A highly flexible tRNA acylation method for non-natural polypeptide synthesis (original) (raw)
- Brief Communication
- Published: 20 April 2006
Nature Methods volume 3, pages 357–359 (2006)Cite this article
- 11k Accesses
- 461 Citations
- 18 Altmetric
- Metrics details
A Corrigendum to this article was published on 01 August 2006
This article has been updated
Abstract
Here we describe a de novo tRNA acylation system, the flexizyme (Fx) system, for the preparation of acyl tRNAs with nearly unlimited selection of amino and hydroxy acids and tRNAs. The combination of the Fx system with an appropriate cell-free translation system allows us to readily perform mRNA-encoded synthesis of proteins and short polypeptides involving multiple non-natural amino acids.
*Note: In the version of this article initially published, the authors did not declare competing financial interests. They have filed a patent covering some of the information described in the paper and now declare competing financial interests. This error has been corrected in the PDF version of the article.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Additional access options:
Figure 1: Acylation of tRNA by dFx and eFx.

The alternative text for this image may have been generated using AI.
Figure 2: Synthesis of a non-natural peptide by the PURE system.
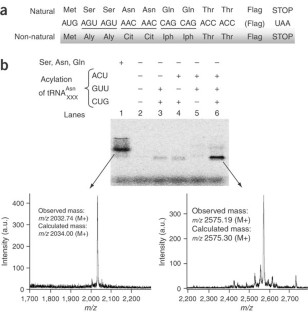
The alternative text for this image may have been generated using AI.
Similar content being viewed by others
Change history
19 July 2006
In the version of this article initially published, the authors did not declare competing financial interests. They have filed a patent covering some of the information described in the paper and now declare competing financial interests. This error has been corrected in the PDF version of the article.
References
- Forster, A.C. et al. Proc. Natl. Acad. Sci. USA 100, 6353–6357 (2003).
Article CAS Google Scholar - Josephson, K., Hartman, M.C. & Szostak, J.W. J. Am. Chem. Soc. 127, 11727–11735 (2005).
Article CAS Google Scholar - Hecht, S.M., Alford, B.L., Kuroda, Y. & Kitano, S. J. Biol. Chem. 253, 4517–4520 (1978).
CAS PubMed Google Scholar - Robertson, S.A., Ellman, J.A. & Schultz, P.G. J. Am. Chem. Soc. 113, 2711–2729 (1991).
Article Google Scholar - Wang, L., Brock, A., Herberich, B. & Schultz, P.G. Science 292, 498–500 (2001).
Article CAS Google Scholar - Xie, J. & Schultz, P.G. Methods 36, 227–238 (2005).
Article CAS Google Scholar - Link, A.J. & Tirrell, D.A. Methods 36, 291–298 (2005).
Article CAS Google Scholar - Murakami, H., Saito, H. & Suga, H. Chem. Biol. 10, 655–662 (2003).
Article CAS Google Scholar - Murakami, H., Kourouklis, D. & Suga, H. Chem. Biol. 10, 1077–1084 (2003).
Article CAS Google Scholar - Bain, J.D., Glabe, C.G., Dix, T.A. & Chamberlin, A.R. J. Am. Chem. Soc. 111, 8013–8014 (1989).
Article CAS Google Scholar - Noren, C.J., Anthony-Cahill, S.J., Griffith, M.C. & Schultz, P.G. Science 244, 182–188 (1989).
Article CAS Google Scholar - Hohsaka, T., Ashizuka, Y., Murakami, H. & Sisido, M. J. Am. Chem. Soc. 118, 9778–9779 (1996).
Article CAS Google Scholar - Shimizu, Y. et al. Nat. Biotechnol. 19, 751–755 (2001).
Article CAS Google Scholar - Murakami, H., Bonzagni, N.J. & Suga, H. J. Am. Chem. Soc. 124, 6834–6835 (2002).
Article CAS Google Scholar - Dedkova, L.M., Fahmi, N.E., Golovine, S.Y. & Hecht, S.M. J. Am. Chem. Soc. 125, 6616–6617 (2003).
Article CAS Google Scholar
Acknowledgements
We thank M. Komiyama for the use of MALDI instrumentation. This work was supported by grants from Japan Society for the Promotion of Science Grants-in-Aid for Scientific Research (S) (16101007) and from the US National Institutes of Health (GM59159).
Author information
Authors and Affiliations
- Research Center for Advanced Science and Technology, The University of Tokyo, Tokyo, 153-8904, Japan
Hiroshi Murakami, Atsushi Ohta, Hiroshi Ashigai & Hiroaki Suga - Department of Chemistry and Biotechnology, Graduate School of Engineering, The University of Tokyo, Tokyo, 113-8656, Japan
Atsushi Ohta, Hiroshi Ashigai & Hiroaki Suga
Authors
- Hiroshi Murakami
- Atsushi Ohta
- Hiroshi Ashigai
- Hiroaki Suga
Corresponding author
Correspondence toHiroaki Suga.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Murakami, H., Ohta, A., Ashigai, H. et al. A highly flexible tRNA acylation method for non-natural polypeptide synthesis.Nat Methods 3, 357–359 (2006). https://doi.org/10.1038/nmeth877
- Received: 12 December 2005
- Accepted: 22 March 2006
- Published: 20 April 2006
- Issue date: May 2006
- DOI: https://doi.org/10.1038/nmeth877