Comparative pathogenesis of tissue culture-adapted and wild-type Cowden porcine enteric calicivirus (PEC) in gnotobiotic pigs and induction of diarrhea by intravenous inoculation of wild-type PEC - PubMed (original) (raw)
Comparative Study
Comparative pathogenesis of tissue culture-adapted and wild-type Cowden porcine enteric calicivirus (PEC) in gnotobiotic pigs and induction of diarrhea by intravenous inoculation of wild-type PEC
M Guo et al. J Virol. 2001 Oct.
Abstract
Porcine enteric calicivirus (PEC/Cowden) causes diarrhea in pigs, grows in cell culture, and is morphologically and genetically similar to the Sapporo-like human caliciviruses. Genetic analysis revealed that the tissue culture-adapted (TC) Cowden PEC has one distant and three clustered amino acid substitutions in the capsid region and 2 amino acid changes in the RNA polymerase region compared to wild-type (WT) PEC (M. Guo, K.-O. Chang, M. E. Hardy, Q. Zhang, A. V. Parwani, and L. J. Saif, J. Virol. 73:9625-9631, 1999). In this study, the TC PEC, passaged in a porcine kidney cell line, and the WT PEC, passaged in gnotobiotic (Gn) pigs, were used to orally inoculate 13 4- to 6-day-old Gn pigs. No diarrhea developed in the TC-PEC-exposed pigs, whereas moderate diarrhea developed in the WT-PEC orally inoculated pigs, persisting for 2 to 5 days. Fecal virus shedding persisting for at least 7 days was detected by both reverse transcription (RT)-PCR and antigen-enzyme-linked immunosorbent assay (antigen-ELISA) in both TC-PEC and WT-PEC orally inoculated pigs but not in mock-inoculated pigs. The PEC particles were detected by immunoelectron microscopy (IEM) in intestinal contents from all the WT-PEC-inoculated pigs, but not from the TC-PEC-inoculated pigs. Mild (duodenum and jejunum) or no (ileum) villous atrophy was observed in histologic sections of the small intestines of TC-PEC-inoculated pigs, whereas WT PEC caused mild to severe (duodenum and jejunum) villous atrophy and fusion. Scanning electron microscopy confirmed mild shortening and blunting of villi in the duodenum and jejunum of the TC-PEC-inoculated pigs, in contrast to moderate to severe villous shortening and blunting in the duodenum and jejunum of WT-PEC-inoculated pigs. Higher numbers of PEC antigen-positive villous enterocytes were detected by immunofluorescent (IF) staining in the proximal small intestine of the WT-PEC-inoculated pigs, in contrast to low numbers of PEC antigen-positive enterocytes in only one of four TC-PEC-inoculated pigs. No PEC antigen-positive cells were observed in the colon or extraintestinal tissues of all inoculated pigs or in the small intestine of one mock-inoculated pig. Thus, the TC PEC was at least partially attenuated (no diarrhea, mild lesions) after serial passage in cell culture. In further experiments, three 4- to 6-day-old Gn pigs were intravenously (i.v.) inoculated with WT PEC, and all pigs developed diarrhea and villous atrophy in the small intestines resembling that observed in the orally inoculated pigs. Fecal viral shedding persisting for 8 days was detected by both RT-PCR and antigen-ELISA, and PEC was detected by IEM in feces or intestinal contents. The PEC RNA and antigens (at low titers) were detected in acute-phase sera from all the WT-PEC i.v.-inoculated pigs and also from seven of nine of the WT-PEC orally inoculated pigs. Oral or i.v. inoculation of four additional pigs with the PEC-positive acute-phase sera induced diarrhea, small intestinal lesions, PEC shedding in feces, and seroconversion to PEC, confirming the occurrence of viremia during PEC infection, with infectious PEC present in acute-phase sera. No diarrhea, histopathologic changes, or IF staining in the small intestine or fecal or serum detection of PEC was evident in two pigs i.v. mock-inoculated or a pig inoculated i.v. with inactivated WT PEC. To our knowledge, this is the first report of an attenuated enteric calicivirus, the induction of diarrhea, and intestinal lesions in Gn pigs caused by i.v. inoculation of WT PEC and the presence of viremia following PEC infection.
Figures
FIG. 1
Histologic lesions in the duodenum or jejunum of Gn pigs following oral or intravenous inoculation with PEC/Cowden. Hematoxylin and eosin stain. (A) Normal-appearing, long villi of the jejunum from a mock-inoculated Gn pig (pig 13-5). (B) Villi of the duodenum showing mild villous atrophy change from the TC-PEC orally inoculated pig (pig 4-6). (C) The severe blunting, shortening, and fusion of jejunal villi and crypt hyperplasia from the WT-PEC orally inoculated Gn pig (pig 8-9). (D) The severe, widespread villous atrophy and crypt hyperplasia and elongation in the jejunum from the WT-PEC i.v.-inoculated Gn pig (pig 8-1). Bar, 100 μm.
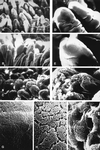
FIG. 2
Scanning electron micrographs of duodenum segments from Gn pigs following oral inoculation with TC PEC/Cowden or WT PEC/Cowden. (A) Typical, long, finger-like villi showing circular transverse grooves on their surface from a mock-inoculated pig (pig 13-5). Magnification, ×130. (B) Finger-like villi with mildly irregular outer surface appearance from the TC-PEC-inoculated pig (pig 4-6). Magnification, ×120. (C) Shortened and fused villi with irregular appearance from the WT-PEC-inoculated pig (pig 4-4). Magnification, ×140. (D) Typical villous apex showing smooth surface from a mock-inoculated pig (pig 13-5). Magnification, ×490. (E) Villous apex with mildly irregular surface appearance from the TC-PEC-inoculated pig (pig 4-6). Magnification, ×510. (F) Shortened and fused villous apex, showing swollen enterocytes, exfoliation, and loss of enterocytes from the WT-PEC-inoculated pig (pig 4-4). Magnification, ×490. (G) Dense, uniform microvillous coat of the enterocytes from a mock-inoculated pig (pig 13-5). Magnification, ×3,870. (H) Smooth microvillous coat of enterocytes, showing mild irregularity, from the TC-PEC-inoculated pig (pig 4-6). Magnification, ×3,870. (I) Irregular microvillous coat of enterocytes from WT-PEC-inoculated pig (pig 4-4). Microvilli are fused, shortened, and more sparse. Magnification, ×3,870.
Similar articles
- Pathogenesis of a genogroup II human norovirus in gnotobiotic pigs.
Cheetham S, Souza M, Meulia T, Grimes S, Han MG, Saif LJ. Cheetham S, et al. J Virol. 2006 Nov;80(21):10372-81. doi: 10.1128/JVI.00809-06. J Virol. 2006. PMID: 17041218 Free PMC article. - Pathogenesis of porcine enteric calicivirus-like virus in four-day-old gnotobiotic pigs.
Flynn WT, Saif LJ, Moorhead PD. Flynn WT, et al. Am J Vet Res. 1988 Jun;49(6):819-25. Am J Vet Res. 1988. PMID: 2840841 - Porcine sapoviruses: Pathogenesis, epidemiology, genetic diversity, and diagnosis.
Nagai M, Wang Q, Oka T, Saif LJ. Nagai M, et al. Virus Res. 2020 Sep;286:198025. doi: 10.1016/j.virusres.2020.198025. Epub 2020 May 26. Virus Res. 2020. PMID: 32470356 Free PMC article. Review. - Porcine enteric caliciviruses: genetic and antigenic relatedness to human caliciviruses, diagnosis and epidemiology.
Wang QH, Costantini V, Saif LJ. Wang QH, et al. Vaccine. 2007 Jul 26;25(30):5453-66. doi: 10.1016/j.vaccine.2006.12.032. Epub 2006 Dec 29. Vaccine. 2007. PMID: 17234307 Free PMC article. Review.
Cited by
- Comprehensive review of human sapoviruses.
Oka T, Wang Q, Katayama K, Saif LJ. Oka T, et al. Clin Microbiol Rev. 2015 Jan;28(1):32-53. doi: 10.1128/CMR.00011-14. Clin Microbiol Rev. 2015. PMID: 25567221 Free PMC article. Review. - Molecular detection of enteric viruses and the genetic characterization of porcine astroviruses and sapoviruses in domestic pigs from Slovakian farms.
Salamunova S, Jackova A, Mandelik R, Novotny J, Vlasakova M, Vilcek S. Salamunova S, et al. BMC Vet Res. 2018 Oct 19;14(1):313. doi: 10.1186/s12917-018-1640-8. BMC Vet Res. 2018. PMID: 30340595 Free PMC article. - Prevalence of noroviruses and sapoviruses in swine of various ages determined by reverse transcription-PCR and microwell hybridization assays.
Wang QH, Souza M, Funk JA, Zhang W, Saif LJ. Wang QH, et al. J Clin Microbiol. 2006 Jun;44(6):2057-62. doi: 10.1128/JCM.02634-05. J Clin Microbiol. 2006. PMID: 16757598 Free PMC article. - Identification of a porcine calicivirus related genetically to human sapoviruses.
Martella V, Lorusso E, Banyai K, Decaro N, Corrente M, Elia G, Cavalli A, Radogna A, Costantini V, Saif LJ, Lavazza A, Di Trani L, Buonavoglia C. Martella V, et al. J Clin Microbiol. 2008 Jun;46(6):1907-13. doi: 10.1128/JCM.00341-08. Epub 2008 Apr 16. J Clin Microbiol. 2008. PMID: 18417656 Free PMC article. - Norovirus: targets and tools in antiviral drug discovery.
Rocha-Pereira J, Neyts J, Jochmans D. Rocha-Pereira J, et al. Biochem Pharmacol. 2014 Sep 1;91(1):1-11. doi: 10.1016/j.bcp.2014.05.021. Epub 2014 Jun 2. Biochem Pharmacol. 2014. PMID: 24893351 Free PMC article. Review.
References
- Agus S G, Dolin R, Wyatt R G, Tousimis A J, Northrup R S. Acute infectious nonbacterial gastroenteritis: intestinal histopathology. Ann Intern Med. 1973;79:18–25. - PubMed
- Blacklow N R, Dolin R, Fedson D S, Dupont H, Northrup R S, Hornick R B, Chanock R M. Acute infectious nonbacterial gastroenteritis: etiology and pathogenesis. Ann Intern Med. 1972;76:993–1008. - PubMed
- Bridger J C. Small viruses associated with gastroenteritis in animals. In: Saif L J, Theil K W, editors. Viral diarrheas of man and animals. Boca Raton, Fla: CRC Press; 1990. pp. 161–182.
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Miscellaneous