Membrane receptor trafficking: evidence of proximal and distal zones conferred by two independent endoplasmic reticulum localization signals - PubMed (original) (raw)
Membrane receptor trafficking: evidence of proximal and distal zones conferred by two independent endoplasmic reticulum localization signals
Sojin Shikano et al. Proc Natl Acad Sci U S A. 2003.
Abstract
The generic membrane trafficking signals of internal RXR and carboxyl-terminal KKXX motifs direct intracellular endoplasmic reticulum (ER) localization of the signal-bearing proteins. These signaling motifs play a critical role in partitioning proteins into designated subcellular compartments by functioning as an intracellular "zip code." In the process of determining the potential distinctions between these two otherwise functionally identical motifs, two functional zones of these signals were revealed. The KKXX signal was effective only when it was positioned closer to the membrane surface. In contrast, under identical conditions, the internal RXR signal was functional when it was positioned distally from the membrane. Different from the C-terminal KKXX signal, the internal RXR motif may be present in multiple copies. The receptor with multivalent RXR motifs displayed similar trafficking behavior to that of the same receptor with one copy of the RXR motif. The distinctive operating ranges from their anchored membrane surface provide experimental evidence for the notion that there are functional zoning layers within which membrane protein signal motifs are active.
Figures
Figure 1
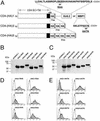
(A) A schematic diagram showing the coding regions of different CD4 constructs: CD4-(HA)1, CD4-(HA)3, and CD4-(HA)5. Amino acid positions are indicated at the top of the CD4-(HA)1 construct. Each of the CD4 constructs is fused with the listed protein sequences containing either the RKR motif from Kir6.2 (24) or the KKTN motif from WBP1. Specific site-directed mutations change RKR to RAA or KKTN to AATN. The number of residues that separate the transmembrane domain from the 36-aa Kir6.2 motif or WBP1 motif is indicated below each construct. (B and C) Immunoblot analyses of fusion proteins from the transfected cells. Protein lysates of transfected cells are identified by the constructs indicated at the top. The fusion protein expression was detected by anti-CD4 antibody. The molecular mass standard in kDa is indicated on the left of the gel. (D and E) FACS analyses of the surface expression are shown. The horizontal axis represents the logarithmic fluorescence intensity. The vertical axis represents the number of events (cells). Unfilled areas are signals from mock-transfected cells (negative control), and the filled areas are signals from cells transfected with the indicated CD4 constructs.
Figure 2
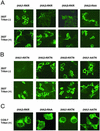
Immunocytochemistry analyses of surface expression chimeric CD4 receptors by confocal microscopy. (A) Immunostaining of 293T cells that were transiently transfected with the indicated RKR constructs (indicated on the top). Cells were stained before (Upper) and after (Lower) detergent treatments. (B) Immunostaining of 293T cells that were transiently transfected with the indicated KKTN constructs (indicated on the top). Cells were stained before (Upper) and after (Lower) detergent-mediated membrane permeation. (C) Immunostaining of COS-7 cells that were transiently transfected with the indicated RKR/RAA and KKTN/AATN constructs (indicated on the top). Cells were stained after detergent treatments.
Figure 3
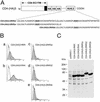
(A) A schematic diagram of chimeric CD4 receptor with one or two copies of the Kir6.2 RKR signal. (B) FACS analyses of the surface expression are shown. The horizontal axis represents the logarithmic florescence intensity. The vertical axis represents the number of events (cells). Unfilled areas are signals from mock-transfected cells (negative control), and the filled areas are signals from cells transfected with the indicated CD4 constructs. (C) Immunoblot analyses of fusion proteins from the transfected cells. Protein lysates of transfected cells are identified by the constructs indicated at the top. The fusion protein expression was detected by anti-CD4 antibody. An overexposed blot is shown to demonstrate the absence of construct-specific degradation. The molecular mass standard in kDa is indicated on the left of the gel.
Figure 4
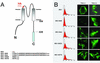
(A) A schematic diagram of the five IRK1 chimeric constructs. The site of the HA epitope insertion is as indicated. The peptide sequences fused to the C terminus are shown (Lower). (B) Different chimeric constructs were expressed and indicated (Left). The surface expression was detected by anti-HA antibody followed by FACS analyses (Left). The horizontal axis represents the logarithmic fluorescence intensity. The vertical axis represents the number of events (cells). Unfilled areas are signals from mock-transfected cells (negative control). Filled areas are the anti-HA signal. (Right) Immunofluorescence microscopy to detect the localization of indicated chimera in transfected cells by anti-HA under either permeabilized (Triton +) or nonpermeabilized (Triton −) condition.
Figure 5
(A) Histogram of quantification of surface expression of CD4 chimeric receptor as a function of spacing. The normalized surface expression of CD4-KKTN (Left) and RKR (Right) was determined by dividing the MESF values for RKR and KKTN constructs by those of RAA and AATN constructs (with the corresponding number of HA copies), respectively (see Materials and Methods). (B) A schematic diagram to show that the KKTN and RKR signals are active in proximal and distal zones. Repositioning the signals into a different zone by a posttranslational event including modification, folding, and protein interaction may cause the receptor to alter its subcellular localization. The membrane stop transfer signal (KR zone) immediately followed transmembrane segment is also shown.
Similar articles
- Scavenging of 14-3-3 proteins reveals their involvement in the cell-surface transport of ATP-sensitive K+ channels.
Heusser K, Yuan H, Neagoe I, Tarasov AI, Ashcroft FM, Schwappach B. Heusser K, et al. J Cell Sci. 2006 Oct 15;119(Pt 20):4353-63. doi: 10.1242/jcs.03196. J Cell Sci. 2006. PMID: 17038548 - Genetic isolation of transport signals directing cell surface expression.
Shikano S, Coblitz B, Sun H, Li M. Shikano S, et al. Nat Cell Biol. 2005 Oct;7(10):985-92. doi: 10.1038/ncb1297. Epub 2005 Sep 11. Nat Cell Biol. 2005. PMID: 16155591 - Role of ER export signals in controlling surface potassium channel numbers.
Ma D, Zerangue N, Lin YF, Collins A, Yu M, Jan YN, Jan LY. Ma D, et al. Science. 2001 Jan 12;291(5502):316-9. doi: 10.1126/science.291.5502.316. Science. 2001. PMID: 11209084 - Cell biology. Tracking an elusive receptor.
Kelly RB. Kelly RB. Nature. 1990 Jun 7;345(6275):480-1. doi: 10.1038/345480a0. Nature. 1990. PMID: 2161498 Review. No abstract available. - Receptor cross talk: ligand-gated ion channels start to communicate.
Brandon NJ, Moss SJ. Brandon NJ, et al. Sci STKE. 2000 Oct 24;2000(55):pe1. doi: 10.1126/stke.2000.55.pe1. Sci STKE. 2000. PMID: 11752615 Review.
Cited by
- Orthogonalized human protease control of secreted signals.
Aldrete CA, Call CC, Sant'Anna LE, Vlahos AE, Pei J, Cong Q, Gao XJ. Aldrete CA, et al. bioRxiv [Preprint]. 2024 Oct 21:2024.01.18.576308. doi: 10.1101/2024.01.18.576308. bioRxiv. 2024. PMID: 39484520 Free PMC article. Updated. Preprint. - Function recovery after chemobleaching (FRAC): evidence for activity silent membrane receptors on cell surface.
Sun H, Shikano S, Xiong Q, Li M. Sun H, et al. Proc Natl Acad Sci U S A. 2004 Nov 30;101(48):16964-9. doi: 10.1073/pnas.0404178101. Epub 2004 Nov 17. Proc Natl Acad Sci U S A. 2004. PMID: 15548608 Free PMC article. - Cytosolic N-terminal arginine-based signals together with a luminal signal target a type II membrane protein to the plant ER.
Boulaflous A, Saint-Jore-Dupas C, Herranz-Gordo MC, Pagny-Salehabadi S, Plasson C, Garidou F, Kiefer-Meyer MC, Ritzenthaler C, Faye L, Gomord V. Boulaflous A, et al. BMC Plant Biol. 2009 Dec 8;9:144. doi: 10.1186/1471-2229-9-144. BMC Plant Biol. 2009. PMID: 19995436 Free PMC article. - Research progress in the role and mechanism of LPCAT3 in metabolic related diseases and cancer.
Shao G, Qian Y, Lu L, Liu Y, Wu T, Ji G, Xu H. Shao G, et al. J Cancer. 2022 May 1;13(8):2430-2439. doi: 10.7150/jca.71619. eCollection 2022. J Cancer. 2022. PMID: 35711841 Free PMC article. Review. - Identification of Receptor Ligands in Apo B100 Reveals Potential Functional Domains.
Guevara J Jr, Romo J Jr, Hernandez E, Guevara NV. Guevara J Jr, et al. Protein J. 2018 Dec;37(6):548-571. doi: 10.1007/s10930-018-9792-8. Protein J. 2018. PMID: 30259240 Free PMC article.
References
- Mellman I, Warren G. Cell. 2000;100:99–112. - PubMed
- Teasdale R D, Jackson M R. Annu Rev Cell Dev Biol. 1996;12:27–54. - PubMed
- Standley S, Roche K W, McCallum J, Sans N, Wenthold R J. Neuron. 2000;28:887–898. - PubMed
- Ma D, Jan L Y. Curr Opin Neurobiol. 2002;12:287–292. - PubMed
- Nilsson T, Jackson M, Peterson P A. Cell. 1989;58:707–718. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources