The effect of cholesterol on the lateral diffusion of phospholipids in oriented bilayers - PubMed (original) (raw)
Comparative Study
The effect of cholesterol on the lateral diffusion of phospholipids in oriented bilayers
Andrey Filippov et al. Biophys J. 2003 May.
Abstract
Pulsed field gradient NMR was utilized to directly determine the lipid lateral diffusion coefficient for the following macroscopically aligned bilayers: dimyristoylphosphatidylcholine (DMPC), sphingomyelin (SM), palmitoyloleoylphosphatidylcholine (POPC), and dioleoylphosphatidylcholine (DOPC) with addition of cholesterol (CHOL) up to approximately 40 mol %. The observed effect of cholesterol on the lipid lateral diffusion is interpreted in terms of the different diffusion coefficients obtained in the liquid ordered (l(o)) and the liquid disordered (l(d)) phases occurring in the phase diagrams. Generally, the lipid lateral diffusion coefficient decreases linearly with increasing CHOL concentration in the l(d) phase for the PC-systems, while it is almost independent of CHOL for the SM-system. In this region the temperature dependence of the diffusion was always of the Arrhenius type with apparent activation energies (E(A)) in the range of 28-40 kJ/mol. The l(o) phase was characterized by smaller diffusion coefficients and weak or no dependence on the CHOL content. The E(A) for this phase was significantly larger (55-65 kJ/mol) than for the l(d) phase. The diffusion coefficients in the two-phase regions were compatible with a fast exchange between the l(d) and l(o) regions in the bilayer on the timescale of the NMR experiment (100 ms). Thus, strong evidence has been obtained that fluid domains (with size of micro m or less) with high molecular ordering are formed within a single lipid bilayer. These domains may play an important role for proteins involved in membrane functioning frequently discussed in the recent literature. The phase diagrams obtained from the analysis of the diffusion data are in qualitative agreement with earlier published ones for the SM/CHOL and DMPC/CHOL systems. For the DOPC/CHOL and the POPC/CHOL systems no two-phase behavior were observed, and the obtained E(A):s indicate that these systems are in the l(d) phase at all CHOL contents for temperatures above 25 degrees C.
Figures
FIGURE 1
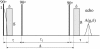
A schematic of the stimulated echo pulse sequence used in this work. RF pulses are shown as filled rectangles, whereas the gradient pulses are hatched. The NMR signal is acquired from the top of the echo.
FIGURE 2
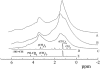
1H-NMR spectra of POPC (A), DOPC (B), and SM (C) in oriented bilayers with 20 wt % 2H2O obtained from the pfg-NMR stimulated echo after suppression of the signal from the residual water protons. T = 333 K. Parameters of the stimulated echo pulse sequence are δ = 1.5 ms, τ = 11 ms, and _τ_1 = 100 ms. Solution 1H-NMR spectra of DOPC (D) and SM (E) are shown below for reference.
FIGURE 3
Diffusion decay of an oriented POPC bilayer with 30 wt % 2H2O at 303 K and a diffusion time of 100 ms. The early, fast decay is attributed to water diffusion, whereas the latter, slow decay corresponds to the lipid diffusion.
FIGURE 4
(A) Phase diagram of the SM/CHOL system according to Sankaram and Thompson (1990). The circles mark experimental points investigated by us. Open and filled circles correspond to samples from one-phase and two-phase regions, respectively. (B) Lateral diffusion coefficients at different CHOL concentrations for the SM/CHOL system with 35 wt % 2H2O and at 313 K (circle), 318 K (triangle top down), 323 K (square), 328 K (diamond), and 333 K (triangle top up). The thick solid lines are estimations of the extension of the two-phase area. The solid and dotted lines are linear estimations of _D_L in the one-phase and two-phase areas, respectively. (C) Arrhenius plots of the temperature dependence of the lateral diffusion coefficient for the SM/CHOL system with 35 wt % 2H2O and CHOL contents varying as 0 (circles), 5 (triangle top down), 10.5 (square), 16.3 (diamond), 31 (triangle top up), and 42.5 mol % (hexagon). The solid lines are Arrhenius fits to the data in the one-phase regions, whereas the dotted lines are the calculated _D_L according to the lever rule in the two-phase area. In these calculations the phase borders obtained by us have been used (thick lines in Fig. 4 B).
FIGURE 4
(A) Phase diagram of the SM/CHOL system according to Sankaram and Thompson (1990). The circles mark experimental points investigated by us. Open and filled circles correspond to samples from one-phase and two-phase regions, respectively. (B) Lateral diffusion coefficients at different CHOL concentrations for the SM/CHOL system with 35 wt % 2H2O and at 313 K (circle), 318 K (triangle top down), 323 K (square), 328 K (diamond), and 333 K (triangle top up). The thick solid lines are estimations of the extension of the two-phase area. The solid and dotted lines are linear estimations of _D_L in the one-phase and two-phase areas, respectively. (C) Arrhenius plots of the temperature dependence of the lateral diffusion coefficient for the SM/CHOL system with 35 wt % 2H2O and CHOL contents varying as 0 (circles), 5 (triangle top down), 10.5 (square), 16.3 (diamond), 31 (triangle top up), and 42.5 mol % (hexagon). The solid lines are Arrhenius fits to the data in the one-phase regions, whereas the dotted lines are the calculated _D_L according to the lever rule in the two-phase area. In these calculations the phase borders obtained by us have been used (thick lines in Fig. 4 B).
FIGURE 4
(A) Phase diagram of the SM/CHOL system according to Sankaram and Thompson (1990). The circles mark experimental points investigated by us. Open and filled circles correspond to samples from one-phase and two-phase regions, respectively. (B) Lateral diffusion coefficients at different CHOL concentrations for the SM/CHOL system with 35 wt % 2H2O and at 313 K (circle), 318 K (triangle top down), 323 K (square), 328 K (diamond), and 333 K (triangle top up). The thick solid lines are estimations of the extension of the two-phase area. The solid and dotted lines are linear estimations of _D_L in the one-phase and two-phase areas, respectively. (C) Arrhenius plots of the temperature dependence of the lateral diffusion coefficient for the SM/CHOL system with 35 wt % 2H2O and CHOL contents varying as 0 (circles), 5 (triangle top down), 10.5 (square), 16.3 (diamond), 31 (triangle top up), and 42.5 mol % (hexagon). The solid lines are Arrhenius fits to the data in the one-phase regions, whereas the dotted lines are the calculated _D_L according to the lever rule in the two-phase area. In these calculations the phase borders obtained by us have been used (thick lines in Fig. 4 B).
FIGURE 5
(A) Phase diagram of the DMPC/CHOL system according to Almeida et al. (1992). The crosses mark experimental points investigated by us. (B) Lipid lateral diffusion coefficients at different CHOL concentrations for the DMPC/CHOL system with 35 wt % 2H2O and at 298 K (circle), 303 K (triangle top down), 308 K (square), 313 K (diamond), 323 K (triangle top up), and 333 K (hexagon). The solid lines are drawn through the concentrations for which _D_L is approximately constant. These points are tentatively assigned to the _l_o one-phase area. (C) Arrhenius plots of the temperature dependence of the lipid lateral diffusion coefficient for the DMPC/CHOL system with 35 wt % 2H2O and varying CHOL contents of 0 (circles), 5 (triangle top down), 15 (square), 25 (diamond), and 35 mol % (triangle top up). The solid lines are Arrhenius fits to the data in the one-phase regions, whereas the dotted lines are the calculated _D_L according to the lever rule in the two-phase area. In these calculations the phase borders given by Almeida et al. (1992) have been used (thick lines in Fig. 5, A and B).
FIGURE 5
(A) Phase diagram of the DMPC/CHOL system according to Almeida et al. (1992). The crosses mark experimental points investigated by us. (B) Lipid lateral diffusion coefficients at different CHOL concentrations for the DMPC/CHOL system with 35 wt % 2H2O and at 298 K (circle), 303 K (triangle top down), 308 K (square), 313 K (diamond), 323 K (triangle top up), and 333 K (hexagon). The solid lines are drawn through the concentrations for which _D_L is approximately constant. These points are tentatively assigned to the _l_o one-phase area. (C) Arrhenius plots of the temperature dependence of the lipid lateral diffusion coefficient for the DMPC/CHOL system with 35 wt % 2H2O and varying CHOL contents of 0 (circles), 5 (triangle top down), 15 (square), 25 (diamond), and 35 mol % (triangle top up). The solid lines are Arrhenius fits to the data in the one-phase regions, whereas the dotted lines are the calculated _D_L according to the lever rule in the two-phase area. In these calculations the phase borders given by Almeida et al. (1992) have been used (thick lines in Fig. 5, A and B).
FIGURE 5
(A) Phase diagram of the DMPC/CHOL system according to Almeida et al. (1992). The crosses mark experimental points investigated by us. (B) Lipid lateral diffusion coefficients at different CHOL concentrations for the DMPC/CHOL system with 35 wt % 2H2O and at 298 K (circle), 303 K (triangle top down), 308 K (square), 313 K (diamond), 323 K (triangle top up), and 333 K (hexagon). The solid lines are drawn through the concentrations for which _D_L is approximately constant. These points are tentatively assigned to the _l_o one-phase area. (C) Arrhenius plots of the temperature dependence of the lipid lateral diffusion coefficient for the DMPC/CHOL system with 35 wt % 2H2O and varying CHOL contents of 0 (circles), 5 (triangle top down), 15 (square), 25 (diamond), and 35 mol % (triangle top up). The solid lines are Arrhenius fits to the data in the one-phase regions, whereas the dotted lines are the calculated _D_L according to the lever rule in the two-phase area. In these calculations the phase borders given by Almeida et al. (1992) have been used (thick lines in Fig. 5, A and B).
FIGURE 6
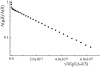
Concentration dependences of lipid lateral diffusion coefficients in oriented bilayers of the DOPC/CHOL system (A) and the POPC/CHOL system (B) with 30 wt % 2H2O and at 298 K (circles), 303 K (triangle top down), 308 K (square), 313 K (diamond), 323 K (triangle top up), and 333 K (hexagon).
FIGURE 6
Concentration dependences of lipid lateral diffusion coefficients in oriented bilayers of the DOPC/CHOL system (A) and the POPC/CHOL system (B) with 30 wt % 2H2O and at 298 K (circles), 303 K (triangle top down), 308 K (square), 313 K (diamond), 323 K (triangle top up), and 333 K (hexagon).
Similar articles
- Lipid lateral diffusion in ordered and disordered phases in raft mixtures.
Filippov A, Orädd G, Lindblom G. Filippov A, et al. Biophys J. 2004 Feb;86(2):891-6. doi: 10.1016/S0006-3495(04)74164-8. Biophys J. 2004. PMID: 14747324 Free PMC article. - Lipid lateral diffusion in bilayers with phosphatidylcholine, sphingomyelin and cholesterol. An NMR study of dynamics and lateral phase separation.
Lindblom G, Orädd G, Filippov A. Lindblom G, et al. Chem Phys Lipids. 2006 Jun;141(1-2):179-84. doi: 10.1016/j.chemphyslip.2006.02.011. Epub 2006 Mar 20. Chem Phys Lipids. 2006. PMID: 16580657 - Lateral diffusion coefficients of separate lipid species in a ternary raft-forming bilayer: a Pfg-NMR multinuclear study.
Orädd G, Westerman PW, Lindblom G. Orädd G, et al. Biophys J. 2005 Jul;89(1):315-20. doi: 10.1529/biophysj.105.061762. Epub 2005 Apr 29. Biophys J. 2005. PMID: 15863478 Free PMC article. - Lipid lateral diffusion and membrane heterogeneity.
Lindblom G, Orädd G. Lindblom G, et al. Biochim Biophys Acta. 2009 Jan;1788(1):234-44. doi: 10.1016/j.bbamem.2008.08.016. Epub 2008 Sep 6. Biochim Biophys Acta. 2009. PMID: 18805393 Review. - Cholesterol Bilayer Domains in the Eye Lens Health: A Review.
Widomska J, Subczynski WK, Mainali L, Raguz M. Widomska J, et al. Cell Biochem Biophys. 2017 Dec;75(3-4):387-398. doi: 10.1007/s12013-017-0812-7. Epub 2017 Jun 29. Cell Biochem Biophys. 2017. PMID: 28660427 Free PMC article. Review.
Cited by
- Vesicles surfing on a lipid bilayer: self-induced haptotactic motion.
Solon J, Streicher P, Richter R, Brochard-Wyart F, Bassereau P. Solon J, et al. Proc Natl Acad Sci U S A. 2006 Aug 15;103(33):12382-7. doi: 10.1073/pnas.0601400103. Epub 2006 Aug 4. Proc Natl Acad Sci U S A. 2006. PMID: 16891411 Free PMC article. - Lyophilization of Liposomal Formulations: Still Necessary, Still Challenging.
Franzé S, Selmin F, Samaritani E, Minghetti P, Cilurzo F. Franzé S, et al. Pharmaceutics. 2018 Aug 28;10(3):139. doi: 10.3390/pharmaceutics10030139. Pharmaceutics. 2018. PMID: 30154315 Free PMC article. Review. - Convergence and Sampling in Determining Free Energy Landscapes for Membrane Protein Association.
Domański J, Hedger G, Best RB, Stansfeld PJ, Sansom MSP. Domański J, et al. J Phys Chem B. 2017 Apr 20;121(15):3364-3375. doi: 10.1021/acs.jpcb.6b08445. Epub 2016 Nov 17. J Phys Chem B. 2017. PMID: 27807980 Free PMC article. - Phase diagram and tie-line determination for the ternary mixture DOPC/eSM/cholesterol.
Bezlyepkina N, Gracià RS, Shchelokovskyy P, Lipowsky R, Dimova R. Bezlyepkina N, et al. Biophys J. 2013 Apr 2;104(7):1456-64. doi: 10.1016/j.bpj.2013.02.024. Biophys J. 2013. PMID: 23561522 Free PMC article. - Effects of the lung surfactant protein B construct Mini-B on lipid bilayer order and topography.
Palleboina D, Waring AJ, Notter RH, Booth V, Morrow M. Palleboina D, et al. Eur Biophys J. 2012 Sep;41(9):755-67. doi: 10.1007/s00249-012-0850-4. Epub 2012 Aug 19. Eur Biophys J. 2012. PMID: 22903196 Free PMC article.
References
- Almeida, P. F. F., W. L. C. Vaz, and T. E. Thompson. 1992. Lateral diffusion in the liquid phases of dimyristoylphosphatidylcholine/cholesterol bilayers: a free volume analysis. Biochemistry. 31:6739–6747. - PubMed
- Brown, D. A. 1998. Structure and origin of ordered lipid domains in biological membranes. J. Membr. Biol. 164:103–114. - PubMed
- Callaghan, P. T. 1991. Principles of Nuclear Magnetic Resonance Microscopy. Clarendon, Oxford.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials