Voudourisite, Cd(SO4)·H2O, and lazaridisite, Cd3(SO4)3·8H2O, two new minerals from the Lavrion Mining District, Greece | Mineralogical Magazine | Cambridge Core (original) (raw)
Abstract
Voudourisite, Cd(SO4)·H2O, and lazaridisite, Cd3(SO4)3·8H2O, are two new minerals from the Esperanza Mine, Kaminiza Area, Lavrion Mining District, Greece. This small ancient mine is also the type locality of niedermayrite and katerinopoulosite. Further associated minerals are sphalerite, galena, edwardsite, chalcanthite, gypsum and greenockite. Both secondary minerals form tiny clusters or crusts and are among the latest to form in this paragenetic sequence. They are colourless or white, transparent to translucent, with a white streak and vitreous lustre. No fluorescence is observed. The minerals exhibit conchoidal fracture, no cleavage or preferred parting. The Mohs hardness is ~3 and both have brittle tenacity. Voudourisite is biaxial (–) with refractive indices at 589(1) nm of α = 1.580(2), β = 1.624(2), γ = 1.640(2), 2Vmeas. = 70(5)°, 2Vcalc. = 61° and β ~ || [010]. Lazaridisite is biaxial neutral with refractive indices at 589(1) nm of α = 1.552(2), β = 1.561(2), γ = 1.570(2), 2Vmeas. = 90(5)° and 2Vcalc. = 90°. The chemical compositions of voudourisite and lazaridisite are close to ideal with only minor amounts of copper detectable. The atomic arrangement in voudourisite (space group P21/c with a = 7.633(2), b = 7.458(2), c = 8.151(2) Å, β = 122.35(1)° and V = 392.0(2) Å3) is related to that of kieserite while lazaridisite (space group C2/c with a = 14.813(3), b = 11.902(2), c = 9.466(2) Å, β = 97.38(1)° and V = 1655.2(6) Å3) is a distinct structure-type. Calculated densities are 3.838 and 3.088 g/cm3, respectively.
The strongest lines in the powder X-ray pattern [d in Å (I) (hkl)] are: 4.890 (66) (110); 3.741 (25) (020); 3.578 (100) (11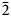 $\bar 2$); 3.230 (43) (200); 2.525 (33) (022); 2.395 (29) (112) for voudourisite and 6.860 (100) (200); 6.317 (72) (111); 5.965 (84) (020); 4.512 (58) (310); 3.727 (78) (202); 3.608(82)(13
$\bar 2$); 3.230 (43) (200); 2.525 (33) (022); 2.395 (29) (112) for voudourisite and 6.860 (100) (200); 6.317 (72) (111); 5.965 (84) (020); 4.512 (58) (310); 3.727 (78) (202); 3.608(82)(13 $\bar 1$); 3.109 (83) (40
$\bar 1$); 3.109 (83) (40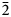 $\bar 2$); 3.020 (50) (33
$\bar 2$); 3.020 (50) (33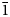 $\bar 1$) for lazaridisite, respectively.
$\bar 1$) for lazaridisite, respectively.
References
Bechtold, A. and Wildner, M. (2016) Crystal chemistry of the kieserite–cobaltkieserite solid solution, Mg1–xCox(SO4)·H2O: well-behaved oddities. European Journal of Mineralogy, 28, 43–52.Google Scholar
Brégeault, J.M. and Herpin, P. (1970) Etude structurale du sulfate de cadmium monohydrate CdSO4 (H2O). Bulletin de la Societe Francaise de Mineralogie et de Cristallographie, 93, 37–42.Google Scholar
Caminiti, R. and Johansson, G. (1981) A refinement of the crystal structure of the cadmium sulfate (CdSO4)3(H2O)8. Acta Chemica Scandinavica, 35A, 451–455.Google Scholar
Conophagos, C.E. (1980) Le Laurium Antique et la Technique Grecque de la Production de l'Argent. Ekdotike Hellados S.A., Athens, Greece, 458 pp.Google Scholar
Dowty, E. (2016) Atoms V6.5.0 for Atomic Structure Display. Shape Software, Kingsport, Tennessee, USA.Google Scholar
Giester, G. (1988) The crystal structures of CuSO4·H2O and CuSeO4·H2O, and their relationship to kieserite. Mineralogy and Petrology, 38, 277–284.Google Scholar
Giester, G. and Rieck, B. (1995) Mereiterite, K2Fe[SO4]2·4H2O, a new leonite-type mineral from the Lavrion Mining District, Greece. European Journal of Mineralogy, 7, 559–566.Google Scholar
Giester, G. and Rieck, B. (2010) Drobecite, a novel mineral from the Lavrion, Greece, deposit. Acta Mineralogica-Petrographica, IMA 2010, Abstract Series 6, 20th General Meeting of the International Mineralogical Association, 21–27 August, Budapest, Hungary, 496.Google Scholar
Giester, G. and Rieck, B. (2014) Crystal structure refinement of aurichalcite, (Cu,Zn)5(CO3)2(OH)6, from the Lavrion Mining District, Greece. Neues Jahrbuch für Mineralogie – Abhandlungen: Journal of Mineralogy and Geochemistry, 191, 225–232.Google Scholar
Giester, G and Wildner, M. (1992) The crystal structures of kieserite-type compounds. II. Crystal structures of Me(II)SeO4·H2O [Me = Mg, Mn, Co, Ni, Zn]. Neues Jahrbuch für Mineralogie Monatshefte, 1992, 135–144.Google Scholar
Giester, G., Lengauer, C.L. and Redhammer, G. (1994) Characterization of the FeSO4·H2O – CuSO4·H2O solid-solution series, and the nature of poitevinite, (Cu,Fe)SO4·H2O. The Canadian Mineralogist, 32, 873–884.Google Scholar
Giester, G., Rieck, B. and Brandstätter, F. (1998) Niedermayrite, Cu4Cd(SO4)2(OH)6·4H2O, a new mineral from the Lavrion Mining District, Greece. Mineralogy and Petrology, 63, 19–34.Google Scholar
Giester, G., Lengauer, C.L. and Rieck, B. (2000) The crystal structure of nesquehonite, MgCO3·3H2O, from Lavrion, Greece. Mineralogy and Petrology, 70, 153–163.Google Scholar
Giester, G., Kolitsch, U., Leverett, P., Turner, P. and Williams, P.A. (2007) The crystal structures of lavendulan, sampleite, and a new polymorph of sampleite. European Journal of Mineralogy, 19, 75–93.Google Scholar
Gladstone, J.H. and Dale, T.P. (1863) Researches on the refraction, dispersion, and sensitiveness of liquids. Philosophical Transactions of the Royal Society of London, 153, 317–343.Google Scholar
Gunter, M.E., Bandli, B.R., Bloss, F.D., Evans, S.H., Su, S.-C. and Weaver, R. (2004) Results from a McCrone spindle stage short course, a new version of EXCALIBR, and how to build a spindle stage. The Microscope, 52, 23–39.Google Scholar
Hawthorne, F.C., Groat, L.A., Raudsepp, M. and Ercit, T.S. (1987) Kieserite, Mg(SO4)·(H2O), a titanite-group mineral. Neues Jahrbuch für Mineralogie Abhandlungen, 157, 121–132.Google Scholar
Holland, T.J.B. and Redfern, S.A.T. (1997) Unit cell refinement from powder diffraction data: the use of regression diagnostics. Mineralogical Magazine, 61, 65–77.Google Scholar
Holtstam, D. (2002) Cobaltkieserite, CoSO4·H2O, a new mineral from Bastnäs, Skinnskatteberg, Sweden. Geologiska Förenigens I Stockholm Förhandlinger, 124, 117–119.Google Scholar
Kolitsch, U. and Giester, G. (2013) The crystal structure of a new secondary zinc mineral from Lavrion, Greece: Zn9(SO4)2(OH)12Cl2·6H2O. Mitteilungen Österreichischen Mineralogischen Gesellschaft, 159, 74.Google Scholar
Kolitsch, U., Rieck, B., Brandstätter, F., Schreiber, F., Fabritz, K.H., Blaß, G. and Gröbner, J. (2014 a) Neufunde aus dem altem Bergbau und den Schlacken von Lavrion (I). Mineralien-Welt, 25, 60–75.Google Scholar
Kolitsch, U., Rieck, B., Brandstätter, F., Schreiber, F., Fabritz, K.H., Blaß, G. and Gröbner, J. (2014 b): Neufunde aus dem altem Bergbau und den Schlacken von Lavrion (II). Mineralien-Welt, 25, 82–95.Google Scholar
Kolitsch, U., Rieck, B. and Voudouris, P. (2015) Mineralogy and genesis of the Lavrion ore deposit: new insights from the study of ore and accessory minerals. Mitteilungen Österreichischen Mineralogischen Gesellschaft, 161, 66.Google Scholar
Kolitsch, U., Sejkora, J., Topa, D., Kampf, A.R., Plášil, J., Rieck, B. and Fabritz, K.H. (2018) Prachařite, IMA 2018-081. CNMNC Newsletter No. 46, December 2018, page 1371; Mineralogical Magazine, 82, 1369–1379.Google Scholar
Krause, W., Bernhardt, H.-J., Braithwaite, R.S.W., Kolitsch, U. and Pritchard, R. (2006) Kapellasite, Cu3Zn(OH)6Cl2, a new mineral from Lavrion, Greece. Mineralogical Magazine, 70, 329–340.Google Scholar
Lengauer, C.L. and Giester, G. (1995) Rietveld refinement of the solid-solution series: (Cu,Mg)SO4·H2O. Powder Diffraction, 10, 189–194.Google Scholar
Le Fur, Y., Coing-Boyat, J. and Bassi, G. (1966) Structure des sulfates monohydratés, monocliniques, des métaux de transition, MSO4, H2O (M = Mn, Fe, Co, Ni et Zn). Comptes Rendus Academie Sciences Paris, 262C, 632–635.Google Scholar
Leonhardt, J. and Weiss, R. (1957) Das Kristallgitter des Kieserits MgSO4·H2O. Naturwissenschaften, 44, 338–339.Google Scholar
Lipson, H. (1936) The crystal structure of (CdSO4)3(H2O)8. Proceedings of the Royal Society of London, Series A: Mathematical and Physical Sciences, 156, 462–470.Google Scholar
Mandarino, J.A. (1976) The Gladstone-Dale Relationship – Part I: Derivation of new constants. The Canadian Mineralogist, 14, 498–502.Google Scholar
Mandarino, J.A. (1981) The Gladstone-Dale Relationship: Part IV. The compatibility concept and its application. The Canadian Mineralogist, 19, 441–450.Google Scholar
Momma, K. and Izumi, F. (2011) VESTA 3 for three-dimensional visualization of crystal, volumetric and morphology data. Journal of Applied Crystallography, 44, 1272–1276.Google Scholar
Neubauer, F. (2005) Structural control of mineralization in metamorphic core complexes. Pp. 561–564 in: Mineral Deposit Research: Meeting the Global Challenge (Mao, J. and Bierlein, F.P., editors). Springer, Berlin.Google Scholar
Rieck, B. (1999) Seltene Arsenate aus der Kamariza und weitere Neufunde. LAPIS, 24, 68–76.Google Scholar
Rieck, B. (2012) Neue Minerale aus dem Lagerstätten-Bezirk Lavrion/Griechenland und den Kalahari Mangan Feldern/Republik Südafrika. PhD dissertation, University of Vienna, Austria.Google Scholar
Rieck, B. and Giester, G. (2012 a) Voudourisite, IMA 2012-042. CNMNC Newsletter No. 14, October 2012, page 1287; Mineralogical Magazine, 76, 1281–1288.Google Scholar
Rieck, B. and Giester, G. (2012 b) Lazaridisite, IMA 2012-043. CNMNC Newsletter No. 14, October 2012, page 1287; Mineralogical Magazine, 76, 1281–1288.Google Scholar
Rieck, B. and Rieck, P. (1999) Silber, Arsen und Antimon: Vererzungen im Revier Plaka (Teil II). LAPIS, 24, 59–63.Google Scholar
Rieck, B. and Wendel, W. (1999) Sounion – Hilarion Top aktuell: Funde vom Frühjahr ’99. LAPIS, 24, 77–78.Google Scholar
Rieck, B., Kolitsch, U., Voudouris, P., Giester, G. and Tzeferis, P. (2018) Weitere Neufunde aus Lavrion. Mineralien-Welt, 29, 32–77 [German].Google Scholar
Rieck, B., Giester, G., Lengauer, C. and Topa, D. (2019) IMA 2018-051a. CNMNC Newsletter No. 47, February 2019, page 146; Mineralogical Magazine, 83, 143–147.Google Scholar
Sejkora, J. and Kotrlý, M. (1998) A new cadmium sulfate natural phase from Radvanice near Trutnov (Czech Republic). Bulletin of the Czech Geological Survey, 73, 321–326.Google Scholar
Sheldrick, G.M. (2008) A short history of SHELX. Acta Crystallographica, 64A, 112–122.Google Scholar
Skarpelis, N. (2002) Geodynamics and evolution of the Miocene mineralization in the Cycladic–Pelagonian belt, Hellenides. Bulletin of the Geological Society of Greece, 34, 2191–2209.Google Scholar
Ståhlhandske, C. (1978) A neutron diffraction study of mercury(II) selenate monohydrate. Acta Crystallographica, 34B, 1408–1411.Google Scholar
Ståhlhandske, C. (1981) Structure of cadmium selenate monohydrate. Acta Crystallographica, 37B, 2055–2057.Google Scholar
Strunz, H. and Nickel, E.H. (2001) Strunz Mineralogical Tables. Chemical-Structural Mineral Classification System. 9th Edition, Schweitzerbart, Stuttgart, GermanyGoogle Scholar
Talla, D. and Wildner, M. (2017) Crystal chemical and IR-spectroscopic investigation of the kieserite –szomolnokite solid solution series with relevance to Mars. Mitteilungen Österreichischen Mineralogischen Gesellschaft, 163, 86.Google Scholar
Theppitak, C. and Chainok, K. (2015) Crystal structure of CdSO4(H2O): a redetermination. Acta Crystallographica, 71E, i8–i9.Google Scholar
Tombros, S.F., Seymour, K. and Spry, P. (2004) Description and conditions of formation of new unnamed Ag-Cu and Ag-Cu-Au sulfotellurides in epithermal polymetallic Ag-Au-Te mineralization, Tinos Island, Hellas. Neues Jahrbuch für Mineralogie Abhandlungen, 179, 295–310.Google Scholar
Tombros, S.F., Seymour, K., Williams-Jones, A. and Spry, P. (2007) The genesis of epithermal Au-Ag-Te mineralization, Panormos Bay, Tinos Island, Cyclades, Greece. Economic Geology, 102, 1269–1294.Google Scholar
Vavelidis, M. (1997) Au-bearing quartz veins and placer gold on Sifnos Island, Aegean Sea, Greece. Pp 335–338 in: Mineral Deposits: Research and Exploration—Where do They Meet? (Papunen, H., editor). Balkema, The Netherlands.Google Scholar
Voudouris, P. (2005) Gold and silver mineralogy of the Lavrion deposit Attika/Greece. Pp 1089–1092 in: Mineral Deposit Research: Meeting the Global Challenge (Mao, J. and Bierlein, F.P., editors) Springer, Berlin.Google Scholar
Wendel, W. and Rieck, B. (1999 a) Die wichtigsten Mineralfundstellen Lavrions. LAPIS, 24, 25–33.Google Scholar
Wendel, W. and Rieck, B. (1999 b) Silber, Arsen und Antimon: Vererzungen im Revier Plaka (Teil I). LAPIS, 24, 53–58.Google Scholar
Wildner, M. and Giester, G. (1991) The crystal structures of kieserite-type compounds. I. Crystal structures of Me(II)SO4·H2O [Me = Mn, Fe, Co, Ni, Zn]. Neues Jahrbuch für Mineralogie Monatshefte, 1991, 296–306.Google Scholar
Witzke, T., Wegner, R., Doering, T., Pöllmann, H. and Schuckmann, W. (2000) Serrabrancaite, MnPO4·H2O, a new mineral from the Alto Serra Branca pegmatite, Pedra Lavrada, Paraiba, Brazil. American Mineralogist, 85, 847–849.Google Scholar