The Novel Dopamine D3 Receptor Antagonist NGB 2904 Inhibits Cocaine's Rewarding Effects and Cocaine-Induced Reinstatement of Drug-Seeking Behavior in Rats (original) (raw)
INTRODUCTION
Dopamine (DA) D3 receptors have recently drawn attention as a potential target for medication development for drug addiction, in part because of their unique regional distribution in the mesolimbic DA system (Murray et al, 1994; Levant, 1997; Stanwood et al, 2000). Virtually all addictive drugs activate the mesolimbic DA system (see for a review Gardner, 2005), and this action is believed to underlie drug reward and relapse (Wise, 1996a; Shalev et al, 2002; Wise and Gardner, 2002). The D3 receptor has the highest binding affinity to endogenous DA of all known receptors (see for a review Sokoloff et al, 1992a; Levant, 1997), suggesting a vital role in the normal functioning of the mesolimbic DA system. D3 receptor activation appears to enhance cocaine-induced reinforcement (Parsons et al, 1996). Congruently, it has been hypothesized that blockade of D3 receptors may selectively block drug reward and relapse (Sokoloff et al, 1992a, 1992b; Caine and Koob, 1993).
However, this hypothesis had been difficult to test due to the lack of D3-receptor-selective ligands suitable for in vivo study. Recently, the D3 partial agonist BP-897 (Pilla et al, 1999) and several D3-selective antagonists such as SB-277011A, S33084, A-437203, and NGB 2904 (_N_-(4-[4-{2,3-dichlorophenyl}-1-piperazinyl]butyl)-3-fluorenylcarboxamide) have become available (Yuan et al, 1998; Stemp et al, 2000; Reavill et al, 2000; Millan et al, 2000), and some have been evaluated in animal models of addiction (for a review see Heidbreder et al, 2005). SB-277011A is the best characterized of these (see reviews by Le Foll et al, 2000; Schwartz et al, 2000; Remington and Kapur, 2001; Heidbreder et al, 2005). It significantly inhibits the rewarding effects of cocaine, heroin, and nicotine, as assessed by intravenous (i.v.) drug self-administration, intracranial brain stimulation reward (BSR), and conditioned place preference in rats (Vorel et al, 2002; [Campos et al, 2003](/articles/1300912#ref-CR14 "Campos AC, Xi Z-X, Gilbert J, Ashby Jr CR, Heidbreder CA, Gardner EL (2003). The dopamine D3 receptor antagonist SB277011A antagonizes nicotine-enhanced brain-stimulation reward in rat. Abstracts of the 33rd Annual Meeting of the Society for Neuroscience, 2003 November 7–12; New Orleans, LA, 2003 Abstract Viewer/Itinerary Planner. Online at http://sfn.scholarone/itin2003/index.html
Washington, DC (Abstract No. 322.8)."); [Ashby et al, 2003](/articles/1300912#ref-CR4 "Ashby Jr CR, Paul M, Gardner EL, Heidbreder CA, Hagan JJ (2003). Acute administration of the selective D3 receptor antagonist SB-277011A blocks the acquisition and expression of the conditioned place preference response to heroin in male rats. Synapse 48: 154–156."); [Gilbert et al, 2003](/articles/1300912#ref-CR27 "Gilbert J, Xi Z-X, Campos AC, Ashby Jr CR, Heidbreder CA, Gardner EL (2003). The dopamine D3 receptor antagonist SB277011A inhibits cocaine reinforcement under fixed-ratio and progressive-ratio schedules. Abstracts of the 33rd Annual Meeting of the Society for Neuroscience, 2003 November 7–12; New Orleans, LA, 2003 Abstract Viewer/Itinerary Planner. Online at
http://sfn.scholarone.com/itin2003/index.html
Washington, DC (Abstract No. 422.10).")). It also inhibits drug-seeking behavior as measured by second-order reinforcement and by cocaine-, nicotine-, or stress-triggered relapse to drug-seeking in the reinstatement model ([Vorel et al, 2002](/articles/1300912#ref-CR81 "Vorel SR, Ashby Jr CR, Paul M, Liu X, Hayes R, Hagan JJ et al (2002). Dopamine D3 receptor antagonism inhibits cocaine-seeking and cocaine-enhanced brain reward in rats. J Neurosci 22: 9595–9603."); [Andreoli et al, 2003](/articles/1300912#ref-CR2 "Andreoli M, Tessari M, Pilla M, Valerio E, Hagan JJ, Heidbreder CA (2003). Selective antagonism at dopamine D3 receptors prevents nicotine-triggered relapse to nicotine-seeking behavior. Neuropsychopharmacology 28: 1272–1280."); [Di Ciano et al, 2003](/articles/1300912#ref-CR21 "Di Ciano P, Underwood RJ, Hagan JJ, Everitt BJ (2003). Attenuation of cue-controlled cocaine-seeking by a selective D3 dopamine receptor antagonist SB-277011-A. Neuropsychopharmacology 28: 329–338."); [Cervo et al, 2003](/articles/1300912#ref-CR16 "Cervo L, Cocco A, Petrella C, Heidbreder CA, Bendotti C, Mennini T (2003). SB-277011-A, a selective dopamine D3 receptor antagonist, reduces cocaine-seeking behavior in response to drug-associated stimuli in rats. Abstracts of the 33rd Annual Meeting of the Society for Neuroscience, 2003 November 7–12; New Orleans, LA, 2003 Abstract Viewer/Itinerary Planner. Online at
http://sfn.scholarone.com/itin2003/index.html
Washington, DC (Abstract No. 420.3)."); [Xi et al, 2004](/articles/1300912#ref-CR95 "Xi Z-X, Gilbert J, Campos AC, Kline N, Ashby Jr CR, Hagan JJ et al (2004). Blockade of mesolimbic dopamine D3 receptors inhibits stress-induced reinstatement of cocaine-seeking in rats. Psychopharmacology 176: 57–65.")). These data strongly support the potential of SB-277011A in the treatment of drug addiction. However, clinical development of SB-277011A has been terminated, due to its short half-life and poor bioavailability in primates ([Austin et al, 2001](/articles/1300912#ref-CR5 "Austin NE, Baldwin SJ, Cutler L, Deeks N, Kelly PJ, Nash M et al (2001). Pharmacokinetics of the novel, high-affinity and selective dopamine D3 receptor antagonist SB-277011 in rat, dog and monkey: in vitro/in vivo correlation and the role of aldehyde oxidase. Xenobiotica 31: 677–686."); [Heidbreder et al, 2005](/articles/1300912#ref-CR31 "Heidbreder CA, Gardner EL, Xi Z-X, Thanos PK, Mugnaini M, Hagan JJ et al (2005). The role of central dopamine D3 receptors in drug addiction: a review of pharmacological evidence. Brain Res Rev 49: 77–105.")).Another novel highly selective DA D3 receptor antagonist, NGB 2904, has been synthesized (Yuan et al, 1998). This compound has structural similarity to BP-897 (Pilla et al, 1999; Wood et al, 2000; Wicke and Garcia-Ladona, 2001), and binds with high affinity to cloned primate D3 receptors (_K_i 1.4 nM) (Yuan et al, 1998; Robarge et al, 2001). NGB 2904 is reported to have 155-fold selectivity for primate D3 over primate D2 receptors, and >800-fold selectivity for rat D3 vs D2 receptors (Yuan et al, 1998; Newman et al, 2003). Also, it has >5000-fold selectivity over D1, D4, and D5 receptors and 200- to 600-fold selectivity over _α_1 and 5HT2 receptors (Yuan et al, 1998). Preliminary pharmacokinetic studies show that NGB 2904 is orally bioavailable and can penetrate the blood–brain barrier (unpublished data from GlaxoSmithKline by Heidbreder et al, personal communication). However, only limited behavioral evaluation of NGB 2904 in animal models of drug reward and relapse has been reported.
Therefore, in this study, we sought to behaviorally evaluate NGB 2904 in animal models of drug reward and relapse by determining: (1) whether blockade of D3 receptors by NGB 2904 alters cocaine's rewarding effects, measured by i.v. cocaine self-administration under both fixed-ratio (FR) and progressive-ratio (PR) reinforcement schedules, and by BSR; (2) whether NGB 2904 itself has reinforcing effects, as assessed by NGB 2904 replacement for cocaine in self-administration, and as assessed by the effects of NGB 2904 itself on BSR; and (3) whether NGB 2904 alters cocaine-triggered reinstatement (relapse) of drug-seeking behavior, as compared to natural reinforcer-triggered reinstatement of goal-seeking behavior.
MATERIALS AND METHODS
Animals
Experimentally naïve male Long–Evans rats (Charles River Laboratories, Raleigh, NC, USA) weighing 250–300 g were used for all experiments. They were housed individually in a climate-controlled animal colony room on a reversed light–dark cycle (lights on at 19 00 hours, lights off at 0700 hours) with free access to food and water. The animals were maintained in a facility fully accredited by the Association for Assessment and Accreditation of Laboratory Animal Care. All experimental procedures were conducted in accordance with the Guide for the Care and Use of Laboratory Animals (National Academy of Sciences, 1996) and were approved by the Animal Care and Use Committee of the National Institute on Drug Abuse of the US National Institutes of Health.
Experimental Set 1: Cocaine Self-Administration
Surgery
All animals were prepared for experimentation by surgical catheterization of the right external jugular vein. The venous catheters were constructed of microrenathane (Braintree Scientific Inc., Braintree, MA, USA), and catheterization was performed under sodium pentobarbital anesthesia (65 mg/kg intraperitoneal (i.p.)) with aseptic surgical technique. After exiting the jugular, the catheter passed subcutaneously to the top of the skull, where it exited into a connector (a modified 24 G cannula; Plastics One, Roanoke, VA, USA) mounted to the skull with jeweler's screws and dental acrylic. During experimental sessions, the catheter was connected to the injection pump via tubing encased in a protective metal spring from the head-mounted connector to the top of the experimental chamber. To help prevent clogging, the catheters were flushed daily with a gentamicin–heparin–saline solution (30 IU/ml heparin; ICN Biochemicals, Cleveland, OH, USA).
Apparatus
The i.v. self-administration experiments were conducted in operant response test chambers (32 × 25 × 33 cm) from MED Associates Inc. (Georgia, VT, USA). Each test chamber had two levers located 6.5 cm above the floor, one active and one inactive. Depression of the active lever activated the infusion pump; depression of the inactive lever was counted but had no consequence. A cue-light and a speaker were located 12 cm above the active lever. The house light was turned on at the start of each 3 h test session. When the animal performed a lever-press that resulted in a drug infusion, it was exposed to two drug-paired environmental cues: a cue-light and a cue-sound (tone) that lasted for the duration of the infusion. Scheduling of experimental events and data collection were accomplished using MED Associates software.
General procedure
After recovery from surgery, each rat was placed into a test chamber and allowed to lever-press for i.v. cocaine (1 mg/kg/injection) delivered in 0.08 ml over 4.6 s, on an FR1 reinforcement schedule. During the 4.6 s injection time, additional responses on the active lever were recorded but did not lead to additional infusions. Each session lasted 3 h. The FR1 reinforcement schedule was used for 3–5 days until stable cocaine self-administration was established. The initial cocaine dose of 1 mg/kg/infusion was chosen on the basis of our previous experience that this dose produces the most rapid and facile acquisition of cocaine self-administration behavior. Subsequently, subjects were randomly assigned to one of the following four experiments: (1) cocaine self-administration under an FR2 reinforcement schedule, (2) cocaine self-administration under a PR reinforcement schedule, (3) NGB 2904 or saline replacement testing in experienced cocaine self-administering rats, or (4) cocaine-triggered reinstatement of drug-seeking behavior. In all experiments, NGB 2904 was given 30 min prior to testing because preliminary data showed that NGB 2904's effects occurred approximately 30 min after systemic administration.
Cocaine self-administration under FR2 reinforcement
After transition from FR1 reinforcement, subjects (_n_=10) were allowed to continue cocaine (0.5 mg/kg/infusion) self-administration under FR2 reinforcement until the following criteria for stable cocaine-maintained responding were met: <10% variability in inter-response interval and <10% variability in the number of presses on the active lever for at least 3 consecutive days. The dose of cocaine was chosen on the basis of previous findings that rats self-administering cocaine at 0.5 mg/kg/infusion display highly stable self-administration behavior. In addition, previous studies have shown that 0.5 or 1 mg/kg/infusion cocaine lies within the range of the descending limb of the cocaine dose–response self-administration curve, where stable and reliable dose-dependent effects have been observed (Weissenborn et al, 1998; Parsons et al, 1998; Xi et al, 2005). Furthermore, we chose 0.5 mg/kg rather than 1 mg/kg of cocaine in order to increase the work demand (ie lever presses) of the animals for the same amount of drug intake. In our previous experience, this approach increases the sensitivity of measuring changes in drug-taking or drug-seeking behavior. To avoid cocaine overdose during the self-administration period, each animal was limited to a maximum of 50 cocaine injections per session. After stable rates of responding were established, each subject randomly received one of four doses of NGB 2904 (0.1, 1, 5, and 10 mg/kg i.p.) or vehicle (1 ml of 25% 2-hydroxypropyl-_β_-cyclodextrin solution) 30 min prior to the test session. Animals then received an additional 5–7 days of self-administration of cocaine alone until the baseline response rate was re-established prior to testing the next dose of NGB 2904. The order of testing for the various doses of NGB 2904 was counterbalanced according to a Latin square design.
Cocaine self-administration under PR reinforcement
Initial cocaine self-administration under FR1 and FR2 reinforcement was identical to that outlined above. After stable cocaine self-administration under FR2 reinforcement was established, the subjects were switched to cocaine self-administration (0.5 mg/kg/injection) under a PR schedule, during which the work requirement of lever presses needed to receive a single i.v. cocaine infusion was progressively raised within each test session (see details in Richardson and Roberts, 1996) according to the following PR series: 1, 2, 4, 6, 9, 12, 15, 20, 25, 32, 40, 50, 62, 77, 95, 118, 145, 178, 219, 268, 328, 402, 492, and 603 until the break-point was reached. The break-point was defined as the maximal workload (ie number of lever presses) completed for the last cocaine infusion prior to a 1-h period during which no infusions were obtained by the animal. Animals were allowed to continue daily sessions of cocaine self-administration under PR reinforcement conditions until day-to-day variability in break-point fell within 1–2 ratio increments for 3 consecutive days. Once a stable break-point was established, subjects were assigned to four subgroups to determine the effects of three different doses of NGB 2904 (0.1, 1, and 5 mg/kg i.p.) or vehicle (1 ml 25% 2-hydroxypropyl-_β_-cyclodextrin solution) on PR break-point for cocaine self-administration. Since it is relatively difficult to reachieve basal break-point levels after each drug test, we chose to use a between-subjects design rather than a within-subjects design for this experiment.
NGB 2904 or saline self-administration in rats formerly self-administering cocaine
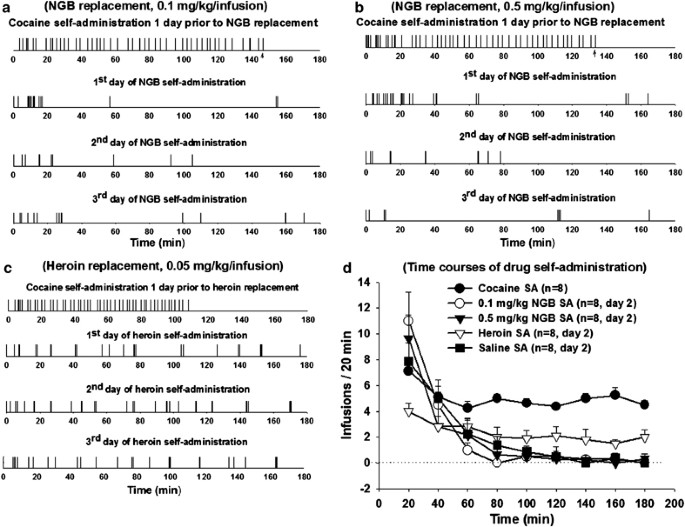
After a stable pattern of daily cocaine self-administration (0.5 mg/kg/infusion) under FR2 reinforcement was established for at least 3 consecutive days, the animals were divided into five groups (_n_=8 each): (1) cocaine (0.5 mg/kg/infusion) was available for self-administration on the following days in the usual 3-h test sessions; (2) cocaine was replaced by NGB 2904 (0.1 mg/kg/infusion); (3) cocaine was replaced by NGB 2904 (0.5 mg/kg/infusion); (4) cocaine was replaced by heroin (0.05 mg/kg/infusion); and (5) cocaine was replaced by saline (0.08 ml/infusion). Since animals might take several days to support self-administration for a novel reinforcer, each replacement test was repeated for 3–5 days. The doses of NGB 2904 were chosen on two grounds. First, NGB 2904's maximum solubility in the 5% 2-hydroxypropyl-_β_-cyclodextrin solution used as vehicle in this experiment is approximately 3 mg/ml, making 0.5 mg/kg/infusion the maximum feasible unit dose. Second, the cumulative i.v. NGB 2904 dose within the initial 30 min (5–8 infusions × 0.1 or 0.5 mg/kg/infusion, see Figure 2) was approximately 0.5–4 mg/kg, which is higher than the i.p. doses (0.1–5 mg/kg) of NGB 2904 found to be effective in the self-administration, BSR, and reinstatement of drug-seeking experiments detailed below, making failure to self-administer an a fortiori finding. Heroin and saline were chosen as positive and neutral reinforcer controls.
Figure 2
Effect of substituting NGB 2904, heroin, or saline for cocaine in animals proficient at cocaine self-administration behavior. (a–c) Representative event records of drug infusions (each vertical line represents one drug infusion), illustrating that NGB 2904 (0.1 or 0.5 mg/kg/infusion; a and b) failed to sustain, but heroin (0.05 mg/kg/infusion; c) did sustain drug self-administration behavior during three repeated days of substitution testing for cocaine. (d) Time courses of mean drug-taking behavior by 20 min intervals for 0.5 mg/kg cocaine (•—•), 0.1 mg/kg NGB 2904 (○—○), 0.5 mg/kg NGB 2904 (▾—▾), 0.05 mg/kg heroin (▿—▿), or saline (▪—▪) on the second substitution test day. Two-way ANOVA for repeated measures over time revealed statistically significant differences in drug-taking behavior between cocaine and NGB 2904 (0.1 or 0.5 mg/kg/infusion), between cocaine and heroin, between cocaine and saline, between heroin and NGB 2904, and between heroin and saline substitution groups. However, there were no significant differences in drug-taking behavior between any dose of NGB 2904 and the saline substitution group. Further, the extinction-like pattern of responding (a, b, and d) after substitution of NGB 2904 for cocaine suggests that NGB 2904 itself has no reinforcing effect. SA, self-administration; NGB, NGB 2904.
Experimental Set 2: Electrical Brain Stimulation Reward (BSR)
Surgery
Under the same anesthesia as used in Experimental Set 1, rats were placed in a stereotaxic frame, and a unilateral monopolar stainless-steel stimulating electrode (Plastics One, Roanoke, VA, USA) was placed into the lateral hypothalamus using standard aseptic surgical and stereotaxic techniques. The implant coordinates for the tips of the electrodes were AP=−2.56, ML=±1.9, and DV=−8.6, according to the rat brain stereotaxic Atlas of Paxinos and Watson (1998). The electrode was attached to the skull with jeweler's screws and dental acrylic. A wire leading from the electrode was wrapped around a skull screw to serve as a current return.
Apparatus
The experiments were conducted in standard MED Associates operant chambers (32 × 25 × 33 cm). Each operant chamber had a lever located 6.5 cm above the floor, connected to an electrical stimulator.
General procedure
The general procedures for electrical BSR were the same as we have reported previously (Vorel et al, 2002; Hayes et al, 2003). Briefly, after 7 days of recovery from surgery, rats were allowed to self-train (autoshape) to lever-press for rewarding BSR. Each press on the lever resulted in a 500-ms train of 0.1-ms rectangular cathodal pulses through the electrode in the rat's lateral hypothalamus, followed by a 500 ms ‘timeout’ in which further presses did not produce brain stimulation. The initial stimulation parameters were 72 Hz and 200 μA. If the animal did not learn to lever-press, the stimulation intensity was increased daily by 50 μA until the animal learned to press (45–60 responses/30 s) or a maximum of 800 μA was reached. Animals that did not lever-press at 800 μA or in which the stimulation produced unwanted effects (eg gross head or body movements, spinning, vocalization, or jumping) were removed from the experiment.
Rate-frequency BSR procedure
Following establishment of lever-pressing for BSR, animals were presented with a series of 16 different pulse frequencies, ranging from 141 to 25 Hz in descending order. At each pulse frequency, animals responded for two 30-s time periods (‘bins’), following which the pulse frequency was decreased by 0.05 log units. Following each 30-s bin, the lever retracted for 5 s. Throughout the experiments, animals were run for three sessions a day. Response rate for each frequency was defined as the mean number of lever responses during two 30-s bins. Since lever-pressing behavior was variable during the first session (the ‘warm-up’ session), but was stable during the second and third sessions, the data from the first session were discarded, and the data from the second and third sessions were designated as the baseline session data and test session data, respectively. The BSR threshold (_θ_0) was defined as the minimum frequency at which the animal responded for rewarding stimulation.
Testing the effects of cocaine and/or NGB 2904 on BSR
Once a baseline _θ_0 value was achieved (<15% variation in _θ_0 over 5 continuous days), the effects of cocaine and/or NGB 2904 on BSR were assessed. On test days, animals randomly received one of three different doses of NGB 2904 (0.1, 1, and 5 mg/kg i.p.) or vehicle (1 ml 25% 2-hydroxypropyl-_β_-cyclodextrin) 30 min prior to a cocaine injection (1, 2, or 10 mg/kg i.p.). After each test, animals received an additional 5–7 days of BSR restabilization until a new baseline _θ_0 was established. The order of testing for various doses of NGB 2904 was counterbalanced according to a Latin square design. The effect of NGB 2904 on cocaine-enhanced BSR was evaluated by comparing cocaine-induced alterations in _θ_0 value in the presence or absence of each dose of NGB 2904 pretreatment.
Experimental Set 3: Reinstatement of Drug-Seeking Behavior
Surgery, apparatus, and general procedure
The surgery, apparatus, and general procedure to establish stable cocaine-taking behavior were the same as in Experimental Set 1, above.
Extinction and testing for reinstatement
After stable cocaine self-administration was established, animals were exposed to extinction conditions, during which cocaine was replaced by saline, and the cocaine-associated cue-light and tone were turned off. Active lever-pressing led only to saline infusion. Daily 3 h extinction sessions for each rat continued until that rat lever-pressed <10 times per 3 h session for at least 3 consecutive days. After successful achievement of extinction, animals were divided into four groups for reinstatement testing. On the reinstatement test day, each group of rats received either the vehicle (25% 2-hydroxypropyl-_β_-cyclodextrin) or one dose of NGB 2904 (0.1, 1, and 5 mg/kg i.p.). At 30 min after vehicle or NGB 2904 administration, all rats were given a priming injection of cocaine (10 mg/kg i.p.) immediately before the reinstatement testing began. During the reinstatement test, the conditions were identical to those in extinction sessions. Cocaine-induced active lever-pressing responses (reinstatement) were recorded, although these lever-pressing responses did not lead to either cocaine infusions or presentation of the conditioned cues. Reinstatement test sessions lasted 3 h.
Sucrose-plus-cue-triggered reinstatement of sucrose-seeking behavior
The procedures for oral sucrose self-administration, extinction, and reinstatement testing were identical to the procedures used in the cocaine-triggered reinstatement test above, except for the following minor differences: (1) no surgery was carried out on the rats in this experiment; (2) active lever presses led to delivery of 0.1 ml of 5% sucrose solution into a liquid food tray on the operant chamber wall; and (3) reinstatement was triggered initially by one to two ‘free’ sucrose deliveries, and subsequent lever presses led to the presentation of the conditioned cue-light and tone. Since sucrose-triggered reinstatement is significantly weaker than cocaine-triggered reinstatement, we chose to use cues plus sucrose priming to facilitate reinstatement of sucrose-seeking behavior.
Drugs
Cocaine HCl or heroin HCl (Sigma Chemical Co., Saint Louis, MO, USA) was dissolved in physiological saline. NGB 2904 was synthesized as reported (Yuan et al, 1998) in the Medicinal Chemistry Section, Medications Discovery Research Branch, Intramural Research Program, National Institute on Drug Abuse. 2-Hydroxypropyl-_β_-cyclodextrin (25%) (Sigma/RBI, St Louis, MO) was used as vehicle for i.p. injections. To decrease injection resistance and solution osmotic pressure, 5% 2-hydroxypropyl-_β_-cyclodextrin was used as vehicle for i.v. NGB 2904 self-administration. We have previously observed no differences in behavioral effects between 5 and 25% 2-hydroxypropyl-_β_-cyclodextrin.
Data Analyses
All behavioral data are presented as means (±SEM), and were subjected to standard univariate or multivariate statistical analyses (Winer, 1962; Kirk, 1982). After subjecting data to preliminary testing to assure homogeneity of variance by Bartlett's (1937) test and to assure that other mathematical requirements for parametric analyses were met, one-way analysis of variance (ANOVA) was used to analyze the data reflecting the effects of NGB 2904 on PR cocaine self-administration, and the data on cocaine- or sucrose-triggered reinstatement of goal-seeking behavior. One-way ANOVA for repeated measures was used to analyze the effects of NGB 2904 on FR2 cocaine self-administration and on cocaine-enhanced BSR. Two-way ANOVA with repeated measures on one factor was used to analyze the data reflecting the ability of NGB 2904, heroin, or saline to sustain self-administration. Post-ANOVA individual group comparisons were carried out using the Tukey (a) statistical procedure (also known as the Tukey honestly significant difference procedure).
RESULTS
Experimental Set 1: Cocaine Self-Administration
Effects of NGB 2904 on cocaine self-administration under FR2 reinforcement
NGB 2904 (0, 0.1, 1, 5, or 10 mg/kg i.p.) administered 30 min prior to the beginning of daily cocaine self-administration sessions had no significant effect on cocaine self-administration behavior at a unit cocaine reinforcement dose of 0.5 mg/kg/infusion compared to vehicle-treated animals (data not shown). One-way ANOVA for repeated measures over the NGB 2904 dose range revealed no statistically significant effect of NGB 2904 on cocaine self-administration (ie number of cocaine infusions) under FR2 reinforcement conditions (F4,29=0.54; _p_=0.71).
Effects of NGB 2904 on cocaine self-administration under PR reinforcement
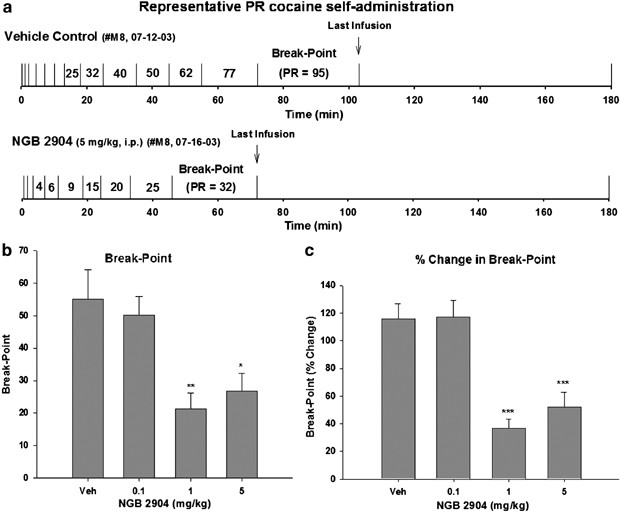
Figure 1a shows representative individual response records for cocaine self-administration under PR reinforcement conditions after vehicle or NGB 2904 administration in the same animal, illustrating a PR break-point (ie completed lever presses for the last cocaine infusion) of 95 for cocaine self-administration after vehicle administration (Figure 1a, upper trace) and a significantly lower PR break-point of 32 for cocaine self-administration after 5 mg/kg NGB 2904 administration (Figure 1a, lower trace). Figure 1b and c illustrate the group data for the observed NGB 2904-induced decrease in PR break-point itself (F3,28=6.07, _P_=0.003) and the percent change in break-point (F3,28=16.42, P<0.001). NGB 2904 significantly lowered the break-point for cocaine self-administration behavior reinforced under PR conditions. Individual group comparisons using the Tukey (a) statistical test revealed a statistically significant difference between PR break-point levels for cocaine self-administration after vehicle vs after 1 mg/kg NGB 2904 (_q_=4.96, p<0.01 for Figure 1b; _q_=7.76, p<0.001 for Figure 1c), and after vehicle vs after 5 mg/kg NGB 2904 (_q_=4.17, p<0.05 for Figure 1b; _q_=6.26, p<0.001 for Figure 1c).
Figure 1
Effect of NGB 2904 on cocaine self-administration under PR reinforcement conditions. (a) Representative records of an individual animal illustrating a reduction in the PR break-point for cocaine self-administration from 95 after vehicle (1 ml 25% 2-hydroxypropyl-_β_-cyclodextrin i.p.; upper trace) to 32 after NGB 2904 (5 mg/kg i.p., 30 min prior to test; lower trace) pretreatment. Each vertical line indicates a cocaine infusion (0.5 mg/kg/infusion). The number between the vertical lines indicates the work demand (progressively increased PR ratio, that is, number of lever presses) for a subsequent cocaine infusion. The PR break-point was defined as the completed work requirement (lever presses) to receive the last cocaine infusion. (b) Depicts the changes in break-point itself, and (c) depicts the percent changes in PR break-point for cocaine self-administration after pretreatment with NGB 2904 (0.1, 1, or 5 mg/kg) or vehicle (25% 2-hydroxypropyl-_β_-cyclodextrin) on test day. One-way ANOVA revealed that NGB 2904 produced a statistically significant reduction in PR break-point for cocaine self-administration. *p<0.05, **p<0.01, and ***p<0.001, individual group comparisons using the Tukey (a) statistic, when compared to the vehicle (Veh) pretreatment group.
Ability of NGB 2904 itself to sustain self-administration
Figure 2 shows the results of the replacement tests with NGB 2904 (0.1 or 0.5 mg/kg/infusion), heroin, cocaine, or saline in five separate groups of rats (_n_=8 each) already experienced and displaying behaviorally stable cocaine self-administration under FR2 reinforcement conditions. Each substitution drug was tested repeatedly for 3–5 days. Figure 2a–c show representative single records of NGB 2904 (0.1 and 0.5 mg/kg/infusion) or heroin (0.05 mg/kg/infusion) replacement for cocaine. NGB 2904 at either dose failed to sustain a stable pattern of self-administration. In fact, the self-administration behavior underwent gradual extinction over the 3-h test period (Figure 2d). This pattern of extinction was essentially identical to that seen when saline (0.08 ml/infusion) was substituted for cocaine (Figure 2d). In contrast, both cocaine and heroin maintained self-administration behavior (Figure 2c and d), showing the typical loading phase of increased drug self-administration during the first 20 min of self-administration opportunity, followed by stable cocaine or heroin self-administration thereafter (Figure 2d). Two-way ANOVA for repeated measures over time revealed a significant main effect of substituting NGB 2904, cocaine, heroin, or saline for cocaine (F4,27=64.86, p<0.001), a significant main effect over time (F8,32=35.31, p<0.001), and a significant time × drug interaction effect (F32,216=1.73, _p_=0.012). Individual group comparisons revealed statistically significant differences between drug-taking behavior for cocaine and drug-taking behavior for 0.1 mg/kg NGB 2904 (_q_=17.39, p<0.001), for 0.5 mg/kg NGB 2904 (_q_=17.93, p<0.001), for heroin (_q_=11.76, p<0.001), or for saline (_q_=18.68, p<0.001). Individual group comparisons revealed statistically significant differences between drug-taking behavior for heroin and drug-taking behavior for 0.1 mg/kg NGB 2904 (_q_=5.08, p<0.05), for 0.5 mg/kg NGB 2904 (_q_=4.39, p<0.05), or for saline (_q_=5.06, p<0.05). Further group comparisons revealed that there were no statistically significant differences between drug-taking behavior for NGB 2904 (0.1 or 0.5 mg/kg) and drug-taking behavior for saline at any 20-min test point over the entire 3-h test period (Figure 2d).
Experimental Set 2: Electrical BSR
Effects of NGB 2904 on cocaine-enhanced BSR
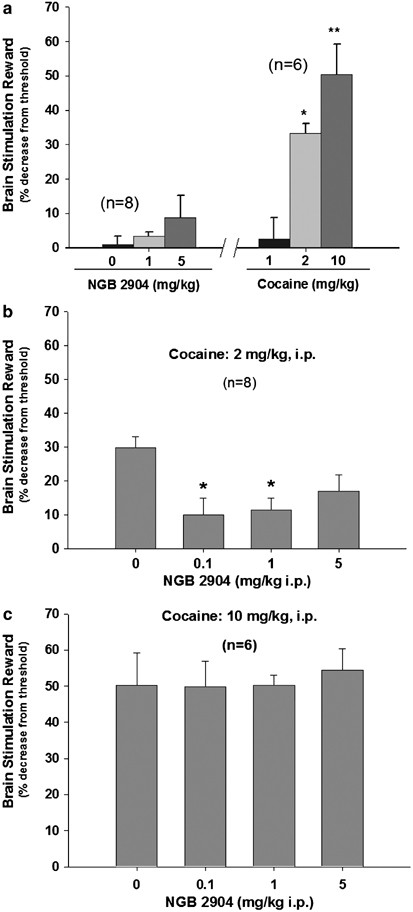
As shown in Figure 3 and as reported previously (Bauco and Wise, 1997; Gilliss et al, 2002), systemic administration of cocaine (1, 2, and 10 mg/kg i.p.) produced significant (and dose-dependent) enhancement of BSR (F2,10=11.01, _p_=0.003) (Figure 3a, right panel), manifested as a decrease in BSR threshold (_θ_0 value). NGB 2904 alone (1 and 5 mg/kg i.p.) had no effect on BSR (F2,14=1.345, _p_=0.292) (Figure 3a, left panel). Pretreatment with NGB 2904 (0.1, 1 mg/kg, but not 5 mg/kg) significantly inhibited the enhanced BSR produced by 2 mg/kg, but not by 10 mg/kg, of cocaine (Figure 3b and c). One-way ANOVA for repeated measures revealed a statistically significant main effect of NGB 2904 for the 2 mg/kg cocaine treatment group (F3,21=4.57, _p_=0.013) (Figure 3b), but not for the 10 mg/kg cocaine treatment group (F3,15=0.18, _p_=0.91) (Figure 3c). Individual group comparisons revealed statistically significant differences in the effect on cocaine-enhanced BSR between vehicle and 0.1 mg/kg NGB 2904 (_q_=4.68, _p_=0.016) and between vehicle and 1 mg/kg NGB 2904 (_q_=4.37, _p_=0.026), but not between vehicle and 5 mg/kg NGB 2904 treatment groups (_q_=3.06, _p_=0.066) (Figure 3b).
Figure 3
Effects of cocaine and NGB 2904 on electrical brain stimulation reward (BSR). (a) NGB 2904 alone (1, 5 mg/kg i.p.) had no significant effect on BSR, while cocaine (1, 2, and 10 mg/kg i.p.) produced a dose-dependent enhancement in BSR, as assessed by a statistically significant decrease in stimulation threshold for brain reward. (b) The mean percentage change in BSR threshold produced by 2 mg/kg cocaine in the absence or presence of NGB 2904 (0.1, 1, and 5 mg/kg i.p., 30 min prior to test). (c) The ineffectiveness of the same doses of NGB 2904 on 10 mg/kg cocaine-enhanced BSR. *p<0.05, **P<0.01, individual group comparisons using the Tukey (a) statistic, when compared with vehicle or baseline.
Experimental Set 3: Reinstatement of Drug-Seeking Behavior
Effects of NGB 2904 on cocaine- or sucrose-plus-sucrose-cue-triggered reinstatement of cocaine-seeking or sucrose-seeking behavior
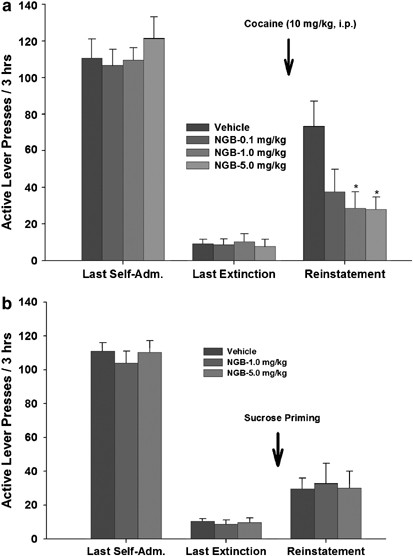
Figure 4 shows the total numbers of active lever presses observed during the last session of cocaine self-administration, during the last session of extinction, and during the reinstatement test session in the four different NGB 2904 dose groups. A single, noncontingent cocaine injection (10 mg/kg i.p.) produced robust reinstatement of extinguished operant behavior previously reinforced by i.v. cocaine infusions. Pretreatment with NGB 2904 produced a significant attenuation of this cocaine-triggered reinstatement of drug-seeking (F3,24=4.00, _p_=0.019). Individual group comparisons revealed a statistically significant difference in cocaine-induced reinstatement of cocaine-seeking behavior between vehicle and 1 mg/kg NGB 2904 (_q_=4.16, _p_=0.034) and between vehicle and 5 mg/kg NGB 2904 (_q_=4.24, _p_=0.03), but not between vehicle and 0.1 mg/kg NGB 2904 (_q_=3.34, _p_=0.113). There was no difference in inactive lever responses between vehicle and any dose of NGB 2904 (data not shown). Finally, as shown in Figure 4b, NGB 2904 (1 or 5 mg/kg i.p.) had no effect on sucrose-plus-sucrose-cue-triggered reinstatement of sucrose-seeking behavior (F2,15=0.038, _p_=0.96).
Figure 4
Effects of NGB 2904 on cocaine- or sucrose-plus-sucrose-cue-triggered reinstatement of reinforcer-seeking behavior. (a) Illustrates that pretreatment with NGB 2904 (0.1, 1, and 5 mg/kg i.p., 30 min prior to test) significantly inhibited cocaine-triggered reinstatement of drug-seeking behavior in rats extinguished from daily cocaine self-administration. *p<0.05, when compared with the vehicle pretreatment group. (b) Illustrates the ineffectiveness of NGB 2904 (1 and 5 mg/kg i.p.) on sucrose-plus-sucrose-cue-triggered reinstatement of sucrose-seeking behavior. Last Self-Adm, last session of cocaine self-administration; Last Extinction, last session of extinction 24 h before reinstatement testing.
DISCUSSION
The present study, for the first time, demonstrates that systemic administration of the highly selective DA D3 receptor antagonist NGB 2904 attenuates cocaine's rewarding effects, as assessed by both the PR self-administration and electrical BSR paradigms. In the reinstatement animal model of relapse, NGB 2904 also significantly inhibited cocaine-triggered drug-seeking behavior, but not sucrose-plus-sucrose-cue-triggered sucrose-seeking behavior. NGB 2904 itself had no reinforcing effect in rats previously experienced at cocaine self-administration, and did not by itself produce any significant effect on BSR. Together, these data support the hypothesis that DA D3 receptors play a critical role in acute cocaine-induced reinforcement and reinstatement (relapse) of drug-seeking behavior. The data also provide additional support for the potential utility of NGB 2904 or other highly selective DA D3 antagonists in the treatment of cocaine dependence.
NGB 2904's Inhibition of Cocaine Self-Administration under PR Reinforcement Conditions
Drug self-administration paradigms in animals offer the most face valid models of drug-taking behavior in humans (Wise and Gardner, 2004). There are many self-administration paradigms at the animal level, of which the FR reinforcement variant is the most widely used. In this model, the addictive drug is readily available to animals under low-effort (low work demand) and high-payoff (high unit dose of drug) conditions. In the PR paradigm, the highest completed work demand for a reward (the PR break-point) is considered a measure of rewarding efficacy (Roberts, 1989; Roberts et al, 1989; Richardson and Roberts, 1996). In the present study, we found that NGB 2904 had no significant effect on cocaine self-administration under FR2 reinforcement conditions, but significantly lowered the break-point for cocaine self-administration under PR reinforcement conditions. This is congruent with previous findings that the selective D3 receptor antagonist SB-277011A inhibits cocaine self-administration under a PR reinforcement schedule, but not under a low-cost high-payoff FR reinforcement schedule (Di Ciano et al, 2003; [Gilbert et al, 2003](/articles/1300912#ref-CR27 "Gilbert J, Xi Z-X, Campos AC, Ashby Jr CR, Heidbreder CA, Gardner EL (2003). The dopamine D3 receptor antagonist SB277011A inhibits cocaine reinforcement under fixed-ratio and progressive-ratio schedules. Abstracts of the 33rd Annual Meeting of the Society for Neuroscience, 2003 November 7–12; New Orleans, LA, 2003 Abstract Viewer/Itinerary Planner. Online at http://sfn.scholarone.com/itin2003/index.html
Washington, DC (Abstract No. 422.10)."); [Gál and Gyertyán, 2003](/articles/1300912#ref-CR23 "Gál K, Gyertyán I (2003). Targeting the dopamine D3 receptor cannot influence continuous reinforcement cocaine self-administration in rats. Brain Res Bull 61: 595–601."); [Xi et al, 2005](/articles/1300912#ref-CR96 "Xi Z-X, Gilbert JG, Pak AC, Ashby Jr CR, Heidbreder CA, Gardner EL (2005). Selective dopamine D3 receptor antagonism by SB-277011A attenuates cocaine reinforcement as assessed by progressive-ratio and variable-cost–variable-payoff fixed-ratio cocaine self-administration in rats. Eur J Neurosci 21: 3427–3438.")). There are three possible explanations for the ineffectiveness of NGB 2904 on FR cocaine self-administration. First, the FR2 reinforcement schedule demands less work and provides a much higher cumulative cocaine-dose payoff than does the PR reinforcement schedule. Thus, the stronger rewarding effects produced by the higher cumulative dose of cocaine may overcome NGB 2904's antagonism of cocaine's effect. Second, animals may compensate for NGB 2904's action by increasing their drug intake or their self-administration rate under FR2 conditions. We consider this second possibility to be unlikely, as we did not observe such a compensatory increase in cocaine intake in the present study. Although we limited the number of maximal drug infusions, which may have masked a compensatory response, we saw neither a significant difference in the total time spent to gain the maximal 50 infusions nor a change in cocaine self-administration rate in the presence or absence of NGB 2904 pretreatment (data not shown). Third, it is possible that FR cocaine self-administration is relatively insensitive to changes in reinforcement efficacy, as suggested by several authorities in the field (eg [Roberts, 1989](/articles/1300912#ref-CR64 "Roberts DCS (1989). Breaking points on a progressive ratio schedule reinforced by intravenous apomorphine increase daily following 6-hydroxydopamine lesions of the nucleus accumbens. Pharmacol Biochem Behav 32: 43–47."); [Katz, 1990](/articles/1300912#ref-CR36 "Katz JL (1990). Models of relative reinforcing efficacy of drugs and their predictive utility. Behav Pharmacol 1: 283–301."); [Arnold and Roberts, 1997](/articles/1300912#ref-CR3 "Arnold JM, Roberts DCS (1997). A critique of fixed ratio and progressive ratio schedules used to examine the neural substrates of drug reinforcement. Pharmacol Biochem Behav 57: 441–447.")), who note that FR reinforcement measures the _fact_ of drug reinforcement, but not the _degree_ of reinforcing efficacy (see also [Gardner, 2000](/articles/1300912#ref-CR24 "Gardner EL (2000). What we have learned about addiction from animal models of drug self-administration. Am J Addict 9: 285–313."); [Wise and Gardner, 2004](/articles/1300912#ref-CR91 "Wise RA, Gardner EL (2004). Animal models of addiction. In: Charney DS, Nestler EJ (eds) Neurobiology of Mental Illness, 2nd edn. Oxford Univ Press: London. pp 683–697."); [Xi et al, 2005](/articles/1300912#ref-CR96 "Xi Z-X, Gilbert JG, Pak AC, Ashby Jr CR, Heidbreder CA, Gardner EL (2005). Selective dopamine D3 receptor antagonism by SB-277011A attenuates cocaine reinforcement as assessed by progressive-ratio and variable-cost–variable-payoff fixed-ratio cocaine self-administration in rats. Eur J Neurosci 21: 3427–3438.")).In contrast to FR reinforcement, the PR break-point shift paradigm is extremely sensitive to dose–response functions that reflect a given drug's reinforcing efficacy (Roberts et al, 1989; Roberts and Bennett, 1993; French et al, 1995; Arnold and Roberts, 1997; Stafford et al, 1998). Based on the assumption that high reward stimulates high motivation and high-effort behavior, the PR self-administration paradigm is believed to also measure motivation to self-administer addictive drugs (Richardson and Roberts, 1996; Arnold and Roberts, 1997; Stafford et al, 1998; Rowlett, 2000). Thus, the present finding that blockade of D3 receptors by NGB 2904 significantly inhibits cocaine self-administration under PR reinforcement conditions suggests that NGB 2904 antagonizes cocaine's rewarding efficacy and therefore its incentive motivational properties. This finding is consistent with previous studies demonstrating that the selective D3 receptor antagonist SB-277011A also inhibits cocaine-seeking behavior under PR and second-order reinforcement conditions, but not under FR reinforcement conditions (Di Ciano et al, 2003; [Gilbert et al, 2003](/articles/1300912#ref-CR27 "Gilbert J, Xi Z-X, Campos AC, Ashby Jr CR, Heidbreder CA, Gardner EL (2003). The dopamine D3 receptor antagonist SB277011A inhibits cocaine reinforcement under fixed-ratio and progressive-ratio schedules. Abstracts of the 33rd Annual Meeting of the Society for Neuroscience, 2003 November 7–12; New Orleans, LA, 2003 Abstract Viewer/Itinerary Planner. Online at http://sfn.scholarone.com/itin2003/index.html
Washington, DC (Abstract No. 422.10)."); [Xi et al, 2005](/articles/1300912#ref-CR96 "Xi Z-X, Gilbert JG, Pak AC, Ashby Jr CR, Heidbreder CA, Gardner EL (2005). Selective dopamine D3 receptor antagonism by SB-277011A attenuates cocaine reinforcement as assessed by progressive-ratio and variable-cost–variable-payoff fixed-ratio cocaine self-administration in rats. Eur J Neurosci 21: 3427–3438.")).NGB 2904's Attenuation of Cocaine-Enhanced BSR
The electrical BSR paradigm is believed to measure a neural substrate of reward that summates with, and is highly sensitive to, drug-induced reward (Stein and Ray, 1960; Wise, 1996b; Wise and Gardner, 2004; Gardner, 2005). In the present study, cocaine dose-dependently decreased stimulation thresholds for brain reward, indicating summation or synergism between the reward provided by the electrical brain stimulation and the cocaine-induced reward (for further discussion of such summation effects, see Bauco and Wise, 1997; Gilliss et al, 2002). Pretreatment with NGB 2904 (0.1 and 1 mg/kg) significantly inhibited the enhanced BSR produced by 2 mg/kg cocaine. As shown in Figure 3, the apparent inhibitory effect produced by 5 mg/kg NGB 2904 on cocaine-enhanced BSR is not statistically significant, and the reasons for this are unclear. Since only those subjects who received all four different test doses were included in our data analysis, the relatively variable data generated from some of the animals may have contributed to this nonsignificant statistical result. However, we cannot exclude the possibility that high doses of NGB 2904 may produce other non-D3 receptor-mediated effects that, in turn, may mask D3 receptor-mediated inhibition of cocaine's enhancement of BSR. However, this seems unlikely because the same dose of NGB 2904 (5 mg/kg) inhibited cocaine self-administration under PR reinforcement conditions and also inhibited cocaine-triggered reinstatement of drug-seeking behavior.
The same dose range of NGB 2904 had no effects on the enhanced BSR produced by 10 mg/kg cocaine, suggesting that the antagonism by NGB 2904 of cocaine-enhanced brain reward is surmountable by increased cocaine dose. Two possible mechanisms may underlie such dependency on cocaine dose. Since D3 receptors have the highest binding affinity to endogenous DA (Levant, 1997), we suggest that D3 receptors may play a critical role in the regulation of reward tone under physiological or mildly elevated DA transmission conditions, such as those created by low doses of cocaine (see also Parsons et al, 1996). However, when extracellular DA levels are very high, for example, under high cocaine dose conditions, other DA receptor subtypes may be affected. Self et al (1996) have reported that activation of DA D2 receptors enhances cocaine-induced drug-seeking behavior, an effect that would be congruent with an enhancement of cocaine-induced reward, as some evidence suggests that drug-seeking behavior is correlated with increased DA function in the nucleus accumbens (Stewart and Vezina, 1988; Ranaldi et al, 1999; Weiss et al, 2000; Di Ciano et al, 2001). Therefore, it is possible that at high cocaine doses, activation of other DA receptors may diminish the attenuation of cocaine-induced reward produced by D3 receptor antagonism. Alternatively, high levels of extracellular DA produced by high doses of cocaine may bind to D3 receptors in a competitive manner, attenuating the binding of NGB 2904 to the same D3 receptors. This suggestion is consistent with our findings that NGB 2904 or SB-277011A had no effect on cocaine self-administration under continuous FR reinforcement conditions in which a high cumulative dose is easily built up, but significantly inhibited cocaine self-administration under PR reinforcement conditions in which a high cumulative dose of drug is difficult to achieve (Di Ciano et al, 2003; [Gilbert et al, 2003](/articles/1300912#ref-CR27 "Gilbert J, Xi Z-X, Campos AC, Ashby Jr CR, Heidbreder CA, Gardner EL (2003). The dopamine D3 receptor antagonist SB277011A inhibits cocaine reinforcement under fixed-ratio and progressive-ratio schedules. Abstracts of the 33rd Annual Meeting of the Society for Neuroscience, 2003 November 7–12; New Orleans, LA, 2003 Abstract Viewer/Itinerary Planner. Online at http://sfn.scholarone.com/itin2003/index.html
Washington, DC (Abstract No. 422.10).")). Similarly, BP-897 (a D3 partial agonist whose antagonist properties may predominate; see [Wood et al, 2000](/articles/1300912#ref-CR93 "Wood MD, Boyfield I, Nash DJ, Jewitt FR, Avenell KY, Riley GJ (2000). Evidence for antagonist activity of the dopamine D3 receptor partial agonist, BP 897, at human dopamine D3 receptor. Eur J Pharmacol 407: 47–51."); [Wicke and Garcia-Ladona, 2001](/articles/1300912#ref-CR84 "Wicke K, Garcia-Ladona J (2001). The dopamine D3 receptor partial agonist, BP 897, is an antagonist at human D3 receptors and at rat somatodendritic dopamine D3 receptors. Eur J Pharmacol 424: 85–90.")) also fails to inhibit cocaine self-administration under FR reinforcement conditions, but significantly inhibits cocaine-seeking behavior under second-order reinforcement conditions ([Pilla et al, 1999](/articles/1300912#ref-CR57 "Pilla M, Perachon S, Sautel F, Garrido F, Mann A, Wermuth CG et al (1999). Selective inhibition of cocaine-seeking behaviour by a partial dopamine D3 receptor agonist. Nature 400: 371–375.")), under which the brain's DA response to the cocaine-associated cues that drive second-order reinforced drug-seeking behavior is substantially lower than to cocaine itself (ie approximately a 25% increase in accumbens DA produced by cocaine-associated cues ([Gerasimov et al, 2001](/articles/1300912#ref-CR26 "Gerasimov MR, Schiffer WK, Gardner EL, Marsteller DA, Lennon IC, Taylor SJC et al (2001). GABAergic blockade of cocaine-associated cue-induced increases in nucleus accumbens dopamine. Eur J Pharmacol 414: 205–209.")) _vs_ approximately a 200–800% increase in accumbens DA produced by either exogenously or self-administered cocaine ([Wise et al, 1995](/articles/1300912#ref-CR92 "Wise RA, Newton P, Leeb K, Burnette B, Pocock D, Justice Jr JB (1995). Fluctuations in nucleus accumbens dopamine concentration during intravenous cocaine self-administration in rats. Psychopharmacology 120: 10–20."); [Dewey et al, 1997](/articles/1300912#ref-CR19 "Dewey SL, Chaurasia CS, Chen C-E, Volkow ND, Clarkson FA, Porter SP et al (1997). GABAergic attenuation of cocaine-induced dopamine release and locomotor activity. Synapse 25: 393–398."))).Noteworthily, the present data show that NGB 2904 itself produces neither a significant reward-like leftward shift nor a rightward shift in BSR rate-frequency functions. This is similar to our findings with SB-277011A ([Campos et al, 2003](/articles/1300912#ref-CR14 "Campos AC, Xi Z-X, Gilbert J, Ashby Jr CR, Heidbreder CA, Gardner EL (2003). The dopamine D3 receptor antagonist SB277011A antagonizes nicotine-enhanced brain-stimulation reward in rat. Abstracts of the 33rd Annual Meeting of the Society for Neuroscience, 2003 November 7–12; New Orleans, LA, 2003 Abstract Viewer/Itinerary Planner. Online at http://sfn.scholarone/itin2003/index.html
Washington, DC (Abstract No. 322.8)."), [2004](/articles/1300912#ref-CR15 "Campos AC, Xi Z-X, Gilbert J, Ashby Jr CR, Heidbreder CA, Newman AH et al (2004). Blockade of dopamine D3 receptors by SB277011A, NGB2904 or BP897 attenuates nicotine-enhanced brain stimulation reward in rat. Abstracts of the 34rd Annual Meeting of the Society for Neuroscience, 2004 November 23–27; San Diego, CA, 2004 Abstract Viewer/Itinerary Planner. Online at
http://sfn.scholarone.com/itin2004/index.html
Washington, DC (Abstract No. 691.6).")). This intermodel, inter-D3 antagonist consistency further supports the conclusion that highly selective DA D3 receptor antagonists diminish cocaine's reinforcing efficacy or reward value.NGB 2904's Inhibition of Cocaine-Triggered Reinstatement of Drug-Seeking Behavior
Drug reward and craving-driven relapse are believed to constitute critically important mechanistic substrates underlying addiction, with relapse believed to be well modeled at the laboratory animal level by cocaine-triggered reinstatement of cocaine-seeking behavior (Shalev et al, 2002). Using this animal relapse model, we found that pretreatment with NGB 2904 (0.1, 1, and 5 mg/kg) significantly attenuated cocaine-triggered reinstatement of cocaine-seeking behavior at doses that failed to reduce sucrose-plus-sucrose-cue-induced reinstatement of sucrose-seeking behavior, suggesting that NGB 2904-induced reduction in cocaine-seeking behavior is not caused by a generalized suppression of operant behavior. This finding is consistent with the previous finding that SB-277011A inhibits cocaine-triggered reinstatement of cocaine-seeking, but not food-triggered reinstatement of food-seeking (Vorel et al, 2002).
As noted above, NGB 2904 (1 and 5 mg/kg) significantly inhibited reinstatement (maximally by around 50%), but not the enhanced BSR produced by 10 mg/kg i.p. of cocaine. It has been reported that in rats undergoing reinstatement testing or extinction from cocaine self-administration, there is significant attenuation of cocaine-induced increases in extracellular DA in the nucleus accumbens (Neisewander et al, 1996; Mateo et al, 2005; but see Hooks et al, 1994). In contrast, electrical BSR per se appears to enhance nucleus accumbens DA (You et al, 2001). Thus, it is possible that cocaine plus electrical BSR may produce an additive or synergistic effect on DA efflux, and that, therefore, the same dose of cocaine may produce a stronger DA response in rats undergoing BSR than in rats undergoing reinstatement testing. Further, as we noted above, NGB 2904's antagonism of cocaine's actions may depend on endogenous DA levels, which in turn may explain why the same doses of NGB 2904 that produce a 50% inhibition of 10 mg/kg cocaine-triggered reinstatement of drug-seeking behavior had no effect on 10 mg/kg cocaine-enhanced BSR.
Importantly, the presently observed reduction in cocaine self-administration or cocaine-triggered drug-seeking behavior caused by NGB 2904 seems unlikely to have resulted from impaired motor function, because NGB 2904, at the dose range tested, alters neither cocaine self-administration behavior under FR reinforcement (data not shown) nor sucrose-plus-sucrose-cue-triggered sucrose-seeking behavior (see Figure 4). It also has no effect on the enhancement of BSR produced by 10 mg/kg of cocaine (see Figure 3) or responding on the inactive lever in all of these operant behavioral experiments (data not shown).
NGB 2904's Selective Blockade of DA D3 Receptors
A number of studies have shown that D1- or D2-preferring antagonists inhibit cocaine self-administration, cocaine-enhanced BSR, and cocaine-triggered reinstatement of drug-seeking behavior (Wilson and Schuster, 1972; de Wit and Wise, 1977; Koob et al, 1987; Bergman et al, 1990; Corrigall and Coen, 1991; McGregor and Roberts, 1993; Caine and Koob, 1994; Caine et al, 1995, 2002; Self et al, 1996; Awasaki et al, 1997; Ikemoto et al, 1997; Kita et al, 1999; Spealman et al, 1999; Khroyan et al, 2000, 2003; Hummel and Unterwald, 2002; Norman et al, 2002; Anderson et al, 2003; Sanchez et al, 2003; for reviews see Platt et al, 2002; Kapur and Mamo, 2003; Gorelick et al, 2004). Consequently, this raises the issue of whether the presently observed attenuating effects of NGB 2904 in these three behavioral paradigms might be attributable to D1 or D2 receptor-selective antagonism rather than to D3 receptor-selective antagonism. We believe that is unlikely, because (1) multiple lines of in vitro evidence indicate that NGB 2904 is a highly selective D3 receptor antagonist (Yuan et al, 1998; Robarge et al, 2001; Newman et al, 2003, 2005; see Introduction); (2) in the BSR paradigm, D1- and D2-preferring DA receptor antagonists inhibit brain reward functions, in a manner opposite to the brain reward enhancement produced by addictive drugs (Stein and Ray, 1960; Stein, 1962; Wise, 1982; Panagis and Spyraki, 1996; Gardner, 2005), while selective blockade of DA D3 receptors by either NGB 2904 or SB-277011A does not alter electrical brain reward thresholds (Vorel et al, 2002; [Campos et al, 2003](/articles/1300912#ref-CR14 "Campos AC, Xi Z-X, Gilbert J, Ashby Jr CR, Heidbreder CA, Gardner EL (2003). The dopamine D3 receptor antagonist SB277011A antagonizes nicotine-enhanced brain-stimulation reward in rat. Abstracts of the 33rd Annual Meeting of the Society for Neuroscience, 2003 November 7–12; New Orleans, LA, 2003 Abstract Viewer/Itinerary Planner. Online at http://sfn.scholarone/itin2003/index.html
Washington, DC (Abstract No. 322.8)."), [2004](/articles/1300912#ref-CR15 "Campos AC, Xi Z-X, Gilbert J, Ashby Jr CR, Heidbreder CA, Newman AH et al (2004). Blockade of dopamine D3 receptors by SB277011A, NGB2904 or BP897 attenuates nicotine-enhanced brain stimulation reward in rat. Abstracts of the 34rd Annual Meeting of the Society for Neuroscience, 2004 November 23–27; San Diego, CA, 2004 Abstract Viewer/Itinerary Planner. Online at
http://sfn.scholarone.com/itin2004/index.html
Washington, DC (Abstract No. 691.6).")); and finally (3) NGB 2904 does not significantly alter locomotor activity at the dose range tested in the present study ([Newman et al, 2005](/articles/1300912#ref-CR51 "Newman AH, Grundt P, Nader MA (2005). Dopamine D3 receptor partial agonists and antagonists as potential drug abuse therapeutic agents. J Med Chem 48: 3663–3679.")), again unlike D1 or D2 receptor antagonists.In conclusion, NGB 2904 attenuates cocaine's rewarding effects and selectively inhibits relapse to drug-seeking behavior, and has no apparent rewarding, aversive, or locomotor effects. Therefore, NGB 2904 or other highly selective D3 antagonists may be promising as pharmacotherapeutic agents to treat cocaine abuse and may provide in vivo tools with which to further characterize the role of DA D3 receptors in drug addiction.
References
- Anderson SM, Bari AA, Pierce RC (2003). Administration of the D1-like dopamine receptor antagonist SCH-23390 into the medial nucleus accumbens shell attenuates cocaine priming-induced reinstatement of drug-seeking behavior in rats. Psychopharmacology 168: 132–138.
CAS PubMed Google Scholar - Andreoli M, Tessari M, Pilla M, Valerio E, Hagan JJ, Heidbreder CA (2003). Selective antagonism at dopamine D3 receptors prevents nicotine-triggered relapse to nicotine-seeking behavior. Neuropsychopharmacology 28: 1272–1280.
CAS PubMed Google Scholar - Arnold JM, Roberts DCS (1997). A critique of fixed ratio and progressive ratio schedules used to examine the neural substrates of drug reinforcement. Pharmacol Biochem Behav 57: 441–447.
Article CAS PubMed Google Scholar - Ashby Jr CR, Paul M, Gardner EL, Heidbreder CA, Hagan JJ (2003). Acute administration of the selective D3 receptor antagonist SB-277011A blocks the acquisition and expression of the conditioned place preference response to heroin in male rats. Synapse 48: 154–156.
CAS PubMed Google Scholar - Austin NE, Baldwin SJ, Cutler L, Deeks N, Kelly PJ, Nash M et al (2001). Pharmacokinetics of the novel, high-affinity and selective dopamine D3 receptor antagonist SB-277011 in rat, dog and monkey: in vitro/in vivo correlation and the role of aldehyde oxidase. Xenobiotica 31: 677–686.
CAS PubMed Google Scholar - Awasaki Y, Nishida N, Sasaki S, Sato S (1997). Dopamine D1 antagonist SCH23390 attenuates self-administration of both cocaine and fentanyl in rats. Environ Toxicol Pharmacol 3: 115–122.
CAS PubMed Google Scholar - Bartlett MS (1937). Properties of sufficiency and statistical tests. Proc R Soc Lond Ser A 160: 268–282.
Google Scholar - Bauco P, Wise RA (1997). Synergistic effects of cocaine with lateral hypothalamic brain stimulation reward: lack of tolerance or sensitization. J Pharmacol Exp Ther 283: 1160–1167.
CAS PubMed Google Scholar - Bergman J, Kamien JB, Spealman RD (1990). Antagonism of cocaine self-administration by selective dopamine D1 and D2 antagonists. Behav Pharmacol 1: 355–363.
PubMed Google Scholar - Caine SB, Heinrichs SC, Coffin VL, Koob GF (1995). Effects of the dopamine D-1 antagonist SCH 23390 microinjected into the accumbens, amygdala or striatum on cocaine self-administration in the rat. Brain Res 692: 47–56.
CAS PubMed Google Scholar - Caine SB, Koob GF (1993). Modulation of cocaine self-administration in the rat through D-3 dopamine receptors. Science 260: 1814–1816.
CAS PubMed Google Scholar - Caine SB, Koob GF (1994). Effects of dopamine D-1 and D-2 antagonists on cocaine self-administration under different schedules of reinforcement in the rat. J Pharmacol Exp Ther 270: 209–218.
CAS PubMed Google Scholar - Caine SB, Negus SS, Mello NK, Patel S, Bristow L, Kulagowski J et al (2002). Role of dopamine D2-like receptors in cocaine self-administration in the rat: studies with D2 receptor mutant mice and novel D2 receptor antagonists. J Neurosci 22: 2977–2988.
CAS PubMed PubMed Central Google Scholar - Campos AC, Xi Z-X, Gilbert J, Ashby Jr CR, Heidbreder CA, Gardner EL (2003). The dopamine D3 receptor antagonist SB277011A antagonizes nicotine-enhanced brain-stimulation reward in rat. Abstracts of the 33rd Annual Meeting of the Society for Neuroscience, 2003 November 7–12; New Orleans, LA, 2003 Abstract Viewer/Itinerary Planner. Online at http://sfn.scholarone/itin2003/index.html Washington, DC (Abstract No. 322.8).
Google Scholar - Campos AC, Xi Z-X, Gilbert J, Ashby Jr CR, Heidbreder CA, Newman AH et al (2004). Blockade of dopamine D3 receptors by SB277011A, NGB2904 or BP897 attenuates nicotine-enhanced brain stimulation reward in rat. Abstracts of the 34rd Annual Meeting of the Society for Neuroscience, 2004 November 23–27; San Diego, CA, 2004 Abstract Viewer/Itinerary Planner. Online at http://sfn.scholarone.com/itin2004/index.html Washington, DC (Abstract No. 691.6).
Google Scholar - Cervo L, Cocco A, Petrella C, Heidbreder CA, Bendotti C, Mennini T (2003). SB-277011-A, a selective dopamine D3 receptor antagonist, reduces cocaine-seeking behavior in response to drug-associated stimuli in rats. Abstracts of the 33rd Annual Meeting of the Society for Neuroscience, 2003 November 7–12; New Orleans, LA, 2003 Abstract Viewer/Itinerary Planner. Online at http://sfn.scholarone.com/itin2003/index.html Washington, DC (Abstract No. 420.3).
Google Scholar - Corrigall WA, Coen KM (1991). Cocaine self-administration is increased by both D1 and D2 dopamine antagonists. Pharmacol Biochem Behav 39: 799–802.
CAS PubMed Google Scholar - de Wit H, Wise RA (1977). Blockade of cocaine reinforcement in rats with the dopamine receptor blocker pimozide, but not with the noradrenergic blockers phentolamine or phenoxybenzamine. Can J Psychol 31: 195–203.
CAS PubMed Google Scholar - Dewey SL, Chaurasia CS, Chen C-E, Volkow ND, Clarkson FA, Porter SP et al (1997). GABAergic attenuation of cocaine-induced dopamine release and locomotor activity. Synapse 25: 393–398.
CAS PubMed Google Scholar - Di Ciano P, Blaha CD, Phillips AG (2001). Changes in dopamine efflux associated with extinction, CS-induced and _d_-amphetamine-induced reinstatement of drug-seeking behavior by rats. Behav Brain Res 120: 147–158.
CAS PubMed Google Scholar - Di Ciano P, Underwood RJ, Hagan JJ, Everitt BJ (2003). Attenuation of cue-controlled cocaine-seeking by a selective D3 dopamine receptor antagonist SB-277011-A. Neuropsychopharmacology 28: 329–338.
CAS PubMed Google Scholar - French ED, Lopez M, Peper S, Kamenka J-M, Roberts DCS (1995). A comparison of the reinforcing efficacy of PCP, the PCP derivatives TCP and BTCP, and cocaine using a progressive ratio schedule in the rat. Behav Pharmacol 6: 223–228.
CAS PubMed Google Scholar - Gál K, Gyertyán I (2003). Targeting the dopamine D3 receptor cannot influence continuous reinforcement cocaine self-administration in rats. Brain Res Bull 61: 595–601.
PubMed Google Scholar - Gardner EL (2000). What we have learned about addiction from animal models of drug self-administration. Am J Addict 9: 285–313.
CAS PubMed Google Scholar - Gardner EL (2005). Brain-reward mechanisms. In: Lowinson JH, Ruiz P, Millman RB, Langrod JG (eds). Substance Abuse: A Comprehensive Textbook, 4th edn. Lippincott Williams & Wilkins: Philadelphia. pp 48–97.
Google Scholar - Gerasimov MR, Schiffer WK, Gardner EL, Marsteller DA, Lennon IC, Taylor SJC et al (2001). GABAergic blockade of cocaine-associated cue-induced increases in nucleus accumbens dopamine. Eur J Pharmacol 414: 205–209.
CAS PubMed Google Scholar - Gilbert J, Xi Z-X, Campos AC, Ashby Jr CR, Heidbreder CA, Gardner EL (2003). The dopamine D3 receptor antagonist SB277011A inhibits cocaine reinforcement under fixed-ratio and progressive-ratio schedules. Abstracts of the 33rd Annual Meeting of the Society for Neuroscience, 2003 November 7–12; New Orleans, LA, 2003 Abstract Viewer/Itinerary Planner. Online at http://sfn.scholarone.com/itin2003/index.html Washington, DC (Abstract No. 422.10).
Google Scholar - Gilliss B, Malanga CJ, Pieper JO, Carlezon Jr WA (2002). Cocaine and SKF-82958 potentiate brain stimulation reward in Swiss–Webster mice. Psychopharmacology 163: 238–248.
CAS PubMed Google Scholar - Gorelick DA, Gardner EL, Xi Z-X (2004). Agents in development for the management of cocaine abuse. Drugs 64: 1547–1573.
CAS PubMed Google Scholar - Hayes RJ, Vorel SR, Spector J, Liu X, Gardner EL (2003). Electrical and chemical stimulation of the basolateral complex of the amygdala reinstates cocaine-seeking behavior in the rat. Psychopharmacology 168: 75–83.
CAS PubMed Google Scholar - Heidbreder CA, Gardner EL, Xi Z-X, Thanos PK, Mugnaini M, Hagan JJ et al (2005). The role of central dopamine D3 receptors in drug addiction: a review of pharmacological evidence. Brain Res Rev 49: 77–105.
CAS PubMed Google Scholar - Hooks MS, Duffy P, Striplin C, Kalivas PW (1994). Behavioral and neurochemical sensitization following cocaine self-administration. Psychopharmacology 115: 265–272.
CAS PubMed Google Scholar - Hummel M, Unterwald EM (2002). D1 dopamine receptor: a putative neurochemical and behavioral link to cocaine addiction. J Cell Physiol 191: 17–27.
CAS PubMed Google Scholar - Ikemoto S, Glazier BS, Murphy JM, McBride WJ (1997). Role of dopamine D1 and D2 receptors in the nucleus accumbens in mediating reward. J Neurosci 17: 8580–8587.
CAS PubMed PubMed Central Google Scholar - Kapur S, Mamo D (2003). Half a century of antipsychotics and still a central role for dopamine D2 receptors. Prog Neuropsychopharmacol Biol Psychiatry 27: 1081–1090.
CAS PubMed Google Scholar - Katz JL (1990). Models of relative reinforcing efficacy of drugs and their predictive utility. Behav Pharmacol 1: 283–301.
PubMed Google Scholar - Khroyan TV, Barrett-Larimore RL, Rowlett JK, Spealman RD (2000). Dopamine D1- and D2-like receptor mechanisms in relapse to cocaine-seeking behavior: effects of selective antagonists and agonists. J Pharmacol Exp Ther 294: 680–687.
CAS PubMed Google Scholar - Khroyan TV, Platt DM, Rowlett JK, Spealman RD (2003). Attenuation of relapse to cocaine seeking by dopamine D1 receptor agonists and antagonists in non-human primates. Psychopharmacology 168: 124–131.
CAS PubMed Google Scholar - Kirk RE (1982). Experimental Design: Procedures for the Behavioral Sciences, 2nd edn. Brooks/Cole: Belmont, CA.
Google Scholar - Kita K, Shiratani T, Takenouchi K, Fuzazako H, Takigawa M (1999). Effects of D1 and D2 dopamine receptor antagonists on cocaine-induced self-stimulation and locomotor activity in rats. Eur Neuropsychopharmacol 9: 1–7.
CAS PubMed Google Scholar - Koob GF, Le HT, Creese I (1987). The D1 dopamine receptor antagonist SCH 23390 increases cocaine self administration in the rat. Neurosci Lett 79: 315–320.
CAS PubMed Google Scholar - Le Foll B, Schwartz J-C, Sokoloff P (2000). Dopamine D3 receptor agents as potential new medications for drug addiction. Eur Psychiatry 15: 140–146.
CAS PubMed Google Scholar - Levant B (1997). The D3 dopamine receptor: neurobiology and potential clinical relevance. Pharmacol Rev 49: 231–252.
CAS PubMed Google Scholar - Mateo Y, Lack CM, Morgan D, Roberts DCS, Jones SR (2005). Reduced dopamine terminal function and insensitivity to cocaine following cocaine binge self-administration and deprivation. Neuropsychopharmacology 30: 1455–1463.
CAS PubMed Google Scholar - McGregor A, Roberts DCS (1993). Dopaminergic antagonism within the nucleus accumbens or the amygdala produces differential effects on intravenous cocaine self-administration under fixed and progressive ratio schedules of reinforcement. Brain Res 624: 245–252.
CAS PubMed Google Scholar - Millan MJ, Dekeyne A, Rivet J-M, Dubuffet T, Lavielle G, Brocco M (2000). S33084, a novel, potent, selective, and competitive antagonist at dopamine D3-receptors: II. Functional and behavioral profile compared with GR218,231 and L741,626. J Pharmacol Exp Ther 293: 1063–1073.
CAS PubMed Google Scholar - Murray AM, Ryoo HL, Gurevich E, Joyce JN (1994). Localization of dopamine D3 receptors to mesolimbic and D2 receptors to mesostriatal regions of human forebrain. Proc Natl Acad Sci USA 91: 11271–11275.
CAS PubMed PubMed Central Google Scholar - National Academy of Sciences (National Research Council, Commission on Life Sciences, Institute of Laboratory Animal Resources) (1996). Guide for the Care and Use of Laboratory Animals. National Academy Press: Washington, DC.
- Neisewander JL, O'Dell LE, Tran-Nguyen LTL, Castañeda E, Fuchs RA (1996). Dopamine overflow in the nucleus accumbens during extinction and reinstatement of cocaine self-administration behavior. Neuropsychopharmacology 15: 506–514.
CAS PubMed Google Scholar - Newman AH, Cao J, Bennett CJ, Robarge MJ, Freeman RA, Luedtke RR (2003). _N_-{4-[4-(2,3-dichlorophenyl)piperazin-1-yl]butyl, butenyl and butynyl}arylcarboxamides as novel dopamine D3 receptor antagonists. Bioorg Med Chem Lett 13: 2179–2183.
CAS PubMed Google Scholar - Newman AH, Grundt P, Nader MA (2005). Dopamine D3 receptor partial agonists and antagonists as potential drug abuse therapeutic agents. J Med Chem 48: 3663–3679.
CAS PubMed Google Scholar - Norman AB, Welge JA, Tsibulsky VL (2002). Characterization of the distribution of the cocaine priming threshold and the effect of SCH23390. Brain Res 946: 253–261.
CAS PubMed Google Scholar - Panagis G, Spyraki C (1996). Neuropharmacological evidence for the role of dopamine in ventral pallidum self-stimulation. Psychopharmacology 123: 280–288.
CAS PubMed Google Scholar - Parsons LH, Caine SB, Sokoloff P, Schwartz J-C, Koob GF, Weiss F (1996). Neurochemical evidence that postsynaptic nucleus accumbens D3 receptor stimulation enhances cocaine reinforcement. J Neurochem 67: 1078–1089.
CAS PubMed Google Scholar - Parsons LH, Weiss F, Koob GF (1998). Serotonin1B receptor stimulation enhances cocaine reinforcement. J Neurosci 18: 10078–10089.
CAS PubMed PubMed Central Google Scholar - Paxinos G, Watson C (1998). The Rat Brain in Stereotaxic Coordinates, 4th edn. Academic Press: San Diego, CA.
Google Scholar - Pilla M, Perachon S, Sautel F, Garrido F, Mann A, Wermuth CG et al (1999). Selective inhibition of cocaine-seeking behaviour by a partial dopamine D3 receptor agonist. Nature 400: 371–375.
CAS PubMed Google Scholar - Platt DM, Rowlett JK, Spealman RD (2002). Behavioral effects of cocaine and dopaminergic strategies for preclinical medication development. Psychopharmacology 163: 265–282.
CAS PubMed Google Scholar - Ranaldi R, Pocock D, Zereik R, Wise RA (1999). Dopamine fluctuations in the nucleus accumbens during maintenance, extinction, and reinstatement of intravenous D-amphetamine self-administration. J Neurosci 19: 4102–4109.
CAS PubMed PubMed Central Google Scholar - Reavill C, Taylor SG, Wood MD, Ashmeade T, Austin NE, Avenell KY et al (2000). Pharmacological actions of a novel, high-affinity, and selective human dopamine D3 receptor antagonist, SB-277011-A. J Pharmacol Exp Ther 294: 1154–1165.
CAS PubMed Google Scholar - Remington G, Kapur S (2001). SB-277011 GlaxoSmithKline. Curr Opin Invest Drugs 2: 946–949.
CAS Google Scholar - Richardson NR, Roberts DCS (1996). Progressive ratio schedules in drug self-administration studies in rats: a method to evaluate reinforcing efficacy. J Neurosci Methods 66: 1–11.
CAS PubMed Google Scholar - Robarge MJ, Husbands SM, Kieltyka A, Brodbeck R, Thurkauf A, Newman AH (2001). Design and synthesis of [(2,3-dichlorophenyl)piperazin-1-yl]alkylfluorenylcarboxamides as novel ligands selective for the dopamine D3 receptor subtype. J Med Chem 44: 3175–3186.
CAS PubMed Google Scholar - Roberts DCS (1989). Breaking points on a progressive ratio schedule reinforced by intravenous apomorphine increase daily following 6-hydroxydopamine lesions of the nucleus accumbens. Pharmacol Biochem Behav 32: 43–47.
CAS PubMed Google Scholar - Roberts DCS, Bennett SAL (1993). Heroin self-administration in rats under a progressive ratio schedule of reinforcement. Psychopharmacology 111: 215–218.
CAS PubMed Google Scholar - Roberts DCS, Loh EA, Vickers G (1989). Self-administration of cocaine on a progressive-ratio schedule in rats: dose-response relationship and effect of haloperidol pretreatment. Psychopharmacology 97: 535–538.
CAS PubMed Google Scholar - Rowlett JK (2000). A labor-supply analysis of cocaine self-administration under progressive-ratio schedules: antecedents, methodologies, and perspectives. Psychopharmacology 153: 1–16.
CAS PubMed Google Scholar - Sanchez CJ, Bailie TM, Wu W-R, Li N, Sorg BA (2003). Manipulation of dopamine D1-like receptor activation in the rat medial prefrontal cortex alters stress- and cocaine-induced reinstatement of conditioned place preference behavior. Neuroscience 119: 497–505.
CAS PubMed Google Scholar - Schwartz J-C, Diaz J, Pilon C, Sokoloff P (2000). Possible implications of the dopamine D3 receptor in schizophrenia and in antipsychotic drug actions. Brain Res Rev 31: 277–287.
CAS PubMed Google Scholar - Self DW, Barnhart WJ, Lehman DA, Nestler EJ (1996). Opposite modulation of cocaine-seeking behavior by D1- and D2-like dopamine receptor agonists. Science 271: 1586–1589.
CAS PubMed Google Scholar - Shalev U, Grimm JW, Shaham Y (2002). Neurobiology of relapse to heroin and cocaine seeking: a review. Pharmacol Rev 54: 1–42.
CAS PubMed Google Scholar - Sokoloff P, Giros B, Martres M-P, Andrieux M, Besancon R, Pilon C et al (1992a). Localization and function of the D3 dopamine receptor. Arzneimittelforschung 42: 224–230.
CAS PubMed Google Scholar - Sokoloff P, Martres M-P, Giros B, Bouthenet ML, Schwartz J-C (1992b). The third dopamine receptor (D3) as a novel target for antipsychotics. Biochem Pharmacol 43: 659–666.
CAS PubMed Google Scholar - Spealman RD, Barrett-Larimore RL, Rowlett JK, Platt DM, Khroyan TV (1999). Pharmacological and environmental determinants of relapse to cocaine-seeking behavior. Pharmacol Biochem Behav 64: 327–336.
CAS PubMed Google Scholar - Stafford D, LeSage MG, Glowa JR (1998). Progressive-ratio schedules of drug delivery in the analysis of drug self-administration: a review. Psychopharmacology 139: 169–184.
CAS PubMed Google Scholar - Stanwood GD, Artymyshyn RP, Kung M-P, Kung HF, Lucki I, McGonigle P (2000). Quantitative autoradiographic mapping of rat brain dopamine D3 binding with [125I]7-OH-PIPAT: evidence for the presence of D3 receptors on dopaminergic and nondopaminergic cell bodies and terminals. J Pharmacol Exp Ther 295: 1223–1231.
CAS PubMed Google Scholar - Stein L (1962). Effects and interactions of imipramine, chlorpromazine, reserpine, and amphetamine on self-stimulation: possible neurophysiological basis of depression. Recent Adv Biol Psychiatry 4: 288–309.
Google Scholar - Stein L, Ray OS (1960). Brain stimulation reward ‘thresholds’ self-determined in rat. Psychopharmacologia 1: 251–256.
CAS PubMed Google Scholar - Stemp G, Ashmeade T, Branch CL, Hadley MS, Hunter AJ, Johnson CN et al (2000). Design and synthesis of _trans_-_N_-[4-[2-(6-cyano-1,2,3,4-tetrahydroisoquinolin-2-yl)ethyl]cyclohexyl]-4-quinolinecarboxamide (SB-277011): a potent and selective dopamine D3 receptor antagonist with high oral bioavailability and CNS penetration in the rat. J Med Chem 43: 1878–1885.
CAS PubMed Google Scholar - Stewart J, Vezina P (1988). A comparison of the effects of intra-accumbens injections of amphetamine and morphine on reinstatement of heroin intravenous self-administration behavior. Brain Res 457: 289–294.
Google Scholar - Vorel SR, Ashby Jr CR, Paul M, Liu X, Hayes R, Hagan JJ et al (2002). Dopamine D3 receptor antagonism inhibits cocaine-seeking and cocaine-enhanced brain reward in rats. J Neurosci 22: 9595–9603.
CAS PubMed PubMed Central Google Scholar - Weiss F, Maldonado-Vlaar CS, Parsons LH, Kerr TM, Smith DL, Ben-Shahar O (2000). Control of cocaine-seeking behavior by drug-associated stimuli in rats: effects on recovery of extinguished operant-responding and extracellular dopamine levels in amygdala and nucleus accumbens. Proc Natl Acad Sci USA 97: 4321–4326.
CAS PubMed PubMed Central Google Scholar - Weissenborn R, Whitelaw RB, Robins TW, Everitt BJ (1998). Excitotoxic lesions of the mediodorsal thalamic nucleus attenuate intravenous cocaine self-administration. Psychopharmacology 140: 225–232.
CAS PubMed Google Scholar - Wicke K, Garcia-Ladona J (2001). The dopamine D3 receptor partial agonist, BP 897, is an antagonist at human D3 receptors and at rat somatodendritic dopamine D3 receptors. Eur J Pharmacol 424: 85–90.
CAS PubMed Google Scholar - Wilson MC, Schuster CR (1972). The effects of chlorpromazine on psychomotor stimulant self-administration in the rhesus monkey. Psychopharmacology 26: 115–126.
CAS Google Scholar - Winer BJ (1962). Statistical Principles in Experimental Design. McGraw-Hill: New York.
Google Scholar - Wise RA (1982). Neuroleptics and operant behavior: the anhedonia hypothesis. Behav Brain Sci 5: 39–87.
Google Scholar - Wise RA (1996a). Neurobiology of addiction. Curr Opin Neurobiol 6: 243–251.
CAS PubMed Google Scholar - Wise RA (1996b). Addictive drugs and brain stimulation reward. Annu Rev Neurosci 19: 319–340.
CAS PubMed Google Scholar - Wise RA, Gardner EL (2002). Functional anatomy of substance-related disorders. In: D'haenen H, den Boer JA, Willner P (eds). Biological Psychiatry. Wiley: New York. pp 509–522.
Google Scholar - Wise RA, Gardner EL (2004). Animal models of addiction. In: Charney DS, Nestler EJ (eds) Neurobiology of Mental Illness, 2nd edn. Oxford Univ Press: London. pp 683–697.
Google Scholar - Wise RA, Newton P, Leeb K, Burnette B, Pocock D, Justice Jr JB (1995). Fluctuations in nucleus accumbens dopamine concentration during intravenous cocaine self-administration in rats. Psychopharmacology 120: 10–20.
CAS PubMed Google Scholar - Wood MD, Boyfield I, Nash DJ, Jewitt FR, Avenell KY, Riley GJ (2000). Evidence for antagonist activity of the dopamine D3 receptor partial agonist, BP 897, at human dopamine D3 receptor. Eur J Pharmacol 407: 47–51.
CAS PubMed Google Scholar - Xi Z-X, Gilbert J, Campos AC, Ashby Jr CR, Gardner EL, Newman AH (2003). The dopamine D3 receptor antagonist NGB 2904 inhibits cocaine reward and cocaine-triggered reinstatement of cocaine-seeking behavior. Abstracts of the 33rd Annual Meeting of the Society for Neuroscience, 2003 November 7–12; New Orleans, LA, 2003 Abstract Viewer/Itinerary Planner. Online at http://sfn.scholarone.com/itin2003/index.html Washington, DC. (Abstract No. 422.9).
Google Scholar - Xi Z-X, Gilbert J, Campos AC, Kline N, Ashby Jr CR, Hagan JJ et al (2004). Blockade of mesolimbic dopamine D3 receptors inhibits stress-induced reinstatement of cocaine-seeking in rats. Psychopharmacology 176: 57–65.
CAS PubMed PubMed Central Google Scholar - Xi Z-X, Gilbert JG, Pak AC, Ashby Jr CR, Heidbreder CA, Gardner EL (2005). Selective dopamine D3 receptor antagonism by SB-277011A attenuates cocaine reinforcement as assessed by progressive-ratio and variable-cost–variable-payoff fixed-ratio cocaine self-administration in rats. Eur J Neurosci 21: 3427–3438.
PubMed PubMed Central Google Scholar - You Z-B, Chen Y-Q, Wise RA (2001). Dopamine and glutamate release in the nucleus accumbens and ventral tegmental area of rat following lateral hypothalamic self-stimulation. Neuroscience 107: 629–639.
CAS PubMed Google Scholar - Yuan J, Chen X, Brodbeck R, Primus R, Braun J, Wasley JWF et al (1998). NGB 2904 and NGB 2849: two highly selective dopamine D3 receptor antagonists. Bioorg Med Chem Lett 8: 2715–2718.
CAS PubMed Google Scholar
Acknowledgements
This research was supported by the Intramural Research Program of the National Institute on Drug Abuse, US National Institutes of Health. Christina Bennett and Jianjing Cao are acknowledged for synthesizing multigram quantities of NGB 2904 for these studies. Preliminary reports on portions of these experiments were presented at the 2003 and 2004 Annual Meetings of the College on Problems of Drug Dependence, and at the 2003 Annual Meeting of the Society for Neuroscience ([Xi et al, 2003](/articles/1300912#ref-CR94 "Xi Z-X, Gilbert J, Campos AC, Ashby Jr CR, Gardner EL, Newman AH (2003). The dopamine D3 receptor antagonist NGB 2904 inhibits cocaine reward and cocaine-triggered reinstatement of cocaine-seeking behavior. Abstracts of the 33rd Annual Meeting of the Society for Neuroscience, 2003 November 7–12; New Orleans, LA, 2003 Abstract Viewer/Itinerary Planner. Online at http://sfn.scholarone.com/itin2003/index.html
Washington, DC. (Abstract No. 422.9).")).Author information
Authors and Affiliations
- Neuropsychopharmacology Section, Behavioral Neuroscience Research Branch, Intramural Research Program, National Institute on Drug Abuse, National Institutes of Health, DHHS, Baltimore, MD, USA
Zheng-Xiong Xi, Jeremy G Gilbert, Arlene C Pak, Xiao-Qing Peng, Leah Gitajn & Eliot L Gardner - Medicinal Chemistry Section, Medications Discovery Research Branch, Intramural Research Program, National Institute on Drug Abuse, National Institutes of Health, DHHS, Baltimore, MD, USA
Amy Hauck Newman - Department of Pharmaceutical Sciences, Saint John's University, Jamaica, NY, USA
Charles R Ashby Jr
Authors
- Zheng-Xiong Xi
You can also search for this author inPubMed Google Scholar - Amy Hauck Newman
You can also search for this author inPubMed Google Scholar - Jeremy G Gilbert
You can also search for this author inPubMed Google Scholar - Arlene C Pak
You can also search for this author inPubMed Google Scholar - Xiao-Qing Peng
You can also search for this author inPubMed Google Scholar - Charles R Ashby Jr
You can also search for this author inPubMed Google Scholar - Leah Gitajn
You can also search for this author inPubMed Google Scholar - Eliot L Gardner
You can also search for this author inPubMed Google Scholar
Corresponding author
Correspondence toZheng-Xiong Xi.
Rights and permissions
About this article
Cite this article
Xi, ZX., Newman, A., Gilbert, J. et al. The Novel Dopamine D3 Receptor Antagonist NGB 2904 Inhibits Cocaine's Rewarding Effects and Cocaine-Induced Reinstatement of Drug-Seeking Behavior in Rats.Neuropsychopharmacol 31, 1393–1405 (2006). https://doi.org/10.1038/sj.npp.1300912
- Received: 01 April 2005
- Revised: 15 August 2005
- Accepted: 25 August 2005
- Published: 05 October 2005
- Issue Date: 01 July 2006
- DOI: https://doi.org/10.1038/sj.npp.1300912