Establishment of conditional vectors for hairpin siRNA knockdowns - PubMed (original) (raw)
Establishment of conditional vectors for hairpin siRNA knockdowns
Shiro Matsukura et al. Nucleic Acids Res. 2003.
Abstract
Small interference RNA (siRNA) is an emerging methodology in reverse genetics. Here we report the development of a new tetracycline-inducible vector-based siRNA system, which uses a tetracycline-responsive derivative of the U6 promoter and the tetracycline repressor for conditional in vivo transcription of short hairpin RNA. This method prevents potential lethality immediately after transfection of a vector when the targeted gene is indispensable, or the phenotype of the knockdown is lethal or results in a growth abnormality. We show that the controlled knockdown of DNA methyltransferase 1 (DNMT1) in human cancer resulted in growth arrest. Removal of the inducer, doxycycline, from treated cells led to re-expression of the targeted gene. Thus the method allows for a highly controlled approach to gene knockdown.
Figures
Figure 1
(A) Sequence comparison of the human U6 promoter and the modified U6 promoter with the bacterial tetracycline operon (tetO). The modified portion is indicated in bold. (B–E) Schematic illustrations of the vector construction process. (B) The U6 promoter sequence with an SphI recognition sequence (GCATG|C) was ligated into a TA cloning vector. (C) To preserve the first transcribed nucleotide as guanosine, the plasmid was digested with SphI and blunted with Klenow fragment. (D) To avoid hairpin formation of integrated sequences (3), DNA oligomers were ligated in two separate ligation reactions. Oligo1 was ligated following AatII digestion. (E) Oligo2 including the transcriptional termination signal T5 was ligated following AatII and ApaI digestion. (F–J) Schematic maps of vectors used in this study. (F) U6pro/DNMT1. DNMT1-targeting hairpin siRNA is driven by the human U6 promoter. (G) U6pro/Blank. As a control, the transcriptional termination signal T5 was ligated immediately downstream of the human U6 promoter. (H) U6pro/tetO/DNMT1. DNMT1-targeting hairpin siRNA is driven by the modified U6 promoter regulated by tetracycline. (I) U6pro/tetO/DNMT1/Hygro. U6pro/tetO/DNMT1 with the hygromycin resistance gene. (J) tetR/Neo. The tetR coding region was ligated in an expression vector with the neomycin resistance gene.
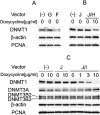
Figure 2
Western blot analysis of DNMT1 expression in cells transfected with various vectors. Vectors transfected into HCT116 cells are indicated with the symbols used in Figure 1. As a control, expression of β-actin and PCNA is shown. (A) Suppression of DNMT1 by siRNA targeting DNMT1 transcribed in vivo under non-selective conditions. (B) Suppression of DNMT1 by hairpin siRNA with inducible vectors. DNMT1 expression in HCT116 cells transfected with the plasmids indicated with or without treatment with doxycycline is shown. (C) DNMT1 knockdown with the inducible vector and selection marker. Expression of DNMT1, DNMT3A and DNMT3B was determined by western blot analysis. HCT116 cells transfected with the various plasmids indicated with or without doxycycline treatment are shown. Western blot analysis showed that DNMT1 levels were decreased by addition of doxycycline in a dose-dependent fashion, with complete suppression at 10 µg/ml. Expression of DNMT3A and DNMT3B was not affected by 10 µg/ml of doxycycline, showing the specificity of the knockdown.
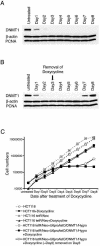
Figure 3
Reversibility of DNMT1 knockdowns by controlled expression of siRNA. (A) Western blot analysis of cell extracts from HCT116 cells containing the inducible vector. DNMT1 protein is not evident 3 days after addition of 10 µg/ml of doxycycline. β-Actin and PCNA are not affected. (B) DNMT1 protein reappears on day 7 following removal of doxycycline on day 3. (C) Growth rates of the indicated cell types show growth arrest in cells with the knockdown vector in the presence but not the absence of doxycycline. Note that these effects are reversible following removal of the doxycycline on day 3.
Similar articles
- Comparison of RNAi efficiency mediated by tetracycline-responsive H1 and U6 promoter variants in mammalian cell lines.
Henriksen JR, Løkke C, Hammerø M, Geerts D, Versteeg R, Flaegstad T, Einvik C. Henriksen JR, et al. Nucleic Acids Res. 2007;35(9):e67. doi: 10.1093/nar/gkm193. Epub 2007 Apr 10. Nucleic Acids Res. 2007. PMID: 17426119 Free PMC article. - Rapid construction of small interfering RNA-expressing adenoviral vectors on the basis of direct cloning of short hairpin RNA-coding DNAs.
Mizuguchi H, Funakoshi N, Hosono T, Sakurai F, Kawabata K, Yamaguchi T, Hayakawa T. Mizuguchi H, et al. Hum Gene Ther. 2007 Jan;18(1):74-80. doi: 10.1089/hum.2006.129. Hum Gene Ther. 2007. PMID: 17187476 - Establishment and utilization of a tetracycline-controlled inducible RNA interfering system to repress gene expression in chronic myelogenous leukemia cells.
Yang F, Zhang Y, Cao YL, Wang SH, Liu L. Yang F, et al. Acta Biochim Biophys Sin (Shanghai). 2005 Dec;37(12):851-6. doi: 10.1111/j.1745-7270.2005.00112.x. Acta Biochim Biophys Sin (Shanghai). 2005. PMID: 16331330 - Optimization of an siRNA-expression system with an improved hairpin and its significant suppressive effects in mammalian cells.
Miyagishi M, Sumimoto H, Miyoshi H, Kawakami Y, Taira K. Miyagishi M, et al. J Gene Med. 2004 Jul;6(7):715-23. doi: 10.1002/jgm.556. J Gene Med. 2004. PMID: 15241778 - Generation of an shRNAi expression library against the whole human transcripts.
Miyagishi M, Matsumoto S, Taira K. Miyagishi M, et al. Virus Res. 2004 Jun 1;102(1):117-24. doi: 10.1016/j.virusres.2004.01.022. Virus Res. 2004. PMID: 15068887 Review.
Cited by
- Histone methylation-mediated silencing of miR-139 enhances invasion of non-small-cell lung cancer.
Watanabe K, Amano Y, Ishikawa R, Sunohara M, Kage H, Ichinose J, Sano A, Nakajima J, Fukayama M, Yatomi Y, Nagase T, Ohishi N, Takai D. Watanabe K, et al. Cancer Med. 2015 Oct;4(10):1573-82. doi: 10.1002/cam4.505. Epub 2015 Aug 8. Cancer Med. 2015. PMID: 26256448 Free PMC article. - Comparison of RNAi efficiency mediated by tetracycline-responsive H1 and U6 promoter variants in mammalian cell lines.
Henriksen JR, Løkke C, Hammerø M, Geerts D, Versteeg R, Flaegstad T, Einvik C. Henriksen JR, et al. Nucleic Acids Res. 2007;35(9):e67. doi: 10.1093/nar/gkm193. Epub 2007 Apr 10. Nucleic Acids Res. 2007. PMID: 17426119 Free PMC article. - Tat-regulated expression of RNA interference: triggers for the treatment of HIV infection.
Unwalla H, Rossi JJ. Unwalla H, et al. Curr HIV/AIDS Rep. 2008 Feb;5(1):40-3. doi: 10.1007/s11904-008-0007-3. Curr HIV/AIDS Rep. 2008. PMID: 18417034 Free PMC article. - Metabotropic glutamate receptor 1 (Grm1) is an oncogene in epithelial cells.
Martino JJ, Wall BA, Mastrantoni E, Wilimczyk BJ, La Cava SN, Degenhardt K, White E, Chen S. Martino JJ, et al. Oncogene. 2013 Sep 12;32(37):4366-76. doi: 10.1038/onc.2012.471. Epub 2012 Oct 22. Oncogene. 2013. PMID: 23085756 Free PMC article. - Preparation of cell-lines for conditional knockdown of gene expression and measurement of the knockdown effects on E4orf4-induced cell death.
Brestovitsky A, Sharf R, Kleinberger T. Brestovitsky A, et al. J Vis Exp. 2012 Oct 21;(68):4442. doi: 10.3791/4442. J Vis Exp. 2012. PMID: 23117279 Free PMC article.
References
- Elbashir S.M., Harborth,J., Lendeckel,W., Yalcin,A., Weber,K. and Tuschl,T. (2001) Duplexes of 21-nucleotide RNAs mediate RNA interference in cultured mammalian cells. Nature, 411, 494–498. - PubMed
- Paul C.P., Good,P.D., Winer,I. and Engelke,D.R. (2002) Effective expression of small interfering RNA in human cells. Nat. Biotechnol., 20, 505–508. - PubMed
- Miyagishi M. and Taira,K. (2002) U6 promoter-driven siRNAs with four uridine 3′ overhangs efficiently suppress targeted gene expression in mammalian cells. Nat. Biotechnol., 20, 497–500. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources