, M. et al. First observation of atomic levels for the element fermium (Z = 100). Phys. Rev. Lett. 90, 163002 (2003)
Article CAS ADS Google Scholar - Rothe, S. et al. Measurement of the first ionization potential of astatine by laser ionization spectroscopy. Nature Commun. 4, 1835 (2013)
Article CAS ADS Google Scholar - Desclaux, J.-P. & Fricke, B. Relativistic prediction of the ground state of atomic lawrencium. J. Phys. 41, 943–946 (1980)
Article CAS Google Scholar - Eliav, E., Kaldor, U. & Ishikawa, Y. Transition energies of ytterbium, lutetium, and lawrencium by the relativistic coupled-cluster method. Phys. Rev. A 52, 291–296 (1995)
Article CAS ADS Google Scholar - Zou, Y. & Fischer, C. F. Resonance transition energies and oscillator strengths in lutetium and lawrencium. Phys. Rev. Lett. 88, 183001 (2002)
Article ADS Google Scholar - Borschevsky, A. et al. Transition energies of atomic lawrencium. Eur. Phys. J. D 45, 115–119 (2007)
Article CAS ADS Google Scholar - Dzuba, V. A., Safronova, M. S. & Safronova, U. I. Atomic properties of superheavy elements No, Lr, and Rf. Phys. Rev. A 90, 012504 (2014)
Article ADS Google Scholar - Sato, T. K. et al. First successful ionization of Lr (Z = 103) by a surface-ionization technique. Rev. Sci. Instrum. 84, 023304(5) (2013)
Article ADS Google Scholar - Stora, T. Radioactive Ion Sources. CERN-2013-007, 331–349 http://cds.cern.ch/record/1693046 (accessed, November 2014)
Google Scholar - Zandberg, É. Y. & Ionov, N. I. Surface ionization. Sov. Phys. Usp. 2, 255–281 (1959)
Article ADS Google Scholar - Kirchner, R. On the thermoionization in hot cavities. Nucl. Instrum. Methods A 292, 203–208 (1990)
Article ADS Google Scholar - Kramida, A., Ralchenko, Y., Reader, J. & Team, N. A. NIST Atomic Spectra Database version 5.1 (2013); http://physics.nist.gov/asd (accessed, July 2014)
Google Scholar - Shabaev, V. M., Tupitsyn, I. I. & Yerokhin, V. A. QEDMOD: Fortran program for calculating the model Lamb-shift operator. Comput. Phys. Commun. 189, 175–181 (2015)
Article CAS ADS Google Scholar - Maeda, H., Mizugai, Y., Matsumoto, Y., Suzuki, A. & Takami, M. Highly excited even Rydberg series of Lu I studied by two-step laser photoionization spectroscopy. J. Phys. B 22, L511–L516 (1989)
Article CAS ADS Google Scholar - Liu, W., Küchle, W. & Dolg, M. Ab initio pseudopotential and density-functional all-electron study of ionization and excitation energies of actinide atoms. Phys. Rev. A 58, 1103–1110 (1998)
Article CAS ADS Google Scholar - Cao, X., Dolg, M. & Stoll, H. Valence basis sets for relativistic energy-consistent small-core actinide pseudopotentials. J. Chem. Phys. 118, 487–496 (2003)
Article CAS ADS Google Scholar - Sato, N. et al. Production of 256Lr in the 249,250,251Cf + 11B, 243Am + 18O, and 248Cm + 14N reactions. Radiochim. Acta 102, 211–219 (2014)
CAS Google Scholar - Sato, T. K. et al. Development of a He/CdI2 gas-jet system coupled to a surface-ionization type ion-source in JAEA-ISOL: towards determination of the first ionization potential of Lr (Z = 103). J. Radioanal. Nucl. Chem. 303, 1253–1257 (2015)
Article CAS Google Scholar - Visscher, L. et al. DIRAC, a relativistic ab initio electronic structure program. Release DIRAC13 http://www.diracprogram.org (2013)
- Faegri, K. Relativistic Gaussian basis sets for the elements K-Uuo. Theor. Chem. Acc. 105, 252–258 (2001)
Article CAS Google Scholar - Visscher, L. & Dyall, K. G. Dirac–Fock atomic electronic structure calculations using different nuclear charge distributions. At. Data Nucl. Data Tables 67, 207–224 (1997)
Article CAS ADS Google Scholar - Eliav, E., Kaldor, U. & Ishikawa, Y. Open-shell relativistic coupled-cluster method with Dirac-Fock-Breit wave functions: energies of the gold atom and its cation. Phys. Rev. A 49, 1724–1729 (1994)
Article CAS ADS Google Scholar
Acknowledgements
We thank the JAEA tandem accelerator crew for supplying intense and stable beams for the experiments. The 249Cf was made available by H. Nitsche (Univ. California, Berkeley); it was produced in the form of 249Bk through the former Transplutonium Element Production Program at Oak Ridge National Laboratory (ORNL) under the auspices of the Director, Office of Science, Office of Basic Energy Sciences, Chemical Sciences, Geosciences, and Biosciences Division of the US Department of Energy. Financial support by the Helmholtz-Institut Mainz is acknowledged. This work has been partly supported by the Grant-in-Aid for Scientific Research (C) no. 26390119 of the Ministry of Education, Science, Sports and Culture (MEXT).
Author information
Authors and Affiliations
- Japan Atomic Energy Agency (JAEA), Tokai, Ibaraki 319-1195, Japan,
T. K. Sato, M. Asai, N. Sato, Y. Kaneya, K. Tsukada, S. Ichikawa, Y. Nagame, A. Osa, M. Schädel & A. Toyoshima - Centre for Theoretical Chemistry and Physics, New Zealand Institute for Advanced Study, Massey University, 0745 North Shore MSC, Auckland, New Zealand,
A. Borschevsky - Helmholtz-Institut Mainz, 55099 Mainz, Germany
A. Borschevsky, Ch. E. Düllmann, K. Eberhardt & P. Thörle-Pospiech - ISOLDE, CERN, CH-1211 Geneva 23, Switzerland,
T. Stora - Graduate School of Science and Engineering, Ibaraki University, Mito, Ibaraki 310-8512, Japan,
Y. Kaneya & Y. Nagame - GSI Helmholtzzentrum für Schwerionenforschung, 64291 Darmstadt, Germany
Ch. E. Düllmann & J. Runke - Institut für Kernchemie, Johannes Gutenberg-Universität Mainz, 55099 Mainz, Germany,
Ch. E. Düllmann, K. Eberhardt, J. V. Kratz, D. Renisch, P. Thörle-Pospiech & N. Trautmann - School of Chemistry, Tel Aviv University, 69978 Tel Aviv, Israel
E. Eliav & U. Kaldor - Nishina Center for Accelerator-Based Science, RIKEN, 2-1 Hirosawa, Wako, Saitama 351-0198, Japan,
S. Ichikawa - Graduate School of Science, Hiroshima University, Kagamiyama, Higashi-Hiroshima 739-8526, Japan,
S. Miyashita - Institute of Science and Technology, Niigata University, Niigata 910-2181, Japan
K. Ooe
Authors
- T. K. Sato
- M. Asai
- A. Borschevsky
- T. Stora
- N. Sato
- Y. Kaneya
- K. Tsukada
- Ch. E. Düllmann
- K. Eberhardt
- E. Eliav
- S. Ichikawa
- U. Kaldor
- J. V. Kratz
- S. Miyashita
- Y. Nagame
- K. Ooe
- A. Osa
- D. Renisch
- J. Runke
- M. Schädel
- P. Thörle-Pospiech
- A. Toyoshima
- N. Trautmann
Contributions
T.K.S., M.A., Y.N. and M.S. prepared the main part of the manuscript, A.B., E. E. and U.K. contributed to the theory part, and T.S. to the experimental part. C.E.D. and J.V.K. commented on the manuscript. T.K.S, M.A., T.S., N.S., K.T. and S.I. developed the surface ion-source in the ISOL setup at the JAEA tandem accelerator facility. T.K.S. and M.A. were responsible for data acquisition and analysis. T.S. commented on ion-source optimizations and the data analysis procedure. K.T. prepared the 249Cf target. K.E., J.R., P.T.-P., C.E.D. and N.T. separated and provided the 249Cf for the target. The on-line experiments were performed by T.K.S., M.A., N.S., Y.K., K.T., S. I., S.M., Y.N., K.O., A.O., D.R., M.S. and A.T., while theoretical calculations were carried out by A.B., E.E. and U.K. All authors discussed the results and commented on the manuscript.
Corresponding author
Correspondence toY. Nagame.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
PowerPoint slides
Source data
Rights and permissions
About this article
Cite this article
Sato, T., Asai, M., Borschevsky, A. et al. Measurement of the first ionization potential of lawrencium, element 103.Nature 520, 209–211 (2015). https://doi.org/10.1038/nature14342
- Received: 27 November 2014
- Accepted: 06 February 2015
- Issue date: 09 April 2015
- DOI: https://doi.org/10.1038/nature14342
This article is cited by
Editorial Summary
The chemistry of element 103
The most dramatic modern revision of Mendeleev's periodic table of elements came in 1944 when Glenn T. Seaborg placed a new series of elements, the actinides (atomic numbers 89–103), below the lanthanides. In this issue of Nature, Yuichiro Nagame and colleagues report the first measurement of one of the basic atomic properties of element 103 (lawrencium), namely its first ionization potential. Lawrencium is only accessible via atom-at-a-time synthesis in heavy-ion accelerators, so experimental investigations of its properties are rare. Nagame and colleagues were able to reduce the number of atoms required to measure the ionization potential from billions to thousands, and these results — in agreement with the latest theoretical calculations — show that the last valence electron in lawrencium is the most weakly bound one in all actinides and any other element beyond group 1 of the periodic table. This signature — in a region of the periodic table where the sheer size of the atoms means that relativistic effects play a crucial role — confirms the end of the actinide series at element 103.