Recombinant protein therapeutics—success rates, market trends and values to 2010 (original) (raw)
- Feature
- Published: December 2004
Nature Biotechnology volume 22, pages 1513–1519 (2004)Cite this article
- 4118 Accesses
- 275 Citations
- 4 Altmetric
- Metrics details
Despite entering its maturation phase, the recombinant protein sector will continue to drive the growth of medical biotechnology over the next six years.
This is a preview of subscription content, access via your institution
Relevant articles
Open Access articles citing this article.
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Additional access options:
Figure 1: US approval success rates for rDNA therapeutics in two periods.
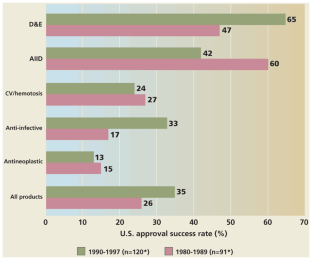
The alternative text for this image may have been generated using AI.
Bob Crimi
Figure 2: Phase transition probabilities by therapeutic category for rDNA products entering clinical study during 1990–1997.
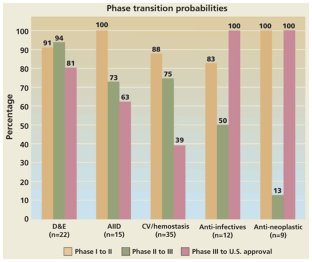
The alternative text for this image may have been generated using AI.
Bob Crimi
Figure 3: Market growth generated by three business waves and recorded between 2001 and 2003.
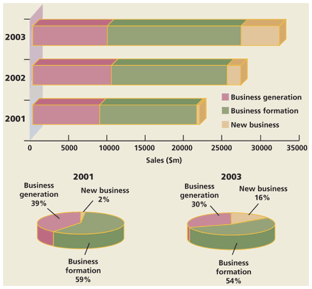
The alternative text for this image may have been generated using AI.
Bob Crimi
Figure 4: Therapeutic market segmentation from 2001 to 2003.
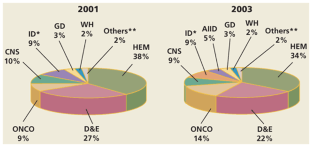
The alternative text for this image may have been generated using AI.
Bob Crimi
Figure 5: Distribution of market subsegments generated by the industry's leading recombinant protein brands within the 57% total market share in 2010.
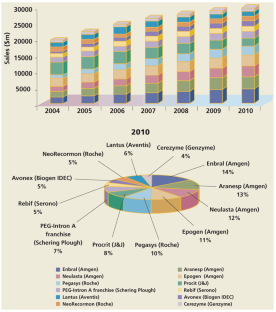
The alternative text for this image may have been generated using AI.
Bob Crimi
Figure 6: Market growth from 2004 to 2010.
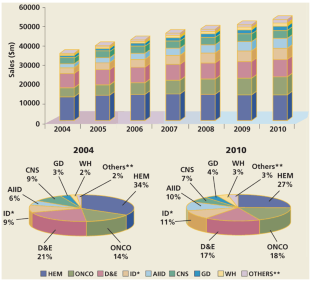
The alternative text for this image may have been generated using AI.
Bob Crimi
References
- Reichert, J.M. & Paquette, C. Therapeutic recombinant proteins: trends in US approvals 1982-2002. Curr. Opin. Mol. Ther. 5, 139–147 (2003).
CAS PubMed Google Scholar - Reichert, J.M. Biopharmaceutical approvals in the US. Reg. Affairs J. Pharma 15, 491–497 (2004).
Google Scholar - Reichert, J.M. & Pavlou, A. Monoclonal antibodies market. Nat. Rev. Drug Discovery 3, 383–384 (2004).
Article CAS Google Scholar - Reichert, J.M. Therapeutic monoclonal antibodies: trends in development and approval in the US. Curr. Opin. Mol. Ther. 4: 110–118 (2002).
CAS PubMed Google Scholar - Reichert, J.M. Monoclonal antibodies in the clinic. Nat. Biotechnol. 19, 819–822 (2001).
Article CAS Google Scholar
Author information
Authors and Affiliations
- Datamonitor plc., Charles House 108-110 Finchley Road, London, NW3 5JJ
Alex K Pavlou - Tufts Center for the Study of Drug Development, 192 South Street, Suite 550, Boston, 02111, Massachusetts, USA
Janice M Reichert
Authors
- Alex K Pavlou
- Janice M Reichert
Rights and permissions
About this article
Cite this article
Pavlou, A., Reichert, J. Recombinant protein therapeutics—success rates, market trends and values to 2010.Nat Biotechnol 22, 1513–1519 (2004). https://doi.org/10.1038/nbt1204-1513
- Issue date: December 2004
- DOI: https://doi.org/10.1038/nbt1204-1513