Injection therapy for subacute and chronic low‐back pain (original) (raw)
Abstract
Background
The effectiveness of injection therapy for low‐back pain is still debatable. Heterogeneity of target tissue, pharmacological agent and dosage generally found in randomized controlled trials (RCTs) points to the need for clinically valid comparisons in a literature synthesis.
Objectives
To determine if injection therapy is more effective than placebo or other treatments for patients with subacute or chronic low‐back pain.
Search methods
We updated the search of the earlier systematic review and searched the Cochrane Central Register of Controlled Trials, MEDLINE and EMBASE databases from January 1999 to March 2007 for relevant trials reported in English, French, German, Dutch and Nordic languages. We also screened references from trials identified.
Selection criteria
RCTs on the effects of injection therapy involving epidural, facet or local sites for subacute or chronic low‐back pain were included. Studies which compared the effects of intradiscal injections, prolotherapy or Ozone therapy with other treatments, were excluded unless injection therapy with another pharmaceutical agent (no placebo treatment) was part of one of the treatment arms. Studies about injections in sacroiliac joints and studies evaluating the effects of epidural steroids for radicular pain were also excluded.
Data collection and analysis
Two review authors independently assessed the quality of the trials. If study data were clinically and statistically too heterogeneous to perform a meta‐analysis, we used a best evidence synthesis to summarize the results. The evidence was classified into five levels (strong, moderate, limited, conflicting or no evidence), taking into account the methodological quality of the studies.
Main results
18 trials (1179 participants) were included in this updated review. The injection sites varied from epidural sites and facet joints (i.e. intra‐articular injections, peri‐articular injections and nerve blocks) to local sites (i.e. tender‐ and trigger points). The drugs that were studied consisted of corticosteroids, local anesthetics and a variety of other drugs. The methodological quality of the trials was limited with 10 out of 18 trials rated as having a high methodological quality. Statistical pooling was not possible due to clinical heterogeneity in the trials. Overall, the results indicated that there is no strong evidence for or against the use of any type of injection therapy.
Authors' conclusions
There is insufficient evidence to support the use of injection therapy in subacute and chronic low‐back pain. However, it cannot be ruled out that specific subgroups of patients may respond to a specific type of injection therapy.
Plain language summary
Injection therapy for subacute and chronic low‐back pain
Injection therapy is one of many treatments available for patients with subacute (longer than six weeks) and chronic (longer than 12 weeks) low‐back pain. Where the injection is given, what drug is used and why the injection is given can all vary.
The injection can be given into different parts of the spine (the space between the vertebrae, around the nerve roots, or into the disc), ligaments, muscles or trigger points (spots in the muscles that when pressed firmly will produce pain). Drugs that reduce swelling (corticosteroids, non‐steroidal anti‐inflammatory (NSAIDs)) and pain (morphine, anaesthetics) are used. Injection therapy can be used for individuals with low‐back pain with or without pain and other symptoms in the leg.
A number of electronic databases of healthcare articles were searched up to March 2007. This search identified 18 randomized controlled trials (RCTs; 1179 participants) that looked at injections with a variety of drugs compared to a placebo drug or other drugs. The injections were given into the epidural space (between the vertebrae of the back and outside the coverings that surround the spinal cord), the facet joints (the joints of two vertebrae), or tender spots in the ligaments or muscles.
The review authors rated ten of the 18 RCTs as having a low risk of bias in the way the trials were conducted and reported. They were unable to statistically pool the results because the injection sites, drugs used and outcomes measured were too varied. Only five of the 18 trials reported significant results in favour of one of the treatment arms. The reviewer authors considered the likely treatment benefits to be worth the potential harms in only two studies. In nine out of the 18 studies, side effects such as headache, dizziness, transient local pain, tingling and numbness and nausea were reported in small numbers of patients. The use of morphine was more frequently associated with itching, nausea and vomiting. Rare but more serious complications of injection therapy have been mentioned in the literature, such as cauda equina syndrome, septic facet joint arthritis, discitis, paraplegia, paraspinal abscesses. Although the absolute frequency of these complications may be rare, these risks should be taken into consideration. Based on these results, the review authors concluded that there is no strong evidence for or against the use of any type of injection therapy for individuals with subacute or chronic low‐back pain.
Background
Numerous treatments have been promoted as useful to reduce the individual and socio‐economic burden of low‐back pain, but many controversies still exist on the effectiveness of the available treatment options (Weinstein 2004; Airaksinen 2006). Injection therapy is one of the treatments for patients with subacute and chronic low‐back pain that needs careful evaluation with respect to its effectiveness for short‐ and long‐term pain relief. In order to investigate the effects of injection therapy for low‐back pain in a systematic review, it should be realized that the content of this treatment method, as studied in randomized controlled trials (RCTs), may show a large degree of variation. The same holds for daily clinical practice, which underlines the need to make clinically valid comparisons of injection therapy interventions. The first source of variation in the content of injection therapy is the location of the injection and the tissue that is targeted for injection. The injection can be given into the facet joints, the epidural space, the spinal nerve root, the intervertebral disc, the lumbar sympathetic chain, sacro‐iliac joints and also into local ligaments, muscles or trigger points (Airaksinen 2006). Second, the pharmacological agent that is used for the injection varies from different types of corticosteroids aimed at reducing inflammation and anaesthetics aimed at pain relief, to a variety of other drugs, all with potentially different dosages. Third, the indications for the injection therapy may also be different. Injection therapy can be applied in cases of acute, subacute or chronic low‐back pain with or without signs of nerve root compression. For obvious reasons, the specific characteristics of the injection therapy with respect to target tissue, pharmacological agent and dosage depends on the presumed pathophysiological mechanisms leading to pain. In some cases, injections are used for diagnostic purposes rather than treatment. Injections into facet joints, for example, have been presented as a diagnostic test for the so‐called lumbar facet joint syndrome (Mooney 1976; Mooney 1987). Furthermore, in some RCTs, injection therapy is only one part of a multimodal treatment package. These issues may challenge a valid assessment and summation of the effects of injection therapy in patients with low‐back pain.
The present systematic review is an update of an earlier Cochrane review on the effectiveness of injection therapy for subacute and chronic low‐back pain (Nelemans 2000). Many of the 21 studies that were included in this prior systematic review were rated as being of low methodological quality and in 11 of the 21 studies, injection was compared with placebo. The authors concluded that convincing evidence for the effectiveness of injection therapy was still lacking and recommended large well‐designed trials in this field, preferably placebo‐controlled (Nelemans 2000).
Since Nelemans 2000 et al was published, several new RCTs on the effects of injections for low‐back pain were published and the method guidelines for systematic reviews in the field of low‐back pain were updated (van Tulder 2003). In order to reduce the clinical heterogeneity identified in the earlier review, several more specific injection therapies have been covered in separate Cochrane reviews. Therefore, the present systematic review does not include the effects of intradiscal therapy (Niemisto 2003), prolotherapy (i.e. injection of sclerosants) (Dagenais 2007) or epidural steroids for radiculopathy due to disc herniation (registered as a title for an upcoming Cochrane review). Although some overlap cannot always be prevented since radiculopathy is not an exclusive diagnosis, we aim to exclude studies dealing specifically with epidural steroids for this particular study population from the present update. Nelemans 2000 et al had included studies on epidural steroids for radiculopathy in the earlier review. This update further differs from the previous review with regard to the internal validity criteria that were used. We rated the studies according to the eleven internal validity items recommended by van Tulder 2003 whereas Nelemans 2000 et al used a fifteen item list according to ter Riet 1990 et al. In addition, we also added an assessment of clinical relevance to the present review.
Because many studies in the review by Nelemans 2000 et al were clinically heterogeneous, a major concern for the present update, as stated before, was to come up with comparisons that made sense from a clinical perspective. Therefore, we chose to classify studies first according to the target tissue of the injection and second according to the therapeutic agent that was used. If possible, these strata were further subdivided into placebo‐controlled trials and those that compared injections with other treatments.
Objectives
The objective of this systematic review was to asses the effectiveness of injection therapy for patients with subacute or chronic low‐back pain.
Methods
Criteria for considering studies for this review
Types of studies
Only RCTs were included. Non‐randomized studies were excluded.
Types of participants
Patients aged from 18 to 70 years were included if they had low‐back pain with symptoms persisting for at least one month. Subacute low‐back pain was defined as lasting for four weeks or longer and chronic as lasting for 12 weeks or longer.
Types of interventions
The treatment had to include injection therapy for pain relief. The injection therapy was studied at three different injection sites: facet joints, epidural sites, and local sites. Studies that compared the effects of epidural steroids for radicular pain, intradiscal injections, prolotherapy or Ozone therapy with other treatments were excluded unless injection therapy with another pharmaceutical agent (no placebo treatment) was part of one of the treatment arms. Studies about injections into sacroiliac joints and studies in which the drugs were administered by means of a catheter and not directly by means of an injection were also excluded. Additional treatments were allowed.
Types of outcome measures
Pain was considered to be the most important outcome measure. In order to be included for the present review, studies should at least have incorporated this outcome measure. This could be expressed as the percentage of patients with pain relief or as mean improvement on a continuous scale. Other important outcome measures were: a global measure of improvement (e.g. overall improvement, proportion of patients recovered, subjective improvement of symptoms), back‐specific disability (expressed on a back‐specific index, such as the Roland Disability Questionnaire or the Oswestry Disability Index), generic health status or well‐being, disability for work, and patient satisfaction (Bombardier 2000; van Tulder 2003). These outcomes were evaluated on both a short‐term (less than six weeks) and long‐term basis (longer than six weeks). Side effects were also considered.
Search methods for identification of studies
RCTs published in English, French, German, Dutch and Nordic languages were included. The literature searches carried out by Nelemans et al were updated and reiterated in the same databases for the period from 1999 to March 2007 (Nelemans 2000). MEDLINE and EMBASE searches were based on the search strategies recommended by the Cochrane Back Review Group (van Tulder 2003). The Cochrane CENTRAL database was also screened for relevant RCTs. In addition, citation tracking of the studies retrieved by the search was performed until no new studies were found. See Appendix 1 for the PubMed and EMBASE search strategies.
Data collection and analysis
Study selection and data extraction
For the 2007 update, two authors (JBS, RdB) independently selected new studies, assessed the methodological quality, and extracted the data (using a standardized form). This was conducted in the same way, described in the following sections, as in the previously published systematic review (Nelemans 2000). A consensus method was used to solve disagreements about the selection of RCTs, and a third author (PN) was consulted if disagreement persisted. First, the two authors screened the titles, abstracts and keywords of all references identified by the literature search to determine if they met the inclusion criteria. Second, the full text of the article was retrieved for studies for which it was unclear from the screening procedure whether or not the study had to be included.
Methodological quality assessment
The methodological quality of the RCTs was independently assessed by two authors (JBS, RdB), using the methodological criteria recommended by the Cochrane Back Review Group (van Tulder 2003). A consensus method was used to resolve disagreements and a third author was consulted if disagreements persisted (PN). The items were scored as either positive (+), negative (‐) or unclear (?). The criteria used, and their operationalization are listed in Table 8.
1. Definitions of internal validity criteria ‐ risks of bias.
| criteria/definition |
|---|
| Method of randomisation adequate: A random (unpredictable) assignment sequence. Examples of adequate methods are computer generated random number table and use of sealed opaque envelopes. Methods of allocation using date of birth, date of admission, hospital numbers, or alternation should not be regarded as appropriate. |
| Concealment of treatment allocation: Assignment generated by an independent person not responsible for determining the eligibility of the patients; this person has no information about the persons included in the trial and has no influence on the assignment sequence or on the decision about eligibility of the patient. |
| Blinding of patients: The reviewer determines if there was enough information about the blinding of the care provider to score a yes. |
| Blinding of care providers: The reviewer determines if there was enough information about the blinding of the patient to score a yes. |
| Blinding of outcome assessors: The reviewer determines if there was enough information about the blinding of the outcome assessor to score a yes. |
| Drop‐out rate described and acceptable: The number of participants who were included in the study but did not complete the observation period or were not included in the analysis must be described and reasons given. If the percentage of drop‐outs does not exceed 20% for short‐term follow‐up and 30% for long‐term follow‐up and does not lead to substantial bias, a yes is scored. (N.B., these percentages are arbitrary, not supported by literature). |
| Intention‐to‐treat analyses: All randomized patients are reported/analyzed in the group to which they were allocated by randomization for the most important moments of effect measurement (minus missing values) irrespective of non‐compliance and co‐interventions. |
| Similarity of baseline characteristics: In order to receive a yes, groups have to be similar at baseline regarding demographic factors, duration and severity of complaints and value of main outcome measure(s). |
| Co‐interventions avoided or similar: Co‐interventions should either be avoided in the trial design or comparable between the index and control groups. |
| Compliance acceptable: The reviewer determines if the compliance to the interventions is acceptable, based on the reported intensity, duration, number, and frequency of sessions for both the index intervention(s) and control intervention(s). |
| Timing outcome assessments similar: Timing of outcome assessment be identical for all intervention groups and for all important outcome assessments. |
Clinical relevance
Two authors (JBS and RdB) independently assessed the clinical relevance of the included studies according to several questions which were recommended by the Cochrane Back Review Group (van Tulder 2003). Each question was scored positive (+) if the clinical relevance item was met, negative (‐) if the item was not met and unclear (?) if data were not available to answer the question. We considered a 20% improvement in pain scores (Salaffi 2004) and a 10% improvement in functioning outcomes (Bombardier 2000) to be clinically important. See Table 9 for the questions used.
2. Clinical Relevance questions.
| questions |
|---|
| A) Are the patients described in detail so that you can decide whether they are comparable to those that you see in your practice? B) Are the interventions and treatment settings described well enough so that you can provide the same for your patients? C) Were all clinically relevant outcomes measured and reported? D) Is the size of the effect clinically important? We considered a 20 % improvement in pain scores (Salaffi 2004) and a 10% improvement in functioning outcomes (Bombardier 2000) to be clinically important. E) Are the likely treatment benefits worth the potential harms? |
Analysis
The results of each RCT were plotted as point estimates with corresponding 95% confidence intervals (95% CI) as far as possible. The clinical homogeneity was evaluated by exploring the differences between the RCTs, taking into consideration the study population, the injection sites, the content of the injection therapy with regard to medication and dosage, reference treatments, timing of follow‐up measurements and measurement instruments. Furthermore, a distinction was made between studies that compared injection with a placebo injection and studies that compared two or more active agents (Nelemans 2000). Attempts were made to statistically pool the data of homogeneous studies for the outcome measures pain, overall improvement and disability for short and long‐term follow‐up. If subgroups of studies were considered to be clinically homogeneous, the statistical homogeneity was formally tested. If studies were considered clinically heterogeneous and/or studies did not report their results in a way that enabled us to perform statistical pooling (for example, for continuous data, means were presented but no standard deviations) we refrained from following a formal meta‐analytic approach. In stead of this, we summarized the results according to a rating system with five levels of evidence (best evidence synthesis), based on the quality and the outcome of the studies (van Tulder 2003). The following levels of evidence were distinguished:
- Strong evidence ‐ Consistent findings among multiple high quality RCTs;
- Moderate evidence ‐ Consistent findings among multiple low quality RCTs or one high quality RCT;
- Limited evidence ‐ one low quality RCT;
- Conflicting evidence ‐ inconsistent findings among multiple trials;
- No evidence ‐ no RCTs.
High quality studies were defined as RCT which fulfilled six or more of the internal validity criteria. The results were considered to be consistent if 75% of the studies showed results in the same direction.
Results
Description of studies
Study selection
The original 1998 search yielded 40 relevant papers (Nelemans 2000). After detailed reading, 19 studies were excluded because they included acute patients (pain for less than one month), they did not describe the duration of the reported symptoms, they appeared to be non‐randomized or the anesthetic agent was injected into the wound during surgery (Nelemans 2000). From the remaining 19 studies that were covered by Nelemans 2000 et al, nine studies were excluded from the present update due to the modified inclusion criteria. As stated earlier, studies on the effects of corticosteroids for radiculopathy will be summarized in another Cochrane review. This means that four studies which investigated the effects of corticosteroids in patients with radiculopathy in comparison with a placebo injection (Cuckler 1985; Mathews 1987; Bush 1991; Rogers 1992) were excluded. Furthermore, one study dealt with intradiscal injection (Simmons 1992), one study dealt with prolotherapy (Ongley 1987), both of which are covered in another Cochrane review, and another study included patients who had cervical symptoms and did not present separate results for the lower back (Hameroff 1981). Two studies were excluded because the drugs were administered by means of a catheter (Dallas 1987; Glynn 1988) and not directly by means of an injection. In addition, the study population of one of them consisted of patients with chronic pain with only a small subgroup of patients labeled as having low‐back pain (Glynn 1988). Consequently, from the previous search by Nelemans 2000 et al, only 10 studies were incorporated into the present update.
The March 2007 literature search update resulted in the identification of 11 new references from MEDLINE, eight references from EMBASE and one reference from additional reference checking. Four references were included in both the MEDLINE and EMBASE search results, leaving 16 references potentially relevant for the purpose of the review. After careful reading of the papers, the two reviewers (JBS and RdB) decided that eight studies had to be excluded. Two of them compared two different needling techniques (Stojanovic 2005) or injection sites (Pneumaticos 2006). One study compared radiofrequency denervation with an injection of a small amount of lidocaine which was considered a sham treatment (Oh 2004). Other studies dealt with patients with radiculopathy (sciatic nerve pain) (Bonetti 2005), with acute patients (Tuzun 2003), patients with osteoporosis (Ofluoglu 2007), or made use of epidural catheterization (Fredman 1999). One study appeared not to be a RCT (Ney 2006). Together with 10 studies from the earlier review, we ended up with 18 studies that were considered relevant for the purpose of this review.
Characteristics of included studies
The number of participants in the included studies varied from 16 to 206 patients. Only three studies included more than 100 participants (Lilius 1990; Carette 1991; Aldrete 2003). The study population in most of the included studies consisted of patients who had low‐back pain with or without sciatica. Two studies dealt only with post‐laminectomy patients (Rocco 1989; Aldrete 2003) and in one of these studies, some of the patients also had radicular symptoms (Aldrete 2003). In two other studies, populations were mixed: patients were included with low‐back pain and/or sciatica whether or not combined with neurological signs (Beliveau 1971; Mauro 2000). In some studies, participation was limited to patients who responded positively to a facet block with an anesthetic (Carette 1991; Manchikanti 2001) or to patients with radiologically confirmed facet joint osteoarthritis (Fuchs 2005). The injection sites of the studies included can be subdivided into epidural sites (Beliveau 1971; Breivik 1976; Rocco 1989; Serrao 1992; Aldrete 2003; Lierz 2004; Takada 2005), facet joints (Lilius 1990; Carette 1991; Marks 1992; Revel 1999; Mayer 2004; Fuchs 2005) and local sites (Sonne 1985; Garvey 1989; Collee 1991; Mauro 2000). Facet injections consisted of intra‐articular injections (Lilius 1990; Carette 1991; Marks 1992; Revel 1999; Mayer 2004; Fuchs 2005), peri‐articular injections (Lilius 1990; Revel 1999) or nerve blocks (Marks 1992; Manchikanti 2001). Local injections sites varied from tender‐ and trigger points (Garvey 1989; Collee 1991) to muscles (Mauro 2000) and ligaments (Sonne 1985). The drugs that were studied were mostly corticosteroids (i.e. methylprednisolone, triamcinolone and prednisolone) (Beliveau 1971; Breivik 1976; Sonne 1985; Garvey 1989; Lilius 1990; Rocco 1989; Carette 1991; Marks 1992; Aldrete 2003; Mayer 2004; Fuchs 2005) or local anesthetics (i.e. procaine, lidocaine, bupivacaine, lignocaine and ropivacaine) (Collee 1991;Revel 1999; Lierz 2004; Takada 2005). Other drugs that were studied were non‐steroidal anti‐inflammatories (NSAIDs) (Aldrete 2003), sodium hyaluronate (Fuchs 2005), Sarapin (i.e. a suspension of powdered pitcher plant in alkaline solution) (Manchikanti 2001), Vitamin B12 (Mauro 2000), morphine (Rocco 1989) and Midazolam (Serrao 1992). Eight studies appeared to be placebo‐controlled (Beliveau 1971; Breivik 1976; Sonne 1985; Garvey 1989; Lilius 1990; Carette 1991; Collee 1991; Mauro 2000) and 10 to be studies that compared injections with other treatments (Rocco 1989; Marks 1992; Serrao 1992; Revel 1999; Manchikanti 2001; Aldrete 2003; Lierz 2004; Mayer 2004; Fuchs 2005; Takada 2005). Among the placebo‐controlled studies, three used an anesthetic (i.e. procaine, bupivacaine and lidocaine) as part of their reference injections (Beliveau 1971; Breivik 1976; Garvey 1989). Nevertheless, we still considered these studies placebo‐controlled since the experimental treatments in these studies consisted of corticosteroids with a similar amount of anesthetic added to the solution as in the reference treatment. Anesthetics in general have a relatively short duration of action and from a pharmacological standpoint it seems unlikely that they will result in lasting pain relief (Nelemans 2000).
In two studies the authors declared they had received their drugs from commercial companies (Breivik 1976; Carette 1991). The authors of three other studies explicitly reported that they had no financial conflict of interest (Fuchs 2005; Lilius 1990; Aldrete 2003). The authors of the other studies provided no explicit information in their publication about funding or eventual conflicts of interest.
Risk of bias in included studies
For the assessment of the methodological quality of the trials, we combined the information from all papers reporting on the same trial. The two reviewers disagreed on 72 of 198 items (36%) scored. After discussing the results, they reached consensus on the scoring of all items. Therefore, the assistance of a third reviewer was not needed. Nine of the 18 studies included met six or more internal validity criteria and were therefore considered to be at an acceptable level of methodological quality (Breivik 1976; Garvey 1989; Rocco 1989; Carette 1991; Collee 1991; Marks 1992; Revel 1999; Lierz 2004; Takada 2005). The most prevalent shortcomings were: a lack of clarity regarding the concealment of random treatment allocation (N = 17), no reporting of co‐interventions (N = 14) and no reporting of an intention‐to‐treat analysis (N = 13). The internal validity criteria that were most frequently met were: the acceptability of the level of compliance of patients to the treatment protocol (N = 18), the description and acceptability of the drop‐out rate (N = 14) and the similarity of the timing of the outcome assessments (N = 13). See details in Figure 1.
1.

Summary of risks of bias
Effects of interventions
Feasibility of statistical pooling
As stated in the method section, statistical pooling was only considered if subgroups of studies were clinically homogeneous and the authors provided sufficient information on study characteristics, outcome measures and study results. After reviewing the study characteristics of the studies included, only two subgroups of two studies seemed to be sufficiently clinically homogeneous to perform statistical pooling. Beliveau 1971 et al and Breivik 1976 et al compared the effect of epidural corticosteroid injections to placebo injections and looked at short‐term results of pain relief, measured on a subjective dichotomous scale. However, in the study by Breivik 1976 et al, the patients who were allocated to a particular treatment and who did not improve were also treated with injections of the alternative group. The timing of the outcome measurements in this study was not clear either. Therefore, we thought these two studies were too heterogeneous to pool their results. Carette 1991 et al and Lilius 1990 et al compared the effects of facet joint injections with corticosteroids to placebo injections (Lilius 1990; Carette 1991). However, the study by Carette 1991 et al used a dichotomous outcome for pain improvement, whereas Lilius 1990 et al reported mean pain scores and no proportions of patients with pain relief. Therefore, it was not possible to pool the results of these two studies. Consequently, instead of statistical pooling, we had to perform a best‐evidence synthesis for both subgroups of studies.
Effectiveness of injection therapy
1. Epidural injections
Epidural corticosteroids versus placebo injections
Two studies compared the short term effects of epidural corticosteroid injections to placebo injections (Beliveau 1971; Breivik 1976). One of them was rated as a high quality study (Breivik 1976) and the other one was rated as a low quality study (Beliveau 1971). These two studies did not show a significant result for pain relief or other outcomes in favour of the corticosteroids group.
There is limited evidence that epidural corticosteroid injections are not significantly different from placebo injections for general improvement in the short term (1 trial; 48 patients). There is moderate for pain relief and limited evidence for work disability that epidural corticosteroid injections are not significantly different from placebo injections in the short term (1 trial; 35 patients). Back‐specific disability, generic health status and patient satisfaction were not measured.
Epidural corticosteroid injections versus other treatments
Three studies compared the effects of epidural corticosteroid injections to other treatments. The low quality study by Aldrete 2003 et al compared corticosteroids to NSAIDs (i.e. indomethacin) and showed no significant differences in short‐term pain progress and physical activity in post‐laminectomy patients. In the low quality study by Serrao 1992 et al, corticosteroids were combined with dextrose solution and compared to an intrathecal benzodiazepine (i.e. Midazolam) combined with a dextrose solution. No significant differences between the groups were reported for pain relief at two weeks and two months, or general improvement at two months. In a high quality study, Rocco 1989 et al compared corticosteroids to morphine and to a combination of corticosteroids and morphine in post‐laminectomy patients but found no significant differences in pain relief at one or six months.
There is limited evidence that the effect of epidural corticosteroid injections are not significantly different from NSAIDs for pain relief in the short term in post‐laminectomy patients (1 trial; 206 patients), benzodiazepine for pain relief and general improvement both in the short and intermediate term (1 trial; 28 patients), and from morphine eventually combined with corticosteroids for pain relief in the short and intermediate term in post‐laminectomy patients (1 trial; 22 patients). Back‐specific disability, generic health status, disability for work and patient satisfaction were not measured.
Epidural injections with local anaesthetics versus other treatments
In a high quality study, Lierz 2004 et al compared the effects of epidural blocks with ropivacaine to epidural blocks with bupivacaine. The purpose of these blocks with local anaesthesia was to provide good analgesia to allow active physiotherapy. There were no significant differences found between the groups in post‐treatment analgesia. In a low quality randomized cross‐over study, Takada 2005 et al also investigated the effects of epidural blocks with ropivacaine of two different dosages and epidural blocks of bupivacaine. No significant differences in pain scores were found after the different epidural blockades.
There is moderate evidence (2 trials; 56 patients) that there is no significant difference in the amount of analgesia provided between epidural blocks with ropivacaine and epidural blocks with bupivacaine. General improvement, back‐specific disability, generic health status, disability for work and patient satisfaction were not measured.
2. Facet joint injections
Facet joint injections with corticosteroids versus placebo injections
One high quality study and one low quality study compared the effects of facet joint injections with corticosteroids to placebo injections. In their high quality study, Carette 1991 et al pre‐selected and only included patients who responded positively to a facet joint injection with lidocaine (more than 50% reduction in pain score). These patients were randomly allocated to either corticosteroids or placebo injections. No significant differences for self‐rated improvement, pain or functional status were found between the groups at one and three months. At six months, significant differences were found with regard to self‐rated improvement, pain and functional status in favour of the corticosteroids group (Carette 1991). In a low quality study, Lilius 1990 et al compared corticosteroids injected intra‐articularly to corticosteroids injected peri‐capsularly and to placebo injections. No significant differences between the groups were reported for pain, disability and work attendance at one hour, two weeks, six weeks and three months (Lilius 1990).
There is moderate evidence (2 trials; 210 patients) that facet joint injections with corticosteroids are not significantly different from placebo injections for short term pain relief and improvement of disability. There is conflicting evidence (2 trials; 210 patients) whether facet joint injections with corticosteroids are more effective for intermediate term pain reduction and improvement of disability than placebo injections. There is limited evidence (1 trial; 109 patients) that there is no significant difference between the effects of facet joint injections with corticosteroids and placebo injection on work attendance. Generic health status and patient satisfaction were not measured.
Facet joint injections with corticosteroids versus other treatments
In four studies, the effects of corticosteroids injections into the facet joints were compared with other treatments. In a low quality study, Manchikanti 2001 et al compared the effects of multiple medial branch blocks, consisting of corticosteroids combined with local anaesthetics and Sarapin, to multiple medial branch blocks consisting of only local anaesthetics and Sarapin. No significant differences between the groups were found in pain relief, overall health, physical, functional, and psychological status and return‐to‐work at different follow‐up moments throughout a 2.5‐year follow‐up period. In a low quality study, Mayer 2004 et al made a comparison between facet joint injections on one to three levels bilaterally, with mixtures of local anaesthetics and corticosteroids combined with a home stretching exercise program, and the home stretching exercise program only. No significant post‐treatment differences between the groups were found for pain and disability. In the high quality study by Marks 1992 et al, facet joint injections with corticosteroids and lignocaine were compared with facet nerve blocks using similar medication. The facet joint injections provided slightly better pain relief than facet nerve blocks, although statistical significance was only reached at one month, not immediately post‐treatment, at two weeks or three months (Marks 1992). In a low quality study, Fuchs 2005 et al compared the effects of facet joint injections with sodium hyaluronate to similar injections with corticosteroids. No significant differences in pain relief, disability and quality of life between the groups were found at different follow‐up points over a 180‐day period.
There is limited evidence (1 trial; 84 patients) that facet joint injections consisting of corticosteroids, local anaesthetics and Sarapin are not more effective than similar injections, consisting of local anaesthetics and Sarapin, in influencing pain relief, overall health, physical, functional, psychological status and return‐to‐work at short‐ and long‐ term follow‐up. There is limited evidence (1 trial; 70 patients) that facet joint injections with mixtures of corticosteroids and local anaesthetics combined with a home stretching exercise program are not more effective than a home stretching exercise program alone in providing long term effects on pain and disability. There is moderate evidence (1 trial; 86 patients) that facet joint injections with corticosteroids and local anaesthetics are not more effective in improving short‐ and long‐term pain scores, disability scores and work attendance than facet nerve blocks using similar medication. There is limited evidence (1 trial; 60 patients) that facet joint injections with sodium hyalorunate are not more effective than similar injections with corticosteroids in providing short‐ and long‐ term pain relief, better function and improved quality of life. Patient satisfaction was not measured.
Facet joint injections with local anaesthetics versus other treatments.
One high quality study by Revel 1999 et al, which was designed from a diagnostic perspective, compared facet joint injections with lidocaine to facet joint injections with saline. In both groups these injections were followed by an injection with corticosteroids (i.e. cortivazol) near the joints. The lidocaine group had significantly higher pain relief post‐treatment than the saline group (Revel 1999).
There is moderate evidence (1 trial; 80 patients) that facet joint injections with lidocaine combined with peri‐articular corticosteroid injections are more effective for short‐term pain relief than facet joint injections with saline combined with peri‐articular corticosteroid injections. General improvement, back‐specific disability, generic health status, disability for work and patient satisfaction were not measured.
3. Local injections
Local injections with corticosteroids versus placebo injections
In a high quality study, Garvey 1989 et al compared trigger point injections with corticosteroids (i.e. Aristopan) to placebo injections, a dry needle stick (acupuncture) and a spray of ethylchloride and acupressure in patients with persistent non‐radiating low‐back pain, who were able to localize a point of maximal tenderness. No significant differences between the groups in self‐reported improvements were found at two weeks. In a low quality study, Sonne 1985 et al studied the effects of local iliolumbar ligament injections with corticosteroids versus placebo and found no significant differences in pain relief at two weeks. However, self‐reported improvement at two weeks was significantly higher in the corticosteroid group than in the placebo group (Sonne 1985).
There is moderate evidence (1 trial; 63 patients) that trigger point injections with corticosteroids are not significantly different from placebo injections for self‐reported improvements at short‐term follow‐up. There is limited evidence (1 trial; 30 patients) that local injections into the iliolumbar ligament are more effective than placebo for self‐reported improvement in the short term but not for pain relief in the short term. Back‐specific disability, generic health status, disability for work and patient satisfaction were not measured.
Local injections with anaesthetics versus placebo injections
The high quality study by Collee 1991 et al investigated the effects of a single local injection of lignocaine into the point of maximal tenderness over the medial part of the iliac crest versus a placebo injection. At two weeks, the mean pain score was significantly lower in the lignocaine groups compared to the placebo group. A non‐significant difference in favour of the lignocaine group was also found for self‐reported improvement (Collee 1991).
There is moderate evidence (1 trial; 41 patients) that an injection with a local anaesthetic at the point of maximal tenderness over the medial part of the iliac crest, in patients with iliac crest pain syndrome is more effective for short‐term pain relief than a placebo injection. Back‐specific disability, generic health status, disability for work and patient satisfaction were not measured.
Intramuscular injections with Vitamin B12 versus placebo injections
In a low quality study, Mauro 2000 et al compared the effects of intramuscular Vitamin B12 injections to intramuscular placebo injections and found significant effects for post‐treatment pain and disability in favour of the Vitamin B12 group.
There is limited evidence (1 trial; 60 patients) that intramuscular Vitamin B12 injections are more effective than intramuscular placebo injections for short‐term pain relief and disability improvement. Generic health status, disability for work and patient satisfaction were not measured.
Clinical relevance
There was disagreement between the two review authors with regard to the scoring of 27 of 90 (30%) clinical relevance items (Table 10). Consensus was reached on all scorings after discussion. In only two studies, the review authors considered the likely treatment benefits to be worth the potential harms (Revel 1999; Mauro 2000) and in only four studies, the size of the effect was considered to be clinically important (Breivik 1976; Sonne 1985; Revel 1999; Mauro 2000). According to the review authors, most of the studies described the interventions and treatment settings well enough to enable clinicians to replicate the treatment in clinical practice.
3. Clinical relevance.
| reference | A | B | C | D | E |
|---|---|---|---|---|---|
| Aldrete 2003 | ‐ | + | + | ‐ | ‐ |
| Beliveau 1971 | ? | + | ? | ‐ | ‐ |
| Breivik 1976 | + | + | + | + | ? |
| Carette 1991 | + | + | + | ‐ | ‐ |
| Collee 1991 | + | + | ‐ | ‐ | ‐ |
| Fuchs 2005 | + | + | + | ‐ | ‐ |
| Garvey 1989 | ‐ | + | ‐ | ‐ | ‐ |
| Lierz 2004 | ? | + | ‐ | ‐ | ‐ |
| Lilius 1990 | + | + | + | ‐ | ? |
| Manchikanti 2001 | ‐ | + | + | ‐ | ‐ |
| Marks 1992 | + | + | ‐ | ‐ | ‐ |
| Mauro 2000 | + | ? | + | + | + |
| Mayer 2004 | + | + | + | ‐ | ‐ |
| Revel 1999 | + | + | ‐ | + | + |
| Rocco 1989 | ‐ | + | ‐ | ‐ | ‐ |
| Serrao 1992 | + | + | + | ‐ | ‐ |
| Sonne 1985 | ‐ | ‐ | + | + | ‐ |
| Takada 2005 | ‐ | + | ‐ | ‐ | ? |
Side effects
In nine of the 18 studies, side effects such as headache, dizziness, transient local pain, paresthesia and nausea were reported in small numbers of patients (Table 2). The use of morphine was more frequently associated with pruritis, nausea and vomiting (Rocco 1989).
Discussion
The overall methodological quality of the studies included in this review was limited and only six of the 18 trials (Sonne 1985; Collee 1991; Carette 1991; Marks 1992; Revel 1999; Mauro 2000) showed significant results for at least one outcome at a follow‐up time point in favour of one of the treatment arms. Four of these six studies reported effects that could be considered to be clinically important (Breivik 1976; Mauro 2000; Revel 1999; Sonne 1985). We conclude there is no strong evidence to support the use of any type of injection therapy (epidural, facet joint or local sites) for subacute or chronic‐low‐back pain in patients without radicular pain. In the earlier Cochrane review on this topic, Nelemans 2000 et al also concluded that convincing evidence was lacking on the effects of injection therapy for low‐back pain, which means that the general picture on the effectiveness of injection therapy has not changed since then. The scientific basis for these injections for low‐back pain has not improved.
Nelemans 2000 et al proposed several recommendations for future research. These recommendations involved the priority for placebo‐controlled trials instead of trials that compared injections to other treatments, the need for methodologically sound RCTs, and more of a focus on long‐term effects of the treatment instead of only short‐term effects (Nelemans 2000). Among the 18 studies included in this review, seven new studies were covered in the present update (Mauro 2000; Manchikanti 2001; Aldrete 2003; Lierz 2004; Fuchs 2005; Takada 2005 ). Of these seven new studies, only Mauro 2000 et al compared intramuscular Vitamin B12 with placebo injections, which resulted in a total of seven placebo‐controlled versus 12 studies that compared injections to other treatments or to each other.
According to the methodological quality assessment, the new studies in this update had no higher methodological quality than the studies of the earlier review. On the contrary, the mean number of positive items was even lower (4.1 versus 6.3 positive scores; Table 2; Table 3) in this group of studies compared to the studies included in the earlier review. The most prevalent shortcomings, which were a lack of clarity regarding the concealment of random treatment allocation, no reporting of co‐interventions and no reporting of an intention‐to‐treat analysis, could easily have been avoided and nowadays, researchers should be aware that these are generally accepted principles of methodological quality for studying the effectiveness of treatments. Only four out of the 18 studies included had outcome assessments performed at six months post‐randomization and beyond (Rocco 1989; Carette 1991; Manchikanti 2001; Fuchs 2005). The outcome assessments of the other studies were either performed post‐treatment or at another short‐term time interval. Of course, from a pharmacological standpoint, the agent under study may act upon the target tissue rather quickly, for example, by reducing inflammation. However, it is advantageous that the favourable effects on pain and disability will also stand at the longer term. Since the course of sub‐acute and chronic low‐back pain can be described as consisting of spontaneous remissions and exacerbations (Croft 1998) short‐term effects of injections do not necessarily reflect a successful treatment result in clinical practice. Outcome measures which were used in the studies included visual analogues scales for pain relief and questions regarding self‐perceived improvement or other outcomes. Only six studies used well‐known and validated questionnaires for measuring disability (Rocco 1989; Carette 1991; Serrao 1992; Mauro 2000 ; Mayer 2004; Fuchs 2005).
For the purpose of this review, we subdivided the studies according to injection sites (i.e. epidural, facet joint and local sites), to pharmaceutical agents (i.e. corticosteroids, anaesthetics and other agents) and according to the comparison that was made (i.e. placebo‐controlled versus comparison of two or more treatments). By following this approach, our aim was to come up with groups that were clinically comparable without ending up with groups consisting of only one study. Despite the fact that this approach seemed logical, there were still differences among the studies that ended up in a group with regard to inclusion criteria, target tissue (e.g. intra‐articular facet joint injections, peri‐capsular facet joint injections and medial branch blocks), injection dosage, co‐interventions, reference treatments and outcome measures. This should be kept in mind when reading the evidence statements in the results section of this paper. Furthermore, it should be emphasized that we only separated trials on the basis of whether the comparator was a placebo injection or other treatment. Other aspects of the studies, such as meticulous patient selection by diagnostic injections or experience and skills of the treatment providers were not assessed. One of the main problems when studying the effects of injection therapy in low‐back pain is the lack of diagnostic criteria for determining the injection site. According to our view, it is extremely difficult to accurately diagnose and determine the localisation of tissues that cause the pain and other symptoms. Some researchers have tackled this problem, for example, by only including patients for facet joint injections who responded positively to facet blocks with anaesthetics (Carette 1991; Manchikanti 2001) or patients with radiologically confirmed facet joint osteoarthritis (Fuchs 2005). From these three studies, only the study by Carette 1991 et al had positive results. They showed significant effects for pain and the physical dimension of the Sickness Impact Profile in favour of a methylprednisolone group compared to a placebo group, even after six‐month follow‐up (Carette 1991).
The rationale behind the application of a particular pharmaceutical agent among the included studies was variable. In 13 studies, the effects of corticosteroids were investigated. This may be a reasonable choice since this agent aims to reduce inflammation. In four studies, the main pharmaceutical agent under study was anaesthetics (Collee 1991; Revel 1999; Lierz 2004; Takada 2005). The reason for choosing an injection with anaesthetics is not always immediately clear. Due to the relatively short duration of action of these pharmaceutical agents, they do not seem to be the first choice for pain relief in sub‐acute and chronic patients. Lierz 2004 et al and Takada 2005 et al compared the short‐ term analgesic effects of epidural blocks with ropivacaine and bupivacaine and found no significant differences between the groups. The authors of both studies explained that epidural blocks may provide analgesia to allow for exercise and active physiotherapy, thus facilitating more active therapies. Collee 1991 et al compared a local injection of lignocaine versus saline into a tender point over the medial part of the iliac crest. The study authors state that the injection with lignocaine may somehow affect the ascending or sensory input and its varying modulating systems. However, no significant differences in improvement rates were found. The study by Revel 1999 et al was carried out from a diagnostic perspective. They compared facet joint injections with lidocaine to placebo injections in order to determine several clinical criteria which were predictive of significant relief of back pain after facet joint anaesthesia (Revel 1999). Other applied pharmaceutical agents were: indomethacin (Aldrete 2003), sodium hyaluronate (Fuchs 2005), Sarapin (Manchikanti 2001), morphine (Rocco 1989) and Vitamin B12 (Mauro 2000). In the study by Mauro 2000 et al, daily intramuscular injections for a two‐week period with Vitamin B12 appeared to be effective for short‐term pain and disability compared to placebo injections.
Half of the included studies reported side effects in small numbers of patients (see Characteristics of included studies table). These side effects ranged from headache, dizziness and transient local pain to nausea and vomiting. Although these are not life threatening complications and have only been reported in a limited number of patients, this does not mean that the application of injection therapy in low‐back pain is always without risk. Rare but more serious complications of injection therapy have been mentioned in the literature, such as, cauda equina syndrome (Bilir 2006), septic facet joint arthritis (Weingarten 2006), discitis (Hooten 2006), paraplegia (Houten 2002) and paraspinal abscesses (Cook 1999). In another study, it was found that eight out of 128 consecutive patients referred to a hospital with meningitis had a history of single or repeated injections for low‐back pain one to three weeks before their hospital admission (Gaul 2005). Although the absolute frequency of these complications may be rare, these risks should be taken into consideration, especially given the fact that convincing evidence on the effects of injection therapy for low‐back pain is still lacking.
Seven out of the 18 trials included in this review reported significant but short‐lasting effects in favour of the intervention group, with interventions varying from facet joint injections with corticosteroids (Carette 1991; Mayer 2004) and anaesthetics (Revel 1999), epidural injections with corticosteroids (Breivik 1976), local injections with anaesthetics (Sonne 1985; Collee 1991) to intramuscular injections with Vitamin B12 (Mauro 2000). The overall picture of the results of this review reveals that there is currently insufficient scientific evidence on the effects of injection therapy, regardless of type and dosage, for patients with subacute and chronic low‐back pain. It is discouraging that the average methodological quality of the trials published since the earlier review has not improved while the validity criteria for effectiveness research have become more familiar to back pain researchers. Most studies dealt with heterogeneous groups of patients, which on average, may mask potential treatment effects. Therefore, we can not rule out that some type of injection therapy might be effective for a particular subgroup of patients. Therefore, accurate diagnostic criteria are needed. However, this remains merely a theoretical consideration, which is not based on the results of the present review.
Authors' conclusions
Implications for practice.
Based on the current state of the literature there is insufficient evidence to support or refute the use of injection therapy, regardless of type and dosage, for patients with subacute and chronic low‐back pain without radicular pain.
Implications for research.
We can not rule out that some type of injection therapy is effective for a particular subgroup of patients. More research is needed to study the accuracy of diagnostic criteria to identify which, if any, subgroups of patients are likely to respond to injection therapy and if established, the effectiveness of injection therapy for these subgroups. Corticosteroids seem to be the most logical therapeutic agent, while anaesthetics seem only useful for diagnostic purposes. If injection therapy turns out to be an effective treatment strategy for a subgroup of low‐back pain patients, it has to be embedded in the current, widely supported active approach directed at reassurance and self management.
What's new
| Date | Event | Description |
|---|---|---|
| 19 January 2011 | Amended | Contact details updated. |
History
Protocol first published: Issue 3, 1996 Review first published: Issue 4, 1999
| Date | Event | Description |
|---|---|---|
| 23 November 2009 | Amended | Contact details updated. |
| 12 May 2008 | Amended | converted to new review format |
| 12 May 2008 | New citation required but conclusions have not changed | This review is an update of a previously withdrawn review. In an attempt to increase clinical homogeneity, the withdrawn review was split, and the current review no longer contains trials on the effects of intradiscal therapy, prolotherapy, or epidural steroids for radiculopathy due to disc herniation. These are topics of current or future Cochrane reviews.As before, the review authors concluded that there was insufficient evidence to support the use of injection therapy in subacute and chronic low‐back pain. However, it cannot be ruled out that specific subgroups of patients may respond to a specific type of injection therapy. |
| 12 May 2008 | New search has been performed | The search was updated and eight new RCTs were included for the scope of the current review. In the current review, the internal validity of the RCTs was measured using the eleven criteria outlined in the 2003 Updated Cochrane Back Review Group method guidelines. Half of the RCTs were rated as having a low risk of bias. |
| 25 January 2006 | New citation required but conclusions have not changed | Review was withdrawn from publication because it was out of date. |
Acknowledgements
The review team wishes to thank Professor Ferd Sturmans, The Netherlands, for his contribution to the original review.
Appendices
Appendix 1. Search Strategies
PubMed
Phase1: (randomized controlled trial[pt] OR controlled clinical trial[pt] OR randomized controlled trials[mh] OR random allocation[mh] OR double‐blind method[mh] OR single‐blind method[mh] OR clinical trial[pt] OR clinical trials[mh] OR "clinical trial"[tw]) OR ((single*[tw] OR doubl*[tw] OR trebl*[tw] OR tripl*[tw]) AND (mask*[tw] OR blind*[tw])) OR ("latin square"[tw]) OR placebos[mh] OR placebo*[tw] OR random*[tw] OR research design[mh:noexp] OR comparative study[mh] OR evaluation studies[mh] OR follow‐up studies[mh] OR prospective studies[mh] OR cross‐over studies[mh] OR control*[tw] OR prospective*[tw] OR volunteer*[tw] NOT (animal[mh] NOT human[mh])
Phase 2: (low back pain OR backache[tw] OR Lumbago[tw])
Phase 3: (injections) OR (injection) OR (chemonucleolysis) OR (rhizotomy) OR (facet denervation) OR (thermolesions)
Phase 4: 1 AND 2 AND 3
EMBASE
#1 clinical article #2 clinical study #3 clinical trial #4 controlled study #5 randomized controlled trial #6 major clinical study #7 double blind procedure #8 multicentre study #9 single blind procedure #10 crossover procedure #11 placebo #12 (1 or 2 or 3 or 4 or 5 or 6 or 7 or 8 or 9 or 10 or 11) #13 allocat$ #14 assign$ #15 blind$ #16 (clinic$ adj25 (study or trial)) #17 compar$ #18 control$ #19 cross?over #20 factorial$ #21 follow?up #22 placebo$ #23 prospectiv$ #24 random$ #25 ((singl$ or doubl$ or trebl$ or tripl$) adj25 (blind$ or mask$)) #26 trial #27 (versus or vs) #28 (13 or 14 or 15 or 16 or 17 or 18 or 19 or 20 or 21 or 22 or 23 or 24 or 25 or 26 or 27 or 28) #29 (12 or 28) #30 human #31 nonhuman #32 animal #33 animal experiment #34 (31 or 32 or 33) #35 (30 and 34) #36 (29 not 34) #37 (29 and 35) #38 (36 or 37) #39 low back pain #40 backache #41 lumbago #42 (#39 or #40 or #41) #43 injection #44 injections #45 chemonucleolysis #46 rhizotomy #47 facet denervation #48 thermolesion #49 (#43 or #44 or #45 or #46 or #47 or #48) #50 (#49 and #42 and #38)
Data and analyses
Comparison 1. epidural corticosteroids versus placebo injections.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Percentage improved over 1 to 3 months | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 2 Percentage pain relief over 3 to 5 weeks | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected |
1.1. Analysis.
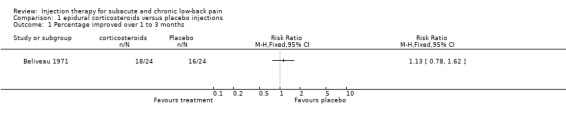
Comparison 1 epidural corticosteroids versus placebo injections, Outcome 1 Percentage improved over 1 to 3 months.
1.2. Analysis.
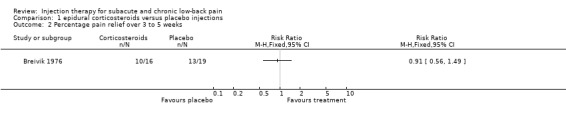
Comparison 1 epidural corticosteroids versus placebo injections, Outcome 2 Percentage pain relief over 3 to 5 weeks.
Comparison 2. epidural corticosteroid injections versus other treatments.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 General improvement at 2 months | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected |
2.1. Analysis.
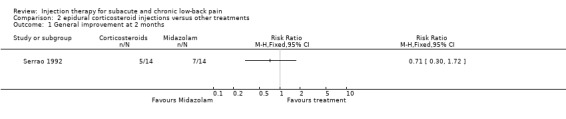
Comparison 2 epidural corticosteroid injections versus other treatments, Outcome 1 General improvement at 2 months.
Comparison 3. Facet joint injections with corticosteroids versus placebo injections.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Self‐rated improvement at 1 month | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 2 Self rated improvement at 6 months | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected |
3.1. Analysis.
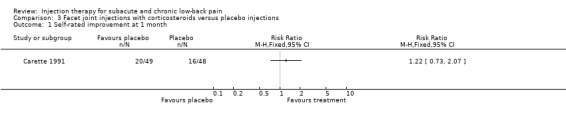
Comparison 3 Facet joint injections with corticosteroids versus placebo injections, Outcome 1 Self‐rated improvement at 1 month.
3.2. Analysis.
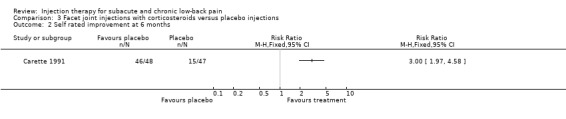
Comparison 3 Facet joint injections with corticosteroids versus placebo injections, Outcome 2 Self rated improvement at 6 months.
Comparison 4. Facet joint injections with corticosteroids versus other injections.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Improvement in pain severity at 1 month | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 2 Improvement in pain severity at 3 months | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected |
4.1. Analysis.
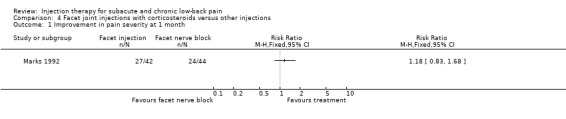
Comparison 4 Facet joint injections with corticosteroids versus other injections, Outcome 1 Improvement in pain severity at 1 month.
4.2. Analysis.
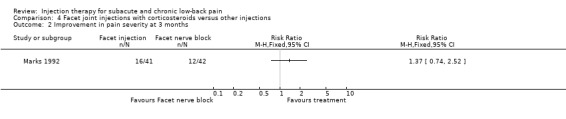
Comparison 4 Facet joint injections with corticosteroids versus other injections, Outcome 2 Improvement in pain severity at 3 months.
Comparison 5. Facet joint injections with local anaesthetics versus other injections.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Pain reduction post‐treatment in positive group (VAS) (facet joint assumed to be the primary source of pain) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected |
5.1. Analysis.
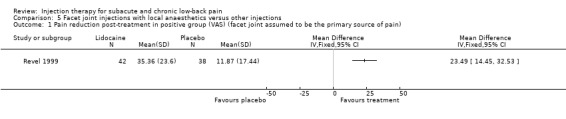
Comparison 5 Facet joint injections with local anaesthetics versus other injections, Outcome 1 Pain reduction post‐treatment in positive group (VAS) (facet joint assumed to be the primary source of pain).
Comparison 6. Local injections with corticosteroids versus placebo injections.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Self‐reported improvement at 2 weeks | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 2 Self‐reported improvement posttreatment | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected |
6.1. Analysis.
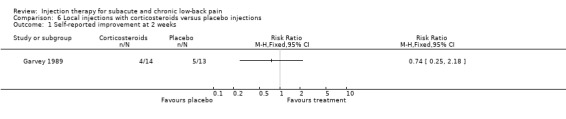
Comparison 6 Local injections with corticosteroids versus placebo injections, Outcome 1 Self‐reported improvement at 2 weeks.
6.2. Analysis.
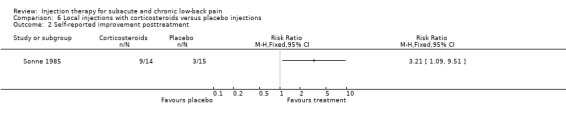
Comparison 6 Local injections with corticosteroids versus placebo injections, Outcome 2 Self‐reported improvement posttreatment.
Comparison 7. Local injections with anaesthetics versus placebo injections.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Pain intensity at 2 weeks (VAS) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 2 Self‐reported improvement at 2 weeks | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected |
7.1. Analysis.
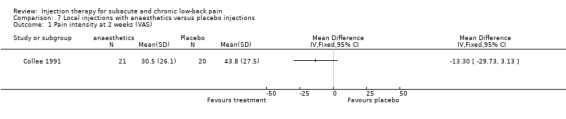
Comparison 7 Local injections with anaesthetics versus placebo injections, Outcome 1 Pain intensity at 2 weeks (VAS).
7.2. Analysis.
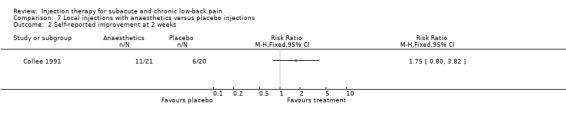
Comparison 7 Local injections with anaesthetics versus placebo injections, Outcome 2 Self‐reported improvement at 2 weeks.
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Aldrete 2003.
| Methods | RCT; Randomization procedure not described. | |
|---|---|---|
| Participants | Pain care center, Florida, USA. 206 patients with recurrent LBP and radiculopathy after laminectomy, at least 6 months post operation, lower back and extremity pain > 7 points on VAS scale (0 to 10). The duration of pain was not specified. Exclusion criteria: pseudomeningocele, arachnoiditis and/or recurrent pain from free disk fragments as confirmed by MRI study. | |
| Interventions | (1) 2 epidural injections of indomethacin 1 mg. (2) 2 epidural injections of indomethacin 2 mg. (3) 2 epidural injections of methylprednisolone 80 mg. In every case, the interval between the injections was 2 weeks and medication was diluted in 3 ml of 0.5% bupivacaine. | |
| Outcomes | Timing of outcomes: before and 2 weeks after randomization. No significant differences were reported between the groups in pain progress and physical activity at 2 weeks. | |
| Notes | Side‐effects: no instances of apparent dural puncture or high sensory or motor block were noted, nor were other effects typical of NSAIDs therapy such as rash, epigastric discomfort, or bruising. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | Randomization procedure not described |
| Allocation concealment? | Unclear risk | B ‐ Unclear |
| Blinding? All outcomes ‐ patients? | Unclear risk | unclear from text |
| Blinding? All outcomes ‐ care providers? | High risk | no |
| Blinding? All outcomes ‐ outcome assessors | Unclear risk | unclear from text |
| Incomplete outcome data addressed? All outcomes ‐ drop‐outs? | High risk | |
| Incomplete outcome data addressed? All outcomes ‐ ITT analysis | High risk | |
| Similarity of baseline characteristics? | Unclear risk | unclear from data given |
| Co‐interventions avoided or similar? | Unclear risk | unclear from text |
| Compliance acceptable? | Low risk | |
| Timing outcome assessments similar? | Low risk |
Beliveau 1971.
| Methods | RCT; Patients were allocated alternately to treatment groups. | |
|---|---|---|
| Participants | Department of Rheumatology and Physical medicine, UK. 48 patients with unilateral sciatica, thought to be caused by an intervertebral disc lesion, with or without neurological signs, before or after other conservative treatments had been tried. The duration of pain was not specified. | |
| Interventions | (1) Epidural injection of 42 ml of procaine 0.5% in normal saline. (2) Epidural injection of 40 ml of procaine 0.5% in normal saline to which 2 ml of methylprednisolone 80 mg had been added. In both groups injections were repeated in patients who had an improvement after the first injection. | |
| Outcomes | Timing of outcomes: before and 1 week after randomization. (1) 16 of 24 patients improved at 1 week after the injection; (2) 18 of 24 patients improved at 1 week after the injection; no other outcomes reported. | |
| Notes | Side effects: Mild headache and dizziness occurred in about ten cases; the symptoms were transient lasting up to 30 minutes after the injection. In two cases the theca was penetrated by the needle during the injection and the procedure was stopped. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | High risk | Patients were allocated alternately to treatment groups |
| Allocation concealment? | High risk | C ‐ Inadequate |
| Blinding? All outcomes ‐ patients? | High risk | |
| Blinding? All outcomes ‐ care providers? | High risk | |
| Blinding? All outcomes ‐ outcome assessors | High risk | |
| Incomplete outcome data addressed? All outcomes ‐ drop‐outs? | Low risk | |
| Incomplete outcome data addressed? All outcomes ‐ ITT analysis | Low risk | |
| Similarity of baseline characteristics? | High risk | |
| Co‐interventions avoided or similar? | High risk | |
| Compliance acceptable? | Low risk | |
| Timing outcome assessments similar? | Low risk |
Breivik 1976.
| Methods | RCT; Patients were allocated according to a list of random numbers. | |
|---|---|---|
| Participants | Departments of Anesthesiology and Neurology, Akershus Central Hospital and outpatient clinic, Norway 35 patients with incapacitating chronic low‐back pain and sciatica unresponsive to conservative treatment for several months to several years. Diagnoses based on radiculography: arachnoiditis (N=8), prolapsed disc (N=8), no abnormality (N=11), inconclusive findings (N=5). | |
| Interventions | (1) Caudal epidural injection of 20 ml bupivacaine 0.25% with 80 mg depo‐methylprednisolone. (2) Caudal epidural injection of 20 ml bupivacaine 0.25% followed by 100 ml saline. Frequency: up to three injections at weekly intervals eventually followed by up three injections of the alternative group. | |
| Outcomes | Timing of outcomes: 2 to 5 weeks after randomization Reported outcomes were pain relief, objective improvements (i.e. neural symptoms) and work status. Pain relief was obtained in 63% of the patients in the intervention group and 68% of the patients in the reference group which was no statistically significant difference. | |
| Notes | If there was no improvement 3 weeks after the last injection, up to three injections of the alternative type were given. Side effects: not mentioned. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | Patients were allocated according to a list of random numbers |
| Allocation concealment? | High risk | C ‐ Inadequate |
| Blinding? All outcomes ‐ patients? | Low risk | |
| Blinding? All outcomes ‐ care providers? | High risk | |
| Blinding? All outcomes ‐ outcome assessors | Low risk | |
| Incomplete outcome data addressed? All outcomes ‐ drop‐outs? | Low risk | |
| Incomplete outcome data addressed? All outcomes ‐ ITT analysis | High risk | |
| Similarity of baseline characteristics? | Unclear risk | data not provided |
| Co‐interventions avoided or similar? | Low risk | |
| Compliance acceptable? | Low risk | |
| Timing outcome assessments similar? | High risk |
Carette 1991.
| Methods | RCT; Randomization by random numbers generation, balancing after every 8 patients. | |
|---|---|---|
| Participants | Centre Hospitalier de l'Université Laval, Quebec city, Canada. 101 patients with pain originating in the facet joints and who had a >50% reduction (on a VAS score for pain) after injection with lidocaine were entered into the trial. The mean duration of the current episode of pain was 18 (intervention group) and 24 months (control group). Exclusion criteria: back pain with a non mechanical origin, previous injections in the facet joints or surgery, pregnancy, known allergy to anesthetic or radiological contrast agents, presence of a blood coagulation disorder. | |
| Interventions | (1) Facet joint injection of 20 mg (1 ml) of methylprednisolone acetate mixed with 1 ml isotonic saline. (2) Facet joint injection of 2 ml isotonic saline. | |
| Outcomes | Timing of outcomes: before and 1, 3 and 6 months after randomization and treatment. At one and three months there were no significant differences in pain, functional status and back flexion between the groups. At one month 42% of the patients in the methylprednisolone group and 33% of the patients in the placebo group reported marked or very marked improvement. At 6 months 46% of the patients in the methylprednisolone group and 15% of the patients in the placebo group reported a significant marked or very marked improvement. Significant differences at 6 months in favor of the methylprednisolone group were also found for pain (VAS‐score) and the physical dimension of the Sickness Impact Profile. The differences were reduced when concurrent interventions in the methylprednisolone group were taken into account. | |
| Notes | Side effects: No adverse effects were reported, other than transient local pain at the injection site. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | Randomization by random numbers generation, balancing after every 8 patients. |
| Allocation concealment? | Unclear risk | B ‐ Unclear |
| Blinding? All outcomes ‐ patients? | Low risk | |
| Blinding? All outcomes ‐ care providers? | Low risk | |
| Blinding? All outcomes ‐ outcome assessors | Low risk | |
| Incomplete outcome data addressed? All outcomes ‐ drop‐outs? | Low risk | |
| Incomplete outcome data addressed? All outcomes ‐ ITT analysis | High risk | |
| Similarity of baseline characteristics? | Low risk | |
| Co‐interventions avoided or similar? | Unclear risk | unclear from text |
| Compliance acceptable? | Low risk | |
| Timing outcome assessments similar? | Low risk |
Collee 1991.
| Methods | RCT; Randomization procedure not described | |
|---|---|---|
| Participants | Department of Rheumatology, University Hospital Leiden, The Netherlands. Patients recruited from a university based outpatient clinic (median duration of pain was 8 years) and a large general practice in a rural area (median duration of pain was 18 days). 41 patients with iliac crest pain syndrome. Exclusion criteria were: diagnosis of sciatica, ankylosing spondylitis, malignancy, infection, spondylolysthesis, severe degenerative disc disease or fibromyalgia. | |
| Interventions | Single local injection at the point of maximal tenderness over the medial part of the iliac crest. (1) Injection of 5 ml lignocaine 0.5%. (2) Injection of 5 ml isotonic saline. | |
| Outcomes | Timing: at baseline and 10 minutes, 7 days, 14 days and 2 months after the injection. At 14 days the mean pain score in the lignocaine group was significantly lower than in the control group. In the lignocaine group 52% of patients improved at 14 days compared to 30% in the control group. This difference was not statistically significant. Among those who improved with lignocaine, the beneficial effect continued for at least 2 months in 80% of the patients. | |
| Notes | Side effects: 2 patients in the lignocaine group and 3 in the saline group complained of a painful injection or increase of pain for some hours after the injection. Nausea some hours after the injection occurred in 2 of the patients treated with lignocaine and in 1 patient treated with saline. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | Randomization procedure not described |
| Allocation concealment? | Unclear risk | B ‐ Unclear |
| Blinding? All outcomes ‐ patients? | Low risk | |
| Blinding? All outcomes ‐ care providers? | Low risk | |
| Blinding? All outcomes ‐ outcome assessors | Low risk | |
| Incomplete outcome data addressed? All outcomes ‐ drop‐outs? | Low risk | |
| Incomplete outcome data addressed? All outcomes ‐ ITT analysis | Low risk | |
| Similarity of baseline characteristics? | Low risk | |
| Co‐interventions avoided or similar? | Unclear risk | unclear from text |
| Compliance acceptable? | Low risk | |
| Timing outcome assessments similar? | Low risk |
Fuchs 2005.
| Methods | RCT; block randomization, blocks of 6 patients generated by computer. | |
|---|---|---|
| Participants | Private orthopedic practice, Goesfeld, Germany 60 patients with chronic non‐radicular low‐back pain (> 3 months) and with radiologic confirmation of facet joint osteoarthritis | |
| Interventions | Injections into the facet joints at three levels of the lumber spine presumably most affected by degenerative changes. Injections under CT guidance at weekly intervals. Each patient received 6 injections. (1) Injection of 10 mg sodium hyaluronate in 1 ml buffer solution. (2) Injection of 10 mg glucocorticoids (triamcolone acetonide) in 1 ml crystalline suspension. | |
| Outcomes | Timing of outcomes: before and 7, 14, 21, 28, 90 and 180 days after randomization. Both groups improved and had pain relief, better function and improved quality of life. No statistically significant differences between the groups in outcomes were reported. | |
| Notes | No adverse effects were reported after administration of both therapies. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | block randomization, blocks of 6 patients generated by computer. |
| Allocation concealment? | Unclear risk | B ‐ Unclear |
| Blinding? All outcomes ‐ patients? | High risk | |
| Blinding? All outcomes ‐ care providers? | High risk | |
| Blinding? All outcomes ‐ outcome assessors | Unclear risk | unclear from text |
| Incomplete outcome data addressed? All outcomes ‐ drop‐outs? | Low risk | |
| Incomplete outcome data addressed? All outcomes ‐ ITT analysis | High risk | |
| Similarity of baseline characteristics? | Low risk | |
| Co‐interventions avoided or similar? | Unclear risk | unclear from text |
| Compliance acceptable? | Low risk | |
| Timing outcome assessments similar? | Low risk |
Garvey 1989.
| Methods | RCT; Allocation by computer‐generated four‐tier list. | |
|---|---|---|
| Participants | Department of Orthopedic Surgery, The George Washington University Medical Center, Washington DC. 63 patients with persistent non‐radiating low‐back pain (> 4 weeks) who were able to localize a point of maximal tenderness. | |
| Interventions | Trigger‐point injections: (1) 1.5 ml lidocaine 1%. (2) 0.75 ml lidocaine 1% and 0.75 ml Aristopan (20 mg/ml). (3) Dry needle stick (acupuncture). (4) 10 second spray of ethylchloride and 20 second acupressure. | |
| Outcomes | Timing of outcomes: patients were followed at 2‐week intervals. Therapy without injected medication (63% improvement rate) was at least as effective as therapy with drug injection (42% improvement rate). The differences in improvement rates between the groups were not statistically significant. | |
| Notes | Side effects: One patient who received lidocaine plus steroid complained about increased pain as well as two patients who received a dry needle stick. Another patient in the dry needle group had complaints of fever, chills and systemic upset on the night of therapy | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | Allocation by computer‐generated four‐tier list |
| Allocation concealment? | Unclear risk | B ‐ Unclear |
| Blinding? All outcomes ‐ patients? | Low risk | |
| Blinding? All outcomes ‐ care providers? | Low risk | |
| Blinding? All outcomes ‐ outcome assessors | Low risk | |
| Incomplete outcome data addressed? All outcomes ‐ drop‐outs? | Low risk | |
| Incomplete outcome data addressed? All outcomes ‐ ITT analysis | Low risk | |
| Similarity of baseline characteristics? | Unclear risk | data not provided |
| Co‐interventions avoided or similar? | Low risk | |
| Compliance acceptable? | Low risk | |
| Timing outcome assessments similar? | Unclear risk | not clearly stated in text |
Lierz 2004.
| Methods | RCT; Randomization performed by computer, no further description of procedure. | |
|---|---|---|
| Participants | Department of Anaesthesiology and Intensive Care, Hospital, Soest, Germany 40 patients with low‐back pain for at least 6 months. | |
| Interventions | Eight single shot epidural blocks at an interval of 2 or 3 days. Active physiotherapy was performed immediately after each session. (1) 10 ml ropivacaine 0.2%. (2) 10 ml bupivacaine 1.125%. | |
| Outcomes | Timing of outcomes: before and post‐treatment. Both groups showed no significant differences in analgesia post‐treatment. | |
| Notes | Side effects: no significant differences were reported in hemodynamic variables; there were three cases of short episodes of headache, two in the ropivacaine group and one in the bupivacaine group | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | Randomization performed by computer, no further description of procedure. |
| Allocation concealment? | Unclear risk | B ‐ Unclear |
| Blinding? All outcomes ‐ patients? | Low risk | |
| Blinding? All outcomes ‐ care providers? | Low risk | |
| Blinding? All outcomes ‐ outcome assessors | Unclear risk | not stated in text |
| Incomplete outcome data addressed? All outcomes ‐ drop‐outs? | Low risk | |
| Incomplete outcome data addressed? All outcomes ‐ ITT analysis | High risk | |
| Similarity of baseline characteristics? | Low risk | |
| Co‐interventions avoided or similar? | Low risk | |
| Compliance acceptable? | Low risk | |
| Timing outcome assessments similar? | Low risk |
Lilius 1990.
| Methods | RCT; Randomization procedure not described. | |
|---|---|---|
| Participants | Department of Physical Medicine and Rehabilitation, Helsinki University Central Hospital. 109 patients with low‐back pain for over three months localized to one side with tenderness and local muscle spasm over the facet joints. 27 of them had undergone previous disc surgery. | |
| Interventions | One facet joint injection: (1) 6 ml (30 mg) bupivacaine hydrochloride mixed with 2 ml (80 mg) methylprednisolone acetate was injected into each of the two facet joints. (2) The same mixture was injected pericapsularly around the facet joints. (3) 8 ml of physiological saline was injected into the two joints. | |
| Outcomes | Timing of outcomes: before and one hour, 6 weeks and 3 months after the injection. All groups improved in work attendance, pain and disability scores. No significant differences between the groups were found at one hour, 2 weeks, 6 weeks and 3 months. | |
| Notes | Side effects: 7 of the 109 patients reported side effects. The nature of the side effects was not reported. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | Randomization procedure not described |
| Allocation concealment? | High risk | C ‐ Inadequate |
| Blinding? All outcomes ‐ patients? | High risk | |
| Blinding? All outcomes ‐ care providers? | High risk | |
| Blinding? All outcomes ‐ outcome assessors | High risk | |
| Incomplete outcome data addressed? All outcomes ‐ drop‐outs? | Low risk | |
| Incomplete outcome data addressed? All outcomes ‐ ITT analysis | High risk | |
| Similarity of baseline characteristics? | Unclear risk | data not provided |
| Co‐interventions avoided or similar? | Unclear risk | information not provided |
| Compliance acceptable? | Low risk | |
| Timing outcome assessments similar? | Low risk |
Manchikanti 2001.
| Methods | RCT; Randomization procedure not described. | |
|---|---|---|
| Participants | Private pain management practice in a non‐university setting, Paducah, Kentucky. 84 patients with low‐back pain with or without lower extremity pain for more than 6 months who did not exhibit neurological deficits and who responded positively to lidocaine blocks. | |
| Interventions | (1) Multiple medial branch blocks with a local anesthetic (0.5% lidocaine or 0.25% bupivacaine) and an equal volume of Sarapin. (2) Multiple medial branch blocks with a mixture of a local anesthetic (0.5% lidocaine or 0.25% bupivacaine), an equal volume of Sarapin and 1 mg of methyl prednisolone. The injections were given throughout the entire follow‐up period. | |
| Outcomes | Timing of outcomes: before the first injection and at different follow‐up moments throughout a 2.5 years period. Both groups in improved in pain relief, overall health status, physical, functional and psychological status and return to work over 2.5 years follow‐up. No significant differences were reported between the groups. | |
| Notes | Side effects: no complications were reported in any of the patients. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | Randomization procedure not described. |
| Allocation concealment? | High risk | C ‐ Inadequate |
| Blinding? All outcomes ‐ patients? | High risk | |
| Blinding? All outcomes ‐ care providers? | High risk | |
| Blinding? All outcomes ‐ outcome assessors | Unclear risk | unclear in text |
| Incomplete outcome data addressed? All outcomes ‐ drop‐outs? | Low risk | |
| Incomplete outcome data addressed? All outcomes ‐ ITT analysis | High risk | |
| Similarity of baseline characteristics? | Low risk | |
| Co‐interventions avoided or similar? | High risk | |
| Compliance acceptable? | Low risk | |
| Timing outcome assessments similar? | High risk |
Marks 1992.
| Methods | RCT; Random number system, concealment of assigned technique from referring clinicians and patients. | |
|---|---|---|
| Participants | Pain or back clinic, Dundee, Scotland. 86 patients with lumbar or lumbosacral pain present most of the time for at least 6 months. Exclusion criteria were a radicular pattern in either lower limb, straight leg raising limited at less than 60 degrees and no evidence of any progressive spinal disorder of non‐degenerative origin. | |
| Interventions | (1) Facet joint injection with 0.5 ml DepoMedrol (20 mg methylprednisolone acetate) followed by 1.5 ml lignocaine. (1%) at the lumbosacral level and at every other level 0.5 ml DepoMedrol followed by 1.0 ml lignocaine. (2) Facet nerve block of the medial articular branch of the posterior primary ramus from L1 to L5. | |
| Outcomes | Timing of outcomes: before and 30‐60 minutes, 1 month and 3 months after infiltration. The facet joints injections did slightly better in relieving pain than the facet nerve blocks. This difference reached statistical significance only at 1 month but not immediately after infiltration, at 2 weeks and at 3 months. | |
| Notes | Side effects: no serious complications. Transient symptoms, such as headache, paresthesia of one leg, nausea and worsening of pain occurred 15 times in the injection group and 18 times in the nerve block group. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | Random number system |
| Allocation concealment? | Low risk | A ‐ Adequate ‐ concealment of assigned technique from referring clinicians and patients |
| Blinding? All outcomes ‐ patients? | Low risk | |
| Blinding? All outcomes ‐ care providers? | Unclear risk | unclear from text |
| Blinding? All outcomes ‐ outcome assessors | Low risk | |
| Incomplete outcome data addressed? All outcomes ‐ drop‐outs? | Low risk | |
| Incomplete outcome data addressed? All outcomes ‐ ITT analysis | High risk | |
| Similarity of baseline characteristics? | Low risk | |
| Co‐interventions avoided or similar? | High risk | |
| Compliance acceptable? | Low risk | |
| Timing outcome assessments similar? | Low risk |
Mauro 2000.
| Methods | RCT; Randomization procedure not described. | |
|---|---|---|
| Participants | Outpatient clinic of the department of Orthopedics of Palermo University Hospital, Palermo, Italy 60 patients with lumbago or sciatic neuritis of mechanical origin without need for surgical procedures. They had to present with a proven medical history for low‐back pain (lasting from 6 months to 5 years) and a pain intensity of at least 60 mm as evaluated on a visual analogue scale. | |
| Interventions | Intramuscular injections once daily for a 2 week period. (1) 2 ml ampoules containing 1000 mg Vitamin B12 (Tricortin 1000). (2) 2 ml placebo ampoules. | |
| Outcomes | Timing of outcomes: before and 2 weeks after randomization. At the end of the treatment period there was a statistically significant difference in improvement in pain (visual analogue scale) and disability (Roland Disability Questionnaire) in favor of the Vitamin B12 group . | |
| Notes | Consumption of paracetamol proved significantly higher in the placebo group compared to the active treatment group. Side effects: No changes in vital signs, or adverse effects were noted. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | Randomization procedure not described. |
| Allocation concealment? | Unclear risk | B ‐ Unclear |
| Blinding? All outcomes ‐ patients? | Unclear risk | unclear from text |
| Blinding? All outcomes ‐ care providers? | Unclear risk | unclear from text |
| Blinding? All outcomes ‐ outcome assessors | Low risk | |
| Incomplete outcome data addressed? All outcomes ‐ drop‐outs? | High risk | |
| Incomplete outcome data addressed? All outcomes ‐ ITT analysis | High risk | |
| Similarity of baseline characteristics? | Low risk | |
| Co‐interventions avoided or similar? | Unclear risk | unclear from text |
| Compliance acceptable? | Low risk | |
| Timing outcome assessments similar? | Low risk |
Mayer 2004.
| Methods | RCT; Assignment based on date of the month of their initial visit alternating with patients allocated to the other group. | |
|---|---|---|
| Participants | Department of Orthopedic Surgery, University of Texas, Southwestern Medical Center, Dallas, Texas. 70 patients with chronic disabling work‐related lumbar spine disorders who had lumbar rigidity between 1 and 3 levels. The mean length of disability varied from 12 (intervention group) to 16 months (control group). | |
| Interventions | (1) Facet joint injection on one to three levels bilaterally. Each point was injected with a mixture of 1 ml 2% lidocaine, 1 ml 0.5% bupivacaine and 1 ml of a depot corticosteroid preparation. In addition, a home stretching exercise program was instructed which was supervised by physiotherapists on each subsequent visit of the patient (on average twice a week). (2) Patients in the control group only received the home stretching exercise program. | |
| Outcomes | Timing of outcomes: before and post‐treatment. No significant differences were found in pain and disability between the groups. The group which combined injections with home exercises had a significantly greater improvement in range of motion at 5 to 7 weeks compared to the control group. | |
| Notes | Side effects were not reported by the study authors. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | High risk | Assignment based on date of the month of their initial visit alternating with patients allocated to the other group. |
| Allocation concealment? | High risk | C ‐ Inadequate |
| Blinding? All outcomes ‐ patients? | High risk | |
| Blinding? All outcomes ‐ care providers? | High risk | |
| Blinding? All outcomes ‐ outcome assessors | Low risk | |
| Incomplete outcome data addressed? All outcomes ‐ drop‐outs? | Low risk | |
| Incomplete outcome data addressed? All outcomes ‐ ITT analysis | Unclear risk | unclear in text |
| Similarity of baseline characteristics? | Low risk | |
| Co‐interventions avoided or similar? | Unclear risk | unclear in text |
| Compliance acceptable? | Low risk | |
| Timing outcome assessments similar? | Unclear risk | unclear in text |
Revel 1999.
| Methods | RCT; Randomization procedure not described. | |
|---|---|---|
| Participants | Department of Rheumatology and Rehabilitation, Hospital Rochin, Paris, France. 80 patients with low‐back pain for more than 3 months and with pain graded higher than 30 mm on a 100 mm visual analogue scale. | |
| Interventions | Facet joint injections (four or six on both sides or two or three on the same side) consisting of: (1) 1 ml 2% lidocaine. (2) 1 ml saline. The injections in both groups were followed by an injection of 2 mg cortivazol near the joint. | |
| Outcomes | Timing of outcomes: before the first injection and half an hour after the last injection. Lidocaine provided significantly greater back pain relief than saline. | |
| Notes | The study was set up from a diagnostic perspective in order to determine clinical criteria that could be used to identify patients with painful facet joints. Side effects were not reported by the study authors. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | Randomization procedure not described |
| Allocation concealment? | Unclear risk | B ‐ Unclear |
| Blinding? All outcomes ‐ patients? | Low risk | |
| Blinding? All outcomes ‐ care providers? | Low risk | |
| Blinding? All outcomes ‐ outcome assessors | Low risk | |
| Incomplete outcome data addressed? All outcomes ‐ drop‐outs? | Low risk | |
| Incomplete outcome data addressed? All outcomes ‐ ITT analysis | Low risk | |
| Similarity of baseline characteristics? | Low risk | |
| Co‐interventions avoided or similar? | Unclear risk | unclear in text |
| Compliance acceptable? | Low risk | |
| Timing outcome assessments similar? | Low risk |
Rocco 1989.
| Methods | RCT; randomization procedure not described. | |
|---|---|---|
| Participants | Pain Treatment Service, Brigham and Women's hospital Boston. 22 patients who had undergone at least one prior laminectomy and who were still symptomatic. The duration of pain at study entry was not specified. | |
| Interventions | Epidural injections which were given at 1 month interval for 3 months: (1) 1.9 ml (75 mg) triamcinolone diacetate plus 1 ml (50 mg) lidocaine. (2) 8 ml (8 mg) morphine plus 1 ml (50 mg) lidocaine. (3) 1.9 ml (75 mg) triamcinolone diacetate plus 8 ml (8 mg) morphine plus 1 ml (50 mg) lidocaine. | |
| Outcomes | Timing of outcomes: before randomization, 1 month after randomization and 3 months after the last injection (6 months after randomization). No significant differences were reported with regard to pain relief at 1 and 6 months as measured with a verbal ordinal scale and a visual analogue scale. | |
| Notes | Side effects: more urinary retention was noted in the triamcinolone plus morphine group. Nausea, vomiting and pruritis were noted more frequently in the patients given morphine or triamcolone combined with morphine than in those given triamcolone alone. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | randomization procedure not described. |
| Allocation concealment? | High risk | C ‐ Inadequate |
| Blinding? All outcomes ‐ patients? | Low risk | |
| Blinding? All outcomes ‐ care providers? | High risk | |
| Blinding? All outcomes ‐ outcome assessors | Low risk | |
| Incomplete outcome data addressed? All outcomes ‐ drop‐outs? | Low risk | |
| Incomplete outcome data addressed? All outcomes ‐ ITT analysis | Low risk | |
| Similarity of baseline characteristics? | Unclear risk | data not provided |
| Co‐interventions avoided or similar? | High risk | |
| Compliance acceptable? | Low risk | |
| Timing outcome assessments similar? | Low risk |
Serrao 1992.
| Methods | RCT; Randomization procedure not described. | |
|---|---|---|
| Participants | York Pain Clinic, Department of Anaesthetics, York District Hospital, York, UK. 28 patients with mechanical low‐back pain were included. The duration of pain ranged from 1 to 35 years. Exclusion criteria were disc lesions and spinal claudication. | |
| Interventions | Epidural injections: (1) 80 mg prednisolone suspended in 10 mL saline (epidural) and 3 mL (5%) dextrose solution (intrathecal). (2) 10 mL normal saline (epidural) and 2 mg midazolam dissolved in 3 mL (5%) dextrose (intrathecal). | |
| Outcomes | Timing of outcomes: before and 2 weeks and 2 months after treatment. No significant differences were reported with regard to pain relief (short form McGill questionnaire and verbal rating scale) and general improvement at 2 week and 2 months after treatment. | |
| Notes | Side effects: Some patients reported headaches (midazolam (N = 7), steroid (N = 8)) or nausea (midazolam (N = 1), steroid (N = 2)). | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | Randomization procedure not described |
| Allocation concealment? | Unclear risk | B ‐ Unclear |
| Blinding? All outcomes ‐ patients? | Low risk | |
| Blinding? All outcomes ‐ care providers? | Unclear risk | unclear in text |
| Blinding? All outcomes ‐ outcome assessors | Low risk | |
| Incomplete outcome data addressed? All outcomes ‐ drop‐outs? | Unclear risk | data not provided |
| Incomplete outcome data addressed? All outcomes ‐ ITT analysis | Unclear risk | data not provided |
| Similarity of baseline characteristics? | Unclear risk | data not provided |
| Co‐interventions avoided or similar? | High risk | |
| Compliance acceptable? | Low risk | |
| Timing outcome assessments similar? | Low risk |
Sonne 1985.
| Methods | RCT; randomization procedure not described. | |
|---|---|---|
| Participants | Department of Rheumatology, Bispebjerg Hospital, Copenhagen, Denmark. 30 patients with low‐back pain of longer than 1 month duration with the exception of patients with herniated disc lesions, osteoporosis, arachnoiditis or ankylosing spondylitis. | |
| Interventions | Local injections at the site of the iliolumbar ligament with a maximum of three injections give at one week intervals. (1) 5 mL lignocaine 1% mixed with 1 mL methylprednisolone. (2) 5 mL isotonic saline. | |
| Outcomes | Timing of outcomes: before the study and at each visit and 2 weeks after completing the study. No significant differences between the groups in pain (visual analogue scale) and spinal flexion at each visit and 2 weeks after completing the study. Self‐reported improvement was assessed 2 weeks after completing the study. 9 out of 15 patients in the methylprednisolone group reported a good or excellent improvement compared to 3 out of 14 patients in the saline group which was a significant difference. | |
| Notes | Side effects were not reported by the study authors. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | randomization procedure not described. |
| Allocation concealment? | High risk | C ‐ Inadequate |
| Blinding? All outcomes ‐ patients? | Unclear risk | unclear in text |
| Blinding? All outcomes ‐ care providers? | High risk | |
| Blinding? All outcomes ‐ outcome assessors | Low risk | |
| Incomplete outcome data addressed? All outcomes ‐ drop‐outs? | Low risk | |
| Incomplete outcome data addressed? All outcomes ‐ ITT analysis | High risk | |
| Similarity of baseline characteristics? | Unclear risk | data not provided |
| Co‐interventions avoided or similar? | High risk | |
| Compliance acceptable? | Low risk | |
| Timing outcome assessments similar? | Low risk |
Takada 2005.
| Methods | Randomized double blind cross‐over study; Randomization procedure not described. | |
|---|---|---|
| Participants | Department of Anesthesia, Nagasaki Rosai Hospital, Sasebo, Japan. 16 patients diagnosed as having degenerative spinal disease. The length of the history of low‐back pain ranged from 1 to 24 months. Patients who had a severe canal stenosis with leg paresis or who had received spinal surgery were excluded from the study. | |
| Interventions | Single epidural injection with: (1) 8 ml ropivacaine 0.375%. (2) 8 ml ropivacaine 0.2%. (3) 8 ml bupivacaine 0.25%. Test solutions were given in a randomized order at intervals of one week or more. | |
| Outcomes | Timing of outcome: before and after epidural blockade. No significant differences in pain scores (visual analogue scale) after epidural blockade were found. | |
| Notes | Side effects: one subject in the 0.375% ropivacaine trial was catheterized because of bladder distension. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | High risk | Randomization procedure not described |
| Allocation concealment? | High risk | C ‐ Inadequate |
| Blinding? All outcomes ‐ patients? | Low risk | |
| Blinding? All outcomes ‐ care providers? | Low risk | |
| Blinding? All outcomes ‐ outcome assessors | Low risk | |
| Incomplete outcome data addressed? All outcomes ‐ drop‐outs? | Unclear risk | data not given |
| Incomplete outcome data addressed? All outcomes ‐ ITT analysis | Unclear risk | data not given |
| Similarity of baseline characteristics? | Low risk | |
| Co‐interventions avoided or similar? | Unclear risk | unclear in text |
| Compliance acceptable? | Low risk | |
| Timing outcome assessments similar? | Low risk |
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Bonetti 2005 | Study population consisted of patients with radiculopathy |
| Bush 1991 | Study was about corticosteroid injections for radiculopathy versus placebo |
| Cuckler 1985 | Study was about corticosteroid injections for radiculopathy versus placebo |
| Dallas 1987 | The drugs were administered by means of a catheter and not directly by means of an injection |
| Fredman 1999 | The drugs were administered by means of a catheter and not directly by means of an injection. |
| Glynn 1988 | The drugs were administered by means of a catheter and not directly by means of an injection. |
| Hameroff 1981 | Patients who had cervical symptoms were also included and no separate results for the lower back were presented |
| Mathews 1987 | Study was about corticosteroid injections for radiculopathy versus placebo |
| Ney 2006 | Study appeared not to be a RCT |
| Ofluoglu 2007 | Study population consisted of patients with osteoporosis |
| Oh 2004 | Radiofrequency denervation was compared with an injection of a small amount of lidocaine which was considered a sham treatment |
| Ongley 1987 | Study was about the effectiveness of prolotherapy |
| Pneumaticos 2006 | Two different injection sites were compared and no comparison was made with regard to the type of injection therapy |
| Rogers 1992 | Study dealt with corticosteroid injections for radiculopathy versus placebo |
| Simmons 1992 | Study was about the effectiveness of intradiscal injections |
| Stojanovic 2005 | Two different needling techniques were compared and no comparison was made with regard to the type of injection therapy |
| Tuzun 2003 | Study population consisted of acute low‐back pain patients |
Contributions of authors
Bart Staal and Rob de Bie updated the searches for new trials, conducted the study selection, quality assessment, data extraction and analysis of all new studies. Patty Nelemans and Rob de Bie conducted the study selection, quality assessment, data extraction and analysis of all studies included in the original review. Bart Staal drafted the present manuscript. All authors critically revised and approved the manuscript.
Declarations of interest
None declared
Edited (no change to conclusions)
References
References to studies included in this review
Aldrete 2003 {published data only}
- Aldrete JA. Epidural injections of indomethacin for postlaminectomy syndrome: a preliminary report. Anesth Analg 2003;96(2):463‐8. [DOI] [PubMed] [Google Scholar]
Beliveau 1971 {published data only}
- Beliveau P. A comparison between epidural anaesthesia with and without corticosteroid in the treatment of sciatica. Rheumatol Phys Med 1971;11(1):40‐3. [DOI] [PubMed] [Google Scholar]
Breivik 1976 {published data only}
- Breivik HH, Hesla PE, Molnar I, Lind B. Treatment of chronic low back pain and sciatica: comparison of caudal epidural injections of bupivacaine and methylprednisolone with bupivacaine followed by saline. Adv Pain Res Ther 1976;1:927‐32. [Google Scholar]
Carette 1991 {published data only}
- Carette S, Marcoux S, Truchon R, Grondin C, Gagnon J, Allard Y, et al. A controlled trial of corticosteroid injections into facet joints for chronic low back pain. N Eng J Med 1991;325(14):1002‐7. [DOI] [PubMed] [Google Scholar]
Collee 1991 {published data only}
- Collee G, Dijkmans BA, Vandenbroucke JP, Cats A. Iliac crest pain syndrome in low back pain. A double blind, randomized study of local injection therapy. J Rheumatol 1991;18(7):1060‐3. [PubMed] [Google Scholar]
Fuchs 2005 {published data only}
- Fuchs S, Erbe T, Fischer HL, Tibesku CO. Intraarticular hyaluronatica acid versus glucocorticoid injections for nonradicular pain in the lumbar spine. J Vasc Interv Radiol 2005;16(11):1493‐8. [DOI] [PubMed] [Google Scholar]
Garvey 1989 {published data only}
- Garvey TA, Marks MR, Wiesel SW. A prospective, randomized, double‐blind evaluation of trigger point injection therapy for low back pain. Spine 1989;14(9):962‐4. [DOI] [PubMed] [Google Scholar]
Lierz 2004 {published data only}
- Lierz P, Gustorff B, Markow G, Felleiter P. Comparison between bupivacaine 0.125% and ropivacaine 0.2% for epidural administration to outpatients with chronic low back pain. Eur J Anaesthesiol 2004;21(1):32‐7. [DOI] [PubMed] [Google Scholar]
Lilius 1990 {published data only}
- Lilius G, Harilainen A, Laasonen EM, Myllynen P. Chronic unilateral low‐back pain. Predictors of outcome of facet joint pain. Spine 1990;15(8):780‐2. [PubMed] [Google Scholar]
- Lillius G, Laasonen EM, Myllynen P, Harilainen A, Gronlund G. Lumbar facet joint syndrome. A randomised clinical trial. J Bone Joint Surg 1989;71:681‐4. [DOI] [PubMed] [Google Scholar]
- Lillius G, Lassonen AM, Myllynen P, Harilainen A, Salo L. The lumbar facet joint syndrome: significance of inappropriate signs: a randomized, placebo‐controlled trial. French Journal of Orthopaedic Surgery 1989;3(4):479‐86. [Google Scholar]
Manchikanti 2001 {published data only}
- Manchikanti L, Pampati V, Bakhit C, Rivera JJ, Beyer CD, Damron KS, et al. Effectiveness of lumbar facet joint nerve blocks in chronic low back pain: a randomized clinical trial. Pain Physician 2001;4(1):101‐7. [PubMed] [Google Scholar]
Marks 1992 {published data only}
- Marks RC, Houston T, Thulbourne T. Facet joint injection and facet nerve block: a randomised comparison in 86 patients with chronic low back pain. Pain 1992;49(3):325‐8. [DOI] [PubMed] [Google Scholar]
Mauro 2000 {published data only}
- Mauro GL, Martorana U, Cataldo P, Brancato G, Letizia G. Vitamin B12 in low back pain: a randomised, double‐blind placebo‐controlled study. Eur Rev Med Pharmacol Sci 2000;4(3):53‐8. [PubMed] [Google Scholar]
Mayer 2004 {published data only}
- Mayer TG, Gatchel RJ, Keeley J, McGeary D, Dersh J, Anagnostis C. A randomized clinical trial of treatment for lumbar segmental rigidity. Spine 2004;29(20):2199‐2205. [DOI] [PubMed] [Google Scholar]
Revel 1999 {published data only}
- Revel M, Poiraudeau S, Auleley GR, Payan C, Nguyen M, Chevrot A, et al. Capacity of the clinical picture to characterize low back pain relieved by facet joint anesthesia. Proposed criteria to identify patients with painful facet joints. Spine 1998;23(18):1972‐6. [DOI] [PubMed] [Google Scholar]
Rocco 1989 {published data only}
- Rocco AG, Frank E, Kaul AF, Lipson SJ, Gallo JP. Epidural steroids, epidural morphine and epidural steroids combined with morphine in the treatment of post‐laminectomy syndrome. Pain 1989;36(3):297‐303. [DOI] [PubMed] [Google Scholar]
Serrao 1992 {published data only}
- Serrao JM, Marks RL, Morley SJ, Goodchild CS. Intrathecal midazolam for the treatment of chronic mechanical low back pain: a controlled comparison with epidural steroid in a pilot study. Pain 1992;48(1):5‐12. [DOI] [PubMed] [Google Scholar]
Sonne 1985 {published data only}
- Sonne M, Christensen K, Hansen SE, Jensen EM. Injection of steroids and local anaesthetics as therapy for low back pain. Scand J Rheumatol 1985;14(4):343‐5. [DOI] [PubMed] [Google Scholar]
Takada 2005 {published data only}
- Takada MF, Fukusaki M, Terrao Y, Yamashita K, Inadomi C, Sumikawa K. Comparative efficacy of ropivaciane and bupivacaine for epidural block in outpatients with degenerative spinal disease and low back pain. Pain Clinic 2005;17(3):275‐81. [Google Scholar]
References to studies excluded from this review
Bonetti 2005 {published data only}
- Bonetti M, Fontana M, Cotticelli B, Volta GD, Guidani M, Leonardi M. Intraforaminal O2‐O3 versus periradicular steroidal infiltrations in lower back pain: Randomized controlled study. AJNR Am J Neuroradiol 2005;26(5):996‐1000. [PMC free article] [PubMed] [Google Scholar]
Bush 1991 {published data only}
- Bush K, Hillier S. A controlled study of caudal epidural injections of triamcinolone plus procaine for the management of intractable sciatica. Spine 1991;16(5):572‐5. [DOI] [PubMed] [Google Scholar]
Cuckler 1985 {published data only}
- Cuckler JM, Bernini PA, Wiesel SW, Booth RE, Rothman RH, Pickens GT. The use of epidural steroids in the treatment of lumbar radicular pain. A prospective randomized, double‐blind study. J Bone Joint Surg Am 1985;67(1):63‐6. [PubMed] [Google Scholar]
Dallas 1987 {published data only}
- Dallas TL, Lin RL, Wu WH, Wolskee P. Epidural morphine and methylprednisolone for low back pain. Anesthesiology 1987;67(3):408‐11. [DOI] [PubMed] [Google Scholar]
Fredman 1999 {published data only}
- Fredman B, Zohar E, Ben Nun M, Iraqi R, Jedeikin R, Gepstein R. The effect of repeated epidural sympathetic nerve block on "failed back surgery syndrome" associated chronic low back pain. J Clin Anesth 1999;11(1):46‐51. [DOI] [PubMed] [Google Scholar]
Glynn 1988 {published data only}
- Glynn C, Dawson D, Sanders R. A double‐blind comparison between epidural morphine and epidural clonidine in patients with chronic non‐cancer pain. Pain 1988;34(2):123‐8. [DOI] [PubMed] [Google Scholar]
Hameroff 1981 {published data only}
- Hameroff SR, Crago BR, Womble J, Kanel J. Comparison of bupivacaine, etidocaine, and saline for trigger point therapy. Anesth Analg 1981;60(10):752‐5. [PubMed] [Google Scholar]
Mathews 1987 {published data only}
- Mathews JA, Mills SB, Jenkins VM, Grimes SM, Morkel MJ, Mathews W, et al. Back pain and sciatica: controlled trials of manipulation, traction, sclerosant and epidural injections. Br J Rheumatol 1987 26;6:416‐23. [DOI] [PubMed] [Google Scholar]
Ney 2006 {published data only}
- Ney JP, Difazio M, Sichani A, Monacci W, Foster L, Jabbari B. Treatment of chronic low back pain with succesive injections of Botulinum Toxin A over 6 months: a prospective trial of 60 patients. Clin J Pain 2006;22(4):363‐9. [DOI] [PubMed] [Google Scholar]
Ofluoglu 2007 {published data only}
- Ofluoglu D, Akyuz G, Unay O, Kayhan O. The effect of calcitonin on beta‐emdorphin levels in postmenopausla osteoporotic patients with back pain. Clin Rheumatol 2007;26(1):44‐9. [DOI] [PubMed] [Google Scholar]
Oh 2004 {published data only}
- Oh WS, Shim JC. A randomized controlled trial of radiofrequency denervation of the ramus communicans nerve for chronic discogenic low back pain. Clin J Pain 2004;20(1):55‐60. [DOI] [PubMed] [Google Scholar]
Ongley 1987 {published data only}
- Ongley MJ, Klein RG, Dorman TA, Eek BC, Hubert LJ. A new approach to the treatment of chronic low back pain. Lancet 1987;2(8551):143‐6. [DOI] [PubMed] [Google Scholar]
Pneumaticos 2006 {published data only}
- Pneumaticos SG, Chatziioannou SN, Hipp JA, Moore WH, Esses SI. Low back pain: prediction of short‐term outcome of facet joint injection with bone scintigraphy. Radiology 2006;238(2):693‐8. [DOI] [PubMed] [Google Scholar]
Rogers 1992 {published data only}
- Rogers P, Nash T, Schiller D, Norman J. Epidural steroids for sciatica. Pain Clin 1992;5(2):67‐72. [Google Scholar]
Simmons 1992 {published data only}
- Simmons JW, McMillin JN, Emery SF, Kimmich SJ. Intradiscal steroids. A prospective double‐blind clinical trial. Spine 1992;17(6 Suppl):S172‐5. [PubMed] [Google Scholar]
Stojanovic 2005 {published data only}
- Stojanovic MP, Dey D, Hord ED, Zhou Y, Cohen SP. A prospective crossover comparison study of the single‐needle and multiple‐needle techniques for facet‐joint medial branch blocks. Reg Anesth Pain Med 2005;30(5):484‐90. [DOI] [PubMed] [Google Scholar]
Tuzun 2003 {published data only}
- Tuzun F, Unalan H, Oner N, Ozguzel H, Kirazli Y, Icagasioglu A, et al. Multicenter, randomized, double‐blinded, placebo‐controlled trial of thiocolchicoside in acute low back pain. Joint Bone Spine 2003;70(5):356‐61. [DOI] [PubMed] [Google Scholar]
Additional references
Airaksinen 2006
- Airaksinen O, Brox JI, Cedraschi C, Hildebrandt J, Klaber‐Moffett J, Kovacs F, et al. Chapter 4. European guidelines for the management of chronic nonspecific low back pain. Eur Spine J 2006;15(Suppl 2):S192‐300. [DOI] [PMC free article] [PubMed] [Google Scholar]
Bilir 2006
- Bilir A, Gulec S. Cauda equina syndrome after epidural steroid injection: a case report. J Manipulative Physiol Ther 2006;29(6):492.e1‐3. [DOI] [PubMed] [Google Scholar]
Bombardier 2000
- Bombardier C. Outcome assessments in the evaluation of treatment of spinal disorders: summary and general recommendations. Spine 2000;25(24):3100‐3. [DOI] [PubMed] [Google Scholar]
Cook 1999
- Cook NJ, Hanrahan P, Song S. Paraspinal abscess following facet joint injection. Clin Rheumatol 1999;18(1):52‐3. [DOI] [PubMed] [Google Scholar]
Croft 1998
- Croft PR, Macfarlane GJ, Papageorgiou AC, Thomas E, Silman AJ. Outcome of low back pain in general practice: a prospective study. BMJ 1998;316(7141):1356‐9. [DOI] [PMC free article] [PubMed] [Google Scholar]
Dagenais 2007
- Dagenais S, Yellans MJ, Mar C, Schoene ML. Prolotherapy injections for chronic low back pain. Cochrane Database of Systematic Reviews 2007, Issue 2. [DOI: 10.1002/14651858] [DOI] [PMC free article] [PubMed] [Google Scholar]
Gaul 2005
- Gaul C, Neundorfer B, Winterholler M. Iatrogenis (para‐) spinal abscesses and meningitis following injection therapy for low back pain. Pain 2005;116(3):407‐10. [DOI] [PubMed] [Google Scholar]
Hooten 2006
- Hooten WM, Mizerak A, Carns PE, Huntoon MA. Discitis after lumbar epidural corticosteroid injection: a case report and analysis of the case report literature. Pain Med 2006;7(1):46‐51. [DOI] [PubMed] [Google Scholar]
Houten 2002
- Houten JK, Errico TJ. Paraplegia after lumbosacral nerve root block: report of three cases. Spine J 2002;2(1):70‐5. [DOI] [PubMed] [Google Scholar]
Mooney 1976
- Mooney V, Robertson J. The facet syndrome. Clin Orthoped 1976;115:149‐56. [PubMed] [Google Scholar]
Mooney 1987
- Mooney, V. Facet joint syndrome. In: Jayson MIV editor(s). The lumbar spine: back pain. 3rd Edition. Edinburgh: Chruchill Livingstone, 1987:370‐82. [Google Scholar]
Niemisto 2003
- Niemisto L, Kalso E, Malmivaara A, Seitsalo S, Hurri H. Radiofrequency denervation for neck and back pain: a systematic review of randomized controlled trials. Cochrane Database of Systematic Reviews 2003, Issue 1. [DOI: 10.1002/14651858] [DOI] [PubMed] [Google Scholar]
Salaffi 2004
- Salaffi F, Stancati A, Silvestri CA, Ciapetti A, Grassi W. Minimal clinically important changes in chronic musculoskeletal pain intensity measured on a numerical rating scale. Eur J Pain 2004;8(4):283‐91. [DOI] [PubMed] [Google Scholar]
ter Riet 1990
- ter Riet G, Kleijnen J, Knipschild P. Acupuncture and chronic pain: a criteria‐based meta‐analysis. J Clin Epidemiology 1990;43:1191‐9. [DOI] [PubMed] [Google Scholar]
van Tulder 2003
- Tulder M, Furlan A, Bombardier C, Bouter L, Editorial Board of the Cochrane Collaboration Back Review Group. Updated method guidelines for systematic reviews in the Cochrane Collaboration Back Review Group. Spine 2003;28(12):1290‐9. [DOI] [PubMed] [Google Scholar]
Weingarten 2006
- Weingarten TN, Hooten WM, Huntoon MA. Septic facet joint arthritis after a corticosteroid facet injection. Pain Med 2006;7(1):52‐6. [DOI] [PubMed] [Google Scholar]
Weinstein 2004
- Weinstein J. Absent from work: nature versus nurture. Ann Intern Med 2004;140(2):142‐3. [DOI] [PubMed] [Google Scholar]
References to other published versions of this review
Nelemans 2000
- Nelemans PJ, Bie RA, Vet HCW, Sturmans F. Injection therapy for subacute and chronic low back pain. Cochrane Database of Systematic Reviews 2000, Issue 2. [DOI: 10.1002/14651858] [DOI] [PubMed] [Google Scholar]