Failure of ICD-9-CM Codes to Identify Patients with Comorbid Chronic Kidney Disease in Diabetes (original) (raw)
Abstract
Objective
To determine prevalence of chronic kidney disease (CKD) in patients with diabetes, and accuracy of International Classification of Diseases, 9th Revision, Clinical Modification (ICD-9-CM) codes to identify such patients.
Data Sources/Study Setting
Secondary data from 1999 to 2000. We linked all inpatient and outpatient administrative and clinical records of U.S. veterans with diabetes dually enrolled in Medicare and the Veterans Administration (VA) health care systems.
Study Design
We used a cross-sectional, observational design to determine the sensitivity and specificity of renal-related ICD-9-CM diagnosis codes in identifying individuals with chronic kidney disease.
Data Collection/Extraction Methods
We estimated glomerular filtration rate (eGFR) from serum creatinine and defined CKD as Stage 3, 4, or 5 CKD by eGFR criterion according to the Kidney Disease Outcomes Quality Initiative guidelines. Renal-related ICD-9-CM codes were grouped by algorithm.
Principal Findings
Prevalence of CKD was 31.6 percent in the veteran sample with diabetes. Depending on the detail of the algorithm, only 20.2 to 42.4 percent of individuals with CKD received a renal-related diagnosis code in either VA or Medicare records over 1 year. Specificity of renal codes for CKD ranged from 93.2 to 99.4 percent. Patients hospitalized in VA facilities were slightly more likely to be correctly coded for CKD than patients hospitalized in facilities reimbursed by Medicare (OR 5.4 versus 4.1, p = .0330)
Conclusions
CKD is a common comorbidity for patients with diabetes in the VA system. Diagnosis codes in administrative records from Medicare and VA systems are insensitive, but specific markers for patients with CKD.
Keywords: Diabetes, chronic kidney disease, diabetic nephropathy, administrative records, ICD-9-CM codes
Comorbid chronic kidney disease (CKD) in patients with diabetes is likely to have profound effects on health care outcomes in the U.S. in the coming decades. The prevalence of diabetes is projected to increase from approximately 16 million adults to almost 22 million by 2025 (Harris et al. 1998). The prevalence of comorbid CKD in diabetic patient populations has not been well characterized, but its presence is likely to increase overall morbidity and mortality. CKD and diabetes are independent predictors for cardiovascular disease, cardiovascular events, complications of cardiovascular procedures, early mortality following myocardial infarction, and hospital-acquired acute renal failure (Anderson et al. 1999; Conlon et al. 1999; Tonelli et al. 2001; Szczech et al. 2002; Go et al. 2004; Keith et al. 2004; Weiner et al. 2004; Thakar et al. 2005).
In addition to increasing the risk of cardiac disease and acute renal failure, CKD in diabetes greatly increases the risk of chronic kidney failure. Although the majority of patients with comorbid CKD and diabetes die prior to reaching end-stage renal disease (ESRD), almost half of the incident cases of ESRD in the U.S. are attributed to diabetes (Keith et al. 2004; USRDS 2004). CKD in diabetes is usually because of diabetic nephropathy. Given that appropriate medical management can slow the progression of established diabetic nephropathy, identifying patients for interventions to slow the rate of loss of glomerular filtration rate (GFR) will be important to forestall the influx of patients into ESRD programs that is predicted by the surge of prevalent diabetes (Lewis et al. 1993, 2001; Brancati et al. 1997; Gaede et al. 1999; Brenner et al. 2001. One-third of adults with type 2 diabetes and comorbid CKD lack proteinuria, which typically characterizes diabetic nephropathy (Kramer et al. 2003). However, even mild renal insufficiency without proteinuria increases the risk for cardiovascular disease in patients with diabetes (Gimeno-Orna et al. 2004). Therefore, better understanding of comorbid CKD of any etiology may inform strategies to prevent cardiovascular disease, acute renal failure, and ESRD in patients with diabetes.
A first step to understanding how CKD in diabetes affects disease outcomes is to develop methods to accurately identify individuals with CKD in patient populations. Investigators in health services research often turn to the International Classification of Diseases, 9th Revision, Clinical Modification (ICD-9-CM) diagnosis codes in administrative records to study the effect of health care delivery upon disease outcomes. The ability of administrative records to identify cases is limited by the completeness and accuracy of diagnosis coding, which in turn is limited by the quality of documentation in the medical record (Fisher et al. 1990, 1992; Mitchell et al. 1994; Daley 1997; Iezzoni 1997; Rosamond et al. 2004). Previous work has validated the use of ICD-9-CM codes from administrative records to identify individuals with diabetes (Hebert et al. 1999; Miller, Safford, and Pogach 2004), but no studies have validated the ability of diagnosis codes to identify individuals with CKD. Despite this limitation, investigators have used ICD-9-CM diagnosis codes to define the presence of comorbid CKD as a risk factor for adverse outcomes in cardiovascular surgery (Mack et al. 2004), estimate CKD prevalence in patient populations with diabetes (Young et al. 2004), and make risk adjustments for renal failure in hospital-based quality assessment systems (Goldfield and Villani 1996). However, it is unknown if renal-related diagnosis codes in administrative records completely and specifically identify patients with CKD.
Although a plethora of codes are used to describe various attributes of kidney disease, most do not indicate the severity of kidney dysfunction. Recognizing the need for standards to both define and assess CKD through all stages of severity, the Work Group of the National Kidney Foundation Kidney Disease Outcomes Quality Initiative (K/DOQI) developed a staging classification based on GFR, defining CKD as GFR less than 60 ml/min/1.73 m2 for at least 3 months duration (NKF 2002). A predictive equation derived from the Modification of Diet in Renal Disease study has been validated as a measure of estimated GFR (eGFR), and applied to the K/DOQI definition of CKD (Levey et al. 1999; NKF 2002; Levey et al. 2003). The methodology enables investigators to identify cases of CKD in populations where serum creatinine and demographic data are available, with the limitation that individuals with proteinuria and eGFR greater than 59 ml/min/1.73 m2 will be misclassified as not having CKD. Therefore, using only an eGFR-based criterion to define CKD will tend to underestimate the prevalence of CKD.
By linking clinical and administrative datasets from the VA and Medicare systems, we determined how completely and accurately ICD-9-CM diagnosis codes from administrative records identify individuals with CKD in a population of patients with diabetes. We defined CKD by eGFR criterion, using definitions set forth by the K/DOQI Work Group.
MATERIALS AND METHODS
Population, Study Sample, and Data Source
The study sample was drawn from the national VA population of 566,509 veterans with diabetes alive on September 30, 1999. A description of the population, and the criteria for defining diabetes, has been described in previous work (Miller, Safford, and Pogach 2004). Patient-level data elements in VA records included demographics, visits and hospitalizations, providers, diagnosis and procedure codes, and values of serum creatinine tests. Medicare records from institutional inpatient and outpatient files (Part A), and physician care (Part B) included the same data elements plus revenue center and beneficiary status codes, but without serum creatinine values. Records were linked for individuals enrolled in both VA and Medicare systems.
We excluded 153,680 individuals who lacked a record of any serum creatinine tests or key demographic variables needed for estimation of GFR. We excluded 143,678 individuals because they either (1) lacked a second serum creatinine value 90 days or more beyond the index creatinine value, or (2) follow-up eGFR was either greater than 89 ml/min/1.73 m2, or less than 60 ml/min/1.73 m2, when the index eGFR was 60–89 ml/min/1.73 m2, or (3) follow-up eGFR was greater than 59 ml/min/1.73 m2 when the index eGFR was less than 60 ml/min/1.73 m2. The purpose of these exclusions was to ensure that individuals classified as having CKD by index eGFR had chronically low eGFR, as opposed to a transient decrease in eGFR. The exclusions also ensured that individuals classified as having an index eGFR of 60 ml/min/1.73 m2 or greater maintained a stable eGFR (neither rising nor declining) for at least 3 months duration. Additionally, we excluded 5,421 individuals with indicators for preexisting ESRD. Preexisting ESRD was defined if any one of the following conditions were present within 2 years before the study period:
- Medicare status codes were 11, 21, or 31, indicating ESRD.
- Diagnosis codes V420, V56, V451, E8791, indicating ESRD, dialysis, or kidney transplant.
- Procedure codes 3993 or 5498, indicating dialysis.
- CPT4 codes 90935, 90937, 90945, 90947, 90989, 90993, 90921, 90925, indicating dialysis or dialysis training.
- VA stop codes 602–611, indicating a visit to the dialysis unit.
- Revenue Center codes 821, 831, 841 or 851, indicating outpatient dialysis.
- Revenue Center codes 801, 802, 803 or 804, indicating inpatient dialysis.
The final study sample of 263,730 veterans had an index creatinine value drawn in fiscal year (FY) 1999, and a subsequent creatinine value drawn 90–365 days following. Of this sample, 71 percent were dually enrolled in VA and Medicare systems; of those with index eGFR less than 60 ml/min/1.73 m2, 87 percent were enrolled in both systems.
The study data is stored at the VA New Jersey Health Care System facility. The research has been approved by the VA New Jersey Health Care System Institutional Review Board.
Variables
We used the four-variable Modification of Diet in Renal Disease equation to derive an eGFR from VA serum creatinine values (Levey et al. 1999; Lamb et al. 2003).
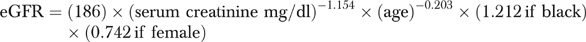
We defined index eGFR as either (1) the first eGFR in FY 1999, if all subsequent eGFR values were greater than 89 ml/min/1.73 m2, or (2) the first eGFR value less than 90 ml/min/1.73 m2 occurring in FY 1999. Veterans whose index eGFR was less than 60 ml/min/1.73 m2 were classified as having CKD according to the definition of the K/DOQI Work Group (NKF 2002). The index eGFR was used to stage CKD according to K/DOQI guidelines:
- Stage 3: Index eGFR 30–59 ml/min/1.73 m2, subsequent eGFR less than 60 ml/min/1.73 m2.
- Stage 4: Index eGFR 15–29 ml/min/1.73 m2, subsequent eGFR less than 60 ml/min/1.73 m2.
- Stage 5: Index eGFR less than 15 ml/min/1.73 m2, subsequent eGFR less than 60 ml/min/1.73 m2
Veterans whose index eGFR was greater than 59 ml/min/1.73 m2 were classified as “no CKD” for the purposes of this study.
From the ICD-9-CM coding manual, we grouped 79 renal-related diagnosis codes into the following seven categories: (A) chronic renal failure, (B) chronic kidney pathophysiology, (C) diabetic nephropathy, (D) acute renal failure and disease, (E) systemic diseases that can cause CKD, (F) kidney or upper urinary tract neoplasm, and (G) kidney or upper urinary tract anatomical abnormalities (Medicode 1999) (Table 1). Codes were initially assigned to groups by the clinical judgment of a nephrologist based on code descriptors. However, if the code had an ambiguous descriptor, the coding practices of a university-affiliated teaching hospital were also used to make group assignment. The groups were empirically ranked by specificity in identifying individuals with low eGFR within the study sample, with Group A being most specific and Group G being least specific. Although all of the renal-related ICD-9-CM codes were assessed for each individual in the study sample over the time span of 1 year, each individual was ultimately assigned to the single, highest ranked (most specific) group according to the codes in their records.
Table 1.
Renal-Related ICD-9-CM Diagnosis Codes, by Common Descriptor Groups
| Description | Group | Codes (79) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Chronic renal failure | A | 403.11 | 403.91 | 404.12 | 404.13 | 404.92 | 404.93 | ||
| 585 | 586 | 587 | |||||||
| Chronic kidney pathophysiology | B | 274.1 | 403.10 | 403.90 | 404.10 | 404.11 | 404.90 | 404.91 | |
| 581 | 582 | 583 | 590.0 | 593.6 | 593.9 | 753.12 | |||
| 753.13 | 753.14 | ||||||||
| Diabetic nephropathy | C | 250.4 | |||||||
| Acute renal failure and disease | D | 403.00 | 403.01 | 404.00 | 404.01 | 404.02 | 404.03 | 405.01 | |
| 453.3 | 580 | 584 | 590.1 | 590.2 | 590.3 | 590.8 | |||
| 593.81 | 866 | ||||||||
| Syatemic disease causing CKD | E | 203.0 | 277.3 | 287.0 | 446.0 | 446.2 | 446.4 | 446.6 | |
| 710.0 | 710.1 | ||||||||
| Upper urinary tract neoplasm | F | 189.0 | 189.1 | 233.9 | 236.91 | 239.5 | |||
| Anatomic abnormality | G | 40.1 | 447.3 | 589 | 591 | 593.4 | 593.5 | 593.7 | 592 |
| 593.0 | 593.1 | 593.2 | 593.3 | 593.82 | 593.89 | 753.0 | |||
| 753.10 | 753.11 | 753.15 | 753.16 | 753.17 | 753.19 | 753.2 | 753.3 |
Study Design
The study design was a cross-sectional analysis over a 1-year time frame. The study period began with the index eGFR, occurring within FY 1999, and ended 365 days later. We searched inpatient and outpatient encounters within both VA and Medicare records for occurrence of renal-related diagnosis codes. Outpatient encounters were limited to physician-attended office visits. The patient was the unit of analysis for determining the sensitivity and specificity of renal-related codes for the presence of CKD. For comparison of VA and Medicare inpatient coding accuracy, the unit of analysis was the hospital episode of care. Because transfers could potentially inflate the number of hospitalizations that were coded for renal-related diagnoses, we excluded 234 hospital stays that were less than or equal to 1 day length of stay, and had a date of admission or discharge that was less than or equal to one day from an adjacent hospitalization.
Statistical Methods
Crude prevalence of chronic kidney disease by eGFR criterion was calculated as the number of individuals with Stage 3, 4, or 5 CKD divided by the total sample size. The crude prevalence of CKD was standardized to the reference population for age and sex distributions using the direct method. Sensitivity of each code group (A–G) was calculated as the percent of patients with Stage 3, 4, or 5 CKD assigned to the code group. Specificity was calculated as the percent of patients with eGFR greater than 59 ml/min/1.73 m2 not assigned to the code group. For hospital episodes of care, Pearson's χ2 was used to test the association of chronic renal failure codes (Group A), versus Group B–G codes, with low eGFR. The homogeneity of the odds ratios for code assignment in VA versus Medicare-reimbursed facilities was evaluated with the Breslow–Day test. Alpha error was considered significant at less than 0.05, two-tailed. Frequency counts and analyses were run on SAS for Windows V9 (Statistical Analysis Systems, Carey, NC, USA).
RESULTS
Prevalence of CKD by eGFR Criterion
The study sample of 263,730 individuals with diabetes contained the following proportions of individuals by stage of CKD: (No CKD) 68.4 percent; (Stage 3) 28.7 percent; (Stage 4) 2.5 percent; (Stage 5) 0.4 percent. The overall prevalence of CKD, defined in our study as Stages 3, 4, or 5, was 31.6 percent (83,338/263,730) (Table 2). When standardized to the age and sex distribution of the U.S. adult population with diabetes in 1999 (Pleis and Coles 2003), the prevalence of CKD fell to 29.0 percent. Both crude and standardized prevalence increased with age, up to 46.1 and 54.8 percent, respectively, in those 70 years and older. Because urine protein values were not available, individuals with Stage 1 or Stage 2 CKD could not be distinguished from nonproteinuric individuals without CKD. Consequently, all individuals with eGFR greater than 59 ml/min/1.73 m2 were combined in the general category of “no CKD.”
Table 2.
Demographic Characteristics of Veterans in the Study Sample, by CKD* Status
| _All (N_=263,730) | |||
|---|---|---|---|
| N | % | CKD (N=83,338) % with CKD | |
| Overall | 263,730 | 100 | 31.6 |
| Men | 258,272 | 97.9 | 31.5 |
| Women | 5,458 | 2.1 | 34.3 |
| <40 years | 4,317 | 1.6 | 3.8 |
| 40–54 years | 52,395 | 19.9 | 11.0 |
| 55–69 years | 103,361 | 39.2 | 28.7 |
| 70+ years | 103,657 | 39.3 | 46.1 |
| White | 191,539 | 72.6 | 35.0 |
| African American | 42,529 | 16.1 | 26.2 |
| Hispanic | 8,351 | 3.2 | 21.8 |
| Other | 5,090 | 1.9 | 30.7 |
| Unknown | 16,221 | 6.2 | 10.9 |
Ability of Renal ICD-9-CM Coding to Identify Individuals with CKD
Frequency counts for individuals with one, and only one, occurrence of any of the 79 renal-related ICD-9-CM codes, from inpatient and outpatient records combined, were ascertained to determine potential random error rates (Table 3). While 13.3 percent (11,067/83,338) of all individuals with Stage 3, 4, or 5 CKD received a renal-related ICD-9-CM code at one, and only one, encounter, the false positive rate was 3.8 percent (6,843/180,392), suggesting that the majority of these “one-time only” code assignments were not random errors.
Table 3.
Identifying Individuals with CKD† Using ICD-9-CM Diagnosis Codes from Inpatient and Outpatient Records: 83,338 Individuals with CKD and 180,392 Individuals without CKD
| ICD-9-CM Diagnosis Code Groups* | True+N | Sensitivity (%) | False+N | Specificity (%) |
|---|---|---|---|---|
| Individuals with 1, and only 1, code from any group | ||||
| ABCDEFG | 11,067 | 13.3 | 6,843 | 96.2 |
| Individuals with at least 1 code from the groups | ||||
| A | 16,864 | 20.2 | 1,034 | 99.4 |
| AB | 29,355 | 35.2 | 4,168 | 97.7 |
| ABC | 32,255 | 38.7 | 8,035 | 95.5 |
| ABCD | 33,454 | 40.1 | 8,736 | 95.2 |
| ABCDE | 33,884 | 40.7 | 9,106 | 95.0 |
| ABCDEF | 34,151 | 41.0 | 9,551 | 93.5 |
| ABCDEFG | 35,318 | 42.4 | 12,317 | 93.2 |
| Total Individuals | 83,338 | 180,392 |
The nine codes in the Group A algorithm are the only ICD-9-CM codes specific for chronic renal failure, either as an isolated diagnosis, or combined with hypertension and/or heart failure. Of individuals with Stage 3, 4, or 5 CKD, 20.2 percent (16,864/83,338) were identified by searching all administrative records for codes from the Group A algorithm: this algorithm had the lowest false positive rate of 0.6 percent (1,034/180,392) compared with any other code group algorithm (Table 3). Including additional individuals with codes in Groups B and C increased the sensitivity to 38.7 percent (32,255/83,338), but lowered the specificity to 95.5 percent as more individuals with eGFR greater than 59 ml/min/1.73 m2 were included. When all individuals with any possible renal-related ICD-9-CM codes were included (Groups A–G), sensitivity increased to 42.4 percent (35,318/83,338), while specificity dropped to 93.2 percent.
Comparing Accuracy of Coding for Inpatient Encounters
We found a renal-related ICD-9-CM diagnosis code contained in the administrative records of 32,529 hospital episodes for 18,755 individuals: 11,253 of these hospitalizations were reimbursed by Medicare, while 21,276 occurred in VA facilities (Table 4). Of hospitalizations occurring in Medicare-reimbursed facilities, 39.6 percent (4,461/11,253) were assigned a Group A code. However, 15.6 percent (233/1,495) of hospitalizations for patients with eGFR greater than 59 ml/min/1.73 m2 were incorrectly assigned a Group A code indicative of chronic renal failure in Medicare-reimbursed facilities. In contrast, only 29.0 percent (6,166/21,276) of hospitalizations in VA facilities received a Group A code. Of these, 7.6 percent (116/1,562) of hospitalizations for patients with eGFR greater than 59 ml/min/1.73 m2 were incorrectly assigned a Group A code. In the VA, the odds of assigning a Group A code to patients with Stage 3, 4, or 5 CKD was 5.4 (95 percent CI: 4.4–6.5; p<.0001) times greater than the odds of assigning a non–Group A renal-related code to patients with eGFR greater than 59 ml/min/1.73 m2. In contrast, the odds ratio was 4.1 (95 percent CI: 3.6–4.8; p<.0001) among Medicare-reimbursed facilities. Comparing odds ratios, the correct assignment of Group A codes to patients with Stage 3, 4, or 5 CKD was significantly more likely in VA facilities compared with Medicare-reimbursed facilities (Breslow–Day test for homogeneity of odds ratios, _p_=.0330).
Table 4.
Accuracy of Inpatient Coding for Chronic Renal Failure (Group A Codes) by Medicare Provider versus VA Provider
| Medicare | VA | |||||
|---|---|---|---|---|---|---|
| All | CKD* (%) | No CKD (%) | All | CKD (%) | No CKD (%) | |
| Coded as group A | 4,461 (39.6) | 4,228 (43.3) | 233 (15.6) | 6,166 (29.0) | 6,050 (30.6) | 116 (7.6) |
| Coded as group B–G | 6,792 (60.4) | 5,530 (56.7) | 1,262 (84.4) | 15,110 (71.0) | 13,700 (69.4) | 1,410 (92.4) |
| Total | 11,253 | 9,758 | 1,495 | 21,276 | 19,750 | 1,526 |
| Odds ratio | 4.1 (3.6–4.8) p<.0001 | 5.4 (4.4–6.5) p<.0001 | ||||
| Breslow–Day test for homogeneity of odds ratios _p_=.0330 |
DISCUSSION
The main finding of this study is that, in adult veterans with diabetes, ICD-9-CM diagnosis codes in VA and Medicare administrative records fail to identify the large majority of individuals with comorbid CKD, when CKD is narrowly defined by eGFR criterion as Stage 3, 4, or 5 CKD. Despite each individual having had, at minimum, two opportunities to diagnose chronic kidney disease within a year, a comprehensive search of all VA and Medicare administrative records revealed that only 20.2 percent of individuals with low eGFR received a code specifically indicating chronic renal failure (Group A code). However, if present, a Group A code indicated that the individual had Stage 3, 4, or 5 CKD with 99.4 percent specificity. Extending the algorithm to 17 additional ICD-9-CM codes for chronic kidney disorders that may not necessarily be associated with low eGFR (Groups B and C) increased sensitivity to 38.7 percent, while specificity declined to 95.5 percent. Extending the algorithm to include individuals with Group D codes, indicating acute renal failure and disease, increased the sensitivity to 40.1 percent, while specificity declined to 95.2 percent. Because underlying CKD is a risk factor for acute renal failure, it is not surprising that including diagnosis codes for acute renal failure in the algorithm identified additional individuals with CKD (Mangoset al. 1995; McCullough et al. 1997; Gruberg et al. 2001). Finally, adding 37 renal-related codes (Groups E–G) for kidney disorders that may, or may not, result in chronically low eGFR increased the sensitivity of the coding algorithm to a maximum value of 42.4 percent, but at a cost of further decline in specificity to 93.2 percent.
If a hospitalized patient is recognized and coded for renal disease, VA hospitals are slightly more accurate than Medicare-reimbursed hospitals in assigning Group A codes, indicating chronic renal failure, to patients with low eGFR. While less likely to assign a Group A code to an individual with low eGFR, VA hospitals were also less likely to assign a Group A code to individuals with eGFR greater than 59 ml/min/1.73 m2. For a medical coder to assign any of the Group A codes for chronic renal failure to a patient's discharge abstract, the word “failure” must be included in the description of chronic kidney disease within the medical record (AHA 2002). Terminology such as “renal insufficiency, chronic kidney disease, or diabetic nephropathy” cannot be coded as any of the Group A codes. The slightly lower coding accuracy of chronic kidney disease by Medicare providers, combined with the finding that 87 percent of individuals with chronic kidney disease were dually enrolled in Medicare and the VA system suggests that the coding gap for CKD includes providers reimbursed by Medicare as well as the VA health care system. The fact that individuals in the study sample had at least two serum creatinine tests 3–12 months apart supports the inference that the lack of CKD coding was not due simply to lack of patient encounters with the medical care system.
Inaccuracy in coding CKD in hospitalized patients may compromise the validity of risk adjustment systems that depend on case mix of the patient population. The presence of comorbid CKD greatly increases the risk of adverse outcomes in patients with myocardial infarction or cardiovascular procedures (Miettinen et al. 1996; Conlon et al. 1999; Gruberg et al. 2001; Mann et al. 2001; Shlipak et al. 2002; Wright et al. 2002; Callahan et al. 2003). If individuals with underlying CKD in these patient groups are not correctly identified, risk adjustment may lead to biased outcome assessments. The Work Group of K/DOQI has proposed that the stage of CKD should be included routinely with each renal-related ICD-9-CM code to facilitate the use of administrative databases for epidemiological and outcomes surveillance (NKF 2002).
An important secondary finding of this study is that the prevalence of CKD, narrowly defined as Stage 3, 4, or 5 CKD, is quite high (31.2 percent) among the population of veterans with diabetes. By excluding individuals with nonexistent or unstable patterns of eGFR from the sample, we attempted to minimize misclassification in order to maximize the validity of our findings regarding the sensitivity and specificity of renal-related ICD-9-CM codes. However, our methods may have introduced selection bias, in that we excluded veterans who had no serum creatinine tests, or veterans without sequential serum creatinine values. If patients with CKD are more likely to get sequential serum creatinine testing than patients without CKD, the sample for our study could have been enriched with patients with CKD. However, even taking the extreme position that none of the veterans with diabetes that we initially excluded from our study sample had CKD, a minimum of 14.7 percent (83,338/566,509) of the national VA population of patients with diabetes have CKD based solely on low eGFR criterion.
CKD is increasingly recognized as a common comorbid condition in selected patient populations. CKD is present in 60 percent of diabetic and nondiabetic patients discharged from hospitalizations for acute myocardial infarction (Langston et al. 2003). The prevalence of CKD was 13 percent among U.S. adults with type 2 diabetes in the population-based sample from the Third National Health and Nutrition Examination Survey (NHANES III), where CKD was defined as Stage 3, 4 or 5 CKD based on eGFR (Kramer et al. 2003). Although the mean age of our sample of veterans was older (mean age of 65 versus 61 years for NHANES III) and predominantly male, the age and sex standardized prevalence of CKD remained high in our study sample, at 29 percent. The lower prevalence of CKD in NHANES III, compared with our study, may possibly be explained by a lower percent of individuals in NHANES III who had diabetes of long enough duration to develop kidney disease because (1) NHANES III excluded individuals with type 1 diabetes, and (2) NHANES III included individuals with previously undiagnosed type 2 diabetes.
The importance of validating methods to identify patients with CKD is highlighted by contrasting our study results with an earlier study that estimated the prevalence of CKD in veterans with diabetes using ICD-9-CM codes. Young et al. (2004) found the prevalence of CKD in the VA diabetic population to be 6 percent, which is much lower than the prevalence of CKD in our study. Therefore, case identification using only renal-related diagnosis codes in administrative records greatly underestimates the true prevalence of disease.
The high prevalence of CKD in our study sample underscores the need for more research on the quality of care and outcomes of kidney disease within populations of patients with diabetes. The failure to code the majority of CKD cases in patients with diabetes may indicate under-diagnosis. The insensitivity of serum creatinine tests to indicate CKD in older patients is evident in our data, as 43 percent of the patients with Stage 3 CKD had an index creatinine value less than 1.5 mg/dl (data not shown). To aid recognition of CKD in individual patients, the Work Group of K/DOQI has advocated routine laboratory reporting of eGFR with serum creatinine tests (NKF 2002).
Our study has limitations. If veterans with CKD are more likely to get tested for serum creatinine, our study sample may be enriched with individuals with CKD. Such a selection bias could inflate our estimates of the prevalence of CKD in the veteran population, but should not affect our conclusions regarding the insensitivity of diagnosis codes to identify individuals with CKD. Because our data did not include values for urine protein, we were unable to classify individuals with proteinuria and eGFR greater than 59 ml/min/1.73 m2 as having CKD. Therefore, our study may overestimate the sensitivity of diagnosis codes to identify individuals with CKD, while underestimating the specificity. Individuals covered by health care plans in addition to Medicare fee-for-service and VA during the year of the study may have had missing claims, possibly diminishing our ability to find encounters with a renal-related code. Our study sample has a high proportion of older, male patients. Age or gender bias in coding renal diagnoses might alter the sensitivity and specificity of our algorithm to identify younger individuals, or women with diabetes and CKD. However, the concomitant use of Medicare and VA claims records extends the generalizability of our diagnosis code algorithm to male individuals enrolled in either system.
In summary, CKD is a common comorbidity in older, adult patients with diabetes. The ICD-9-CM diagnosis codes in administrative records are an insensitive, although reasonably specific marker for individuals with comorbid CKD. Research focused on health care quality and outcomes in diabetes, or case-mix methodology that adjusts for excess risk imposed by CKD, should use eGFR rather than diagnosis codes to identify individuals with CKD.
Acknowledgments
The Department of Veterans Affairs, Veterans Health Administration, Veterans Affairs Medical Center at New Jersey supported the research reported in this article, in addition to grants from VA Medical Service Research and VA Grant IIR 04-205-2 (Dr. Miller and Dr. Pogach), VA Health Services Research Quality Enhancement Research Initiative (Dr. Pogach), and the VA HSR&D Center for Quality Improvement Research (Dr. Aron). The authors gratefully acknowledge the help of Susan Weerts in data management and analysis for this study. Dr. Kern was supported as a fellow in the VA National Quality Scholars Program.
REFERENCES
- AHA. Coding Clinic for ICD-9-CM: Official Guidelines for Coding and Reporting/American Hospital Association. Chicago: American Hospital Association; 2002. [Google Scholar]
- Anderson RJ, O'Brien M, MaWhinney S, VillaNueva CB, Moritz TE, Sethi GK, Henderson WG, Hammermeister KE, Grover FL, Shroyer AL. “Renal Failure Predisposes Patients to Adverse Outcome after Coronary Artery Bypass Surgery. VA Cooperative Study #5.”. Kidney International. 1999;55((3)):1057–62. doi: 10.1046/j.1523-1755.1999.0550031057.x. [DOI] [PubMed] [Google Scholar]
- Brancati FL, Whelton PK, Randall BL, Neaton JD, Stamler J, Klag MJ. “Risk of End-Stage Renal Disease in Diabetes Mellitus: A Prospective Cohort Study of Men Screened for MRFIT. Multiple Risk Factor Intervention Trial.”. Journal of the American Medical Association. 1997;278((23)):2069–74. [PubMed] [Google Scholar]
- Brenner BM, Cooper ME, deZeeuw D, Keane WF, Mitch WE, Parving HH, Remuzzi G, Snapinn SM, Zhan Z, Shahinfar S. “Effects of Losartan on Renal and Cardiovascular Outcomes in Patients with Type 2 Diabetes and Nephropathy.”. New England Journal of Medicine. 2001;345((12)):861–9. doi: 10.1056/NEJMoa011161. [DOI] [PubMed] [Google Scholar]
- Callahan M, Battleman DS, Christos P, Efimba M, Whitelaw G. “Economic Consequences of Renal Dysfunction among Cardiopulmonary Bypass Surgery Patients A Hospital-Based Perspective.”. Value Health. 2003;6((2)):137–43. doi: 10.1046/j.1524-4733.2003.00211.x. [DOI] [PubMed] [Google Scholar]
- Conlon PJ, Stafford-Smith M, White WD, Newman MF, King S, Winn MP, Landolfo K. “Acute Renal Failure following Cardiac Surgery.”. Nephrology, Dialysis, Transplantation. 1999;14((5)):1158–62. doi: 10.1093/ndt/14.5.1158. [DOI] [PubMed] [Google Scholar]
- Daley J. “Validity of Risk-Adjustment Methods.”. In: Iezzoni LI, editor. Risk Adjustment for Measuring Healthcare Outcomes. 2d Edition. Chicago: Health Administration Press; 1997. pp. 331–63. [Google Scholar]
- Fisher ES, Baron JA, Malenka DJ, Barrett J, Bubolz TA. “Overcoming Potential Pitfalls in the Use of Medicare Data for Epidemiologic Research.”. American Journal of Public Health. 1990;80((12)):1487–90. doi: 10.2105/ajph.80.12.1487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fisher ES, Whaley FS, Krushat WM, Malenka DJ, Fleming C, Baron JA, Hsia DC. “The Accuracy of Medicare's Hospital Claims Data: Progress Has Been Made, but Problems Remain.”. American Journal of Public Health. 1992;82((2)):243–8. doi: 10.2105/ajph.82.2.243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gaede P, Vedel P, Parving HH, Pedersen O. “Intensified Multifactorial Intervention in Patients with Type 2 Diabetes Mellitus and Microalbuminuria: The Steno Type 2 Randomised Study.”. Lancet. 1999;353((9153)):617–22. doi: 10.1016/S0140-6736(98)07368-1. [DOI] [PubMed] [Google Scholar]
- Gimeno-Orna JA, Lou-Arnal LM, Boned-Juliani B, Molinero-Herguedas E. “Mild Renal Insufficiency as a Cardiovascular Risk Factor in Non-Proteinuric Type II Diabetes.”. Diabetes Research and Clinical Practice. 2004;64((3)):191–9. doi: 10.1016/j.diabres.2003.10.018. [DOI] [PubMed] [Google Scholar]
- Go AS, Chertow GM, Fan D, McCulloch CE, Hsu CY. “Chronic Kidney Disease and the Risks of Death, Cardiovascular Events, and Hospitalization.”. New England Journal of Medicine. 2004;351((13)):1296–305. doi: 10.1056/NEJMoa041031. [DOI] [PubMed] [Google Scholar]
- Goldfield N, Villani J. “The Use of Administrative Data as the First Step in the Continuous Quality Improvement Process.”. American Journal of Medical Quality. 1996;11((1)):S35–8. [PubMed] [Google Scholar]
- Gruberg L, Mehran R, Dangas G, Mintz GS, Waksman R, Kent KM, Pichard AD, Satler LF, Wu H, Leon MB. “Acute Renal Failure Requiring Dialysis after Percutaneous Coronary Interventions.”. Catheterization and Cardiovascular Interventions. 2001;52((4)):409–16. doi: 10.1002/ccd.1093. [DOI] [PubMed] [Google Scholar]
- Harris MI, Flegal KM, Cowie CC, Eberhardt MS, Goldstein DE, Little RR, Wiedmeyer HM, Byrd-Holt DD. “Prevalence of Diabetes, Impaired Fasting Glucose, and Impaired Glucose Tolerance in U.S. Adults. The Third National Health and Nutrition Examination Survey, 1988–1994.”. Diabetes Care. 1998;21((4)):518–24. doi: 10.2337/diacare.21.4.518. [DOI] [PubMed] [Google Scholar]
- Hebert PL, Geiss LS, Tierney EF, Engelgau MM, Yawn BP, McBean AM. “Identifying Persons with Diabetes Using Medicare Claims Data.”. American Journal of Medical Quality. 1999;14((6)):270–7. doi: 10.1177/106286069901400607. [DOI] [PubMed] [Google Scholar]
- Iezzoni LI. “Risk Adjustment and Current Health Policy Initiatives.”. In: Iezzoni LI, editor. Risk Adjustment for Measuring Healthcare Outcomes. 2d edition. Chicago: Health Administration Press; 1997. pp. 517–95. [Google Scholar]
- Keith DS, Nichols GA, Gullion CM, Brown JB, Smith DH. “Longitudinal Follow-up and Outcomes among a Population with Chronic Kidney Disease in a Large Managed Care Organization.”. Archives of Internal Medicine. 2004;164((6)):659–63. doi: 10.1001/archinte.164.6.659. [DOI] [PubMed] [Google Scholar]
- Kramer HJ, Nguyen QD, Curhan G, Hsu CY. “Renal Insufficiency in the Absence of Albuminuria and Retinopathy among Adults with Type 2 Diabetes Mellitus.”. Journal of the American Medical Association. 2003;289((24)):3273–7. doi: 10.1001/jama.289.24.3273. [DOI] [PubMed] [Google Scholar]
- Lamb EJ, Webb MC, Simpson DE, Coakley AJ, Newman DJ, O'Riordan SE. “Estimation of Glomerular Filtration Rate in Older Patients with Chronic Renal Insufficiency: Is the Modification of Diet in Renal Disease Formula an Improvement?”. Journal of the American Geriatrics Society. 2003;51((7)):1012–7. doi: 10.1046/j.1365-2389.2003.51330.x. [DOI] [PubMed] [Google Scholar]
- Langston RD, Presley R, Flanders WD, McClellan WM. “Renal Insufficiency and Anemia Are Independent Risk Factors for Death among Patients with Acute Myocardial Infarction.”. Kidney International. 2003;64((4)):1398–405. doi: 10.1046/j.1523-1755.2003.00200.x. [DOI] [PubMed] [Google Scholar]
- Levey AS, Bosch JP, Lewis JB, Greene T, Rogers N, Roth D. “A More Accurate Method to Estimate Glomerular Filtration Rate from Serum Creatinine: A New Prediction Equation. Modification of Diet in Renal Disease Study Group.”. Annals of Internal Medicine. 1999;130((6)):461–70. doi: 10.7326/0003-4819-130-6-199903160-00002. [DOI] [PubMed] [Google Scholar]
- Levey AS, Coresh J, Balk E, Kausz AT, Levin A, Steffes MW, Hogg RJ, Perrone RD, Lau J, Eknoyan G. “National Kidney Foundation Practice Guidelines for Chronic Kidney Disease: Evaluation, Classification, and Stratification.”. Annals of Internal Medicine. 2003;139((2)):137–47. doi: 10.7326/0003-4819-139-2-200307150-00013. [DOI] [PubMed] [Google Scholar]
- Lewis EJ, Hunsicker LG, Clarke WR, Berl T, Pohl MA, Lewis JB, Ritz E, Atkins RC, Rohde R, Raz I. “Renoprotective Effect of the Angiotensin-Receptor Antagonist Irbesartan in Patients with Nephropathy Due to Type 2 Diabetes.”. New England Journal of Medicine. 2001;345((12)):851–60. doi: 10.1056/NEJMoa011303. [DOI] [PubMed] [Google Scholar]
- Lewis EJ, Hunsicker LG, Bain RP, Rohde RD. “The Effect of Angiotensin-Converting-Enzyme Inhibition on Diabetic Nephropathy. The Collaborative Study Group.”. New England Journal of Medicine. 1993;329((20)):1456–62. doi: 10.1056/NEJM199311113292004. [DOI] [PubMed] [Google Scholar]
- Mack MJ, Brown PP, Kugelmass AD, Battaglia SL, Tarkington LG, Simon AW, Culler SD, Becker ER. “Current Status and Outcomes of Coronary Revascularization 1999 to 2002: 148,396 Surgical and Percutaneous Procedures.”. Annals of Thoracic Surgery. 2004;77((3)):761–6. doi: 10.1016/j.athoracsur.2003.06.019. discussion 66–8. [DOI] [PubMed] [Google Scholar]
- Mangos GJ, Brown MA, Chan WY, Horton D, Trew P, Whitworth JA. “Acute Renal Failure following Cardiac Surgery: Incidence, Outcomes and Risk Factors.”. Australian and New Zealand Journal of Medicine. 1995;25((4)):284–9. doi: 10.1111/j.1445-5994.1995.tb01891.x. [DOI] [PubMed] [Google Scholar]
- Mann JF, Gerstein HC, Pogue J, Bosch J, Yusuf S. “Renal Insufficiency as a Predictor of Cardiovascular Outcomes and the Impact of Ramipril: The Hope Randomized Trial.”. Annals of Internal Medicine. 2001;134((8)):629–36. doi: 10.7326/0003-4819-134-8-200104170-00007. [DOI] [PubMed] [Google Scholar]
- McCullough PA, Wolyn R, Rocher LL, Levin RN, O'Neill WW. “Acute Renal Failure after Coronary Intervention: Incidence, Risk Factors, and Relationship to Mortality.”. American Journal of Medicine. 1997;103((5)):368–75. doi: 10.1016/s0002-9343(97)00150-2. [DOI] [PubMed] [Google Scholar]
- Medicode . International Classification of Diseases, Ninth Revision, Clinical Modification. Physican ICD-9-CM Coding Manual Physician ICD-9-CM Coding Manual. 5th Edition. 1 and 2. Salt Lake City, UT: Medicode Inc; 1999. Physician ICD-9-CM Coding Manual. [Google Scholar]
- Miettinen H, Haffner SM, Lehto S, Ronnemaa T, Pyorala K, Laakso M. “Proteinuria Predicts Stroke and Other Atherosclerotic Vascular Disease Events in Nondiabetic and Non-Insulin-Dependent Diabetic Subjects.”. Stroke. 1996;27((11)):2033–9. doi: 10.1161/01.str.27.11.2033. [DOI] [PubMed] [Google Scholar]
- Miller DR, Safford MM, Pogach LM. “Who Has Diabetes? Best Estimates of Diabetes Prevalence in the Department of Veterans Affairs Based on Computerized Patient Data.”. Diabetes Care. 2004;27((2 suppl)):B10–21. doi: 10.2337/diacare.27.suppl_2.b10. [DOI] [PubMed] [Google Scholar]
- Mitchell JB, Bubolz T, Paul JE, Pashos CL, Escarce JJ, Muhlbaier LH, Wiesman JM, Young WW, Epstein RS, Javitt JC. “Using Medicare Claims for Outcomes Research.”. Medical Care. 1994;32((7 suppl)):JS38–51. [PubMed] [Google Scholar]
- NKF. “K/DOQI Clinical Practice Guidelines for Chronic Kidney Disease: Evaluation, Classification, and Stratification.”. American Journal of Kidney Diseases. 2002;39((2 suppl 1)):S1–266. [PubMed] [Google Scholar]
- Pleis JR, Coles R. “Summary Health Statistics for U.S. Adults: National Health Interview Survey, 1999.”. Vital Health Statistics. 2003;10((212)):1–145. [PubMed] [Google Scholar]
- Rosamond WD, Chambless LE, Sorlie PD, Bell EM, Weitzman S, Smith JC, Folsom AR. “Trends in the Sensitivity, Positive Predictive Value, False-Positive Rate, and Comparability Ratio of Hospital Discharge Diagnosis Codes for Acute Myocardial Infarction in Four US Communities, 1987–2000.”. American Journal of Epidemiology. 2004;160((12)):1137–46. doi: 10.1093/aje/kwh341. [DOI] [PubMed] [Google Scholar]
- Shlipak MG, Heidenreich PA, Noguchi H, Chertow GM, Browner WS, McClellan MB. “Association of Renal Insufficiency with Treatment and Outcomes after Myocardial Infarction in Elderly Patients.”. Annals of Internal Medicine. 2002;137((7)):555–62. doi: 10.7326/0003-4819-137-7-200210010-00006. [DOI] [PubMed] [Google Scholar]
- Szczech LA, Best PJ, Crowley E, Brooks MM, Berger PB, Bittner V, Gersh BJ, Jones R, Califf RM, Ting HH, Whitlow PJ, Detre KM, Holmes D. “Outcomes of Patients with Chronic Renal Insufficiency in the Bypass Angioplasty Revascularization Investigation.”. Circulation. 2002;105((19)):2253–8. doi: 10.1161/01.cir.0000016051.33225.33. [DOI] [PubMed] [Google Scholar]
- Thakar CV, Worley S, Arrigain S, Yared JP, Paganini EP. “Influence of Renal Dysfunction on Mortality after Cardiac Surgery: Modifying Effect of Preoperative Renal Function.”. Kidney International. 2005;67((3)):1112–9. doi: 10.1111/j.1523-1755.2005.00177.x. [DOI] [PubMed] [Google Scholar]
- Tonelli M, Bohm C, Pandeya S, Gill J, Levin A, Kiberd BA. “Cardiac Risk Factors and the Use of Cardioprotective Medications in Patients with Chronic Renal Insufficiency.”. American Journal of Kidney Diseases. 2001;37((3)):484–9. [PubMed] [Google Scholar]
- USRDS. Bethesda, MD: National Institutes of Health, National Institute for Diabetes, Digestive, and Kidney Diseases; 2004. U.S. Renal Data System 2004 Annual Data Report. Atlas of End-Stage Renal Disease in the United States, (Table A.11) [Google Scholar]
- Weiner DE, Tighiouart H, Amin MG, Stark PC, MacLeod B, Griffith JL, Salem DN, Levey AS, Sarnak MJ. “Chronic Kidney Disease as a Risk Factor for Cardiovascular Disease and All-Cause Mortality: A Pooled Analysis of Community-Based Studies.”. Journal of the American Society of Nephrology. 2004;15((5)):1307–15. doi: 10.1097/01.asn.0000123691.46138.e2. [DOI] [PubMed] [Google Scholar]
- Wright RS, Reeder GS, Herzog CA, Albright RC, Williams BA, Dvorak DL, Miller WL, Murphy JG, Kopecky SL, Jaffe AS. “Acute Myocardial Infarction and Renal Dysfunction: A High-Risk Combination.”. Annals of Internal Medicine. 2002;137((7)):563–70. doi: 10.7326/0003-4819-137-7-200210010-00007. [DOI] [PubMed] [Google Scholar]
- Young BA, Pugh JA, Maynard C, Reiber G. “Diabetes and Renal Disease in Veterans.”. Diabetes Care. 2004;27((2 suppl)):B45–9. doi: 10.2337/diacare.27.suppl_2.b45. [DOI] [PubMed] [Google Scholar]