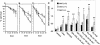Growth retardation and increased apoptosis in mice with homozygous disruption of the Akt1 gene - PubMed (original) (raw)
. 2001 Sep 1;15(17):2203-8.
doi: 10.1101/gad.913901.
P Z Xu, K Gottlob, M L Chen, K Sokol, T Shiyanova, I Roninson, W Weng, R Suzuki, K Tobe, T Kadowaki, N Hay
Affiliations
- PMID: 11544177
- PMCID: PMC312770
- DOI: 10.1101/gad.913901
Growth retardation and increased apoptosis in mice with homozygous disruption of the Akt1 gene
W S Chen et al. Genes Dev. 2001.
Abstract
The serine/threonine kinase Akt has been implicated in the control of cell survival and metabolism. Here we report the disruption of the most ubiquitously expressed member of the akt family of genes, akt1, in the mouse. Akt1(-/-) mice are viable but smaller when compared to wild-type littermates. In addition, the life span of Akt1(-/-) mice, upon exposure to genotoxic stress, is shorter. However, Akt1(-/-) mice do not display a diabetic phenotype. Increased spontaneous apoptosis in testes, and attenuation of spermatogenesis is observed in Akt1(-/-) male mice. Increased spontaneous apoptosis is also observed in the thymi of Akt1(-/-) mice, and Akt1(-/-) thymocytes are more sensitive to apoptosis induced by gamma-irradiation and dexamethasone. Finally, Akt1(-/-) mouse embryo fibroblasts (MEFs) are more susceptible to apoptosis induced by TNF, anti-Fas, UV irradiation, and serum withdrawal.
Figures
Figure 1
Generation of Akt1−/− mice. (A) Targeted disruption strategy to delete the akt1 gene. Shown from top to bottom, the wild-type akt1 allele with indicated exons, the targeting vector, and the disrupted allele. The locations of the PCR primers used to screen for homologous recombination are depicted by small arrows. A short bar spanning the region between exons 4 and 5 depicts the DNA fragment used as a DNA probe for Southern blots. (B) Mouse genotyping by Southern blot analysis. Genomic DNA extracted from mouse tails was digested by _Nco_I and hybridized with a DNA fragment spanning the region between exons 4 and 5. The wild-type fragment is ∼10 kb, whereas the deleted akt1 allele is ∼8.5 kb. (C) Western blot analysis of proteins extracted from mouse embryo fibroblasts (MEFs) isolated from wild-type embryos (+/+), heterozygous (+/−), and homozygous mutant embryos (−/−). (Top panel) Western blot analysis of twofold concentrations of extract using Akt1-specific antibodies (UBI). (Bottom panel) Western blot analysis with Akt1 pleckstrin homology (PH) domain-specific antibodies (UBI). Blots were probed with anti-β-actin as a control. (D) Side view of 5-week-old mice from the same littermate. Wild-type Akt1 (+/+) and Akt1 homozygous deletion (−/−) mice are shown. (E) Relative body weight of wild-type (+/+), heterozygous (+/−), and homozygous (−/−) Akt1 mutant mice 30 d after birth. The weight of each mouse is expressed relative to the mean weight of sex-matched, wild-type littermates in each of seven different litters.
Figure 1
Generation of Akt1−/− mice. (A) Targeted disruption strategy to delete the akt1 gene. Shown from top to bottom, the wild-type akt1 allele with indicated exons, the targeting vector, and the disrupted allele. The locations of the PCR primers used to screen for homologous recombination are depicted by small arrows. A short bar spanning the region between exons 4 and 5 depicts the DNA fragment used as a DNA probe for Southern blots. (B) Mouse genotyping by Southern blot analysis. Genomic DNA extracted from mouse tails was digested by _Nco_I and hybridized with a DNA fragment spanning the region between exons 4 and 5. The wild-type fragment is ∼10 kb, whereas the deleted akt1 allele is ∼8.5 kb. (C) Western blot analysis of proteins extracted from mouse embryo fibroblasts (MEFs) isolated from wild-type embryos (+/+), heterozygous (+/−), and homozygous mutant embryos (−/−). (Top panel) Western blot analysis of twofold concentrations of extract using Akt1-specific antibodies (UBI). (Bottom panel) Western blot analysis with Akt1 pleckstrin homology (PH) domain-specific antibodies (UBI). Blots were probed with anti-β-actin as a control. (D) Side view of 5-week-old mice from the same littermate. Wild-type Akt1 (+/+) and Akt1 homozygous deletion (−/−) mice are shown. (E) Relative body weight of wild-type (+/+), heterozygous (+/−), and homozygous (−/−) Akt1 mutant mice 30 d after birth. The weight of each mouse is expressed relative to the mean weight of sex-matched, wild-type littermates in each of seven different litters.
Figure 1
Generation of Akt1−/− mice. (A) Targeted disruption strategy to delete the akt1 gene. Shown from top to bottom, the wild-type akt1 allele with indicated exons, the targeting vector, and the disrupted allele. The locations of the PCR primers used to screen for homologous recombination are depicted by small arrows. A short bar spanning the region between exons 4 and 5 depicts the DNA fragment used as a DNA probe for Southern blots. (B) Mouse genotyping by Southern blot analysis. Genomic DNA extracted from mouse tails was digested by _Nco_I and hybridized with a DNA fragment spanning the region between exons 4 and 5. The wild-type fragment is ∼10 kb, whereas the deleted akt1 allele is ∼8.5 kb. (C) Western blot analysis of proteins extracted from mouse embryo fibroblasts (MEFs) isolated from wild-type embryos (+/+), heterozygous (+/−), and homozygous mutant embryos (−/−). (Top panel) Western blot analysis of twofold concentrations of extract using Akt1-specific antibodies (UBI). (Bottom panel) Western blot analysis with Akt1 pleckstrin homology (PH) domain-specific antibodies (UBI). Blots were probed with anti-β-actin as a control. (D) Side view of 5-week-old mice from the same littermate. Wild-type Akt1 (+/+) and Akt1 homozygous deletion (−/−) mice are shown. (E) Relative body weight of wild-type (+/+), heterozygous (+/−), and homozygous (−/−) Akt1 mutant mice 30 d after birth. The weight of each mouse is expressed relative to the mean weight of sex-matched, wild-type littermates in each of seven different litters.
Figure 2
(A) Akt1−/− mice are sensitized to genotoxic stress. Survival of Akt1+/+ and Akt1−/− mice was scored for 21 d after exposure to 10 Gy of γ-irradiation. (B) Oral glucose tolerance test (OGTT). Plasma glucose levels of 3-month-old Akt1−/− male mice and their wild-type littermates were measured at 15, 30, 60, and 120 min after oral glucose load. The values are expressed as the mean ±SE (n = 4–6 for each genotype). Similar results were obtained in three independent experiments.
Figure 3
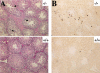
Spontaneous apoptosis in the testes and thymus of Akt1−/− mice. (A) Morphology and histology of testes from Akt1+/+ and Akt1−/−. Testes were fixed with 4% paraformaldehyde, processed with paraffin, sectioned, and stained with hematoxylin/eosin. (Top panel) Seminiferous tubules of Akt null (−/−); (bottom panel) seminiferous tubules of wild-type (+/+) littermate (200× magnification). Arrows indicate multinucleated giant cells. (B) Spontaneous apoptosis in tubules from Akt1−/− mice. Comparison of TUNEL-positive cells in tubules from Akt1−/− mice (top panel) and from wild-type littermate (bottom panel). Cells with dark stained nuclei are apoptotic cells (200× magnification). (C) Sperm count of Akt1+/+ male mice (n = 8, ±SE) and Akt1−/− male mice (n = 10, ±SE; P < 0.001). (D) Spontaneous apoptosis is observed throughout the thymi of Akt1−/− mice. TUNEL assays were performed on sections from thymi of Akt1−/− (top panel) and wild-type (bottom panel) mice. Cells with dark stained nuclei are apoptotic cells. Magnification, 400×. (E) Percentage of apoptotic cells as measured by TUNEL-positive cells in sections from thymi of age-matched (1–2 mo old), wild-type, and Akt1−/− mice as measured by counting 10 fields of each section (P < 0.001).
Figure 3
Spontaneous apoptosis in the testes and thymus of Akt1−/− mice. (A) Morphology and histology of testes from Akt1+/+ and Akt1−/−. Testes were fixed with 4% paraformaldehyde, processed with paraffin, sectioned, and stained with hematoxylin/eosin. (Top panel) Seminiferous tubules of Akt null (−/−); (bottom panel) seminiferous tubules of wild-type (+/+) littermate (200× magnification). Arrows indicate multinucleated giant cells. (B) Spontaneous apoptosis in tubules from Akt1−/− mice. Comparison of TUNEL-positive cells in tubules from Akt1−/− mice (top panel) and from wild-type littermate (bottom panel). Cells with dark stained nuclei are apoptotic cells (200× magnification). (C) Sperm count of Akt1+/+ male mice (n = 8, ±SE) and Akt1−/− male mice (n = 10, ±SE; P < 0.001). (D) Spontaneous apoptosis is observed throughout the thymi of Akt1−/− mice. TUNEL assays were performed on sections from thymi of Akt1−/− (top panel) and wild-type (bottom panel) mice. Cells with dark stained nuclei are apoptotic cells. Magnification, 400×. (E) Percentage of apoptotic cells as measured by TUNEL-positive cells in sections from thymi of age-matched (1–2 mo old), wild-type, and Akt1−/− mice as measured by counting 10 fields of each section (P < 0.001).
Figure 3
Spontaneous apoptosis in the testes and thymus of Akt1−/− mice. (A) Morphology and histology of testes from Akt1+/+ and Akt1−/−. Testes were fixed with 4% paraformaldehyde, processed with paraffin, sectioned, and stained with hematoxylin/eosin. (Top panel) Seminiferous tubules of Akt null (−/−); (bottom panel) seminiferous tubules of wild-type (+/+) littermate (200× magnification). Arrows indicate multinucleated giant cells. (B) Spontaneous apoptosis in tubules from Akt1−/− mice. Comparison of TUNEL-positive cells in tubules from Akt1−/− mice (top panel) and from wild-type littermate (bottom panel). Cells with dark stained nuclei are apoptotic cells (200× magnification). (C) Sperm count of Akt1+/+ male mice (n = 8, ±SE) and Akt1−/− male mice (n = 10, ±SE; P < 0.001). (D) Spontaneous apoptosis is observed throughout the thymi of Akt1−/− mice. TUNEL assays were performed on sections from thymi of Akt1−/− (top panel) and wild-type (bottom panel) mice. Cells with dark stained nuclei are apoptotic cells. Magnification, 400×. (E) Percentage of apoptotic cells as measured by TUNEL-positive cells in sections from thymi of age-matched (1–2 mo old), wild-type, and Akt1−/− mice as measured by counting 10 fields of each section (P < 0.001).
Figure 3
Spontaneous apoptosis in the testes and thymus of Akt1−/− mice. (A) Morphology and histology of testes from Akt1+/+ and Akt1−/−. Testes were fixed with 4% paraformaldehyde, processed with paraffin, sectioned, and stained with hematoxylin/eosin. (Top panel) Seminiferous tubules of Akt null (−/−); (bottom panel) seminiferous tubules of wild-type (+/+) littermate (200× magnification). Arrows indicate multinucleated giant cells. (B) Spontaneous apoptosis in tubules from Akt1−/− mice. Comparison of TUNEL-positive cells in tubules from Akt1−/− mice (top panel) and from wild-type littermate (bottom panel). Cells with dark stained nuclei are apoptotic cells (200× magnification). (C) Sperm count of Akt1+/+ male mice (n = 8, ±SE) and Akt1−/− male mice (n = 10, ±SE; P < 0.001). (D) Spontaneous apoptosis is observed throughout the thymi of Akt1−/− mice. TUNEL assays were performed on sections from thymi of Akt1−/− (top panel) and wild-type (bottom panel) mice. Cells with dark stained nuclei are apoptotic cells. Magnification, 400×. (E) Percentage of apoptotic cells as measured by TUNEL-positive cells in sections from thymi of age-matched (1–2 mo old), wild-type, and Akt1−/− mice as measured by counting 10 fields of each section (P < 0.001).
Figure 4
Akt1−/− thymocytes and MEFs are more susceptible to apoptosis. (A) Survival of thymocytes in the absence of treatment. (B) Survival of thymocytes following exposure to γ-irradiation (500 rad). (C) Survival of thymocytes upon treatment with 1 μM of dexamethasone. Values represent the average of four independent experiments of thymocytes derived from three mice of each genotype (±SE). (█) Percentage survival of wild-type thymocytes; (□) percentage survival of Akt1−/− thymocytes as measured by trypan blue exclusion. (D) Akt1−/− MEFs are sensitized to apoptosis by various apoptotic stimuli. MEFs derived from Akt1+/+, Akt1+/−, and Akt1−/− embryos were treated with 1 μg/mL anti-Fas antibody (Pharmingen), 10 ng/mL TNFα (Life Technologies), 5 μg/mL cycloheximide CHX (Sigma), UV irradiation (60 or 120 J/m2) in the presence (20% FCS) or absence (0% FCS) of fetal calf serum in the medium and apoptosis was quantitated 24 h following treatment. Apoptosis induced just by serum deprivation (0% FCS) was quantitated 48 h following serum deprivation. Apoptosis was measured by quantitation of DNA fragmentation using the cell death detection ELISA method (Boehringer Mannheim). The average of 3 independent experiments (±SE) is shown (*, P < 0.01; **, P < 0.05).
Similar articles
- Overexpression of Akt/AKT can modulate chemotherapy-induced apoptosis.
Page C, Lin HJ, Jin Y, Castle VP, Nunez G, Huang M, Lin J. Page C, et al. Anticancer Res. 2000 Jan-Feb;20(1A):407-16. Anticancer Res. 2000. PMID: 10769688 - Functions of AKT1 and AKT2 potassium channels determined by studies of single and double mutants of Arabidopsis.
Dennison KL, Robertson WR, Lewis BD, Hirsch RE, Sussman MR, Spalding EP. Dennison KL, et al. Plant Physiol. 2001 Nov;127(3):1012-9. Plant Physiol. 2001. PMID: 11706182 Free PMC article. - Akt1 suppresses radiation-induced germ cell apoptosis in vivo.
Rasoulpour T, DiPalma K, Kolvek B, Hixon M. Rasoulpour T, et al. Endocrinology. 2006 Sep;147(9):4213-21. doi: 10.1210/en.2006-0174. Epub 2006 Jun 8. Endocrinology. 2006. PMID: 16763066 - Tumor necrosis factor-alpha and CD95 ligation suppress erythropoiesis in Fanconi anemia C gene knockout mice.
Otsuki T, Nagakura S, Wang J, Bloom M, Grompe M, Liu JM. Otsuki T, et al. J Cell Physiol. 1999 Apr;179(1):79-86. doi: 10.1002/(SICI)1097-4652(199904)179:1<79::AID-JCP10>3.0.CO;2-O. J Cell Physiol. 1999. PMID: 10082135 - Akt2, but not Akt1, is required for cell survival by inhibiting activation of JNK and p38 after UV irradiation.
Kim MA, Kim HJ, Jee HJ, Kim AJ, Bae YS, Bae SS, Yun J. Kim MA, et al. Oncogene. 2009 Mar 5;28(9):1241-7. doi: 10.1038/onc.2008.487. Epub 2009 Jan 19. Oncogene. 2009. PMID: 19151757
Cited by
- Akt: a double-edged sword in cell proliferation and genome stability.
Xu N, Lao Y, Zhang Y, Gillespie DA. Xu N, et al. J Oncol. 2012;2012:951724. doi: 10.1155/2012/951724. Epub 2012 Mar 15. J Oncol. 2012. PMID: 22481935 Free PMC article. - Regulation of Hematopoietic Stem Cell Fate and Malignancy.
Cho HJ, Lee J, Yoon SR, Lee HG, Jung H. Cho HJ, et al. Int J Mol Sci. 2020 Jul 6;21(13):4780. doi: 10.3390/ijms21134780. Int J Mol Sci. 2020. PMID: 32640596 Free PMC article. Review. - Regulation of DNA Damage Response and Homologous Recombination Repair by microRNA in Human Cells Exposed to Ionizing Radiation.
Szatkowska M, Krupa R. Szatkowska M, et al. Cancers (Basel). 2020 Jul 8;12(7):1838. doi: 10.3390/cancers12071838. Cancers (Basel). 2020. PMID: 32650508 Free PMC article. Review. - AKT1 Activation is Obligatory for Spontaneous BCC Tumor Growth in a Murine Model that Mimics Some Features of Basal Cell Nevus Syndrome.
Kim AL, Back JH, Zhu Y, Tang X, Yardley NP, Kim KJ, Athar M, Bickers DR. Kim AL, et al. Cancer Prev Res (Phila). 2016 Oct;9(10):794-802. doi: 10.1158/1940-6207.CAPR-16-0066. Epub 2016 Jul 7. Cancer Prev Res (Phila). 2016. PMID: 27388747 Free PMC article. - Impaired Mitochondrial Fat Oxidation Induces FGF21 in Muscle.
Vandanmagsar B, Warfel JD, Wicks SE, Ghosh S, Salbaum JM, Burk D, Dubuisson OS, Mendoza TM, Zhang J, Noland RC, Mynatt RL. Vandanmagsar B, et al. Cell Rep. 2016 May 24;15(8):1686-99. doi: 10.1016/j.celrep.2016.04.057. Epub 2016 May 12. Cell Rep. 2016. PMID: 27184848 Free PMC article.
References
- Alessi DR, Cohen P. Mechanism of activation and function of protein kinase B. Curr Opin Genet Dev. 1998;8:55–62. - PubMed
- Altomare DA, Guo K, Cheng JQ, Sonoda G, Walsh K, Testa JR. Cloning, chromosomal localization and expression analysis of the mouse Akt2 oncogene. Oncogene. 1995;11:1055–1060. - PubMed
- Altomare DA, Lyons GE, Mitsuuchi Y, Cheng JQ, Testa JR. Akt2 mRNA is highly expressed in embryonic brown fat and the AKT2 kinase is activated by insulin. Oncogene. 1998;116:2407–2411. - PubMed
- Barthel A, Okino ST, Liao J, Nakatani K, Li J, Whitlock J, Jr, Roth RA. Regulation of GLUT1 gene transcription by the serine/threonine kinase Akt1. J Biol Chem. 1999;274:20281–20286. - PubMed
- Blume-Jensen P, Jiang G, Hyman R, Lee KF, O'Gorman S, Hunter T. Kit/stem cell factor receptor-induced activation of phosphatidylinositol 3′-kinase is essential for male fertility. Nat Genet. 2000;24:157–162. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous