ADMET—turning chemicals into drugs (original) (raw)
- Feature
- Published: 01 August 2001
Nature Biotechnology volume 19, pages 722–726 (2001)Cite this article
- 2432 Accesses
- 311 Citations
- 6 Altmetric
- Metrics details
Rapidly resolving the pharmacokinetic and toxicological properties of drug candidates remains a key challenge for drug developers.
This is a preview of subscription content, access via your institution
Relevant articles
Open Access articles citing this article.
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Additional access options:
Figure 1
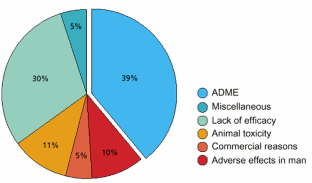
© Amy Center
References
- Lazarou, J., Pomeranz, B.H., & Corey, P.N. Incidence of adverse drug reactions in hospitalised patients. J. Am. Med. Assoc. 279, 1200–1205 (1998).
Article CAS Google Scholar - Lehman Brothers & McKinsey & Co. The Fruits of Genomics. (Lehman Brothers, New York, 2001).
- Bowen, W.P. et al. Measurement of cytochrome P450 gene induction in human hepatocytes using quantitative real-time reverse transcriptase-polymerase chain reaction. Drug Met. Dis. 28, 781–788 (2000).
CAS Google Scholar
Authors
- John Hodgson
You can also search for this author inPubMed Google Scholar
Rights and permissions
About this article
Cite this article
Hodgson, J. ADMET—turning chemicals into drugs.Nat Biotechnol 19, 722–726 (2001). https://doi.org/10.1038/90761
- Published: 01 August 2001
- Issue Date: 01 August 2001
- DOI: https://doi.org/10.1038/90761