Transgenic Organisms: Principles, Steps, Methods, Types, Examples, Applications (original) (raw)
Transgenic organisms are those whose genetic material (DNA) has been deliberately altered in the laboratory by incorporating foreign DNA from another species to introduce a new beneficial trait in the target organism.
The gene that is transferred across species is called a transgene. This transgene transfer across species would not occur naturally and is only possible through advanced genetic engineering techniques.

Transgenic organisms
A transgenic organism differs from cloned or selectively bred organisms in that it does not carry genes from other species. Unlike traditional breeding, which requires selecting organisms with desirable traits over multiple generations, transgenic technology allows direct genetic manipulation without the need for a long period of natural reproduction.
Also, unlike somatic gene therapy, in which the transgene is restricted to the individual who receives the therapy, the germline is altered in transgenic organisms to produce stable, heritable changes that persist after reproduction. The transgene is delivered in such a way that it becomes a stable, functional, and inheritable part of the organism’s genetic makeup.
- History and Development of Transgenic Technology
- Principles for Creating Transgenic Organisms
- Steps in the Production of Transgenic Organisms
- Techniques (Methods) to generate transgenic organisms
- Types of Transgenic Organisms, Examples, and their applications
- Advantages of Transgenic Organisms
- Limitations of Transgenic Organisms
- Biosafety, Ethical, and Regulatory Issues of Transgenic Organisms
- Future Prospects of Transgenic Technology
- Conclusion
- References
History and Development of Transgenic Technology
- In 1972, the development and application of recombinant DNA technology for gene transfer in bacteria led to genetically modified organisms, providing proof of concept for transgenic technology and laying the foundation for developing higher transgenic organisms.
- The first transgenic organism was created in 1974 and contained Staphylococcus aureusgenes in Escherichia coli.
- This was followed by gene transfer in yeast cells (the first transgenic eukaryotic organism) in 1978, mouse cells in 1979, and mouse embryos in 1980.
- Similarly, in 1980, a team led by Gordon demonstrated the DNA microinjection technique, successfully producing the first transgenic mammal: transgenic mice. This research further opened avenues for transgenic research in higher animals, as the experiment showed that a foreign gene introduced can integrate into the germline and be passed on to offspring.
- In 1985, transgenic pigs, sheep, and rabbits followed.
- In 1989, a knockout mouse generated by homologous recombination was developed, enabling researchers to study gene silencing across multiple disease models. This development was awarded the Nobel Prize in Physiology or Medicine in 2007.
- Later, in 1996, Dolly the sheep, the first cloned mammal through nuclear transfer of a somatic cell, revolutionized transgenic technology in larger mammals.
- The late 19th century and early 20th century saw the development of xenotransplantation using organs from non-human primates.
- Similarly, CRISPR-Cas9-based gene editing in the 2010s and 2020s has further advanced transgenic technology.
Principles for Creating Transgenic Organisms
The main principle of transgenesis is gene transfer: the isolation of a target gene, its delivery, and its stable expression. The fundamentals for creating transgenic organisms are based upon multiple biological principles. They are:
Genetic code: The ability of a gene isolated from one species to be read, transcribed, and translated correctly by the cellular system of another species makes inter-species gene transfer feasible.
Integration of transgene: The transgene should stably integrate into the host’s genome at its intended location and be replicated alongside it. The integration of the transgene in germlines (sperm or egg cells) ensures that the transgene is passed on to subsequent generations.
Gene expression: Similarly, the gene must be expressed in the right site, at the right time, and at the right level to generate the desired effect. This is governed by the promoter sequence attached to the transgene, which significantly controls the necessary variables required for gene expression.
Steps in the Production of Transgenic Organisms
Identification and Isolation of the target gene: A specific gene or set of genes responsible for a desirable trait is first identified through gene sequencing. After DNA or RNA extraction, the target gene can be isolated using restriction enzymes. Alternatively, target genes can also be artificially synthesized in the laboratory.
Assembly of the GOI: The GOI is then assembled into a gene construct, comprising the target gene, a tissue-specific promoter for suitable expression, a terminator sequence, and a marker gene to identify transformed cells
Delivery into the host: The gene construct is delivered into host cells using various methods like microinjection, gene gun, Agrobacterium-mediated delivery, electroporation, or by using viral vectors. DNA Pronuclear microinjection, however, is one of the most used methods for transgenic animals, and Agrobacterium-mediated transformation is more relevant in the case of plants.
Selection and confirmation of transformed cells: The transformed cells containing the marker gene, mostly an antibiotic-resistant gene, are grown in-vitro containing the antibiotic, which allows only the transformed cells to grow. Selected cells are then screened again using molecular diagnostic techniques such as PCR, Southern blotting, and sequencing, to confirm correct integration and gene expression.
Development: In transgenic plants, confirmed transformed cells are cultured in a controlled environment and allowed to develop into whole plants by plant tissue culture, through plant organogenesis or somatic embryogenesis. In the case of transgenic animals, the transgenic embryo is implanted into the uterus of a surrogate mother for development.
Breeding: The resulting transgenic organisms are then bred through multiple generations to confirm that the transgene is inherited and expressed in all cells in each offspring.
Techniques (Methods) to generate transgenic organisms
To create transgenic organisms, various physical and biological methods are used to introduce a target gene into the host.
Pronuclear Microinjection: During early fertilization, the larger male pronucleus in the fertilized egg is selected, and the target gene is injected into it. The injected DNA randomly integrates into the host genome, which is then replicated and inherited by the daughter cells.
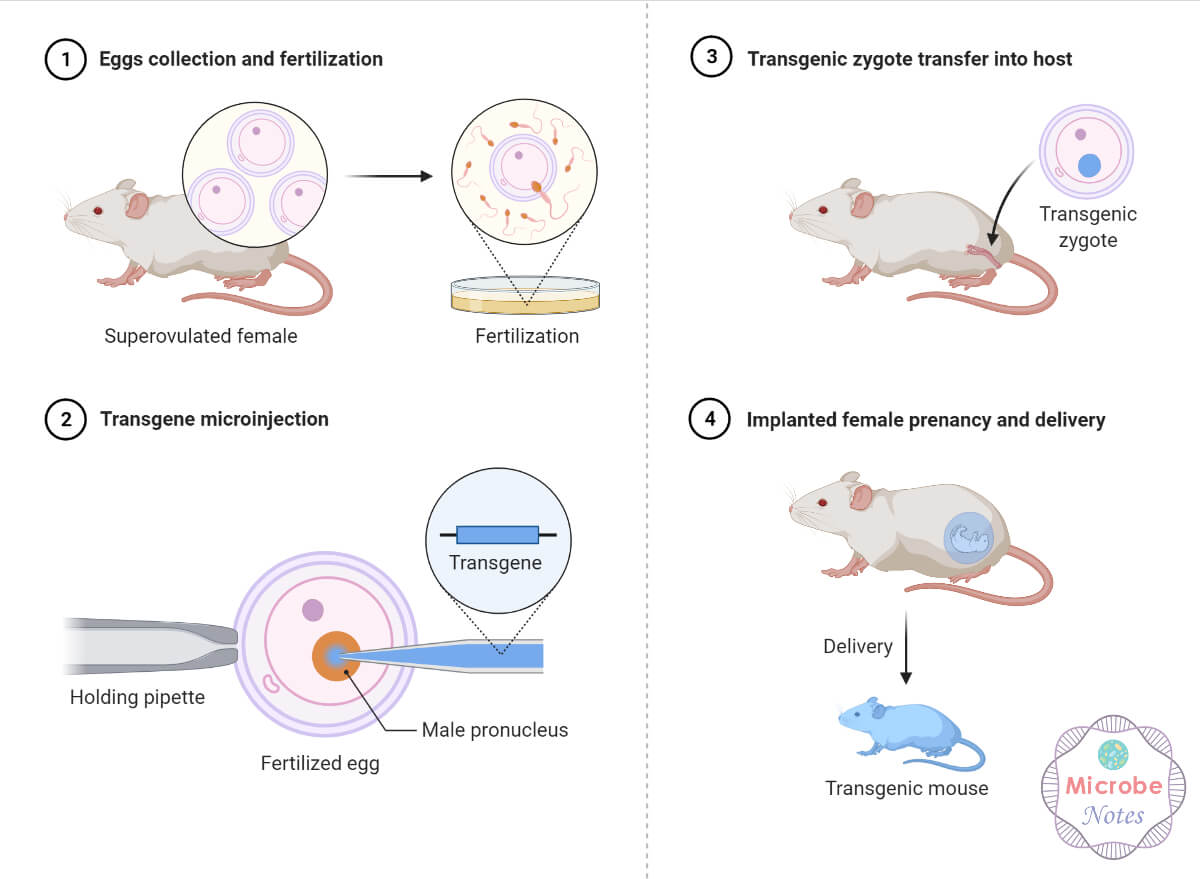
Figure: Transgenic Mice Production-Microinjection.
Embryonic Stem Cells Microinjection: Pluripotent stem cells, harvested from an early-stage blastocyst, are used to deliver the transgene. The desired transgene construct is delivered into embryonic stem cells. The transgene then integrates with the genome. After successful transformation, selection, and screening, these stem cells are then again microinjected into a recipient blastocyst. This develops into a new transgenic animal (with the transgene integrated into germline cells).
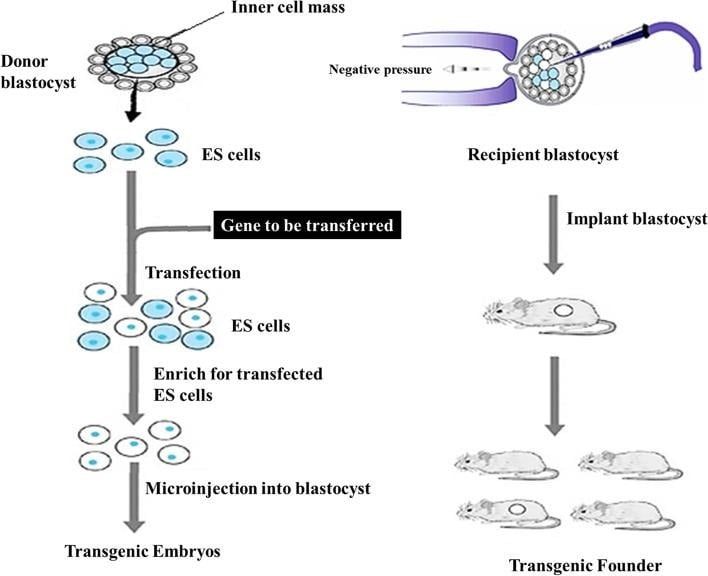
Embryonic Stem Cells Microinjection
Source: Shakweer, W. M. E., Krivoruchko, A. Y., Dessouki, S. M., & Khattab, A. A. (2023). A review of transgenic animal techniques and their applications. Journal, genetic engineering & biotechnology, 21(1), 55. https://doi.org/10.1186/s43141-023-00502-z
Agrobacterium-mediated delivery: Agrobacterium has a special ability to transfer the segment of its own plasmid DNA, called T-DNA, into the plant genome. Normally, this native T-DNA of Agrobacterium is associated with pathogenicity; however, it is replaced with the gene construct for transgenesis.

Agrobacterium-Mediated Transformation
Gene Gun: A gene gun works by the mechanism of physical acceleration of microscopic gold or tungsten particles (microprojectiles, which are coated with the gene construct) into the target cell.
Electroporation: Electroporation uses high-voltage electrical pulses to permeabilize the cell membrane, creating temporary pores through which the gene construct can enter the cell.
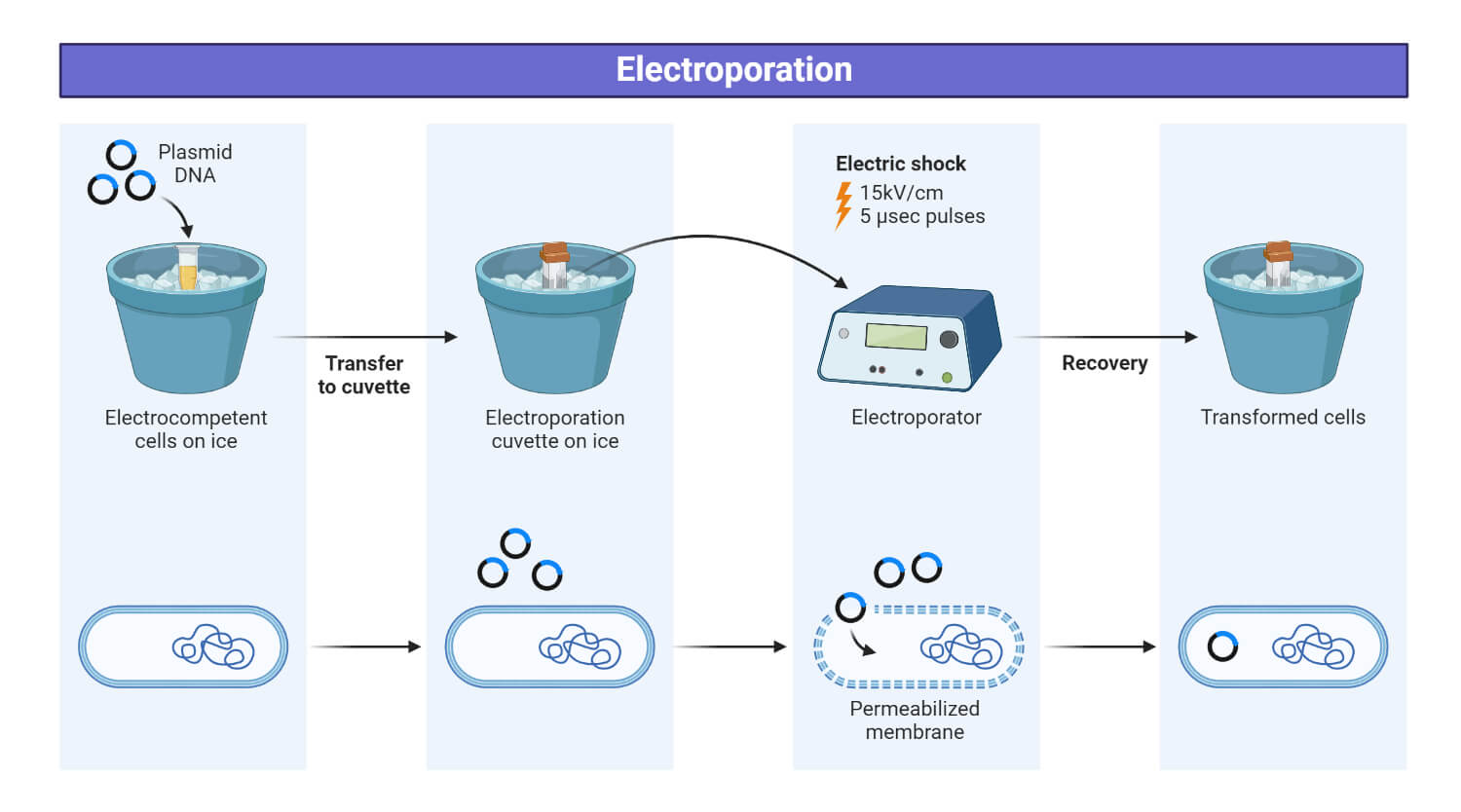
Electroporation Steps
Viral Vectors: The pathogenic genes of viruses, such as retroviruses, adenoviruses, and lentiviruses, are replaced with the desired transgene, producing a vehicle effective in entering the host cells and delivering the required gene without pathogenicity.
Transgenic Plants
Transgenic plants are primarily engineered to express traits that improve agricultural productivity. These traits are resistance against insects, herbicide tolerance, drought and salinity tolerance, resistance against plant pathogens, and enhanced nutritional content.
Examples of Transgenic Plants and Their Applications
BT Crops (Cotton & Corn): These crops carry the insecticidal gene from Bacillus thuringiensis to code Cry proteins, which are used as a natural insecticide.
Golden Rice: Genetically engineered to synthesize β-carotene, and used as an enhanced nutritional food rich in Vitamin A.
Herbicide-resistant Crops (Soybeans & Corns): Crops like Soybeans and corns are engineered to synthesize the compound 5-EPSPS, which can withstand glyphosate herbicide. This causes the herbicide to kill weeds without damaging the crops.
Transgenic animals
Transgenic animals are produced by introducing foreign genes into their genome, typically through microinjection. Transgenic animals are mostly used in biomedical research as bio-models for studying human diseases, as food sources, and for pharmaceutical production.
Examples of Transgenic Animals and Their Applications
Transgenic mice: Highly applicable in biomedical research to serve as model systems. Engineered to carry human genes to study the effect of a gene on development, and also an in-vitro study of diseases such as cancer, arthritis, Alzheimer’s disease, cardiovascular diseases, diabetes, etc.
Transgenic animals as food: GalSafe Pigs are engineered to make their meat suitable to eat for people having allergies to meat (Alpha-gal syndrome). AquAdvantage Salmon is engineered to accelerate their growth, providing for higher demand.
Transpharmers: Animals (sheep, goats, and cows) are engineered to secrete therapeutic proteins in their milk, such as α1-antitrypsin and antithrombin III, providing economic pharmaceutical alternatives.
Transgenic Microorganisms
Microorganisms remain among the most widely used transgenic organisms due to their rapid multiplication rates, ease of manipulation, and ability to produce biological products on a large scale.
Examples of Transgenic Microorganisms and Their Applications
Transgenic bacteria: Recombinant E. coli are used to manufacture large-scale human insulin. Engineered Lactobacillus and Lactococcus strains are directly used in the patient’s gut to treat bowel diseases and diabetes. Additional transgenic bacteria are also used to produce human growth hormones, interferons, interleukins, and antibiotics.
Transgenic yeasts: Saccharomyces cerevisiae and related yeasts are also used to produce therapeutic proteins and hormones at industrial levels, such as antimicrobial peptides, interferons, antigens (for vaccines), modified forms of insulin, etc.
Transgenic fungi: Transgenic fungi, such as Trichoderma and Aspergillus, are used in the production of enzymes (cellulases, proteases) and pharmaceuticals (antibiotics, human proteins). Entomopathogenic fungi, such as Metarhizium and Beauveria, are also genetically engineered to increase their virulence against insects.
Advantages of Transgenic Organisms
- Inter-species gene transfer: Transgenesis is advantageous over traditional selective breeding or hybridization techniques as it allows the transfer of a gene with a desirable trait into a completely unrelated host or species.
- Stable heritability: Transgenesis allows the stable integration of a transgene into the host germline, making sure that the introduced trait is inherited by all future generations.
- Controlled gene expression: Transgenic technology allows precise control over where and when the gene should be expressed. It ensures that the transgene doesn’t interfere with the host’s normal physiological processes.
- Animal models production: Transgenic technology has allowed the creation of animal models expressing specific human diseases. This enables researchers to study organisms that replicate the genetic basis of human conditions and diseases in a working biological system.
**Limitations of Transgenic Organisms
- Random effects: The random integration of a transgene into the host genome disrupts existing gene functions and could also lead to undesired gene expressions.
- Low efficiency: the overall efficiency of generating a transgenic organism remains low, with very few organisms developing into complete organisms.
- Transgene silencing: Due to epigenetic mechanisms, a transgene, even though it is successfully integrated into the genome, can have silenced expression over subsequent generations, raising issues about the long-term stability of the transgenic line.
- Stress to animals: The introduced transgene could cause unintended physical stress, such as pain or abnormalities.
Biosafety, Ethical, and Regulatory Issues of Transgenic Organisms
- Ecological risks: Transgenic organisms can pose unintended transgene flow into the environment, potentially causing genetic disruption in non-target organisms. This could range from simple minor health issues to extreme disruptions in ecosystems. For example, the development of herbicide-resistant weeds due to the acquisition of herbicide-resistant transgenes.
- Consumer rights and equity: Commercialization of transgenic plants and organisms raises questions about food choice and the right to make informed decisions about consumers’ food.
- Animal welfare and rights: Transgenic animals engineered to develop diseases raise questions about the physical suffering experienced by animals involved in biomedical research.
- Global regulatory issues: The difference in stringency and regulation in the use of transgenic organisms differs according to countries, causing international disagreements. This has led to slower progress in the application and commercialization of transgenic organisms.
Future Prospects of Transgenic Technology
- Precision in gene editing: Genome editing techniques such as CRISPR-Cas9, including base editing and prime editing, due to their precision, are further reducing off-target effects and are replacing random gene inserts with site-specific changes, which is highly relevant in transgenesis.
- Climate-resilient crops: Transgenic technology is being directed towards producing heat, drought, flood, and salinity-tolerant crops by simultaneously incorporating multiple traits in a single organism.
- Xenotransplantation: Refinements in transgenic pigs, such as the combination of multiple knockouts and human gene insertions to reduce immune incompatibility, are increasing.
- Industrial biotechnology: In industrial biotechnology, transgenic microorganisms are being engineered to produce next-generation biofuels, bioplastics, and rare pharmaceutical compounds.
Conclusion
Transgenic technology has advanced research, therapeutics, agriculture, and industry through interspecies gene transfer. However, its biosafety, ethical, and regulatory challenges need to be addressed carefully in the long run.
References
- https://www.ebsco.com/research-starters/zoology/transgenic-organisms
- Nicholl, D. S. T. (2008). An introduction to genetic engineering (3rd ed.). Cambridge University Press
- Pray, L. (2008) Recombinant DNA technology and transgenic animals. Nature Education 1(1):51. https://www.nature.com/scitable/topicpage/recombinant-dna-technology-and-transgenic-animals-34513/
- Chang, A. C., & Cohen, S. N. (1974). Genome construction between bacterial species in vitro: replication and expression of Staphylococcus plasmid genes in Escherichia coli. Proceedings of the National Academy of Sciences of the United States of America, 71(4), 1030–1034. https://doi.org/10.1073/pnas.71.4.1030
- Jon W. Gordon, Transgenic Technology and Laboratory Animal Science, ILAR Journal, Volume 38, Issue 1, 1997, Pages 32–41, https://doi.org/10.1093/ilar.38.1.32
- Palmiter, R., Brinster, R., Hammer, R. et al. Dramatic growth of mice that develop from eggs microinjected with metallothionein–growth hormone fusion genes. Nature 300, 611–615 (1982). https://doi.org/10.1038/300611a0
- Vipra, M., Patil, N., Sivaram, A. (2022). Applications of Recombinant DNA Technology. In: A Complete Guide to Gene Cloning: From Basic to Advanced . Techniques in Life Science and Biomedicine for the Non-Expert. Springer, Cham. https://doi.org/10.1007/978-3-030-96851-9\_9
- https://www.nobelprize.org/prizes/medicine/2007/advanced-information/
- Wilmut, I., Schnieke, A., McWhir, J. et al. Viable offspring derived from fetal and adult mammalian cells. Nature 385, 810–813 (1997). https://doi.org/10.1038/385810a0
- Cooper, D. K. C., Ekser, B., & Tector, A. J. (2015). A brief history of clinical xenotransplantation. International Journal of Surgery, 23(Pt B), 205–210. https://doi.org/10.1016/j.ijsu.2015.06.060
- Frangoul, H., Altshuler, D., Cappellini, M. D., Chen, Y. S., Domm, J., Eustace, B. K., Foell, J., de la Fuente, J., Grupp, S., Handgretinger, R., Ho, T. W., Kattamis, A., Kernytsky, A., Lekstrom-Himes, J., Li, A. M., Locatelli, F., Mapara, M. Y., de Montalembert, M., Rondelli, D., Sharma, A., … Corbacioglu, S. (2021). CRISPR-Cas9 Gene Editing for Sickle Cell Disease and β-Thalassemia. The New England journal of medicine, 384(3), 252–260. https://doi.org/10.1056/NEJMoa2031054
- Haruyama, N., Cho, A., & Kulkarni, A. B. (2009). Overview: engineering transgenic constructs and mice. Current protocols in cell biology, Chapter 19, Unit–19.10. https://doi.org/10.1002/0471143030.cb1910s42
- Shakweer, W. M. E., Krivoruchko, A. Y., Dessouki, S. M., & Khattab, A. A. (2023). A review of transgenic animal techniques and their applications. Journal, genetic engineering & biotechnology, 21(1), 55. https://doi.org/10.1186/s43141-023-00502-z
- Xu, Y., Zhao, J., Ren, Y. et al. Derivation of totipotent-like stem cells with blastocyst-like structure forming potential. Cell Res 32, 513–529 (2022). https://doi.org/10.1038/s41422-022-00668-0
- Tiwari, M., Mishra, A. K., & Chakrabarty, D. (2022). Agrobacterium-mediated gene transfer: recent advancements and layered immunity in plants. Planta, 256(2), 37. https://doi.org/10.1007/s00425-022-03951-x
- Transpharmers – Bioreactors for Pharmaceutical Products. (2006). In: The ABCs of Gene Cloning. Springer, Boston, MA. https://doi.org/10.1007/0-387-28679-9\_21
- Wang, C., & Wang, S. (2017). Insect Pathogenic Fungi: Genomics, Molecular Interactions, and Genetic Improvements. Annual review of entomology, 62, 73–90. https://doi.org/10.1146/annurev-ento-031616-035509
- Maksimenko, O. G., Deykin, A. V., Khodarovich, Y. M., & Georgiev, P. G. (2013). Use of transgenic animals in biotechnology: prospects and problems. Acta naturae, 5(1), 33–46.
- Davis, D. J., & Yeddula, S. G. R. (2024). CRISPR Advancements for Human Health. Missouri medicine, 121(2), 170–176.