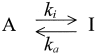Na self inhibition of human epithelial Na channel: temperature dependence and effect of extracellular proteases - PubMed (original) (raw)
Na self inhibition of human epithelial Na channel: temperature dependence and effect of extracellular proteases
Ahmed Chraïbi et al. J Gen Physiol. 2002 Aug.
Abstract
The regulation of the open probability of the epithelial Na(+) channel (ENaC) by the extracellular concentration of Na(+), a phenomenon called "Na(+) self inhibition," has been well described in several natural tight epithelia, but its molecular mechanism is not known. We have studied the kinetics of Na(+) self inhibition on human ENaC expressed in Xenopus oocytes. Rapid removal of amiloride or rapid increase in the extracellular Na(+) concentration from 1 to 100 mM resulted in a peak inward current followed by a decline to a lower quasi-steady-state current. The rate of current decline and the steady-state level were temperature dependent and the current transient could be well explained by a two-state (active-inactive) model with a weakly temperature-dependent (Q(10)act = 1.5) activation rate and a strongly temperature-dependant (Q(10)inact = 8.0) inactivation rate. The steep temperature dependence of the inactivation rate resulted in the paradoxical decrease in the steady-state amiloride-sensitive current at high temperature. Na(+) self inhibition depended only on the extracellular Na(+) concentration but not on the amplitude of the inward current, and it was observed as a decrease of the conductance at the reversal potential for Na(+) as well as a reduction of Na(+) outward current. Self inhibition could be prevented by exposure to extracellular protease, a treatment known to activate ENaC or by treatment with p-CMB. After protease treatment, the amiloride-sensitive current displayed the expected increase with rising temperature. These results indicate that Na(+) self inhibition is an intrinsic property of sodium channels resulting from the expression of the alpha, beta, and gamma subunits of human ENaC in Xenopus oocyte. The extracellular Na(+)-dependent inactivation has a large energy of activation and can be abolished by treatment with extracellular proteases.
Figures
(SCHEME I)
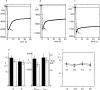
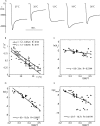
Figure 1.
Sodium self inhibition with ENaC expressed in Xenopus oocyte. Examples of current recordings at −60 mV holding potential showing the peak current and its relaxation after (A) the sudden removal of amiloride in the presence of 100 mM Na+, or (B and C) a 1–100 mM increase in Na+ concentration. The original data (collected at the rate of 5 points/s) are indicated by the small black circles. The solid line, representing the best fitting curve (according to Eq. 1 presented in the
materials and methods
) was obtained by fitting the current values from the inflexion point (arrow) of the current amplitude decline to the 40-s point (∼160 data points). In the example of A, the best fitting parameters were (a) the maximal current extrapolated to time t = 0, I max 8596 nA, (b) the activation constant k a 0.086 s−1, (c) the inactivation constant k i 0.127 s−1, and (d) the run down factor k down 0.0016. The origin of time was set to the inflexion point of the rising phase of the current amplitude (indicated by a black diamond). C presents a similar recording obtained with an oocyte-expressing rat αβγ ENaC following the same experimental maneuver as in B, i.e., a sudden increase of the Na+ concentration from 1–100 mM. A current relaxation with a similar time course as with human ENaC (A and B) was also observed after the initial peak; however, the amplitude of the decrease was smaller. D shows the mean values of the kinetics parameters k a, k i, k down, and I max obtained by model fitting in 11 and 6 measurements using amiloride removal (black bars) and Na+ concentration increase (hatched bars), respectively. There were no statistically significant differences between the mean values obtained by amiloride removal (black bars) and those obtained in Na+ concentration increase experiments. E shows the results obtained for repeated measurements after amiloride removal under the same condition on the same oocyte. A recuperation period of at least 60 s in the amiloride-containing solution was allowed between each measurement. The kinetic parameters k a (black circles) and k i (black triangles) obtained in 10 first, second, and third measurements and in 8 fourth measurements are shown. There was no significant trend with time nor statistically significant differences between the values obtained at the first, second, third or fourth measurement for both these parameters. The number of measurements is indicated in parentheses.
Figure 5.
Effect of protease treatment and _p-_CMB on self inhibition. Mean amiloride-sensitive current during the 30 s after a sudden increase from 1 to 100 mM Na+ (at time t = 0) are shown as absolute values in A and as values relative to the peak current in B. In A, the experimental group is represented by black squares and open squares, before and after trypsin treatment (5 min in a 5 μg/ml trypsin solution), respectively. The results of a subgroup in which trypsin was added alone (n = 11) and another subgroup in which trypsin was added in the presence of 10 μM amiloride (n = 4) were similar and are pooled. The control group (n = 8) is represented by black circles and black triangles, respectively, before and after a 5-min incubation in a solution without trypsin. The dotted line shows the theoretical curve with the best fitting parameters obtained from the mean current data of the experimental group before trypsin treatment (k a 0.15 s−1, k i 0.21 s−1, k down 0.0061, I max 9979 nA). The extrapolated current value at time 0 (I max) is indicated by a star. Because most of the variation in the current values originate in the variable level of expression of the channel in individual oocytes, we have also presented the same results as values normalized to the highest current value recorded during each 30-s measurement (peak current). In this panel the values of the trypsin and trypsin+amiloride groups are represented separately by squares and diamonds, respectively (black symbols before trypsin, open symbols after trypsin). Presence of amiloride during the exposure to trypsin did not make any detectable difference. C and D show the mean current values after a 1–100 mM Na+ concentration change recorded in the absence and in the presence of 0.5 mM _p-_CMB. In all four panels, the solid line is in each case the mean current value (measured at a sampling rate of 3 s−1) and the errors bars indicating ±SEM are shown with the symbols every 2 s. The data are the results of nine pairs of measurements; control are indicated by circles and measurements with _p-_CMB by white squares.
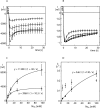
Figure 4.
Sodium concentration dependence of self inhibition. A shows the mean amiloride-sensitive current recorded after 1 to 4 (losanges), 1 to 12 (triangles), 1 to 30 (circles), and 1 to 100 (squares) mM Na+ concentration jumps. The solid line is in each case the mean of nine current values (measured at 0.3-s interval) and the errors bars indicate the SEM every 2 s. Because most of the variation in the current values originate in the variable level of expression of the channel in individual oocytes, we have also presented the same results as values normalized to the highest current value (peak current) recorded during each 30 s measurement in B. (C) Na+ concentration dependence of the extrapolated maximal current (I max, circles) obtained by model fitting of the individual data and of the steady-state current (triangles), i.e., the current value 30 s after the Na+ concentration change. (D) Na+ concentration dependence of the activation and inactivation constant (k a, triangles and k i, circles). In C and D, the solid line is the Michaelis-Menten relation with the best fitting parameters indicated in the equations printed in the graph. This relation is not shown for k a, because the concentration dependence was very weak for this parameter.
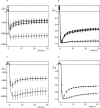
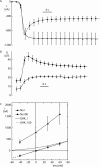
Figure 2.
Temperature dependence of the self inhibition. A shows five sequential current recordings of the effect of amiloride removal in a 100 mM Na+ solution obtained with the same oocyte at different temperatures. Intervals of at least 1 min, during which the temperature was changed, were allowed to occur between each episode of amiloride removal (not depicted). The peak current (I peak, the largest recorded inward current value) and the steady-state current value (I ss, i.e., the value measured at 40 s), and the kinetic parameters (k a, k i, k down, and I max) of the current relaxation were obtained from each current recording. B shows the temperature dependence of steady-state current reported either to the peak current (I ss/I peak, open triangles) or to the maximal current I ss/I max, black dots). C is the Arrhenius plots of the activation rate (k a), D is the plot of the inactivation rate (k i), and E the plot of the run down factor (k down).
Figure 3.
Self inhibition is not related to the inward Na+ current. (A) Mean values of the amiloride-sensitive current recorded upon removal of amiloride at a membrane potential of −15 mV in a 100 mM Na+ solution (black triangles, n = 22) and at −120 mV in a 2 mM Na+ solution (black filled circles, n = 29). The SE values are indicated only for points at 2-s intervals. (B) Mean conductance of the whole oocyte membrane during amiloride removal in a 100 mM (circles, n = 15) and in a 1 mM (triangles, n = 9) Na+ solution. The first data points show the conductance in the presence of 10 μM amiloride. The holding potential was set at the reversal potential of the amiloride-sensitive current and the conductance was measured, three times per second, from the current changes induced by a 10 mV (from 5 to −5 mV from the holding potential) voltage step. (C) Amiloride-sensitive I-V curves for potentials ranging from −40 to 60 mV were obtained by subtraction of I-V curves obtained in the absence and in the presence of 50 μM amiloride, in 1 mM (n = 11) and 100 mM (n = 11) Na+ solutions. The dashed line represents the Goldman-Hodgkin-Katz (GHK) current equation: _I_=P
, for a 100 mM extracellular Na+ concentration ([_Na_]o) and with the following best fitting parameters: Na+ permeability (P) = 1.47 × 10−4 cm.s−1 and intracellular Na+ concentration ([_Na_]i) = 94 mM (V is the membrane potential, and R, T, and F have their usual meaning). The dotted line shows the current values calculated from the GHK equation with the same parameters when the extracellular Na+ concentration has been set to 1 mM.
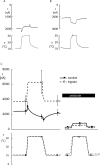
Figure 6.
Effect of temperature on the steady-state amiloride-sensitive current. A shows current and temperature recordings illustrating the biphasic effect of a sudden rise or decrease of temperature in a control oocyte expressing hENaC. B shows the effect of the same change of temperature on an oocyte previously exposed for 10 min to 2 μg/ml trypsin. In this case, the response was monophasic with an increase in current amplitude with higher temperature. C is a scheme showing the summary of measurement on 11 control oocytes and 7 oocytes exposed to trypsin. The thick continuous line (control) and dashed line (trypsin) are schematized current traces and the symbols (black circle, control; open square, trypsin) indicate the mean values of the amiloride-sensitive current recorded at the points indicated by the trace. The right part of the figure shows the effect of temperature change in the presence of 10 μM amiloride. These values were similar in the control and trypsin-treated group. The left part of the figure shows the mean values of the amiloride-sensitive current (i.e., the current values after subtraction of the values recorded in the presence of amiloride). The lower part of the figure shows the mean values of the recorded temperature. Some error bars are not visible because they are smaller than the symbols.
Similar articles
- Feedback inhibition of rat amiloride-sensitive epithelial sodium channels expressed in Xenopus laevis oocytes.
Abriel H, Horisberger JD. Abriel H, et al. J Physiol. 1999 Apr 1;516 ( Pt 1)(Pt 1):31-43. doi: 10.1111/j.1469-7793.1999.031aa.x. J Physiol. 1999. PMID: 10066920 Free PMC article. - Sodium self-inhibition of human epithelial sodium channel: selectivity and affinity of the extracellular sodium sensing site.
Bize V, Horisberger JD. Bize V, et al. Am J Physiol Renal Physiol. 2007 Oct;293(4):F1137-46. doi: 10.1152/ajprenal.00100.2007. Epub 2007 Aug 1. Am J Physiol Renal Physiol. 2007. PMID: 17670907 - Endogenous protease activation of ENaC: effect of serine protease inhibition on ENaC single channel properties.
Adebamiro A, Cheng Y, Johnson JP, Bridges RJ. Adebamiro A, et al. J Gen Physiol. 2005 Oct;126(4):339-52. doi: 10.1085/jgp.200509285. J Gen Physiol. 2005. PMID: 16186561 Free PMC article. - ENaC activation by proteases.
Anand D, Hummler E, Rickman OJ. Anand D, et al. Acta Physiol (Oxf). 2022 May;235(1):e13811. doi: 10.1111/apha.13811. Epub 2022 Mar 21. Acta Physiol (Oxf). 2022. PMID: 35276025 Free PMC article. Review. - A nonconventional look at ionic fluxes in the skin: lessons from genetically modified mice.
Guitard M, Leyvraz C, Hummler E. Guitard M, et al. News Physiol Sci. 2004 Apr;19:75-9. doi: 10.1152/nips.01503.2003. News Physiol Sci. 2004. PMID: 15016907 Review.
Cited by
- Epithelial Na + Channels Function as Extracellular Sensors.
Kashlan OB, Wang XP, Sheng S, Kleyman TR. Kashlan OB, et al. Compr Physiol. 2024 Mar 29;14(2):1-41. doi: 10.1002/cphy.c230015. Compr Physiol. 2024. PMID: 39109974 Review. - Varying Selection Pressure for a Na+ Sensing Site in Epithelial Na+ Channel Subunits Reflect Divergent Roles in Na+ Homeostasis.
Wang XP, Srinivasan P, El Hamdaoui M, Blobner BM, Grytz R, Kashlan OB. Wang XP, et al. Mol Biol Evol. 2024 Aug 2;41(8):msae162. doi: 10.1093/molbev/msae162. Mol Biol Evol. 2024. PMID: 39101592 Free PMC article. - Recording Sodium Self-Inhibition of Epithelial Sodium Channels Using Automated Electrophysiology in Xenopus Oocytes.
Lawong RY, May F, Etang EC, Vorrat P, George J, Weder J, Kockler D, Preller M, Althaus M. Lawong RY, et al. Membranes (Basel). 2023 May 19;13(5):529. doi: 10.3390/membranes13050529. Membranes (Basel). 2023. PMID: 37233590 Free PMC article. - Vascular mechanotransduction.
Davis MJ, Earley S, Li YS, Chien S. Davis MJ, et al. Physiol Rev. 2023 Apr 1;103(2):1247-1421. doi: 10.1152/physrev.00053.2021. Epub 2023 Jan 5. Physiol Rev. 2023. PMID: 36603156 Free PMC article. Review. - Accessibility of ENaC extracellular domain central core residues.
Zhang L, Wang X, Chen J, Kleyman TR, Sheng S. Zhang L, et al. J Biol Chem. 2022 May;298(5):101860. doi: 10.1016/j.jbc.2022.101860. Epub 2022 Mar 23. J Biol Chem. 2022. PMID: 35339489 Free PMC article.
References
- Canessa, C.M., J.-D. Horisberger, and B.C. Rossier. 1993. Functional cloning of the epithelial sodium channel: relation with genes involved in neurodegeneration. Nature. 361:467–470. - PubMed
- Canessa, C.M., L. Schild, G. Buell, B. Thorens, Y. Gautshi, J.-D. Horisberger, and B.C. Rossier. 1994. The amiloride-sensitive epithelial sodium channel is made of three homologous subunits. Nature. 367:463–467. - PubMed