Remarkable muscles, remarkable locomotion in desert-dwelling wildebeest (original) (raw)
- Letter
- Published: 24 October 2018
- Hattie L. A. Bartlam-Brooks1,
- Tatjana Y. Hubel1,
- John C. Lowe1,
- Anthony R. Gardner-Medwin2,
- Emily Bennitt3,
- Stephen J. Amos1,
- Maja Lorenc1,
- Timothy G. West1 &
- …
- Alan M. Wilson1
Nature volume 563, pages 393–396 (2018) Cite this article
- 9606 Accesses
- 35 Citations
- 108 Altmetric
- Metrics details
Subjects
Abstract
Large mammals that live in arid and/or desert environments can cope with seasonal and local variations in rainfall, food and climate1 by moving long distances, often without reliable water or food en route. The capacity of an animal for this long-distance travel is substantially dependent on the rate of energy utilization and thus heat production during locomotion—the cost of transport2,3,4. The terrestrial cost of transport is much higher than for flying (7.5 times) and swimming (20 times)4. Terrestrial migrants are usually large1,2,3 with anatomical specializations for economical locomotion5,6,7,8,9, because the cost of transport reduces with increasing size and limb length5,6,7. Here we used GPS-tracking collars10 with movement and environmental sensors to show that blue wildebeest (Connochaetes taurinus, 220 kg) that live in a hot arid environment in Northern Botswana walked up to 80 km over five days without drinking. They predominantly travelled during the day and locomotion appeared to be unaffected by temperature and humidity, although some behavioural thermoregulation was apparent. We measured power and efficiency of work production (mechanical work and heat production) during cyclic contractions of intact muscle biopsies from the forelimb flexor carpi ulnaris of wildebeest and domestic cows (Bos taurus, 760 kg), a comparable but relatively sedentary ruminant. The energetic costs of isometric contraction (activation and force generation) in wildebeest and cows were similar to published values for smaller mammals. Wildebeest muscle was substantially more efficient (62.6%) than the same muscle from much larger cows (41.8%) and comparable measurements that were obtained from smaller mammals (mouse (34%)11 and rabbit (27%)). We used the direct energetic measurements on intact muscle fibres to model the contribution of high working efficiency of wildebeest muscle to minimizing thermoregulatory challenges during their long migrations under hot arid conditions.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout
Additional access options:
Fig. 1: Locomotion of desert wildebeest.
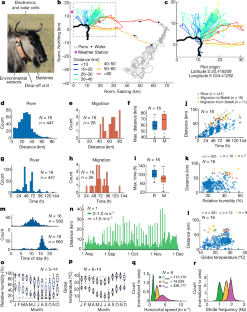
The alternative text for this image may have been generated using AI.
Fig. 2: Example records.
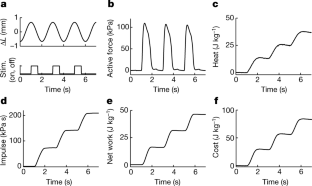
The alternative text for this image may have been generated using AI.
Fig. 3: Muscle mechanical and energetic performance.
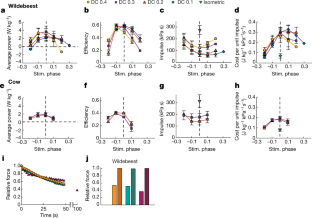
The alternative text for this image may have been generated using AI.
Similar content being viewed by others
Data availability
The authors declare that all relevant processed data supporting the findings of this study are available as Source Data. Further data are available from the corresponding authors upon reasonable request.
References
- Schmidt-Nielsen, K. Desert Animals: Physiological Problems of Heat and Water (Clarendon, Oxford, 1965).
Google Scholar - Hedenström, A. Optimal migration strategies in animals that run: a range equation and its consequences. Anim. Behav. 66, 631–636 (2003).
Article Google Scholar - Hein, A. M., Hou, C. & Gillooly, J. F. Energetic and biomechanical constraints on animal migration distance. Ecol. Lett. 15, 104–110 (2012).
Article Google Scholar - Tucker, V. A. The energetic cost of moving about: walking and running are extremely inefficient forms of locomotion. Much greater efficiency is achieved by birds, fish—and bicyclists. Am. Sci. 63, 413–419 (1975).
ADS CAS PubMed Google Scholar - Taylor, C. R., Heglund, N. C. & Maloiy, G. M. O. Energetics and mechanics of terrestrial locomotion. I. Metabolic energy consumption as a function of speed and body size in birds and mammals. J. Exp. Biol. 97, 1–21 (1982).
CAS PubMed Google Scholar - Kram, R. & Taylor, C. R. Energetics of running: a new perspective. Nature 346, 265–267 (1990).
Article ADS CAS Google Scholar - Wilson, A. M., Watson, J. C. & Lichtwark, G. A. Biomechanics: a catapult action for rapid limb protraction. Nature 421, 35–36 (2003).
Article ADS CAS Google Scholar - Alexander, R., Maloiy, G. M. O., Njau, R. & Jayes, A. Mechanics of running of the ostrich (Struthio camelus). J. Zool. 187, 169–178 (1979).
Article Google Scholar - Alexander, R., Maloiy, G. M. O., Ker, R., Jayes, A. & Warui, C. The role of tendon elasticity in the locomotion of the camel (Camelus dromedarius). J. Zool. 198, 293–313 (1982).
Article Google Scholar - Wilson, A. M. et al. Locomotion dynamics of hunting in wild cheetahs. Nature 498, 185–189 (2013).
Article ADS CAS Google Scholar - Barclay, C. J. Efficiency of fast- and slow-twitch muscles of the mouse performing cyclic contractions. J. Exp. Biol. 193, 65–78 (1994).
CAS PubMed Google Scholar - Hetem, R. S., Maloney, S. K., Fuller, A., Meyer, L.C. & Mitchell, D. Validation of a biotelemetric technique, using ambulatory miniature black globe thermometers, to quantify thermoregulatory behaviour in ungulates. J. Exp. Zool. 307A, 342–356 (2007).
Article Google Scholar - Estes, R. The Behavior Guide to African Mammals Vol. 64 (Univ. California Press, Berkeley, 1991).
Google Scholar - Kuo, A. D. A simple model of bipedal walking predicts the preferred speed-step length relationship. J. Biomech. Eng. 123, 264–269 (2001).
Article CAS Google Scholar - Bertram, J. E. Constrained optimization in human walking: cost minimization and gait plasticity. J. Exp. Biol. 208, 979–991 (2005).
Article Google Scholar - Kohn, T. A., Curry, J. W. & Noakes, T. D. Black wildebeest skeletal muscle exhibits high oxidative capacity and a high proportion of type IIx fibres. J. Exp. Biol. 214, 4041–4047 (2011).
Article CAS Google Scholar - Woledge, R. C. The energetics of tortoise muscle. J. Physiol. 197, 685–707 (1968).
Article CAS Google Scholar - Williams, T. L. Experimental analysis of the gait and frequency of locomotion in the tortoise, with a simple mathematical description. J. Physiol. 310, 307–320 (1981).
Article CAS Google Scholar - Barclay, C. J. Energetics of contraction. Compr. Physiol. 5, 961–995 (2015).
Article CAS Google Scholar - Butler, P. J. et al. Respiratory and cardiovascular adjustments during exercise of increasing intensity and during recovery in thoroughbred racehorses. J. Exp. Biol. 179, 159–180 (1993).
CAS PubMed Google Scholar - Hetem, R. S., Maloney, S. K., Fuller, A. & Mitchell, D. Heterothermy in large mammals: inevitable or implemented? Biol. Rev. Camb. Philos. Soc. 91, 187–205 (2016).
Article Google Scholar - Valeix, M. et al. Behavioral adjustments of African herbivores to predation risk by lions: spatiotemporal variations influence habitat use. Ecology 90, 23–30 (2009).
Article CAS Google Scholar - MacFarlane, W. V., Howard, B., Haines, H., Kennedy, P. & Sharpe, C. M. Hierarchy of water and energy turnover of desert mammals. Nature 234, 483–484 (1971).
Article ADS Google Scholar - Maloiy, G. M. O. Water economy of the Somali donkey. Am. J. Physiol. 219, 1522–1527 (1970).
CAS PubMed Google Scholar - Wilson, A. M. et al. Biomechanics of predator–prey arms race in lion, zebra, cheetah and impala. Nature 554, 183–188 (2018).
Article ADS CAS Google Scholar - Dewhirst, O. P. et al. Improving the accuracy of estimates of animal path and travel distance using GPS drift-corrected dead reckoning. Ecol. Evol. 6, 6210–6222 (2016).
Article Google Scholar - Hubel, T. Y. et al. Energy cost and return for hunting in African wild dogs and cheetahs. Nat. Commun. 7, 11034 (2016).
Article ADS CAS Google Scholar - Hubel, T. Y. et al. Additive opportunistic capture explains group hunting benefits in African wild dogs. Nat. Commun. 7, 11033 (2016).
Article ADS CAS Google Scholar - West, T. G. et al. Power output of skinned skeletal muscle fibres from the cheetah (Acinonyx jubatus). J. Exp. Biol. 216, 2974–2982 (2013).
Article Google Scholar - Barclay, C. J., Woledge, R. C. & Curtin, N. A. Is the efficiency of mammalian (mouse) skeletal muscle temperature dependent? J. Physiol. 588, 3819–3831 (2010).
Article CAS Google Scholar - Kretzschmar, K. M. & Wilkie, D. R. The use of the Peltier effect for simple and accurate calibration of thermoelectric devices. Proc. R. Soc. Lond. B 190, 315–321 (1975).
Article ADS CAS Google Scholar - Woledge, R. C., Curtin, N. A. & Homsher, E. Energetic aspects of muscle contraction. Monogr. Physiol. Soc. 41, 1–357 (1985).
CAS PubMed Google Scholar - Hill, A. Trails and Trials in Physiology (E. Arnold, London, 1965).
Google Scholar - Phillips, S. K., Takei, M. & Yamada, K. The time course of phosphate metabolites and intracellular pH using 31P NMR compared to recovery heat in rat soleus muscle. J. Physiol. 460, 693–704 (1993).
Article CAS Google Scholar - Curtin, N. A. & Woledge, R. C. Efficiency of energy conversion during sinusoidal movement of white muscle fibres from the dogfish, Scyliorhinus canicula. J. Exp. Biol. 183, 137–147 (1993).
Google Scholar - Barclay, C. J. Mechanical efficiency and fatigue of fast and slow muscles of the mouse. J. Physiol. 497, 781–794 (1996).
Article CAS Google Scholar - Curtin, N. A. & Woledge, R. C. Efficiency of energy conversion during shortening of muscle fibres from the dogfish Scyliorhinus canicula. J. Exp. Biol. 158, 343–353 (1991).
CAS PubMed Google Scholar - Rumble, J. R. (ed.) CRC Handbook of Chemistry and Physics 99th edn (CRC, Boca Raton, 2018).
- Hetem, R. S. et al. Variation in the daily rhythm of body temperature of free-living Arabian oryx (Oryx leucoryx): does water limitation drive heterothermy? J. Comp. Physiol. B 180, 1111–1119 (2010).
Article Google Scholar - Maloiy, G. M. O. Water metabolism of East African ruminants in arid and semi-arid regions. Z. Tierzuecht. Zuechtungsbiol. 90, 219–228 (1973).
Article Google Scholar
Acknowledgements
We thank R. Woledge for contributing to early design of experiments; C. Barclay for helping us to fabricate the thermocouple elements; our field assistants, N. Terry and M. Claase; A. R. Wilson for logistical support and editorial contributions; M. Flyman (Department of Wildlife and National Parks) for his support and enthusiasm and J. O’Connor and P. O’Riordan (Dawn Meats, Bedford) for enabling cow muscle collection. Funding was provided by the EPSRC (EP/H013016/1), BBSRC (BB/J018007/1) and ERC (323041). A Botswana Research Permit EWT 8/36/4 was held by A.M.W. and A.M.W. was a registered Botswana veterinarian.
Reviewer information
Nature thanks J. E. A. Bertram, R. Hetem and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Author information
Authors and Affiliations
- Structure & Motion Laboratory, Royal Veterinary College, University of London, Hatfield, UK
Nancy A. Curtin, Hattie L. A. Bartlam-Brooks, Tatjana Y. Hubel, John C. Lowe, Stephen J. Amos, Maja Lorenc, Timothy G. West & Alan M. Wilson - Department of Neuroscience, Physiology & Pharmacology, University College London, London, UK
Anthony R. Gardner-Medwin - Okavango Research Institute, University of Botswana, Maun, Botswana
Emily Bennitt
Authors
- Nancy A. Curtin
- Hattie L. A. Bartlam-Brooks
- Tatjana Y. Hubel
- John C. Lowe
- Anthony R. Gardner-Medwin
- Emily Bennitt
- Stephen J. Amos
- Maja Lorenc
- Timothy G. West
- Alan M. Wilson
Contributions
A.M.W., N.A.C. and H.L.A.B.-B. conceived, designed and led the study; H.L.A.B.-B. and E.B. led and organized field work. A.M.W. performed veterinary procedures and biopsies. N.A.C., A.R.G.-M., M.L. and T.G.W. undertook muscle experiments and N.A.C. and A.R.G.-M. analysed and interpreted muscle data. J.C.L., A.M.W. and S.J.A. designed and built collars and weather stations. T.Y.H., A.M.W. and H.L.A.B.-B. analysed and interpreted collar data, A.M.W. made the water balance model and A.M.W. and N.A.C. wrote paper with input from all authors.
Corresponding authors
Correspondence toNancy A. Curtin or Alan M. Wilson.
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Comparison of temperature maxima and minima recorded in the collars and at weather stations.
The number of working collar sensors varied; therefore, a median was taken from all available data on each day and the maximum and minimum value for each day, which was then averaged over each month. The monthly maximum temperature (mean ± s.d.) was 5.6 ± 0.6 °C higher at the weather stations than the collars and the monthly minimum temperature was, on average, 3.6 ± 1.1 °C lower at the weather stations than the collars. n = 12. Ambient temperature exceeded body temperature of 38 °C (horizontal dashed line) during nine months of the year. Note weather stations were 10 km away from the river in the dry season range, while animals were in the wet season range to the east from November to April approximately. (Fig. 1b, c).
Extended Data Fig. 2 Controlled variables for muscle length and stimulation pattern.
a, Pattern of lever movement. Frequency of 0.5 Hz and peak-to-peak amplitude 18% _L_o (10% _L_o). _L_o is the fibre bundle length at which isometric force was greatest. Values for cow experiments are in parentheses, where they are different than those for wildebeest. b, Stimulus duty cycles used in the experiments. Top to bottom, duty cycle of 0.1, 0.2, 0.3 and 0.4 (0.2 and 0.3). c, Stimulus phases used in the experiments. Top to bottom: phase −0.2, −0.1, 0, 0.1, 0.2 and 0.3. (−0.2 to 0.1). Phase = 0.0 corresponds to the stimulus starting when shortening starts. In this example, DC = 0.4.
Extended Data Fig. 3 Individual data points for muscle mechanical and energetic performance.
Data presented in Fig. 3, but subdivided by duty cycle. The mean is plotted, symbols and line colours are as in Fig. 3, n numbers are given in Extended Data Table 2.
Extended Data Fig. 4 Efficiency versus stimulus phase for individual muscle fibre bundles from wildebeest and cows.
a–f, Data from wildebeest. g–k, Data from cows. Relationship between stimulus phase and efficiency during three cycles of movement at 0.5 Hz for stimulus duty cycles (DC). Circle, DC = 0.4; square, DC = 0.3; triangle, DC = 0.2; diamond, DC = 0.1. Efficiency = power per rate of heat + work output. a, b, d, Data are from a different muscle fibre bundle each from a different wildebeest. c, Data are the average of the values shown in e and f, which are results for two fibre bundles from the same wildebeest. g–k, Data are from a different fibre bundle from a different cow.
Extended Data Table 1 Subject data
Extended Data Table 2 n values
Extended Data Table 3 Minimum cost per unit impulse, values and comparison by species
Extended Data Table 4 Maximum enthalpy efficiency value for muscle fibre bundles from wildebeest and cow
Extended Data Table 5 Values of maximum enthalpy efficiency for locomotor muscles from different species
Extended Data Table 6 Calculation of cross-bridge work from enthalpy efficiency
Supplementary information
Source data
Rights and permissions
About this article
Cite this article
Curtin, N.A., Bartlam-Brooks, H.L.A., Hubel, T.Y. et al. Remarkable muscles, remarkable locomotion in desert-dwelling wildebeest.Nature 563, 393–396 (2018). https://doi.org/10.1038/s41586-018-0602-4
- Received: 12 April 2018
- Accepted: 24 August 2018
- Published: 24 October 2018
- Version of record: 24 October 2018
- Issue date: 15 November 2018
- DOI: https://doi.org/10.1038/s41586-018-0602-4