The unravelling of the ubiquitin system (original) (raw)
- Essay
- Published: 23 April 2015
Nature Reviews Molecular Cell Biology volume 16, pages 322–324 (2015)Cite this article
- 13k Accesses
- 301 Citations
- 5 Altmetric
- Metrics details
Subjects
Abstract
Today, many scientific discoveries are made using a top-down experimental approach. The ubiquitin system was discovered using a 'classic' bottom-up approach to tackle the question: 'how are cellular proteins selectively degraded?' A simple proteolytic assay, which used a crude cell-extract, was all that was required to address this question; it was followed by fractionation and reconstitution experiments to decipher the role of the components in this multi-step process. This 'biochemistry at its best' approach, which was published in a periodical that today would not be regarded as highly visible, provided magnificent findings.
This is a preview of subscription content, access via your institution
Relevant articles
Open Access articles citing this article.
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Additional access options:
Figure 1: The title and abstract of the first manuscript that prompted additional studies and resulted in the discovery of the ubiquitin proteolytic system.

The alternative text for this image may have been generated using AI.
Figure 2: APF1 is covalently conjugated to proteolytic substrates, presumably marking them for degradation by a downstream protease.
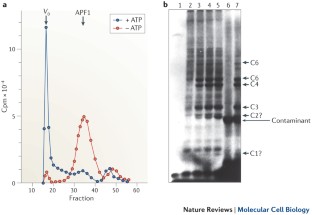
The alternative text for this image may have been generated using AI.
Figure 3: Model of the APF1 (ubiquitin)-mediated proteolytic pathway as proposed in 1980.

The alternative text for this image may have been generated using AI.
References
- Schoenheimer, R. The Dynamic State of Body Constituents. (Harvard University Press, 1942).
Google Scholar - de Duve, C. & Wattiaux, R. Functions of lysosomes. Annu. Rev. Physiol. 28, 435–492 (1966).
Article CAS Google Scholar - Simpson, M. V. The release of labeled amino acids from proteins in liver slices. J. Biol. Chem. 201, 143–154 (1953).
CAS PubMed Google Scholar - Schneider, D. L. ATP-dependent acidification of intact and disrupted lysosomes: evidence for an ATP-driven proton pump. J. Biol. Chem. 256, 3858–3864 (1981).
CAS PubMed Google Scholar - Mandelstam, J. Turnover of protein in growing and non-growing populations of Escherichia coli. Biochem. J. 69, 110–119 (1958).
Article CAS Google Scholar - Poole, B., Ohkuma, S. & Warburton, M. J. Protein Turnover and Lysosome Function (eds. Segal, H. L. & Doyle, D. J.) 43–58 (Academic, 1978).
Book Google Scholar - Rabinovitz, M. & Fisher, J. M. Characteristics of the inhibition of hemoglobin synthesis in rabbit reticulocytes by threo-α-amino-β-chlorobutyric acid. Biochim. Biophys. Acta. 91, 313–322 (1964).
CAS PubMed Google Scholar - Etlinger, J. D., & Goldberg, A. L. A soluble ATP-dependent proteolytic system responsible for the degradation of abnormal proteins in reticulocytes. Proc. Natl Acad. Sci. USA 74, 54–58 (1977).
Article CAS Google Scholar - Ciechanover, A., Hod, Y. & Hershko, A. A heat-stable polypeptide component of an ATP-dependent proteolytic system from reticulocytes. Biochem. Biophys. Res. Commun. 81, 1100–1105 (1978).
Article Google Scholar - Ciechanover, A., Heller, H., Elias, S., Haas, A. L. & Hershko, A. ATP-dependent conjugation of reticulocyte proteins with the polypeptide required for protein degradation. Proc. Natl Acad. Sci. USA 77, 1365–1368 (1980).
Article CAS Google Scholar - Hershko, A., Ciechanover, A., Heller, H., Haas, A. L. & Rose, I. A. Proposed role of ATP in protein breakdown: conjugation of proteins with multiple chains of the polypeptide of ATP-dependent proteolysis. Proc. Natl Acad. Sci. USA 77, 1783–1786 (1980).
Article CAS Google Scholar - Ciechanover, A., Elias, S., Heller, H., Ferber, S. & Hershko, A. Characterization of the heat-stable polypeptide of the ATP-dependent proteolytic system from reticulocytes. J. Biol. Chem. 255, 7525–7528 (1980).
CAS PubMed Google Scholar - Wilkinson, D., Urban, M. K. & Haas, A. L. Ubiquitin is the ATP-dependent proteolysis factor I of rabbit reticulocytes. J. Biol. Chem. 255, 7529–7532 (1980).
CAS PubMed Google Scholar - Goldknopf, I. L. & Busch, H. Isopeptide linkage between nonhistone and histone 2A polypeptides of chromosomal conjugate-protein A24. Proc. Natl Acad. Sci. USA 74, 864–868 (1977).
Article CAS Google Scholar - de Napoles, M. et al. Polycomb group proteins Ring1A/B link ubiquitylation of histone H2A to heritable gene silencing and X inactivation. Dev. Cell 7, 663–676 (2004).
Article CAS Google Scholar - Hershko, A., Eytan, E., Ciechanover, A. & Haas, A. L. Immunochemical analysis of the turnover of ubiquitin-protein conjugates in intact cells. Relationship to the breakdown of abnormal proteins. J. Biol. Chem. 257, 13964–13970 (1982).
CAS PubMed Google Scholar - Hershko, A., Heller, H., Elias, S. & Ciechanover, A. Components of ubiquitin-protein ligase system. Resolution, affinity purification, and role in protein breakdown. J. Biol. Chem. 258, 8206–8214 (1983).
CAS PubMed Google Scholar - Hough, R., Pratt, G. & Rechsteiner, M. Ubiquitin-lysozyme conjugates. Identification and characterization of an ATP-dependent protease from rabbit reticulocyte lysates. J. Biol. Chem. 261, 2400–2408 (1986).
CAS PubMed Google Scholar - Waxman, L., Fagan, J. M. & Goldberg, A. L. Demonstration of two distinct high molecular weight proteases in rabbit reticulocytes, one of which degrades ubiquitin conjugates. J. Biol. Chem. 262, 2451–2457 (1987).
CAS PubMed Google Scholar
Acknowledgements
Research in the author's laboratory is supported by grants from the Dr. Miriam and Sheldon G. Adelson Medical Research Foundation (AMRF), the Israel Science Foundation (ISF), the Israeli Centers for Research Excellence (I-CORE) Program of the Israeli Planning and Budgeting Committee and the Israel Science Foundation (ISF) (Grant1775/12), the EU Treat PolyQ Network, and the Deutsch-Israelische Projektkooperation (DIP). The author is an Israel Cancer Research Fund (ICRF) USA professor.
Author information
Authors and Affiliations
- Aaron Ciechanover is at the Cancer and Vascular Biology Research Center, Faculty of Medicine, Technion – Israel Institute of Technology, Haifa 31096, Israel.,
Aaron Ciechanover
Corresponding author
Correspondence toAaron Ciechanover.
Ethics declarations
Competing interests
The author declares no competing financial interests.
PowerPoint slides
Rights and permissions
About this article
Cite this article
Ciechanover, A. The unravelling of the ubiquitin system.Nat Rev Mol Cell Biol 16, 322–324 (2015). https://doi.org/10.1038/nrm3982
- Published: 23 April 2015
- Issue date: May 2015
- DOI: https://doi.org/10.1038/nrm3982