Versatile and open software for comparing large genomes (original) (raw)
Since the development of MUMmer 1.0 in 1999, several other programs for large-scale genome comparison have been developed, for example, SSAHA [16], AVID [17], MGA [18], BLASTZ [19], and LAGAN [20] (see also [21] for a review). Most of these programs follow an anchor-based approach, which can be divided into three phases: computation of potential anchors; computation of a colinear sequence of non-overlapping potential anchors - these anchors form the basis of the alignment; and alignment of the gaps in between the anchors. The traditional methods to compute potential anchors, that is, maximal matches of some length l or longer, use a generate-and-test approach. In a first step, all matches of some fixed length k < l, called _k_-mers, are generated using a method based on hashing (adopted from [22]). Each such _k_-mer is checked to see whether it can be extended to a maximal exact match of length at least l. The extension is done by pairwise character comparisons, and thus the run time of this approach depends not only on the number of potential anchors, but also on their lengths. This can be illustrated by an example where all maximal matches of length 20 or larger between two different strains of Escherichia coli (strain K12, 4,639,221 base-pairs (bp) and strain O157:H7, 5,528,445 bp) are computed. With k = 10, a typical choice for k, the hashing approach first generates 4.99 × 107 _k_-mers and then performs 1.66 × 107 character comparisons to determine all 46,629 maximal matches of length 20 or larger. Thus, less than 0.1% of the generated _k_-mers are extended to maximal matches of the specified length. For this reason, the generate-and-test approach leads to long running times, if the sequences under consideration share long substrings.
Recognizing this disadvantage of the hashing approach, MUMmer 1.0 was the first software system to use suffix trees to find potential anchors for an alignment. Suffix trees have been studied for almost three decades in computer science (see [23] for an overview). A suffix tree is a data structure for representing all the substrings of a string, whether that string is a DNA sequence, a protein sequence, or plain text. Suffix trees have the following nice features which make them an important data structure for large-scale genome analysis: a suffix tree for a string S of length n can be represented in space proportional to n; fast algorithms have been designed that can construct a suffix tree in time proportional to n [24, 25]; given the suffix tree of S and a query string Q of length m, there are algorithms to compute all unique maximal matches between S and Q of any specified minimum length (the potential anchors) in time proportional to m. All maximal matches, unique or not, can be found in near-optimal time. Note especially that, unlike the hashing approaches, the run-time of the suffix-tree algorithms does not depend on the length of the maximal matches.
Details of the suffix-tree algorithms incorporated in earlier versions of MUMmer have been described in [5, 7]. Here we will focus on novel developments. MUMmer is among the fastest programs for large-scale alignment; one recent test reported times for MUMmer that ranged from 4 to 110 times faster than AVID, BLASTZ, and LAGAN [20]. At its default settings, MUMmer is less sensitive at detecting matches than these programs; however, we have added several command-line options to MUMmer 3.0 that permit the detection of much weaker matches than the system would find otherwise. Note that the modularity of MUMmer, and its availability as open-source code, means that others can now build a hybrid system using, for example, the suffix-tree matching algorithm in MUMmer and the match extension program code from LAGAN or AVID.
Additional features added to MUMmer 3.0 are a new Java viewer, DisplayMUMs; a new graphical output program to generate images in fig-format or PDF, showing the alignment of a set of contigs to a reference chromosome; and new options to find non-unique matches. These will be described below.
Optimized suffix-tree data structure and suffix-tree library
The most significant technical improvement in MUMmer 3.0 is a complete rewrite of the suffix-tree code, based on the compact suffix-tree representation of [26]. This representation was also used in the repeat analysis tool REPuter [27]. However, REPuter could only accommodate sequences up to 134 million bp (Mbp). For MUMmer 3.0 the implementation was improved so that it allows sequences up to 250 Mbp on a PC with 4 gigabytes (GB) of real memory, at the cost of a slightly larger space usage per base pair. For example, one can construct the suffix tree for human chromosome 2 (237.6 Mbp, the largest human chromosome) using 15.4 bytes per base-pair. For processing DNA sequences less than 134 Mbp in length, MUMmer can be compiled so that it uses only about 12.5 bytes per bp [26]. Since suffix trees for DNA sequences are typically larger than for protein sequences, the bytes per base-pair ratio is even better for the latter.
MUMmer now requires approximately 25% less memory than release 2.1 and it runs slightly faster. Compared to the initial release in 1999, the system is more than twice as fast and uses less than half the memory. As in MUMmer 2.1, release 3.0 streams the query sequence against the suffix tree of the reference sequence. Thus the total space requirement of MUMmer is the size of the suffix tree plus the size of the reference and the query sequences. Table 1 shows run times and memory requirements for MUMmer release 2.1 and 3.0, when computing maximal matches for different pairs of genomes or chromosomes.
Table 1 Time and space requirements of MUMmer 2.1 and 3.0 when computing all exact matches of length 20 or longer for different pairs of sequences
While the previous versions of MUMmer implemented the main suffix tree construction and traversal algorithms in one monolithic program of 1,700 lines of code, the current version is based on a well-structured and well-documented software library. This provides data types for handling multiple DNA or protein sequences and their suffix trees. The library contains functions to construct the suffix tree and traverse it. In this way, a programmer who intends to modify or extend the code base can use the well-documented interfaces provided by the library, without the need to learn all of the low-level implementation details of the suffix tree.
With release 3.0, MUMmer now has the ability to run a multi-contig query against a multi-contig reference. Previously this was available by using the Nucmer package, but not directly within the core mummer program. In Table 1, for example, the genome sequence of Aspergillus fumigatus consisted (at the time of this study) of 19 scaffolds that were aligned to 248 contigs from A. nidulans. This comparison was handled by a simple call to the mummer program in release 3.0, but in release 2.1 the reference sequence needs to be first collapsed into a single contig and, after matching, the coordinates have to be re-mapped (by Nucmer) to the correct contig locations. Both releases handle multi-contig query files. Table 1 also shows the times for aligning the 22.2 Mbp chromosome 2L of the fruit fly Drosophila melanogaster to an interim assembly (before the genome project was complete) of D. pseudoobscura. In this case the query sequence, consisting of 4,653 scaffolds containing approximately 150 Mbp of sequence, was much longer than the reference. The program required 485 Mb of total memory, approximately 310 Mb for the suffix tree and the rest to hold the input sequences.
Non-unique maximal matches
Previous versions of MUMmer emphasized maximal unique matches (MUMs) as prospective anchors for an alignment. MUMs are unique in that they occur exactly once in each of the genomes. In some cases, the uniqueness constraint will prevent MUMmer from finding all matches for a repetitive substring. For example, if the reference genome has two exact copies of a particular string and the query has just one copy, then earlier versions of MUMmer would generally miss one of the matching copies, depending on the surrounding sequence. To overcome this problem, the new MUMmer system can find all maximal matches - including non-unique ones - between two input sequences simply by providing a command-line option to the program mummer. Other command-line options allow the user to produce MUMs that are unique in both the query and the reference sequence or MUMs that are unique only in the reference sequence.
Although the algorithm to produce all maximal matches is more complicated than the algorithm to produce unique maximal matches, it still runs in nearly optimal time, where optimal time would be proportional to the sum of the sizes of the input strings and the number of matches found. The run times to produce any of the three types of maximal matches are generally similar. Note, however, that when the program is directed to find all non-unique matches, including short ones, the size of the output can be extremely large, and the time to create the output file will be the dominant part of the computation.
Distant matches
One of the criticisms that has been leveled at MUMmer 1.0 was that it only finds exact matches, whereas in practice we often want to find approximate matches, that is, matches between sequences that are less than 100% identical. We addressed this concern in release 2.1, with the introduction of the Nucmer and Promer packages built on top of MUMmer. These have been substantially improved in the 3.0 release, and now exhibit performance only marginally slower than the basic search itself. The speed-up of Nucmer and Promer compared to release 2.1 is approximately 10-fold.
Both Nucmer and Promer produce a collection of local alignments using the algorithm described below. The difference between the two programs is that Nucmer constructs nucleotide alignments between two sets of DNA sequences, while Promer constructs amino acid alignments. Each set of sequences is a collection of one or more sequences from the same genome, for example, a collection of contigs produced by a genome assembler. Promer first translates both reference and query in all six frames, finds all matches in the amino acid sequences, and then maps the matches back to the original DNA coordinate system. For the extension step below, Promer uses a standard amino acid substitution matrix (BLOSUM62 is the default) to score mismatches.
The Nucmer/Promer alignment algorithm is as follows. First, both programs run MUMmer to find all exact matches longer than a specified length l, measured in nucleotides for Nucmer and amino acids for Promer. Second, the matches are clustered in preparation for extending them. Two matches are joined into the same cluster if they are separated by no more than g nucleotides (Nucmer) or amino acids (Promer). Then from each cluster, the maximum-length colinear chain of matches is extracted and processed further if the combined length of its matches is at least c nucleotides/amino acids. (Note that a chain can consist of a single matching region if l >c.) The parameters l, g, and c can all be set on the command line. The chain matches are then extended using an implementation of the Smith-Waterman dynamic programming algorithm [28], which is applied to the regions between the exact matches and also to the boundaries of the chains, which may be extended outward. This 'match and extend' step in the algorithm is essentially the same as that used by FASTA [29], BLAST [30], and many other sequence-alignment programs.
When two species are very similar, such as the two isolates of the Bacillus anthracis Ames strain sequenced at TIGR [31–33], then MUMmer is ideally suited for aligning the genomes. In that comparison of anthrax isolates, only four single-nucleotide differences separated the two 5.3 Mbp main chromosomes from one another. Similarly, in our comparison of a clinical isolate of Mycobacterium tuberculosis to a laboratory strain [31], MUMmer quickly found the approximately 1,100 SNPs and a handful of IS elements that distinguished the strains. However, when the species being compared are more distant, Nucmer and Promer provide much more detailed and more useful alignments than MUMmer alone. In the examples described below, we show how each of the programs described here may be run for genomes at different evolutionary distances
Fly versus fly
The 130 Mbp genome of D. melanogaster is largely complete, with the six main chromosome arms containing only a few gaps. Recently, the Human Genome Sequencing Center at Baylor College of Medicine completed the shotgun sequencing of D. pseudoobscura, a closely related species with a genome of approximately the same size. These two species are close enough that almost all genes are shared, and exons show a high level of sequence identity. However, they are sufficiently distant that intergenic regions and introns do not align well, and there have been hundreds of large-scale chromosomal rearrangements since the species diverged. Thus, one cannot simply align each chromosome arm to its counterpart. Complicating matters further, the D. pseudoobscura shotgun assembly consists of thousands of scaffolds and contigs. To facilitate comparison, the first computational task is to align all the scaffolds to each of the D. melanogaster arms. (The comprehensive analysis of D. pseudoobscura, organized by the sequencing center scientists and their collaborators, will appear in a future paper. The description here is primarily intended to illustrate the use and capabilities of Nucmer.)
We ran the Nucmer program with a minimum match length of 25, which was adequate to capture virtually all matching exons. Because matching genes are much longer, we required cluster chains to contain at least 100 matching nucleotides. To account for long introns and to allow the program to cluster together multiple genes, we allowed the gap between exact matches to be as long as 3,000 bp. At the time of our analysis (before completion of the sequencing project), the D. pseudoobscura assembly contained 4,653 scaffolds spanning 150 Mbp. We ran Nucmer separately to align the full set of scaffolds to each D. melanogaster chromosome arm. Using these settings, the program takes about 6 minutes per arm and uses approximately 490 Mb of memory on a 2.8 GHz desktop Pentium 4 PC running Linux.
Fly versus mosquito
When the two species are more distantly related, the only means of detecting large-scale similarity is through comparisons on the amino acid level. One example of this phenomenon arose during our comparison of the genomes of the malaria mosquito, Anopheles gambiae, and the fruit fly D. melanogaster. Because Anopheles was the second insect genome to be sequenced, the only available species for comparison was fruit fly. Our detailed analysis, done jointly with colleagues at the European Molecular Biology Laboratory in Heidelberg, was based on a combination of BLAST and MUMmer analysis [34]. These two species diverged about 250 million years ago, and they have an average protein sequence identity of 56%, less than that shared between humans and pufferfish. Although the two insects have the same number of chromosomes, the Anopheles genome is approximately twice as large, and the gene order has been almost completely shuffled, as our alignments revealed. Only small, but numerous, regions of 'microsynteny' remain: we reported 948 regions, the largest containing 8 genes in Anopheles and 31 in Drosophila. An interesting finding, though, was that despite extensive shuffling, each chromosome arm had a clear predominance of homologs on a single arm in the other species, indicating that intrachromosome gene shuffling was the primary force affecting gene order (see Figure 7 of [34]).
Fungus versus fungus
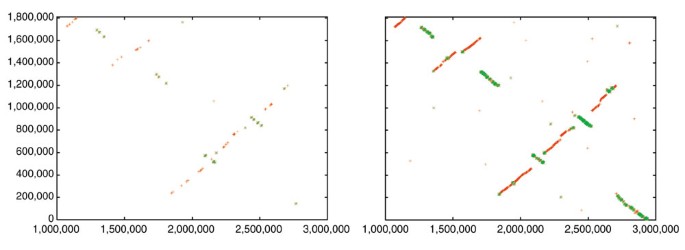
In a current application, we are using both Nucmer and Promer to compare two related fungal genomes, Aspergillus fumigatus (a human pathogen) and A. nidulans (a non-pathogenic model organism). Shotgun sequencing of these two genomes has been completed, and A. fumigatus is in the process of being completely finished; that is, all gaps are being closed. (A. fumigatus is a joint sequencing project of TIGR and The Sanger Institute, while A. nidulans is being sequenced at the Whitehead/MIT Genome Center.) At the time of our most recent comparison, the A. fumigatus genome had progressed to the point where it was assembled into 19 scaffolds spanning 28 Mbp, and the A. nidulans genome was assembled into 238 contigs spanning 30 Mbp. For this comparison, we first ran Nucmer and found that most of the two genomes mapped onto one another quite clearly: there are sufficient matches to reveal large segments of similarity in a simple dot plot. There has been extensive rearrangement of the chromosomes, but large-scale synteny is still present. For example, the largest contig (A1058) in A. fumigatus, at 2.9 Mbp, representing an essentially complete chromosome, maps onto five different scaffolds in A. nidulans. If one looks only at the Nucmer alignment of the largest of these, a 2.1 Mbp scaffold containing 10 contigs, it appears to be rearranged into multiple segments, but the matches are so scattered that it is difficult to tell how many segments there are (Figure 1, left-hand side).
Figure 1
Dot-plot alignments of a 2.9 Mbp chromosome of A. fumigatus (_x_-axis) to a 2.1 Mbp scaffold of A. nidulans (_y_-axis). Left: nucleotide-based alignment with Nucmer. Right: amino-acid-based alignment with Promer. Aligned segments are represented as dots or lines, up to 3,000 bp long in the Nucmer alignment and up to 9,500 bp in the Promer alignment. These alignments were generated by the mummerplot script and the Unix program gnuplot.
The syntenic alignment is much more clearly visible, however, if we use Promer instead. The simplest summary is just the number of bases included in the alignments: if we look at the Nucmer alignment between the scaffolds, the total number of matching bases is 81 kbp. In contrast, the Promer alignment covers 1.87 Mbp of A1058, beginning at nucleotide position 1,000,000 and continuing to the end of the chromosome. A graphical illustration is shown in Figure 1, which displays both the Promer and Nucmer alignments between the 2.1 Mbp scaffold from A. nidulans and scaffold A1058 of A. fumigatus. As the figure makes clear, the amino-acid-based alignment covers much more of the sequence of both species, and is therefore much more useful for determining homologous relationships between genes and chromosomal relationships.
Human versus human
One of the most challenging computational tasks one can perform today is the cross-comparison of mammalian genomes. The human and mouse genomes are sufficiently complete that much ongoing research is based on mappings between these two species. As shown in Table 1, MUMmer 3.0 can compare human and mouse chromosomes in a matter of minutes. The table shows the time (7 minutes 10 seconds, on a 2.4 GHz Pentium processor) required to align mouse chromosome 16 (Mm16) to human chromosome 21 (Hs21). These two were chosen because nearly all of Hs21 maps to one end of Mm16; in fact, researchers have developed a mouse model of Down syndrome that has an extra copy of this part of Mm16.
We ran a benchmark test of MUMmer 3.0 in which we compared the human genome (version of 3 January 2003, downloaded from GenBank) to itself by computing all maximal matches of length at least 300 between each chromosome and all the others. The resulting 631,975 matches allow one to identify both large- and small-scale interchromosomal duplications. Note that the run-times reported in [[6](/article/10.1186/gb-2004-5-2-r12#ref-CR6 "MUMmer: comparative applications. [ http://www.tigr.org/software/mummer/applications.html
]")\] are only for the match-finding part of MUMmer. The time for processing clusters and performing alignments in the gaps between matches are omitted as these vary widely depending on the parameters used.For this test, we needed a maximum of about 4 GB of memory. As we did not have a PC available with this amount of memory, we used a Sun-Sparc computer running the Solaris operating system, with 64 GB of memory and a 950 MHz processor.
We ran the alignment as follows. Each human chromosome was used as a reference, and the rest of the genome was used as a query and streamed against it. To avoid duplication, we only included chromosomes in the query if they had not already been compared; thus we first used chromosome 1 as a reference, and streamed the other 23 chromosomes against it. Then we used chromosome 2 as a reference, and streamed chromosomes 3-22, X, and Y against that, and so on.
The total length of all human chromosomes for this test was 2,839 Mbp. The time required to build all the suffix trees was 4.7 hours. The space requirement for the suffix tree was remarkably constant, with about 15.5 bytes per base-pair (with only one exception). The total query time was 101.5 hours, and memory usage never exceeded 3.9 GB (see [[6](/article/10.1186/gb-2004-5-2-r12#ref-CR6 "MUMmer: comparative applications. [ http://www.tigr.org/software/mummer/applications.html
]")\] for details). Thus, in approximately 4.5 days on a single processor, we matched the human genome against itself. This could easily be divided up among multiple computers, with each chromosome handled separately, bringing the time down to just 11 hours.Graphical viewers
Because the text-format output of MUMmer 3.0 is often voluminous, we have developed two graphical viewers, one for the purpose of comparing two genome assemblies or near-identical sequences, and the other for comparing more distantly related genomes, such as two distinct species. The first viewer, DisplayMUMs, is an open-source, platform-independent Java program. It has been tested on a variety of Unix/Linux platforms and also runs on Apple Macintosh (OS X) or Microsoft Windows computers. The program, which takes as input the results of running MUMmer, allows the user to align and view the results of two different assemblies of the same or very closely related genomes and to tile one set of contigs onto the other. This provides a powerful graphical front end for assembly comparison, a function that is frequently used in the process of assembling and finishing genomes. It allows a user to visualize the tiling of sequence reads onto an assembly in order to understand why contigs might not have properly merged together. Alternatively, one can compare the output of different genome assemblers on the same data, a task that can be quite bewildering when the genome is large and the assemblers disagree.
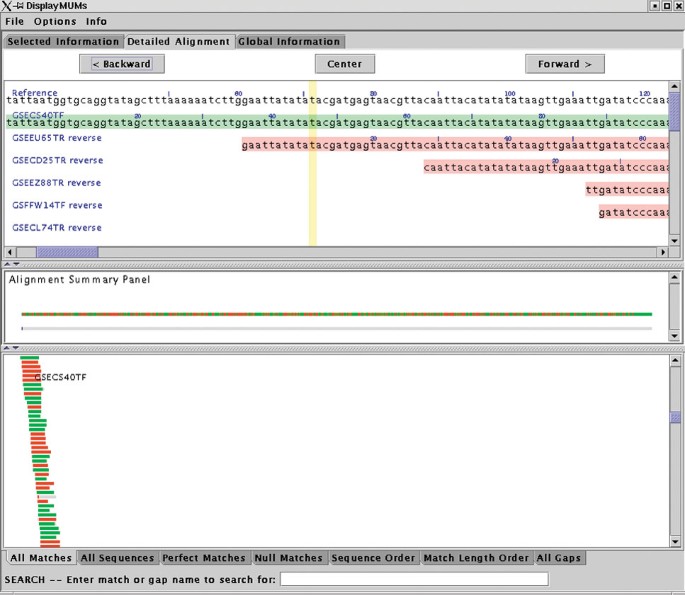
DisplayMUMs creates a stand-alone display, illustrated in Figure 2. It contains three main areas. The upper area can show a variety of types of information, including zoomed-in nucleotide alignments. The central panel shows a summary of the alignment, with the reference shown as a gray bar. The matches of the queries to the reference are shown as green (forward) and red (reverse) rectangles, with gaps indicated in gray. A second gray bar shows the gaps in blue, which may seem redundant but is useful when the scale is zoomed out; for example, if the sequence has only one small gap and the scale shows 1 Mbp, then the small gap will be invisible in the upper bar but will still be visible on the lower bar. The lower panel shows the tiling of all the query sequences on the reference, with red and green colors indicating the forward and reverse matching substrings. As Figure 2 shows, some sequences might match for only a small portion of their length, while others will match across their entire length. DisplayMUMs has many other features, including mouse-over and searching functions, all of which are documented in the software. As this example makes clear, its primary purpose is to improve the utility of MUMmer for genome-assembly analysis.
Figure 2
Sample display from DisplayMUMs, showing whole-genome alignment of individual shotgun reads (query sequences) to a contig from the Staphylococcus epidermidis genome. The display illustrates how exact matches of the tiling reads can be seen against the contig consensus. Green and red colors in the query sequences indicate alignment on the forward and reverse strands, respectively.
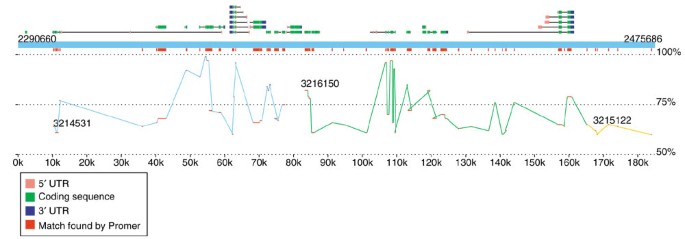
The second viewer, MapView, creates a picture of the mapping between two species based on Nucmer or Promer output. The motivation for creating this viewer was the rapidly increasing number of genome projects that are undertaken to enhance our understanding of another, already completed genome. In these projects, the second genome may have only faint DNA sequence similarity to the first, and in some cases the similarity may be detectable only through protein sequence alignments, such as those produced by Promer. A good example of such a project is the recent effort to sequence D. pseudoobscura mentioned above. The primary motivation for this project is to improve the annotation of D. melanogaster, and MUMmer is one of the tools being used to map the newly assembled D. pseudoobscura onto it. Because the reference genome is well annotated, we included in the viewer the option to display the locations of the genes (and their identifiers) along with the mapping at either the DNA or amino acid sequence level. A snapshot of this alignment by MapView is in Figure 3, which makes it clear that the amino acid conservation between these two species closely matches the annotated exon structure. This viewer can be used to highlight areas of a genome where exons might have been missed in previous analyses.
Figure 3
Sample display created by the MapView program, showing a 185 kbp slice of D. melanogaster chromosome 2L and its alignment to D. pseudoobscura. The alignment, generated by Promer, shows all regions of conserved amino acid sequence. The blue rectangle spanning the figure represents the reference (D. melanogaster), with annotated genes shown above it. Alternative splice variants of the same gene are stacked vertically. Exons are shown as boxes, with intervening introns connecting them. The 5' and 3' UTRs are colored pink and blue to indicate the gene's direction of translation. Promer matches are shown twice, once just below the reference genome, where all matches are collapsed into red boxes, and in a larger display showing the separate matches within each contig, where the contigs are colored differently to indicate contig boundaries. The vertical position of the matches indicates their percent identity, ranging from 50% at the bottom of the display to 100% just below the red rectangles.
The MapView program can produce output in three formats: fig (for viewing with the Unix xfig program), PostScript, or PDF. The most flexible format, fig, allows for unlimited scrolling and zooming, and for export to a wide range of additional formats. This makes it easy to view the mapping between a large collection of contigs and a large chromosome.