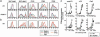Selective contribution of IFN-alpha/beta signaling to the maturation of dendritic cells induced by double-stranded RNA or viral infection - PubMed (original) (raw)
. 2003 Sep 16;100(19):10872-7.
doi: 10.1073/pnas.1934678100. Epub 2003 Sep 5.
Shinya Sakaguchi, Chigusa Nakajima, Ai Watanabe, Hideyuki Yanai, Misako Matsumoto, Toshiaki Ohteki, Tsuneyasu Kaisho, Akinori Takaoka, Shizuo Akira, Tsukasa Seya, Tadatsugu Taniguchi
Affiliations
- PMID: 12960379
- PMCID: PMC196895
- DOI: 10.1073/pnas.1934678100
Selective contribution of IFN-alpha/beta signaling to the maturation of dendritic cells induced by double-stranded RNA or viral infection
Kenya Honda et al. Proc Natl Acad Sci U S A. 2003.
Abstract
A complex mechanism may be operational for dendritic cell (DC) maturation, wherein Toll-like receptor and other signaling pathways may be coordinated differently depending on the nature of the pathogens, in order for DC maturation to be most effective to a given threat. Here, we show that IFN-alpha/beta signaling is selectively required for the maturation of DCs induced by double-stranded RNA or viral infection in vitro. Interestingly, the maturation is still observed in the absence of either of the two target genes of IFN-alpha/beta, TLR3 and PKR (double-stranded-RNA-dependent protein kinase R), indicating the complexity of the IFN-alpha/beta-induced transcriptional program in DCs. We also show that the DCs stimulated in vivo by these agents can migrate into the T cell zone of the spleen but fail to mature without the IFN signal. The immune system may have acquired the selective utilization of this cytokine system, which is essential for innate antiviral immunity, to effectively couple with the induction of adaptive immunity.
Figures
Fig. 1.
Selective requirement of IFN-α/β signaling in DC maturation in response to ds-RNA. (a) WT or IFNAR1–/– DCs were stimulated with poly(I:C), LPS, or CpG for 24 h and analyzed for cell-surface expression of the indicated molecules by flow cytometry. (b) WT DCs were stimulated with poly(I:C) or 1,000 units/ml IFN-β and analyzed by flow cytometry. (c) To measure the allostimulatory activity of DCs, poly(I:C)-, LPS-, or CpG-stimulated or unstimulated WT or IFNAR1–/– DCs were irradiated and incubated with allogeneic BALB/c CD4+ or CD8+ T cells at various cell concentrations as indicated. The percent of control [3H]thymidine incorporation was calculated as follows: (cpm obtained in the presence of stimulated DCs)/(cpm obtained in the presence of unstimulated DCs). Data are expressed as mean ± SD of triplicate samples. A representative of two experiments is shown.
Fig. 2.
IFN-α/β signaling in gene induction events and NF-κB activation in DCs. DCs were stimulated for the indicated time with poly(I:C) or LPS, and the kinetics of cytokine (a) and chemokine receptor (b) mRNA levels was determined by real-time RT-PCR. (c) NF-κB DNA binding activity in poly(I:C)- or LPS-stimulated DCs was measured by electrophoretic mobility-shift assay. The arrowhead indicates the NF-κB complex. The specificity of the NF-κB complex was determined by adding 100-fold molar excess of a specific competitor or a specific antibody to RelA. The same cell lysates were also analyzed by immunoblotting (IB) with the anti-RelA antibody to monitor protein expression level.
Fig. 3.
Role of TLR3 or PKR induced by IFN-α/β in poly(I:C)-stimulated DCs. (a) WT, TLR3–/–, or PKR–/– DCs were stimulated with poly(I:C) or LPS for 24 h and analyzed as in Fig. 1_a_.(b) Real-time RT-PCR analysis of indicated cytokine mRNAs after poly(I:C) stimulation in WT, TLR3–/–, or PKR–/– DCs.
Fig. 4.
Role of IFN-α/β signaling in live-virus-mediated DC maturation. (a) WT, TLR3–/–, PKR–/–, and IFNAR1–/– DCs were infected with NDV (0.5 hemagglutinating units) for 18 h, and the indicated molecules were analyzed by flow cytometry. (b and c) The induction of mRNAs for the indicated cytokines at the indicated time after NDV infection of WT, IFNAR1–/–, PKR–/–, or TLR3–/– DCs was monitored by real-time RT-PCR.
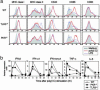
Fig. 5.
IFN signal-dependent amplification of maturation signaling on viral infection. (a) The kinetics of CD86 expression on NDV-stimulated WT or IFNAR1–/– DCs was analyzed by flow cytometry. The numbers indicate the mean fluorescent intensity of the cells. (b) NF-κB or IFN-stimulated gene factor 3 (ISGF3) activation at different time points was analyzed by electrophoretic mobility-shift assay. The filled and open arrowheads indicate the NF-κB and ISGF3 complex with the probe DNA, respectively. Cell lysates were also subjected to immunoblotting to analyze total RelA level. (c) The kinetics of chemokine receptor mRNA levels in NDV-stimulated DCs was determined by real-time RT-PCR. (d) Immunohistochemical analyses of spleens from WT and IFNAR1–/– mice were performed after i.p. injection with control PBS, poly(I:C) (100 μg per body), NDV (250 hemagglutinating units per body), or LPS (200 ng per body). Spleen sections were doubly stained with antibodies against B220 (FITC; green) and CD11c or CD86 (Alexa Fluor 546; red). It is worth noting that CD11c– CD86high cells are detected in the marginal zone of the spleen of both virus-infected WT and IFNAR1–/– mice, but the nature of these cells remains unknown.
Similar articles
- Immunoadjuvant effects of polyadenylic:polyuridylic acids through TLR3 and TLR7.
Sugiyama T, Hoshino K, Saito M, Yano T, Sasaki I, Yamazaki C, Akira S, Kaisho T. Sugiyama T, et al. Int Immunol. 2008 Jan;20(1):1-9. doi: 10.1093/intimm/dxm112. Epub 2007 Nov 1. Int Immunol. 2008. PMID: 17981792 - Role of double-stranded RNA and Npro of classical swine fever virus in the activation of monocyte-derived dendritic cells.
Bauhofer O, Summerfield A, McCullough KC, Ruggli N. Bauhofer O, et al. Virology. 2005 Dec 5;343(1):93-105. doi: 10.1016/j.virol.2005.08.016. Epub 2005 Sep 8. Virology. 2005. PMID: 16154171 - IRF-7 is the master regulator of type-I interferon-dependent immune responses.
Honda K, Yanai H, Negishi H, Asagiri M, Sato M, Mizutani T, Shimada N, Ohba Y, Takaoka A, Yoshida N, Taniguchi T. Honda K, et al. Nature. 2005 Apr 7;434(7034):772-7. doi: 10.1038/nature03464. Epub 2005 Mar 30. Nature. 2005. PMID: 15800576 - Transcriptional signaling by double-stranded RNA: role of TLR3.
Sen GC, Sarkar SN. Sen GC, et al. Cytokine Growth Factor Rev. 2005 Feb;16(1):1-14. doi: 10.1016/j.cytogfr.2005.01.006. Cytokine Growth Factor Rev. 2005. PMID: 15733829 Review. - TLR3: interferon induction by double-stranded RNA including poly(I:C).
Matsumoto M, Seya T. Matsumoto M, et al. Adv Drug Deliv Rev. 2008 Apr 29;60(7):805-12. doi: 10.1016/j.addr.2007.11.005. Epub 2008 Jan 2. Adv Drug Deliv Rev. 2008. PMID: 18262679 Review.
Cited by
- Type I IFN drives a distinctive dendritic cell maturation phenotype that allows continued class II MHC synthesis and antigen processing.
Simmons DP, Wearsch PA, Canaday DH, Meyerson HJ, Liu YC, Wang Y, Boom WH, Harding CV. Simmons DP, et al. J Immunol. 2012 Apr 1;188(7):3116-26. doi: 10.4049/jimmunol.1101313. Epub 2012 Feb 27. J Immunol. 2012. PMID: 22371391 Free PMC article. - Interferon-α is the primary plasma type-I IFN in HIV-1 infection and correlates with immune activation and disease markers.
Hardy GA, Sieg S, Rodriguez B, Anthony D, Asaad R, Jiang W, Mudd J, Schacker T, Funderburg NT, Pilch-Cooper HA, Debernardo R, Rabin RL, Lederman MM, Harding CV. Hardy GA, et al. PLoS One. 2013;8(2):e56527. doi: 10.1371/journal.pone.0056527. Epub 2013 Feb 20. PLoS One. 2013. PMID: 23437155 Free PMC article. - Computational approaches to understanding dendritic cell responses to influenza virus infection.
Zaslavsky E, Hayot F, Sealfon SC. Zaslavsky E, et al. Immunol Res. 2012 Dec;54(1-3):160-8. doi: 10.1007/s12026-012-8322-6. Immunol Res. 2012. PMID: 22544465 Free PMC article. Review. - Age-dependent TLR3 expression of the intestinal epithelium contributes to rotavirus susceptibility.
Pott J, Stockinger S, Torow N, Smoczek A, Lindner C, McInerney G, Bäckhed F, Baumann U, Pabst O, Bleich A, Hornef MW. Pott J, et al. PLoS Pathog. 2012;8(5):e1002670. doi: 10.1371/journal.ppat.1002670. Epub 2012 May 3. PLoS Pathog. 2012. PMID: 22570612 Free PMC article. - Cell-cell fusion induced by measles virus amplifies the type I interferon response.
Herschke F, Plumet S, Duhen T, Azocar O, Druelle J, Laine D, Wild TF, Rabourdin-Combe C, Gerlier D, Valentin H. Herschke F, et al. J Virol. 2007 Dec;81(23):12859-71. doi: 10.1128/JVI.00078-07. Epub 2007 Sep 26. J Virol. 2007. PMID: 17898060 Free PMC article.
References
- Medzhitov, R. & Janeway, C. A., Jr. (1997) Curr. Opin. Immunol. 9, 4–9. - PubMed
- Akira, S., Takeda, K. & Kaisho, T. (2001) Nat. Immunol. 2, 675–680. - PubMed
- Barton, G. M. & Medzhitov, R. (2003) Science 300, 1524–1525. - PubMed
- Banchereau, J. & Steinman, R. M. (1998) Nature 392, 245–252. - PubMed
- Balachandran, S., Roberts, P. C., Brown, L. E., Truong, H., Pattnaik, A. K., Archer, D. R. & Barber, G. N. (2000) Immunity 13, 129–141. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases